Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Edward Weaver | + 2487 word(s) | 2487 | 2021-05-07 11:54:35 | | | |
| 2 | Dimitrios A. Lamprou | + 2 word(s) | 2489 | 2021-05-10 20:15:50 | | | | |
| 3 | Catherine Yang | Meta information modification | 2489 | 2021-05-11 03:22:57 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Weaver, E.; Lamprou, D. Peptide Applications Using Microfluidics. Encyclopedia. Available online: https://encyclopedia.pub/entry/9474 (accessed on 13 May 2026).
Weaver E, Lamprou D. Peptide Applications Using Microfluidics. Encyclopedia. Available at: https://encyclopedia.pub/entry/9474. Accessed May 13, 2026.
Weaver, Edward, Dimitrios Lamprou. "Peptide Applications Using Microfluidics" Encyclopedia, https://encyclopedia.pub/entry/9474 (accessed May 13, 2026).
Weaver, E., & Lamprou, D. (2021, May 10). Peptide Applications Using Microfluidics. In Encyclopedia. https://encyclopedia.pub/entry/9474
Weaver, Edward and Dimitrios Lamprou. "Peptide Applications Using Microfluidics." Encyclopedia. Web. 10 May, 2021.
Copy Citation
Peptides possess multifunctional roles within therapeutic formulations, ranging from enhancing target specificity to acting as the active component of the medicine. By applying the platform of microfluidics, the range of applications for peptides has been seen to rise exponentially. Various therapeutic applications of peptides are listed in this review.
microfluidics
peptides
nanoparticles
nanomedicines
liposomes
1. Introduction
Peptide-based molecules fall into two distinct classes. The first class consist of peptides, which are short chains of amino acids (AAs), generally considered to be 2–50 AAs in length [1][2][3], that depending on their chemical composition, can perform a wide variety of functions, both for diagnostic and therapeutic purposes. With 22 well-known proteinogenic AAs [4] and many more nonproteinogenic AAs, the variability of peptide composition is extremely vast. Proteinogenic AAs are essential for normal human function, regulating metabolism, growth and repair, among other processes [5]; however, they can also be exploited for their medicinal qualities. Over 7000 naturally occurring peptides have been identified in nature [6], with much more able to be synthesized artificially, providing a huge library of molecules with potentially life-sustaining effects. Common peptide-based medicines, such as adalimumab (TNF inhibitor) and insulin, are used daily by millions of people for an extensive range of conditions, namely Crohn’s disease, diabetes mellitus, and rheumatoid arthritis, among others. The second class of peptide-based molecules consists of proteins, generally >50 AAs and can have complex secondary structures. This class includes enzymes and antibodies.
Peptide therapeutics are some of the most valuable medicines available [6]; however, their formulations are far from optimized. All peptide medicines must be delivered parenterally [7], often intravenously, due to the degradative process that occurs upon oral administration, which, despite being less convenient for the patient, still allows the delivery of life-saving medicines. Peptide drugs frequently possess high selectivity and potency while sustaining an agreeable safety profile, for example, novel antimicrobial peptides [8][9] and chemotherapeutic agents [10]. Their chemical nature often makes their metabolism predictable [11], which is very useful for administration, distribution, metabolism, and excretion (ADME) calculations; however, frequently, the unmodified peptide will have a very unfavorable ADME profile. For this reason, administrative formulation methods like nanoencapsulation, pegylation and peptide-implant reservoirs must be exploited to allow optimized delivery of the peptides.
2. Therapeutic Applications of Peptides
The application of peptides for medicinal use has exploded in recent years owing to their immense potential for therapeutic effect. Such applications include monoclonal antibodies (MABs) for cancer therapy and infectious disease prevention [12], vaccine synthesis using RNA (including the COVID-19 vaccine) [13][14] and newer uses for regenerative medicines [15], to name a few.
