Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sonu Gandhi | + 2551 word(s) | 2551 | 2021-05-10 10:21:13 | | | |
| 2 | Bruce Ren | -21 word(s) | 2530 | 2021-05-17 11:31:02 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Gandhi, S. Magnetic Nanoparticles for Diagnosis. Encyclopedia. Available online: https://encyclopedia.pub/entry/9702 (accessed on 24 May 2026).
Gandhi S. Magnetic Nanoparticles for Diagnosis. Encyclopedia. Available at: https://encyclopedia.pub/entry/9702. Accessed May 24, 2026.
Gandhi, Sonu. "Magnetic Nanoparticles for Diagnosis" Encyclopedia, https://encyclopedia.pub/entry/9702 (accessed May 24, 2026).
Gandhi, S. (2021, May 17). Magnetic Nanoparticles for Diagnosis. In Encyclopedia. https://encyclopedia.pub/entry/9702
Gandhi, Sonu. "Magnetic Nanoparticles for Diagnosis." Encyclopedia. Web. 17 May, 2021.
Copy Citation
Magnetic nanoparticles gained considerable attention in last few years due to their remarkable properties. Superparamaganetism, non-toxicity, biocompatibility, chemical inertness, and environmental friendliness are some of the properties that make iron oxide nanoparticles (IONPs) an ideal choice for biomedical applications. Along with being easily tuneable and a tailored surface for conjugation of IONPs, their physio-chemical and biological properties can also be varied by modifying the basic parameters for synthesis that enhances the additional possibilities for designing novel magnetic nanomaterial for theranostic applications.
iron oxide nanoparticles
cancer
superparamagnetism
tumor microenvironment
magnetic nanoparticles
1. Introduction
Early diagnosis and therapy of crucial diseases are pertinent for accurate diagnosis in a timely manner. Nanotechnology-based approaches paved the way for the diagnosis of various diseases. Improvement in the sensitivity and accuracy of diagnosis paved the way for efficient and early diagnosis. Various non-invasive in-vivo imaging techniques were developed for the diagnosis to visualize the progression of abnormal cells by binding to the target site. These non-invasive techniques include magnetic resonance imaging (MRI), single-photon emission computed tomography (SPECT), computed tomography (CT), nuclear imaging of positron emission tomography (PET), and optical (or fluorescence) imaging. The above said techniques are based on in-vivo imaging that assist the clinicians to locate the physiological and anatomical state of the organs.
IONPs majorly are used as a contrast agent in imaging, targeting, diagnostics, and treatment of hyperthermia. Magnetite (Fe3O4), maghemite (γ- Fe2O3) and hematite (α- Fe2O3) are some of the important examples of SPIONs [1]. The presence of Fe2+ and Fe3+ ions make SPIONs ideal candidate for biomedical applications. Superparamagnetic iron oxide nanoparticles (SPIONs) consisted of a ferrimagnetic group of the magnetic particles with major applications in biomedicine and bioengineering. Currently, SPIONs based therapeutics are at clinical, and preclinical trials, and few already reached to the market [2]. However, cellular toxicity occurred by SPIONs could potentially lead to adverse effects such as apoptosis, DNA damage, reactive oxygen species (ROS) generation, inflammation, mitochondrial function impairment, and chromosome condensation.
Polymeric materials such as polyethylene glycol (PEG), starch, and dextran polymers were widely used for the coating of SPIONs due to less toxicity, escaped by macrophages or spleen cells [3]. Advancement in the field of stimuli responsive polymers has occurred, due to a reversible phase transition upon exposure to environmental changes such as temperature, pH, light, enzyme, and the magnetic field [4]. The hydrophilic capsule of lipids due to its amphiphillic properties used for the coating of SPIONs includes various methods such as co-precipitation, thermal decomposition, sol-gel, microemulsion, electrochemical, and biosynthesis [5][6][7]. The present review aims to provide an insight into biomedical and clinical applications such as Magnetic Resonance Imaging (MRI), Magnetic Particle Imaging (MPI), Computed Tomography (CT), Positron Emission Tomography (PET), nanozyme applications for biosensing, gene and drug delivery, and hyperthermia and photothermal therapy, and broad spectrum antimicrobial applications in diagnosis of various diseases.
2. Biomedical and Clinical Applications
2.1. Magnetic Resonance Imaging (MRI)
MRI is an imaging technique that employs a strong magnetic field and radio waves for the creation of high-resolution images of various organs in the human and animal body. It has been used very widely in clinical radiology that uses non-ionizing radiation in experimental settings. Additionally, the modality is tomographic with the penetration of soft tissue with high resolution [4][8].
