
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Alexander Sirotkin | + 2475 word(s) | 2475 | 2021-04-28 11:49:17 | | | |
| 2 | Camila Xu | Meta information modification | 2475 | 2021-04-29 04:45:04 | | |
Video Upload Options
Isoflavones (including daidzein, the glycoside forms of daidzein, and glycitein, the methoxylated form of daidzein) are bioactive compounds that are present in significant quantities in legumes, soybeans, green beans, and mung beans.
1. Provenance and Properties of Daidzein and Its Metabolite Equol
Isoflavones (including daidzein, the glycoside forms of daidzein, and glycitein, the methoxylated form of daidzein) are bioactive compounds that are present in significant quantities in legumes, soybeans, green beans, and mung beans. In grains (raw materials), they are mostly present as simple and complex O-glycosides, β-glucuronides and sulfate esters, and highly polar, water-soluble compounds. They are hardly absorbed by the intestinal epithelium and have insignificant biological activities. Beans are included in various food products owing to their digestibility, taste, bioavailability of nutrients, and higher amounts of bioactives. Processing steps include steaming, cooking, roasting, and microbial fermentation, which destroys protease inhibitors and cleaves the glycoside bond to yield absorbable and bioactive aglycone in the processed products, such as miso, natto, soy milk, tofu, and increases the product’s shelf life. Results are reliable (including the conversion of glucoside daidzein to aglicon daidzein, which hydrolyzes conjugated isoflavones with gut bacteria) [1][2][3][4]. Clinical studies show the differences between the biological activities of aglycone and conjugated forms of daidzein [1]. Processed soy food products were an integral part of diets in many Asia-Pacific countries for centuries, but in the last two decades, soy products were successfully integrated into Western diets because of their health benefits [2].
Daidzein (7-hydroxy-3-(4-hydroxyphenyl)-4H-chromen-4-one) (PubChem ID 5281708, molecular formula C15H10O4; chemical structure indicated in Figure 1a, https://pubchem.ncbi.nlm.nih.gov/ (accessed on 16 April 2021)) is an isoflavonoid derived from L-phenylalanine. Daidzein is an aglycone form of daidzein. Its solubility in water is relatively low; therefore, for medical purposes, it is dissolved in alcohol or an alcohol/water mixture.
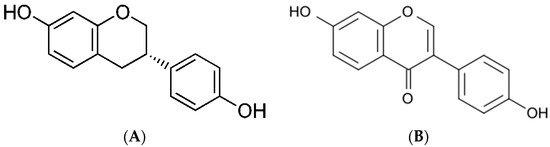
Figure 1. Chemical structures of (A) daidzein and (B) equol (https://pubchem.ncbi.nlm.nih.gov/ (accessed on 16 April 2021)).
The primary biological and health effect of daidzein is because of its metabolite, equol ((3S)-3-(4-hydroxyphenyl)-7-chromanol) (PubChem ID, 6950272 molecular formula C15H14O3; chemical structure indicated in Figure 1b, https://pubchem.ncbi.nlm.nih.gov/ (accessed on 16 April 2021)). Equol is an isoflavandiol nonsteroidal estrogen and a secondary metabolite of daidzein that is the result of daidzein hydrolysis by gut bacteria. It is insoluble in water and acid-labile [5][6][7]. The conversion of daidzein into equol takes place in the intestine via the action of reductase enzymes belonging to incompletely characterized members of the gut microbiota. While all animal species analyzed so far produce equol, only between one-third and one-half of humans (depending on the community) can do so; ostensibly, those that harbor equol-producing microbes. Only 20%–30% of Westerners produce S-equol compared to 50%–70% of Asians; these subjects might be the only ones who can fully benefit from soy or isoflavone consumption [3][5][6]. Besides microflora, raw plant polyphenols can be metabolized by hepatocytes, intestines, and some other cells, including breast tumor cells. After daidzein enters the circulation via intestinal absorption, it may be transported to the liver, where it is conjugated by liver enzymes to form sulfated, glycosylated, and glucuronidated isoflavones, which are more water-soluble [1][2][4]. Additionally, inflammatory cells produce chemical oxidants that can react with polyphenols and inactivate them [4].