A common method to circumvent peptide-based formulation issues is by formulating the peptides within a nanocarrier vessel, for instance, a nanoparticle (NP). Different NPs have been synthesized in an attempt to effectively manipulate peptide APIs, such as hybrid-magnetic NPs [16], nanoemulsions, liposomes, solid lipid nanoparticles (SLNs) [17] and polymer NPs (e.g., chitosan) [18]. All forms of NP have their own merits and drawbacks, and the method of synthesis can have a large impact on particle characteristics, irrespective of the type of NP being produced. The NP allows a certain level of customization to the medicine, offering the opportunity to provide target-specific delivery via either the NP’s shell material properties, or by the modification of the shell with specific chemical groups to increase target receptor affinity. The NP also provides protection for the peptide against the broad-spectrum of peptidases present within the human body by providing a physical barrier to the degradative enzymes [19].
Formulation of peptides into NPs has been achieved in various ways. However, the processes are far from being optimized and often fall short compared to the standards of industrial medicine synthesis that are implemented for other medicines. Methods, such as unilamellar fusion and thin-film hydration, have been proven to encapsulate peptides [20], but the methods have not yet rectified issues surrounding effective control of particle size and polydispersity index (PDI), as well as possessing unfavorable encapsulation efficiencies [21]. Thus, it has been theorized that a highly controllable system like Microfluidics (MFs) could be the solution to providing a high standard of encapsulation of peptide molecules within various NPs. A brief summary of method attributes is shown in Table 1.
Table 1. Advantages and disadvantages of three methods used for peptide nanomedicine synthesis.
| Microfluidics | Unilamellar Vesicle Fusion | Thin-Film Hydration |
|---|---|---|
| Advantages | ||
| Allows synthesis at room temperature | Simple method | Simple method |
| Good control over PDI and particle size | Process can be controlled via electrostatic manipulation [22] | Acceptable encapsulation efficiencies for smaller peptides |
| Easy method to scale-up | ||
| High encapsulation efficiencies [23] | ||
| Continuous process | ||
| Disadvantages | ||
| Material interactions with MF chip | Less control over PDI and particle size | Less control over PDI and particle size |
| Requires initial high-cost system | Affected greatly by temperature | Produces heterogeneous particle population that requires extrusion/sonication [24] |
| Batch process | ||
The introduction of peptides into the MF system to fabricate nanomedicines (NMs) is a novel approach, which is already showing huge potential within the nano-formulation field. In 2020, the COVID-19 virus expedited research into RNA-based vaccines as many companies, including BioNTech/Pfizer and Moderna, relied on the genetically sequenced peptides for the efficacy of their vaccines [25]. The utilization of MFs to synthesize RNA-based vaccines has been shown to be an effective means of RNA encapsulation, achieving favorable synthetic attributes, in particular encapsulation efficiencies of >99% [26].
This area of research represents a huge opportunity for advancing vaccine technology, as MFs can be adapted to allow NP modification, which in turn increases target specificity. Most research performed revolving around RNA manipulation within the MF system is based upon RNA sequencing, which is an extremely effective tool [27][28]. Discovering the heterogeneity of cellular information is essential to many biomedical applications, for instance, vaccine development, suggesting that MFs could provide a “door-to-door” approach for vaccine production, as MFs can provide both the target identification platform via sequence isolation, as well as a synthesis system to formulate the final vaccine. The system’s high throughput capacity allows multiple tests to be conducted within a limited timeframe, which would have been perfect in response to developing a COVID-19 vaccine.
One of the key elements inhibiting using peptides as nanocarriers is the lack of reliable formulation reproducibility and general disadvantageous nanocarrier properties, such as large sizes and high PDI values. By utilizing the controlled, small volume environment provided by the MF platform, researchers have begun to observe a change in this trend. The self-assembly of peptide chains from amino acids highlighted by Chan et al. and proven by Ni et al. indicate a huge potential for implementing peptides into the nanocarrier field. Frequently, the function of peptides for nanoformulations is to act as membrane epitopes attached to a functional polymer, such as chitosan or polylactic co-glycolic acid (PLGA) [29][30]. However, due to the emergence of MFs, it is starting to become feasible to produce NP membranes consisting fully of peptides. These peptide nanocarriers can possess desirable properties, including increased cell permeation [31], region-specific delivery [32] and a high degree of potential for modification based upon their unique chemical makeup. As a result, of the controlled synthesis, using extrusion and sonication is rarely needed for formulations produced by MFs, which ensures that the time taken for production is minimal.