MRI utilizes the magnetic field, radio waves, or electric fields to illustrate the detailed internal structure of the body. MRI contrast agents can be divided into two categories, T1 and T2 agents. T1 agents alter the longitudinal relaxation time of water protons whereas T2 agents alter the transverse relaxation time of water protons. Positive T1—weighted, it shortens the T1 and has a moderate effect on T2 providing a brighter image. On the contrary negative contrast of T2/T2* weighted and shortens the T2 relaxation time and leads to the formation of dark images [9]. Tracking and monitoring of treatment, delivery of cells into the body, its proliferation, diffusion time, and migration rate require in-vivo imaging of the cells. Various imaging techniques are being used for this purpose, such as positron emission tomography (PET), single positron emission computed tomography (SPECT), X-ray based computed tomography (CT), and magnetic resonance imaging (MRI). Among all, MRI has shown to have a high spatial resolution up to 100 µm, in the absence of exposure to ionizing radiation [8], which makes it a great imaging technique for in-vivo cell imaging.
SPIONs are extensively used as contrast agents due to their superparamagnetic properties, biocompatibility, and low cost. SPIONs are used for visualizing tumor and metastatic cancer in liver, spleen, lymph nodes, and as a blood pool agent for angiography as well for inflammatory lesions. The nanoparticles reduce the relaxation time of the surrounding protons due to their superparamagnetic properties, which makes them suitable candidates as MRI contrast agents. IONPs based contrast agents were chosen over gadolinium-based contrast agents due to their low toxicity [10]. Ferucarbotran Resovist by Bayer Schering Pharma AG, contrast the effect of T1/T2, but most of their negative contrast and approved in few countries such as Japan, EU, and Australia [11]. Ferumoxytol (Feraheme) IONPs are approved by the FDA for the treatment of iron deficiency in patients suffering from chronic kidney diseases [12][13].
2.2. Magnetic Particle Imaging (MPI)
MPI is a tomographic technique that is non-invasive and detects tracers particles with superparamagnetic properties. MPI has potential applications in the field of diagnostics, imaging, and materials properties. MPI can be used to locate and measure the concentration of nanoparticles with non-ionizing radiation at any depth within the body to produce a signal. MPI signals are directly generated from the superparamagnetic nanoparticles when magnetized, which is 107 times more sensitive than MRI signals. Magnetic particle spectrometer (MPS) has the potential for 3D in-vivo imaging with high special resolution where SPIONs were employed for lactoferrin conjugation. The MPS signal was recorded using a custom-built magnetic particle spectrometer (MPS) [14][15]. Overexpressed cancer cells secrete specific proteases, such as trypsin, matrix metalloprotease-2 (MMP-2), which was detected via MPS. Neutravidin-coated SPIONs were aggregated in the presence of biotin labeled peptides that were later selectively identified and cleaved by specific proteases which was present in the peptides (protease cleavage site) which led to the dispersion of the SPIONs. The aggregation or dispersion state of the SPIONs were identified by MPS signals through magnetic relaxation characteristics. Hence, MPS can be used in biomedical applications such as rapid detection of proteases in biological samples for example blood, urine, tissue extracts, and cell culture media for diagnosis of various types of cancer [16].
2.3. Computed Tomography (CT)
A computed tomography can be used in radiology for medical imaging to acquire detailed images of the body in a non-invasive manner. A CT scan measures X-ray attenuations of different tissues inside the body with rotating x-ray tube and detectors. The X-ray measurements usually taken multiple times from different angles to process on a computer to reconstruct the algorithms for the generation of tomographic images of a body. Contrast agents for CT are injected intravenously, the widely used agents are iodine conjugated small particles, and gadolinium-based contrast agents. However, patients with renal impairment are reported to be highly sensitive to iodine-based contrast agents, and gadolinium-based agents show toxicity in cell or tissues. Therefore, iron-oxide-based nanoparticles were taken into consideration as an alternate of iodine-based contrast agents [17][18].
Gold-coated iron oxide glycol nanoparticles can be used as an effective contrast agent for imaging in CT. It was confirmed that small size magnetic nanoparticles are highly biocompatible, biodegradable, and possessive of X-ray attenuation characteristics, and represent low toxicity over a longer period of time. These characteristics make them a strong candidate for both CT and MRI imaging [19][20]. Naha et al. synthesized a nanocomposite of bismuth-IONPs with dextran coating to check the cytotoxicity, accumulation, and half-life of the nanoconjugate. Lower cytotoxicity was observed in HepG2 (Human liver cancer cell line), and CT imaging was done after overnight incubation with nanoparticles to observe the contrast in blood vessels and heart. The nanoconjugate was found to have a protracted circulation half-life in the body. It was established that the nanoconjugate had a strong X-ray attenuation property and, biocompatibility that make it a good candidate as contrast agent in CT and MRI [19].