Although isoflavones are nonsteroidal, they have similarities in their chemical structure to that of mammalian estrogens, show estrogenic activities in biological assays, and induce estrogen-like effects in mammalian systems (see below).
Despite the low oral bioavailability, most polyphenols have significant biological effects that highlight the low bioavailability/high bioactivity paradox. This phenomenon can be explained by the high affinity and sensitivity of estrogen receptors, even to a low amount of phytoestrogen [8].
2. Mechanisms of Action of Daidzein and Its Metabolite Equol
Daidzein, like other flavanoid isoflavones, is a phenol plant compound that resembles vertebrate steroid hormones due to its molecular structure. Therefore, in vertebrates, it can bind steroid hormone receptors and exert estrogenic or antiestrogenic effects, thereby affecting physiological processes and illnesses dependent on steroid hormones [1][3][9][10][11]. As an antioxidant, daidzein can prevent oxidative stress and resulting disorders [4][9][8][7]. The manifestation of these illnesses could also be mitigated by daidzein due to its ability to promote antioxidant enzyme cytochromes such as P450 [7], which inhibits tyrosine kinase, a cell-proliferation promoter [9], and to activate the Akt signaling pathway, which promotes DNA fragmentation and cell apoptosis [12].
Equol, a daidzein metabolite with higher biological activity, can affect these processes via the direct binding of reactive oxygen species and the activation of antioxidative enzymes, binding to estrogen receptors, mitogen-activated protein kinase, protein kinase B (Akt), and epidermal growth factor receptor kinase and cyclin B/CDK complex (cell-proliferation promoters), transcription factor NFkB (promoter of inflammatory processes), nitric oxide-dependent intracellular signaling pathway, transcription factors FOXO3a and p53 (promoters of apoptosis), epigenetic mechanisms, including DNA methylation, histone modification, and microRNA regulation, and other intracellular signaling mechanisms [5].
3. Physiological and Therapeutic Actions of Daidzein and Its Metabolite Equol
Epidemiological, animal, and in vitro studies demonstrated the ability of daidzein to reduce the incidence of estrogen-dependent and aging-associated disorders, such as menopause, osteoporosis, cardiovascular diseases, cancer [13][1][2][3][4][9][8][14][7], cognitive disorders, and blood pressure [14]. Higher daidzein concentrations were associated with a lower risk of breast cancer and diabetes [13][9]. Daidzein impairs glucose and lipid metabolism and vascular inflammation associated with Type 2 diabetes [15]. It can promote the expression and activity of detoxifying enzymes [9], and prevent the toxic effects of polycyclic aromatic hydrocarbons [7]. Lastly, daidzein can assist with the treatment of viral infections, including COVID-19 [16].
Results of recent clinical trials and meta-analyses on the effects of equol demonstrated the preventive and curative action of equol on menopause, the cardiovascular system, bone health (prevention of osteoporosis), cancer, central-nervous-system functions, and mental disorders [5][6][12]. Equol may also modulate obesity and Type 2 diabetes by controlling the glycemic index, ameliorate chronic kidney disease, and prevent skin aging [5].
However, the effects of daidzein and equol depend on their income from food. For example, in East Asian countries, where the consumption of daidzein-containing products is 10 times higher than that in the West, the incidence of cardiovascular disease, osteoporosis, mental disorders, certain types of cancer, and menopausal symptoms is several times lower than that of the West [5]. The additional consumption of soy, daidzein, and equol did not affect the expression of menopausal symptoms in Chinese women [17].
Nevertheless, most clinical studies involving isoflavones suffered from small sample sizes, short trial durations, a lack of appropriate controls, the use of isoflavones from various sources, supplements with different aglycone contents, and other methodological flaws. Therefore, both specialists and regulatory agencies concluded that there is still no scientifically sound evidence of isoflavones reducing the risks and symptoms of any disease [5].
Lastly, isoflavones, including daidzein and its metabolites, may also be considered endocrine disruptors with possible negative health effects on certain parts of the population or on the environment [3][5]. Both in vitro and animal studies reported that isoflavones can interfere with different checkpoints of the hypothalamic/pituitary/thyroid system. Further, the estrogenic activity of isoflavones could be a hazard by promoting certain types of tumors; however, evidence for harm is also inconclusive [5].