2.1. Peptide Encapsulation
Formulation of peptides within liposomes is possible using MFs due to the ability of certain lipids, e.g., 1,2-dimyristoyl-sn-glycero-3-phosphocholine (DMPC), to self-assemble into lipid nanostructures upon contact with aqueous media. Due to the amphiphilic nature of phospholipids, when in the presence of an aqueous medium, self-assembly occurs caused by noncovalent interactions forcing lipophilic areas of the molecules to a geometric state of low entropy [33], as seen in Figure 1. This principle has been harnessed during a novel attempt to encapsulate large biologic drugs, for example, ovalbumin and bovine serum albumin (BSA) [23]. The encapsulation of biologic drugs performed in this study produced liposomes of drastically smaller sizes and PDIs than obtained via previous methods, for instance, sonication or extrusion [23], as well as importantly increasing encapsulation efficacy.
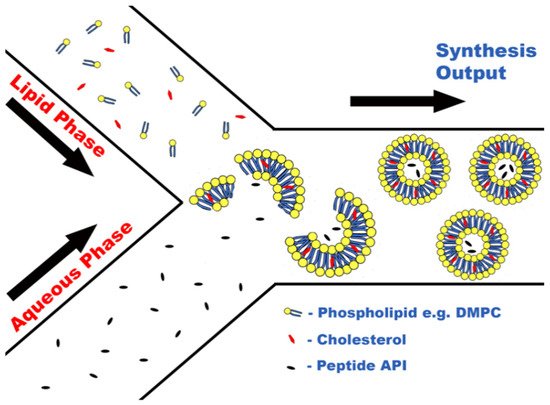
Figure 1. Concept of peptide encapsulation within liposomes via liposome self-assembly.
Increasing the encapsulation efficiency is crucial for biologic molecules due to their cost as even commonly used biologics, such as insulin, can cost over GBP 200 for 50 mg, which has been a limiting factor for scaling up production of previous methods to industrial levels. With further research encapsulating other biologics, it is possible that a financially viable method could be devised to produce these biologic formulations, which could even lead to groundbreaking medicines, including orally active insulin preparations. Evidently, as research in this area is limited, it still requires much work to determine the exact MF parameters that will be optimal for biologic encapsulation, or even that biologics are susceptible to encapsulation via an MF route. However, the evidence suggests so far that MFs offer a very promising synthetic method to produce such formulations.
2.2. Hydrogels and Peptide Antibiotics
The production of peptide-based biomaterials via MFs is attractive as they possess a low level of immunogenicity, and prolonged-release formulations, such as biologic hydrogels, will degrade slowly over time, via natural processes, into non-toxic materials [34][35]. Recent work performed utilizing the prolonged curative properties of peptide hydrogels allowed a double-pronged approach to wound healing via an MF-assisted process [36]. Peptide hydrogels possess a structure similar to the extracellular matrix, making them an ideal biocompatible medium for the controlled release of APIs. MFs were employed to synthesize a compact fibrous network of alginate to provide an optimal setting for the self-assembly of peptides onto the structure. The use of peptides on the structure also allowed the addition of a secondary API, the antibiotic lincomycin, within the hydrogel; hence providing two APIs to provide a therapeutic effect. The novel approach of using peptides within the structure ensured that the wound healing process could be optimized by providing both the anti-inflammatory benefits of prolonged-release peptide therapy, as well as introducing antibiotic activity.
It is possible that the process designed by Jain et al. [37] would be adaptable to a microfluidic approach, which exploited ultrashort peptides (USPs) to control gold NP synthesis within a complex hydrogel structure. USPs are short chains of amino acids (approximately 3–7 amino acids) that can be used to provide unique interactions with external environments. Owing to the predictable chemical behavior of peptides, they have been shown to be exploited to assist with NP shape control. NP shape is an important factor to consider as it affects overall PDI values, and it can also affect the thermal stability of a formulation or encapsulation efficacy. For example, nickel-based NPs in a spherical shape have better heat-transference properties than a disc shape [38].