Reguera et al. designed and synthesized gold-iron-oxid- based Janus magnetic-plasmonic nanoparticles, which were used as a contrast agent for numerous techniques such as CT (Computed tomography), MRI (magnetic resonance imaging), TEM (transmission electron microscopy), PAI (photo acoustic imaging), SERS (surface enhanced Raman spectroscopy), optical imaging. Hence, by using these multifunctional nanoparticle agents, maximum cellular information can be obtained at once making them serve as great imaging tool in biomedical platform [21]. However, the adverse effects caused by the use of ionizing radiations sometimes restrict use in the patients.
2.4. PET (Positron Emission Tomography)
Positron emission tomography (PET) is an imaging technique that employs radioactive material known as radio-tracers to envision and measure changes in metabolic events. PET enables localization and quantification of activities in specific organs or tissues for whole body imaging; however, it cannot reveal about the anatomy or morphology of the tissues. Different tracers were used for imaging, based on the target such as 18F-FDG for cancer, NaF-F18 for bone formation, and oxygen-15 to measure blood flow. In PET, a radioisotope was attached to a drug, followed by injection into the body as a tracer. The emitted gamma rays were detected and used for the reconstruction of 3-D image. PET scanners can be combined with a CT scanner in the same session and are known as PET-CT scanners.
Torres et al. developed a novel contrast agent composed of IONPs (Feridex) labeled with a 64Cu based bi-functional chelator for in-vivo imaging of lymph node [22]. Tri-modality agents were synthesized by conjugation of a PET tracer 64Cu with dextran coated magneto fluorescent nanoparticles for PET, MRI, and fluorescence imaging (Figure 1). Tri-modality nanoparticles were used to detect macrophages in atherosclerotic patients [23]. Stelter et al. functionalized IONPs with Ga-DTPA to synthesize a Ga-DTPA-IONP complex and injected into mouse model. PET and magnetic resonance imaging were performed to observe the accumulation of IONPs in kidney and spleen regions [24]. Glaus et al. synthesized IONPs micelles, which were radioactively labeled by 64Cu using DOTA as a chelating agent. In-vivo biodistribution was observed after 24 h of injection in mice through PET and MRI with 143 min half-life of the radioactive nanoconjugate, leading to accumulation in hepatic and splenic region [25]. Aryal et al. utilized 64Cu labeled PLGA functionalized SPIONs to locate breast cancer cells in a xenograft mouse tumor model. The nanohybrid was selected due to its longer circulation time as the nanohybrid was taken up by the tumor cells via EPR effect and located at the tumor region using PET and T2 weighed MRI [26]. Some SPIONs are approved by US-FDA for imaging applications in clinical settings (Table 1).
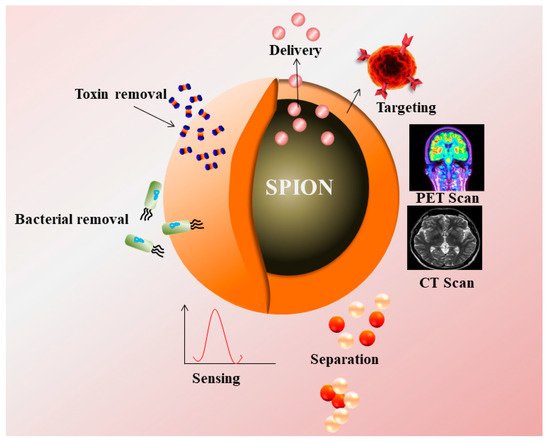
Figure 1. Superparamagnetic nanoparticles (SPIONs) as a contrast agent in PET, CT Scan, and their applications in targeting, drug delivery, removal of toxin, bacteria, and sensing.