4. Effects of Daidzein and Its Metabolite Equol on Female Reproductive Processes
Several laboratories studied the character of daidzein’s influence on animal reproductive systems and fecundity, but results were variable. Lamartiniere et al. [18] failed to find any effect of daidzein on rat ovarian histomorphology, weight, and fertility, the number of male and female offspring, or anogenital distances. Kaludjerovic et al. [19] did not find an influence with dietary daidzein on mice ovarian weight, the number of ovarian follicles, the number of multiple oocyte follicles, or the percentage of ovarian cysts; however, they observed the adverse effect of daidzein on ovarian structure and its ability to reduce the number of ovarian corpus lutea. In rats, daidzein suppressed follicular growth (reduced ovarian weight), but not ovarian folliculogenesis and fecundity (number of corpora lutea) or sexual behavior (lordosis quotient) [20][21]. Talsness et al. [22] reported the inhibitory influence of daidzein on rat ovarian folliculogenesis, which increased follicular atresia; reduced secondary and tertiary follicle numbers, and probably ovarian surface epithelium proliferation; induced cyst formation; and prolonged estrous. The ability to suppress growth and to induce the atresia of murine cultured ovarian follicles was also reported for quercetin metabolite equol [23].
Other studies showed the stimulatory action of daidzein on chicken ovarian germ cells [24], chicken ovarian folliculogenesis, and the ovulation/egg-laying rate [25]. The daidzein-induced promotion of rat ovarian folliculogenesis and a reduction in ovarian follicular atresia were observed by Medigović et al. [26]. Dorward et al. [27] reported that feeding mice a mixture of daidzein and genistein promoted ovarian and uterine growth, and increased the incidence of ovarian tumorigenesis.
The influence of daidzein on nonovarian reproductive organs remains unknown. Dietary daidzein did not affect uterine morphology and weight in rats [18] and mice [19]. No influence of daidzein on the proliferation of murine endometrium and endometrioma cells was found [28]. Kaludjerovic et al. [19] reported that dietary daidzein induced hyperplasia in the murine oviduct and abnormal histomorphological changes in the subjects’ uteri.
Therefore, available information concerning the expression and characterization of daidzein action is inconsistent, although most relevant papers suggest inhibitory action with this polyphenol on rodent and chicken reproductive systems. It remains unknown whether daidzein can influence the human reproductive system.
Daidzein did not affect viability in cultured bovine ovarian granulosa and luteal cells [29]. In some of our experiments [30], daidzein promoted cell viability and proliferation but not apoptosis by cultured porcine ovarian granulosa cells. In other experiments [31], daidzein reduced the viability of these cells, but this effect was not associated with changes in cell proliferation or apoptosis. Daidzein was able to induce the death of ovarian cancer cells both in vivo [32] and in vitro [32][33].
Therefore, daidzein can induce the death of cancer cells, but available data concerning its action on healthy ovarian cells’ viability, proliferation, and apoptosis remain inconclusive.
5. Hormonal Mechanisms of Daidzein and Equol Effects on Female Reproductive Processes
Female reproductive functions, like other functions, are controlled by a hierarchical system of exogenous and endogenous regulators—regulatory molecules produced in the hypothalamus and other areas of the central nervous system (CNS), pituitary hormones (mainly gonadotropins), hormones produced by the ovaries and other peripheral organs, receptors to hormones, and intracellular mediators of their action—and enzymes including protein kinases, transcription factors regulating cell proliferation, apoptosis, viability, and differentiation [34]. The influence of daidzein on some (but not all) of these regulatory molecules is documented.
In the available literature, we failed to find any evidence for daidzein action on the CNS. Furthermore, histomorphological studies did not reveal daidzein influence on morphophysiological indexes of activity in pituitary gonadotropes and lactotropes [26], indicating that daidzein does not affect gonadotropin- and prolactin-producing pituitary cells.