Owing to increased pathogen drug resistance, the production of novel antimicrobials is limited, which is why a new area of antimicrobials in the form of peptide therapeutics should be explored. Despite the categorization of over 3000 AMPs, currently, only 7 antimicrobial peptides (AMPs) are approved for clinical use by the FDA [39], owing mainly to a lack of clinical efficacy. Bioassays performed using an MF design allow detecting antimicrobial activity. However, when compared to the traditional format of measuring via 96-well plate analysis, the results obtained were dissimilar, owing to differing oxygenation levels during incubation subjected to each system. The 96-well plate was exposed to a greater level of oxygen than the MFs assays, causing incomparability of the systems and hence the results of this study were inconclusive [39]. However, there is strong potential for future research in this area to explore different approaches for obtaining AMP activity using MFs. Conceptually, it is clear that MFs can be employed for this purpose, and there is an acute need for a new bioassay method, so this is an area that should be developed further.
2.3. Gene Therapy
Gene therapy is an area of medicine that could potentially have a massive impact on our perceived view of gold-standard healthcare. However, it is constantly shrouded by controversy and negative implications, as well as proving to be a difficult procedure to perfect. The advancement of MF designs could impact this area as current investigations into the MF synthesis of double-stranded DNA (dsDNA) carriers are at the forefront of enhancing gene delivery to specific targets [40]. Carrier materials like graphene oxide (GO) are versatile materials that, when unmodified, possess the ability to integrate single-stranded DNA or RNA onto their structure via distributed π−π
interactions. Modification via MFs is required to alter the charge of the GO structure to allow dsDNA bonding, as seen in Figure 2. Coating of the GO with a cationic lipid, such as 1,2-dioleoyl-3-trimethylammonium-propane (DOTAP), via MFs, increases the overall surface electrostatic charge, complementary to that of dsDNA. This lipid coating increased cellular uptake of DNA from 48.8% ± 6 using unmodified GO to an improved 93.9% ± 0.2. As one of the main inhibitors of gene therapy is achieving suitable gene concentrations for therapeutic effect, this progress is revolutionary. The carrier is required to prevent the susceptible DNA from degradation upon administration, as well as increasing gene transfection [41].
2.4. Oral Administration of Peptide-Based Medicines
Finally, for peptide therapeutics, a zealous approach to the synthesis of orally administered peptide medicines was performed using a multilamellar delivery system for the co-administration of an anti-diabetic peptide, glucagon-like peptide-1 (GLP-1), alongside another molecule, dipeptidyl peptidase 4 inhibitors (DPP4i) [42]. Currently, the only administration route for GLP-1 is parenteral, which leads to lower patient medicine adherence [43]. The natural degradation time for GLP-1 post-administration is approximately 2 min [44] due to rapid degradation by the enzyme dipeptidyl peptidase 4. Upon formulation into the dual-action functionalized NPs, hypoglycemic effects of the GLP-1 could still be observed 6 h after administration.
The synthesis of this formulation is complex, requiring multiple stages and multiple vessel materials: An initial NP was fabricated from PLGA and mesoporous silicon (PSi). This NP had additional surface functional groups of chitosan (CS) (to aid mucoadhesion) and cell-penetrating peptides (CPP), which assist the delivery of NP contents through the lipid bilayer of a cell. This NP contained the API GLP-1 and can be seen in Figure 2. MFs were then employed to further encapsulate this NP within an enteric coating material, hydroxypropylmethylcellulose acetyl succinate (HPMC-AS). The DPPi4 was only added upon final encapsulation alongside the HPMC-AS, which protected the two APIs against the inhospitable conditions exhibited upon oral administration. As research into orally administered peptide-based medicines is developed, a wide range of new patient-friendly, peptide-based medicines could begin to emerge. According to a 2019 study, parenterally administered medicines consistently had greater non-adherence rates than oral formulations [43], potentially leading to a lower quality of life.
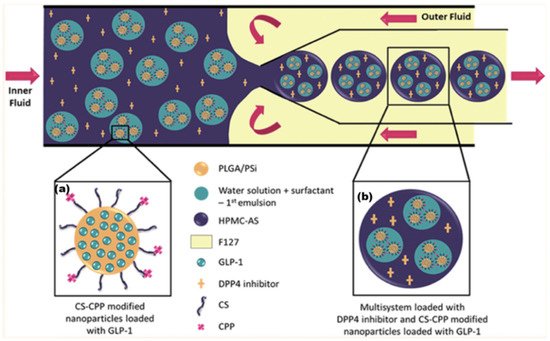
Figure 2. (a) Initial NP produced via the double emulsion method. (b) Final multilayered formulation produced by mixing emulsified particle (a) with HPMC-AS/DPP4i solution. Adapted with permission from Araújo et al. [42] Copyright 2015 American Chemical Society.