Table 1. Clinically approved IONPs conjugated compounds and its applications at various biomedical platform.
| Serial No. | Compound Name | Coating | Applications | Clinically Approved | References |
|---|---|---|---|---|---|
| 1 | Ferumoxtran (Combidex®) |
Dextran | Lymph node imaging, macrophage imaging, blood pool agent, cell labelling, CNS imaging, MRI | In clinical trials | [27][28] |
| 2 | Ferucarbotran (Resovist®) |
Carboxydextran | Liver imaging, cell labelling, CNS imaging, MRI | Approved | [29][30][31] |
| 3 | Ferumoxide (Feridex®) |
Dextran | Liver imaging, cell labelling, CNS imaging, MRI | Withdrawn from market | [32][33][34] |
| 4 | Ferumoxytol (Feraheme®) |
Carboxymethyl-dextran | Iron replacement therapy in patients with chronic kidney diseases | Approved | [35] |
| 5 | Feruglose (Clariscan™) |
PEGylated starch | Blood pool agent, MRI | In clinical trials | [36] |
| 6 | Ferumoxsil (Gastromark®) |
Siloxane | Oral GI imaging | Approved | [13][37] |
2.5. Nanozyme Applications for Biosensing
Nanozymes are the nanoparticles that exhibit the properties of enzymes such as oxidase, peroxidase, catalase, and superoxide dismutase. Various metals as well as metal oxides such as gold, silver, copper, SPIONs, and graphene nanosheets have shown HRP (horse radish peroxidase) like activities, which are currently being used for fabrication of biosensors. These non-enzymatic biosensors were used for detection of cholesterol concentration, creatinine, glucose, glutathione, H2O2, and urea. These biomolecules acted as biomarkers for various diseases, and biosensors developed for these molecules were used for early diagnosis of the diseases [38][39][40][41][42]. Studies showed that coating SPIONs with anionic SPIONs showed a higher affinity for TMB (3,3′,5,5′-tetramethylbenzidine), and cationic SPIONs for ABTS (2,2′-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid) and OPD (O-phenylenediamine). Compared to natural enzymes, the nanozymes have various disadvantages such as slow substrate affinity and specificity. Therefore, novel methods for coating were used to enhance the catalytic activities of the nanozymes. Qian et al. synthesized nanozymes via conjugation of reduced graphene oxide on Fe3O4 nanospheres that was found to be stable for about three months when stored at 4 °C [43].
Super paramagnetic iron oxide nanoparticles (SPIONs) have catalytic properties of peroxides and catalase, which play an important role in preventing the cellular oxidative damage. Magnetite based iron oxide nanoparticles (IONPs) mimic the peroxidase activity and are used for fabrication of peroxidase based biosensors. Unlike the peroxidase enzyme, these IONPs do not degrade at unfavorable temperatures and pH (Table 2). Kacar et al. developed an amperometric sensor for creatinine using Fe3O4 nanoparticles that were modified with carbon paste electrodes. This method required two catalytic reactions, creatinase and sarcosine oxidase, to produce H2O2. The sensor detected the presence of H2O2 to determine the creatine level. The presence of elevated levels of creatine in urine or blood indicated the renal abnormalities or failure [44]. Gao et al. synthesized chitosan coated magnetic nanoparticles (CS-MNPs) for capture detection immunoassay, which detected the carcinoembryonic antigen (CEA) with a limit of detection (LOD) up to 1 ng/mL via sandwich ELISA, and direct ELISA [45]. Currently, various pathogens were detected by novel immunoassays such as IgG, Mycoplasma pneumonia, Vibrio cholera, Rotavirus, Hepatocellular Carcinoma biomarker, Golgi protein 73 (GP73) [46], Human Chorionic Gonadotropin (HCG), and cancer cells with Human Epidermal Growth Factor Receptor 2 (HER2) and Epidermal Growth Factor Receptor (EGFR) [47]. Peter et al. developed positively charged nanoparticles and monitored their localization in-situ by optical biosensor and transmission electron microscope (TEM). The positively charged nanoparticles penetrated well into the cells as compared with negatively charged particles with an optimal size around 5 nm for the cellular uptake [48]. A lateral flow assay was developed with IONPs’ nanozymes strip by Duan et al. for the detection of Ebola virus (EBOV). The nanostrip was capable to detect EBOV glycoprotein as low as 1 ng/mL (Figure 2). The diagnostic capability was compared with ELISA that required less time to detect EBOV [49].