Feeding with daidzein increased the number of follicle stimulating hormone (FSH) and luteinizing hormone (LH) receptors in chicken ovarian follicles [25]. Furthermore, daidzein promoted the stimulatory action of LH on mouse ovarian-cancer development [27]. These observations indicate that daidzein can promote the reception and effect of gonadotropins on ovarian functions. The ability of daidzein to promote the generation of gonadotropin receptors, which was associated with an increase in ovarian folliculogenesis and the egg-laying rate [25] mentioned above, suggests that daidzein promotes ovarian-cell function through the upregulation of gonadotropin receptors. The exact mechanism remains unknown; however, estrogen via estrogen receptors can boost the production of gonadotropin receptors in the ovary [34]. Daidzein can promote the generation of estrogen receptors (see below) [35][36], and mimic gonadotropin action on ovarian cell proliferation [24]. This indicates that daidzein can promote the production of gonadotropin reception through the activation of estrogen receptors. Nevertheless, this hypothesis requires experimental validation.
The action of daidzein on peripheral hormones, including hormones of ovarian origin regulating reproductive functions, is well-documented, although substantial differences are evident in the character of daidzein action, even in similar experiments.
In the in vivo experiments of Medigović et al. [26], injections of diosgenin increased progesterone and estradiol, and decreased testosterone levels in rat serum. On the other hand, in the in vivo experiments of Lamartiniere et al. [18], feeding rats with daidzein reduced the concentration of circulating progesterone but not estrogen.
Some in vitro experiments demonstrated a direct inhibitory action of daidzein and its metabolite equol on steroid hormones‘ release by ovarian cells. Equol inhibited the expression of steroidogenic enzymes, and the synthesis and release of progesterone, testosterone, androstenedione, and estradiol by cultured murine ovarian follicles [23]. Similarly, there was an inhibitory influence with daidzein on progesterone, but not the estradiol release of cultured porcine ovarian granulosa cells, as observed in the in vitro experiments of Nynca et al. [34][35]. Similar experiments by Sirotkin et al. [31] failed to detect daidzein action on progesterone and estradiol release by cultured porcine granulosa cells, but in these experiments, daidzein promoted testosterone output. Other experiments on porcine granulosa cells showed the upregulation of progesterone, but not testosterone and estradiol production [30]. Mlynarczuk et al. [29] did not observe any influence of daidzein on progesterone and estradiol release by cultured bovine granulosa and luteal cells. In their experiments, daidzein was a potent stimulator of enzymes responsible for oxytocin synthesis and release.
Epidemiological studies [37] showed an association between decreased serum daidzein levels and decreased female serum adiponectin levels, and increased serum insulin levels, which could be associated with an elevated risk of ovarian cancer.
The similarity of daidzein and estrogen effects on rat ovarian folliculogenesis [22] and steroidogenesis [26] suggests there is a similar mechanism of action with these molecules on adult ovarian functions via estrogen receptors. This hypothesis was confirmed by the ability of both daidzein and estradiol to bind to estrogen receptors in ovarian and nonovarian tissue [1][2], and to upregulate the expression of estrogen receptors alpha and beta in porcine ovarian cells [35][36]. Moreover, the blockage of estrogen receptors prevented daidzein action on the proliferation of chicken ovarian germ cells [24]. On the contrary, there are reports [27][38] that daidzein can antagonize estrogen action on ovarian-cancer cells. Reports indicate that daidzein affects ovarian-cancer development via both the up- and the downregulation of estrogen receptors. The current literature does not contain direct evidence concerning daidzein action on and via receptors to other peripheral hormones regulating female reproductive processes, but such action cannot be excluded.
Taken together, available data demonstrate the ability of daidzein and equol to regulate the release of steroid and nonsteroid hormones (progestogen, androgens, estrogens, oxytocin, adipokines), and to generate the production of gonadotropin and estrogen receptors. These signaling molecules could be constituents of the integrated endocrine pituitary–gonadal axis. For example, the production of progestogens, androgens, estrogens, and oxytocin is regulated by gonadotropins, while there is also the mutual stimulation of steroid hormones and oxytocin. The gonadotropin–ovarian hormone axis and receptors to these hormones are known regulators of basic ovarian functions and fecundity [34].