3. Conclusions
Microfluidics can used for drug discovery and development, and the market includes drug discovery, drug delivery, and in-vitro diagnostics, among many others. Microfluidics have also all the potentials to be used in the fight against a pandemic for the manufacturing of vaccines or rapid diagnosis. The microfluidic facility at the School of Pharmacy, hosts five microfluidic devices, including temperature controllers and high-speed microscope for the evaluation of the particles and flows in the microfluidic chips, including the manufacturing of chips by 3D printing technologies.
References
- Polypeptides and Proteins. 3 January 2021. Available online: (accessed on 29 April 2021).
- Osorio, D.; Rondón-Villarreal, P.; Torres, R. Peptides: A package for data mining of antimicrobial peptides. Small 2015, 7, 4–14.
- Marqus, S.; Pirogova, E.; Piva, T.J. Evaluation of the use of therapeutic peptides for cancer treatment. J. Biomed. Sci. 2017, 24, 21.
- Hedges, J.B.; Ryan, K.S. Biosynthetic Pathways to Nonproteinogenic α-Amino Acids. Chem. Rev. 2020, 120, 3161–3209.
- Marchese, L.; Nascimento, J.; Damasceno, F.; Bringaud, F.; Michels, P.; Silber, A. The Uptake and Metabolism of Amino Acids, and Their Unique Role in the Biology of Pathogenic Trypanosomatids. Pathogens 2018, 7, 36.
- Fosgerau, K.; Hoffmann, T. Peptide therapeutics: Current status and future directions. Drug Discov. Today 2015, 20, 122–128.
- Vllasaliu, D.; Thanou, M.; Stolnik, S.; Fowler, R. Recent advances in oral delivery of biologics: Nanomedicine and physical modes of delivery. Expert Opin. Drug Deliv. 2018, 15, 759–770.
- Meade, E.; Slattery, M.A.; Garvey, M. Bacteriocins, Potent Antimicrobial Peptides and the Fight against Multi Drug Resistant Species: Resistance Is Futile? Antibiotics 2020, 9, 32.
- Roudi, R.; Syn, N.L.; Roudbary, M. Antimicrobial Peptides As Biologic and Immunotherapeutic Agents against Cancer: A Comprehensive Overview. Front. Immunol. 2017, 8, 1320.
- Cutone, A.; Rosa, L.; Ianiro, G.; Lepanto, M.S.; Bonaccorsi Di Patti, M.C.; Valenti, P.; Musci, G. Lactoferrin’s Anti-Cancer Properties: Safety, Selectivity, and Wide Range of Action. Biomolecules 2020, 10, 456.
- Hazam, P.K.; Jerath, G.; Kumar, A.; Chaudhary, N.; Ramakrishnan, V. Effect of tacticity-derived topological constraints in bactericidal peptides. Biochim. Biophys. Acta Biomembr. 2017, 1859, 1388–1395.
- Pento, J.T. Monoclonal antibodies for the treatment of cancer. Anticancer Res. 2017, 37, 5935–5939.
- Hildebrand, D.; Metz-Zumaran, C.; Jaschkowitz, G.; Heeg, K. Silencing SOCS1 via Liposome-Packed siRNA Sustains TLR4-Ligand Adjuvant. Front. Immunol. 2019, 10, 1279.
- Le, T.T.; Andreadakis, Z.; Kumar, A.; Román, R.G.; Tollefsen, S.; Saville, M.; Mayhew, S. The COVID-19 vaccine development landscape. Nat. Rev. Drug Discov. 2020, 19, 305–306.
- Koutsopoulos, S. Self-assembling peptide nanofiber hydrogels in tissue engineering and regenerative medicine: Progress, design guidelines, and applications. J. Biomed. Mater. Res. Part A 2016, 104, 1002–1016.