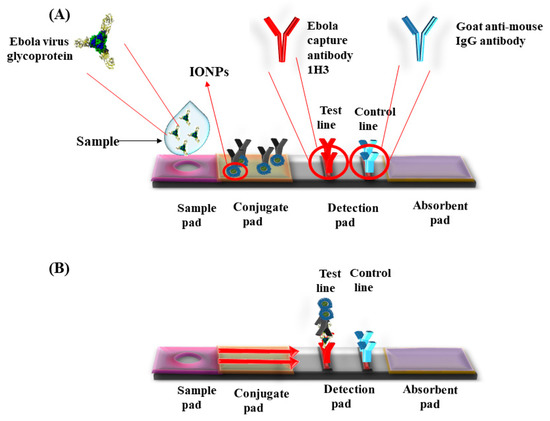
Figure 2. Lateral flow dipstick device for the detection of Ebola virus using Iron oxide nanoparticles (IONPs). (A) Ebola capture antibody 1H3 coated on test line and control line coated with goat anti-mouse IgG antibody. Sample was applied in the sample well. (B) The sample runs via capillary action on the membrane. Presence of specific antigen for Ebola developed color in the detection pad.
Table 2. Chemically synthesized and modified IONPs nanozymes and its application for the detection of various biological compounds.
| Serial No. | IONPs/Conjugated IONPs | Applications | References |
|---|---|---|---|
| 1 | Fe3O4 NPs | Exhibit peroxidase enzyme like activity. Used for fluorescent turn off system for detection of protein in urine | [50] |
| 2 | Chitosan coated IONPs with urease IONPs | Used for detection of urea | [51] |
| 3 | IONPs | Detection of Brucella antibodies with a LOD of 0.05 µg/mL | [52] |
| 4 | Fe3O4 nanocomposites/graphene oxide | Biosensor synthesis for glucose detection with the range 0.5–10 mM | [53] |
| 5 | Fe3O4 NPs loaded in Co3O4 nanocages | Used for glucose detection with the range of 0.5–30 µM with an LOD of 0.05 µM | [54] |
References
- Bowles, J.F.W.; Cornell, R.M.; Schwertmann, U. The Iron Oxides: Structure, Properties Reactions Occurrence and Uses. Weinheim and New York (VCH Verlagsgeseiischaft mbH). Mineralogical Magazine 1996, 61, 740–741.
- Wu, W.; Wu, Z.; Yu, T.; Jiang, C.; Kim, W.S. Recent progress on magnetic iron oxide nanoparticles: Synthesis, surface functional strategies and biomedical applications. Sci. Technol. Adv. Mat. 2015, 16, 023501.
- Tassa, C.; Shaw, S.Y.; Weissleder, R. Dextran-coated iron oxide nanoparticles: A versatile platform for targeted molecular imaging, molecular diagnostics, and therapy. Acc. Chem. Res. 2011, 44, 842–852.
- Sun, S.N.; Wei, C.; Zhu, Z.Z.; Hou, Y.L.; Venkatraman, S.S.; Xu, Z.C. Magnetic iron oxide nanoparticles: Synthesis and surface coating techniques for biomedical applications. Chi. Phy. B 2014, 23, 037503.
- Mohapatra, M.; Anand, S. Synthesis and applications of nano-structured iron oxides/hydroxides—a review. Int. J. Eng. Sci. Technol. 2011, 2.
- Bárcena, C.; Sra, A.K.; Gao, J. Applications of magnetic nanoparticles in biomedicine. Nanoscale Mag. Mat. and Appl. 2009.
- Laurent, S.; Forge, D.; Port, M.; Roch, A.; Robic, C.; Elst, L.V.; Muller, R.N. Magnetic iron oxide nanoparticles: Synthesis, stabilization, vectorization, physicochemical characterizations and biological applications. Chem. Rev. 2008, 108, 2064–2110.
- Li, L.; Jiang, W.; Luo, K.; Song, H.; Lan, F.; Wu, Y.; Gu, Z. Superparamagnetic iron oxide nanoparticles as MRI contrast agents for non-invasive stem cell labeling and tracking. Theranostics 2013, 3, 595–615.
- Wang, G.; Gao, W.; Zhang, X.; Mei, X. Au Nanocage Functionalized with Ultra-small Fe3O4 Nanoparticles for Targeting T1-T2 Dual MRI and CT Imaging of Tumor. Sci Rep. 2016, 28258.
- Waters, E.A.; Wickline, S.A. Contrast agents for MRI. Basic Res. Cardiol. 2008, 103, 114–121.
- Skopalik, J.; Polakova, K.; Havrdova, M.; Justan, I.; Magro, M.; Milde, D.; Knopfova, L.; Jan Smarda, J.; Polakova, H.; Gabrielova, E.; et al. Mesenchymal stromal cell labeling by new uncoated superparamagnetic maghemite nanoparticles in comparison with commercial Resovist--an initial in vitro study. Int. J. Nanomed. 2014, 5355.
- Aghighi, M.; Golovko, D.; Ansari, C.; Marina, N.M.; Pisani, L.; Kurlander, L.; Klenk, C.; Bhaumik, S.; Wendland, M.; Daldrup-Link, H.E. Imaging tumor necrosis with ferumoxytol. PLoS ONE 2015, 10, e0142665.
- Jung, C.W. Surface properties of superparamagnetic iron oxide MR contrast agents: Ferumoxides, ferumoxtran, ferumoxsil. Mag. Res. Imag. 1995, 13, 675–691.
- Weizenecker, J.; Gleich, B.; Rahmer, J.; Dahnke, H.; Borgert, J. Three-dimensional real-time in vivo magnetic particle imaging. Phys. Med. Biol. 2009, 54, L1–L10.
- Tomitaka, A.; Arami, H.; Gandhi, S.; Krishnan, K.M. Lactoferrin conjugated iron oxide nanoparticles for targeting brain glioma cells in magnetic particle imaging. Nanoscale 2015, 7, 16890–16898.
- Gandhi, S.; Arami, H.; Krishnan, K.M. Detection of Cancer-Specific Proteases Using Magnetic Relaxation of Peptide-Conjugated Nanoparticles in Biological Environment. Nano Lett. 2016, 16, 3668–3674.
- Cormode, D.P.; Naha, P.C.; Fayad, Z.A. Nanoparticle contrast agents for computed tomography: A focus on micelles. Contrast. Media Mol. Imag. 2014, 9, 37–52.
- Thomas, R.; Park, I.K.; Jeong, Y.Y. Magnetic iron oxide nanoparticles for multimodal imaging and therapy of cancer. Int. J. Mol. Sci. 2013, 14, 15910–15930.
- Naha, P.C.; Al Zaki, A.; Hecht, E.; Chorny, M.; Chhour, P.; Blankemeyer, E.; Yates, D.M.; Walter, W.R.; Litt, H.I. Dextran coated bismuth-iron oxide nanohybrid contrast agents for computed tomography and magnetic resonance imaging. J. Mat. Chem. B. 2014, 46, 8239–8248.
- Xue, S.; Wang, Y.; Wang, M.; Zhang, L.; Du, X.; Gu, H.; Zhang, C. Iodinated oil-loaded, fluorescent mesoporous silica-coated iron oxide nanoparticles for magnetic resonance imaging/computed tomography/fluorescence trimodal imaging. Int. J. Nanomed. 2014, 9, 2527–2534.
- Reguera, J.; Jiménez, D.; Aberasturi, D.; Henriksen-Lacey, M.; Langer, J.; Espinosa, A.; Szczupak, B.; Wilhelm, C.; Liz-Marzán, L.M. Janus plasmonic-magnetic gold-iron oxide nanoparticles as contrast agents for multimodal imaging. Nanoscale 2017, 9, 9467–9480.
- Torresmartinderosales, R.; Tavare, R.; Paul, R.L.; Jauregui-Osoro, M.; Protti, A.; Glaria, A.; Varma, G.; Szanda, I.; Blower, P.J. Synthesis of 64CuII- bis(dithiocarbamatebisphosphonate) and its conjugation with superparamagnetic iron oxide nanoparticles: In vivo evaluation as dual-modality PET-MRI agent. Angew. Chemie Int. Ed. 2011, 2, 8239–8248.
- Nahrendorf, M.; Zhang, H.; Hembrador, S.; Panizzi, P.; Sosnovik, D.E.; Aikawa, E.; Libby, P.; Swirski, F.K.; Weissleder, R. Nanoparticle PET-CT imaging of macrophages in inflammatory atherosclerosis. Circulation 2008, 117, 379–387.
- Stelter, L.; Pinkernelle, J.G.; Michel, R.; Schwartländer, R.; Raschzok, N.; Morgul, M.H.; Koch, M.; Denecke, T.; Ruf, J.; Bäumler, H.; et al. Modification of aminosilanized superparamagnetic nanoparticles: Feasibility of multimodal detection using 3T MRI, small animal PET, and fluorescence imaging. Mol. Imaging Biol. 2010, 12, 25–34.
- Glaus, C.; Rossin, R.; Welch, M.J.; Bao, G. In vivo evaluation of 64Cu-labeled magnetic nanoparticles as a dual-modality PET/MR imaging agent. Bioconjug. Chem. 2010, 21, 715–722.
- Aryal, S.; Key, J.; Stigliano, C.; Landis, M.D.; Lee, D.Y.; Decuzzi, P. Positron emitting magnetic nanoconstructs for PET/MR imaging. Small 2014, 10, 2688–2696.