- Zhang, W.; Taheri-Ledari, R.; Hajizadeh, Z.; Zolfaghari, E.; Ahghari, M.R.; Maleki, A.; Hamblin, M.R.; Tian, Y. Enhanced activity of vancomycin by encapsulation in hybrid magnetic nanoparticles conjugated to a cell-penetrating peptide. Nanoscale 2020, 12, 3855–3870.
- McClements, D.J. Encapsulation, protection, and delivery of bioactive proteins and peptides using nanoparticle and microparticle systems: A review. Adv. Colloid Interface Sci. 2018, 253, 1–22.
- Wong, C.Y.; Al-Salami, H.; Dass, C.R. The role of chitosan on oral delivery of peptide-loaded nanoparticle formulation. J. Drug Target. 2018, 26, 551–562.
- Ross, C.; Taylor, M.; Fullwood, N.; Allsop, D. Liposome delivery systems for the treatment of Alzheimer’s disease. Int. J. Nanomed. 2018, 13, 8507–8522.
- Ibaraki, H.; Kanazawa, T.; Kurano, T.; Oogi, C.; Takashima, Y.; Seta, Y. Anti-RelA siRNA-Encapsulated Flexible Liposome with Tight Junction-Opening Peptide as a Non-invasive Topical Therapeutic for Atopic Dermatitis. Biol. Pharm. Bull. 2019, 42, 1216–1225.
- Lindqvist, A.; Rip, J.; van Kregten, J.; Gaillard, P.J.; Hammarlund-Udenaes, M. In vivo functional evaluation of increased brain delivery of the opioid peptide DAMGO by Glutathione-PEGylated liposomes. Pharm. Res. 2016, 33, 177–185.
- Oshima, A.; Sumitomo, K. Vesicle fusion with bilayer lipid membrane controlled by electrostatic interaction. Biochem. Biophys. Rep. 2017, 11, 58–63.
- Forbes, N.; Hussain, M.T.; Briuglia, M.L.; Edwards, D.P.; Horst, J.H.T.; Szita, N.; Perrie, Y. Rapid and scale-independent microfluidic manufacture of liposomes entrapping protein incorporating in-line purification and at-line size monitoring. Int. J. Pharm. 2019, 556, 68–81.
- Zhang, H. Thin-Film Hydration Followed by Extrusion Method for Liposome Preparation. Methods Mol. Biol. 2017, 1522, 17–22.
- Corey, L.; Mascola, J.R.; Fauci, A.S.; Collins, F.S. A strategic approach to COVID-19 vaccine R&D. Science 2020, 368, 948–950.
- Leung, A.K.K.; Tam, Y.Y.C.; Chen, S.; Hafez, I.M.; Cullis, P.R. Microfluidic Mixing: A General Method for Encapsulating Macromolecules in Lipid Nanoparticle Systems. J. Phys. Chem. B 2015, 119, 8698–8706.
- Moon, H.-S.; Je, K.; Min, J.-W.; Park, D.; Han, K.-Y.; Shin, S.-H.; Park, W.-Y.; Yoo, C.E.; Kim, S.-H. Inertial-ordering-assisted droplet microfluidics for high-throughput single-cell RNA-sequencing. Lab Chip 2018, 18, 775–784.
- Taylor, N.; Elbaum-Garfinkle, S.; Vaidya, N.; Zhang, H.; Stone, H.A.; Brangwynne, C.P. Biophysical characterization of organelle-based RNA/protein liquid phases using microfluidics. Soft Matter 2016, 12, 9142–9150.
- Lee, M.-H.; Lin, C.-C.; Thomas, J.L.; Chan, C.-K.; Lin, H.-Y. Epitope recognition of magnetic peptide-imprinted chitosan composite nanoparticles for the extraction of CRISPR/dCas9a proteins from transfected cells. Nanotechnology 2021, 32, 18LT02.
- Roozbehani, M.; Falak, R.; Mohammadi, M.; Hemphill, A.; Razmjou, E.; Meamar, A.R.; Masoori, L.; Khoshmirsafa, M.; Moradi, M.; Gharavi, M.J. Characterization of a multi-epitope peptide with selective MHC-binding capabilities encapsulated in PLGA nanoparticles as a novel vaccine candidate against Toxoplasma gondii infection. Vaccine 2018, 36, 6124–6132.