- Kernstine, K.H.; Stanford, W.; Mullan, B.F.; Rossi, N.P.; Thompson, B.H.; Bushnell, D.L.; McLaughlin, K.A.; Kern, J.A. PET, CT, and MRI with Combidex for mediastinal staging in non-small cell lung carcinoma. Ann. Thoracic Surg. 1999, 68, 1022–1028.
- Harisinghani, M.G.; Saini, S.; Weissleder, R.; Hahn, P.F.; Yantiss, R.K.; Tempany, C.; Wood, B.J.; Mueller, P.R. MR lymphangiography using ultrasmall superparamagnetic iron oxide in patients with primary abdominal and pelvic malignancies: Radiographic- pathologic correlation. Americ. J. Roentgenol. 1999, 172, 1347–1351.
- Reimer, P.; Balzer, T. Ferucarbotran (Resovist): A new clinically approved RES-specific contrast agent for contrast-enhanced MRI of the liver: Properties, clinical development, and applications. Europ. Radiol. 2003, 13, 1266–1276.
- Vogl, T.J.; Hammerstingl, R.; Schwarz, W.; Kümmel, S.; Müller, P.K.; Balzer, T.; Lauten, M.J.; Balzer, J.O.; Mack, M.G.; Schimpfky, C.; et al. Magnetic resonance imaging of focal liver lesions: Comparison of the superparamagnetic iron oxide resovist versus gadolinium-DTPA in the same patient. Invest. Radiol. 1996, 31, 696–708.
- Reimer, P.; Rummeny, E.J.; Daldrup, H.E.; Balzer, T.; Tombach, B.; Berns, T.; Peters, P.E. Clinical results with Resovist: A phase 2 clinical trial. Radiology 1995, 195, 489–496.
- Kostura, L.; Kraitchman, D.L.; Mackay, A.M.; Pittenger, M.F.; Bulte, J.M.W. Feridex labeling of mesenchymal stem cells inhibits chondrogenesis but not adipogenesis or osteogenesis. NMR Biomed. 2004, 17, 513–517.
- Briley-Saebo, K.C.; Mani, V.; Hyafil, F.; Cornily, J.C.; Fayad, Z.A. Fractionated Feridex and positive contrast: In vivo MR imaging of atherosclerosis. Mag. Res. Med. 2008, 59, 721–730.
- Johnson, L.; Pinder, S.E.; Douek, M. Deposition of superparamagnetic iron-oxide nanoparticles in axillary sentinel lymph nodes following subcutaneous injection. Histopathology 2013, 62, 481–486.
- Bullivant, J.P.; Zhao, S.; Willenberg, B.J.; Kozissnik, B.; Batich, C.D.; Dobson, J. Materials characterization of feraheme/ferumoxytol and preliminary evaluation of its potential for magnetic fluid hyperthermia. Int. J. Mol. Sci. 2013, 14, 17501–17510.
- Kellar, K.E.; Fujii, D.K.; Gunther, W.H.H.; Briley-Sæbø, K.; Bjørnerud, A.; Spiller, M.; Koenig, S.H. NC 100150 injection, a preparation of optimized iron oxide nanoparticles for positive-contrast MR angiography. J. Mag. Res. Imag. 2000, 11, 488–494.
- Shan, L.; Leung, K.; Zhang, H.; Cheng, K.T.; Pagel, M. Molecular Imaging and Contrast Agent Database. Bethesda (MD) National Center for Biotechnology Information (US) 2004-2013. Available online: (accessed on 20 April 2021).
- Yu, F.; Huang, Y.; Cole, A.J.; Yang, V.C. Biomaterials The artificial peroxidase activity of magnetic iron oxide nanoparticles and its application to glucose detection. Biomaterials 2009, 30, 4716–4722.
- Gandhi, S.; Banga, I.; Maurya, P.K.; Eremin, S.A. Gold nanoparticles-single chain fragment variable antibody as immunoprobe for rapid detection of morphine by dipstick. RSC Adv. 2018, 8, 1511–1518.
- Singh, S.; Mishra, P.; Banga, I.; Parmar, A.S.; Tripathi, P.P.; Gandhi, S. Chemiluminescence based immunoassay for the detection of heroin and its metabolites. Bioimpacts 2018, 8, 57–62.
- Roberts, A.; Tripathi, P.P.; Gandhi, S. Graphene nanosheets as electric mediator for ultrafast sensing of urokinase plasminogen activator receptor-a biomarker of cancer. Biosens. Bioelectron. 2019, 141, 111398.