- Gessner, I.; Neundorf, I. Nanoparticles Modified with Cell-Penetrating Peptides: Conjugation Mechanisms, Physicochemical Properties, and Application in Cancer Diagnosis and Therapy. Int. J. Mol. Sci. 2020, 21, 2536.
- Field, L.D.; Delehanty, J.B.; Chen, Y.; Medintz, I.L. Peptides for Specifically Targeting Nanoparticles to Cellular Organelles: Quo Vadis? Acc. Chem. Res. 2015, 48, 1380–1390.
- Liu, K.; Li, H.; Williams, G.R.; Wu, J.; Zhu, L.-M. pH-responsive liposomes self-assembled from electrosprayed microparticles, and their drug release properties. Colloids Surfaces A Physicochem. Eng. Asp. 2018, 537, 20–27.
- Schiller, J.L.; Lai, S.K. Tuning Barrier Properties of Biological Hydrogels. ACS Appl. Bio Mater. 2020, 3, 2875–2890.
- Chan, K.H.; Tay, J.J.J. Advancement of Peptide Nanobiotechnology via Emerging Microfluidic Technology. Micromachines 2019, 10, 627.
- Cui, T.; Li, X.; He, S.; Xu, D.; Yin, L.; Huang, X.; Deng, S.; Yue, W.; Zhong, W. Instant Self-Assembly Peptide Hydrogel Encapsulation with Fibrous Alginate by Microfluidics for Infected Wound Healing. ACS Biomater. Sci. Eng. 2020, 6, 5001–5011.
- Jain, R.; Khandelwal, G.; Roy, S. Unraveling the Design Rules in Ultrashort Amyloid-Based Peptide Assemblies toward Shape-Controlled Synthesis of Gold Nanoparticles. Langmuir 2019, 35, 5878–5889.
- Gireesha, B.; Sowmya, G.; Gorla, R.S.R. Nanoparticle shape effect on the thermal behaviour of moving longitudinal porous fin. Proc. Inst. Mech. Eng. Part N J. Nanomater. Nanoeng. Nanosyst. 2020, 234, 115–121.
- Parsley, N.C.; Smythers, A.L.; Hicks, L.M. Implementation of Microfluidics for Antimicrobial Susceptibility Assays: Issues and Optimization Requirements. Front. Cell. Infect. Microbiol. 2020, 10, 547177.
- Di Santo, R.; Digiacomo, L.; Palchetti, S.; Palmieri, V.; Perini, G.; Pozzi, D.; Papi, M.; Caracciolo, G. Microfluidic manufacturing of surface-functionalized graphene oxide nanoflakes for gene delivery. Nanoscale 2019, 11, 2733–2741.
- Imani, R.; Emami, S.H.; Faghihi, S. Synthesis and characterization of an octaarginine functionalized graphene oxide nano-carrier for gene delivery applications. Phys. Chem. Chem. Phys. 2015, 17, 6328–6339.
- Araújo, F.; Shrestha, N.; Shahbazi, M.A.; Liu, D.; Herranz-Blanco, B.; Mäkilä, E.M.; Salonen, J.J.; Hirvonen, J.T.; Granja, P.L.; Sarmento, B.; et al. Microfluidic Assembly of a Multifunctional Tailorable Composite System Designed for Site Specific Combined Oral Delivery of Peptide Drugs. ACS Nano 2015, 9, 8291–8302.
- Unni, E.; Sternbach, N.; Goren, A. Using the Medication Adherence Reasons Scale (MAR-Scale) to identify the reasons for non-adherence across multiple disease conditions. Patient Prefer. Adherence 2019, 13, 993–1004.
- Cheang, J.Y.; Moyle, P.M. Glucagon-Like Peptide-1 (GLP-1)-Based Therapeutics: Current Status and Future Opportunities beyond Type 2 Diabetes. ChemMedChem 2018, 13, 662–671.
More
Information
Subjects:
Nanoscience & Nanotechnology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.9K
Revisions:
3 times
(View History)
Update Date:
11 May 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