- Kasoju, A.; Shahdeo, D.; Khan, A.A.; Shrikrishna, N.S.; Mahiri, S.; Alanazi, M.; Bhat, M.A.; Giri, J.; Gandhi, S. Fabrication of microfluidic device for Aflatoxin M1 detection in milk samples with specific aptamers. Sci. Rep. 2020, 10, 4627.
- Qian, J.; Yang, X.; Jiang, L.; Zhu, C.; Mao, H.; Wang, K. Facile preparation of Fe3O4 nanospheres/reduced graphene oxide nanocomposites with high peroxidase-like activity for sensitive and selective colorimetric detection of acetylcholine. Sens. Act. B Chem. 2014, 201, 160–166.
- Kaçar, C.; Erden, P.E.; Pekyardimci, S.; Kiliç, E. An Fe3O4-nanoparticles-based amperometric biosensor for creatine determination. Art. Cells Nanomed. Biotechnol. 2013, 41, 2–7.
- Gao, L.; Wu, J.; Lyle, S.; Zehr, K.; Cao, L.; Gao, D. Magnetite nanoparticle-linked immunosorbent assay. J. Phy. Chem. C. 2008, 112, 17357–17361.
- Yang, M.; Guan, Y.; Yang, Y.; Xie, L.; Xia, T.; Xiong, W.; Guo, C. Immunological detection of hepatocellular carcinoma biomarker GP73 based on dissolved magnetic nanoparticles. Coll. Sur. A: Phys. Eng. Asp. 2014, 443, 280–285.
- Yang, M.; Guan, Y.; Yang, Y.; Xia, T.; Xiong, W.; Wang, N.; Guo, C. Peroxidase-like activity of amino-functionalized magnetic nanoparticles and their applications in immunoassay. J. Coll. Int. Sci. 2013, 405, 291–295.
- Kim, M.I.; Kim, M.S.; Woo, M.A.; Ye, Y.; Kang, K.S.; Lee, J.; Park, H.G. Highly efficient colorimetric detection of target cancer cells utilizing superior catalytic activity of graphene oxide-magnetic-platinum nanohybrids. Nanoscale 2014, 6, 1529–1536.
- Peter, B.; Lagzi, I.; Teraji, S.; Nakanishi, H.; Cervenak, L.; Zámbó, D.; Deák, A.; Molnár, K.; Truszka, M.; Szekacs, I.; et al. Interaction of Positively Charged Gold Nanoparticles with Cancer Cells Monitored by an in Situ Label-Free Optical Biosensor and Transmission Electron Microscopy. ACS Appl. Mater. Interfaces 2018, 10, 26841–26850.
- Duan, D.; Fan, K.; Zhang, D.; Tan, S.; Liang, M.; Liu, Y.; Zhang, J.; Zhang, P.; Liu, W.; Qiu, X.; et al. Nanozyme-strip for rapid local diagnosis of Ebola. Biosens. Bioelectron. 2015, 74, 134–141.
- Yang, Y.C.; Wang, Y.T.; Tseng, W.L. Amplified Peroxidase-Like Activity in Iron Oxide Nanoparticles Using Adenosine Monophosphate: Application to Urinary Protein Sensing. ACS Appl. Mat. Interf. 2017, 9, 10069–10077.
- Ali, A.; Alsalhi, M.S.; Atif, M.; Ansari, A.A.; Israr, M.Q.; Sadaf, J.R.; Ahmed, E.; Nur, O.; Willander, M. Potentiometric urea biosensor utilizing nanobiocomposite of chitosan-iron oxide magnetic nanoparticles. J. Phy. Conf. Series 2013, 414, 012024.
- Fornara, A.; Johansson, P.; Petersson, K.; Gustafsson, S.; Qin, J.; Olsson, E.; Ilver, D.; Krozer, A.; Muhammed, M.; Johansson, C. Tailored magnetic nanoparticles for direct and sensitive detection of biomolecules in biological samples. Nano Lett. 2008, 8, 3423–3428.
- Wang, Y.; Liu, X.-Y.; Xu, X.; Yang, Y.; Huang, L.-H.; He, Z.-Y.; Xu, Y.-H.; Chen, J.-J.; Feng, Z.-S. Preparation and characterization of reduced graphene oxide/Fe3O4 nanocomposite by a facile in-situ deposition method for glucose biosensor applications. Mat. Res. Bull. 2018, 101, 340–346.
More
Information
Subjects:
Nanoscience & Nanotechnology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.1K
Revisions:
2 times
(View History)
Update Date:
17 May 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