Therefore, available data demonstrate that daidzein can affect female reproductive processes via the system of extracellular self-stimulating hormones of pituitary, ovarian, and nonovarian origins and of their receptors. On the other hand, the involvement of only gonadotropin and estrogen receptors in mediating daidzein action is not yet indicated.
References
- Vitale, D.C.; Piazza, C.; Melilli, B.; Drago, F.; Salomone, S. Isoflavones: Estrogenic activity, biological effect and bioavailability. Eur. J. Drug Metab. Pharmacokinet. 2013, 38, 15–25.
- Zaheer, K.; Humayoun Akhtar, M. An updated review of dietary isoflavones: Nutrition, processing, bioavailability and impacts on human health. Crit. Rev. Food Sci. Nutr. 2017, 57, 1280–1293.
- Křížová, L.; Dadáková, K.; Kašparovská, J.; Kašparovský, T. Isoflavones. Molecules 2019, 24, 1076.
- Barnes, S.; Prasain, J.; D’Alessandro, T.; Arabshahi, A.; Botting, N.; Lila, M.A.; Jackson, G.; Janle, E.M.; Weaver, C.M. The metabolism and analysis of isoflavones and other dietary polyphenols in foods and biological systems. Food Funct. 2011, 2, 235–244.
- Mayo, B.; Vázquez, L.; Flórez, A.B. Equol: A Bacterial Metabolite from the Daidzein Isoflavone and Its Presumed Beneficial Health Effects. Nutrients 2019, 11, 2231.
- Sekikawa, A.; Ihara, M.; Lopez, O.; Kakuta, C.; Lopresti, B.; Higashiyama, A.; Aizenstein, H.; Chang, Y.F.; Mathis, C.; Miyamoto, Y.; et al. Effect of S-equol and Soy Isoflavones on Heart and Brain. Curr. Cardiol. Rev. 2019, 15, 114–135.
- Ronis, M.J. Effects of soy containing diet and isoflavones on cytochrome P450 enzyme expression and activity. Drug Metab. Rev. 2016, 48, 331–341.
- Luca, S.V.; Macovei, I.; Bujor, A.; Miron, A.; Skalicka-Woźniak, K.; Aprotosoaie, A.C.; Trifan, A. Bioactivity of dietary polyphenols: The role of metabolites. Crit. Rev. Food Sci. Nutr. 2020, 60, 626–659.
- Zhou, T.; Meng, C.; He, P. Soy Isoflavones and their Effects on Xenobiotic Metabolism. Curr. Drug Metab. 2019, 20, 46–53.
- Cederroth, C.R.; Zimmermann, C.; Nef, S. Soy, phytoestrogens and their impact on reproductive health. Mol. Cell. Endocrinol. 2012, 355, 192–200.
- Van Duursen, M.B.M. Modulation of estrogen synthesis and metabolism by phytoestrogens in vitro and the implications for women’s health. Toxicol. Res. 2017, 6, 772–794.
- Hu, W.S.; Lin, Y.M.; Kuo, W.W.; Pan, L.F.; Yeh, Y.L.; Li, Y.H.; Kuo, C.H.; Chen, R.J.; Padma, V.V.; Chen, T.S.; et al. Suppression of isoproterenol-induced apoptosis in H9c2 cardiomyoblast cells by daidzein through activation of Akt. Chin. J. Physiol. 2016, 59, 323–330.
- Rienks, J.; Barbaresko, J.; Nöthlings, U. Association of isoflavone biomarkers with risk of chronic disease and mortality: A systematic review and meta-analysis of observational studies. Nutr. Rev. 2017, 75, 616–641.
- Chen, L.R.; Ko, N.Y.; Chen, K.H. Isoflavone Supplements for Menopausal Women: A Systematic Review. Nutrients 2019, 11, 2649.
- Das, D.; Sarkar, S.; Bordoloi, J.; Wann, S.B.; Kalita, J.; Manna, P. Daidzein, its effects on impaired glucose and lipid metabolism and vascular inflammation associated with type 2 diabetes. Biofactors 2018, 44, 407–417.
- Adhikari, B.; Marasini, B.P.; Rayamajhee, B.; Bhattarai, B.R.; Lamichhane, G.; Khadayat, K.; Adhikari, A.; Khanal, S.; Parajuli, N. Potential roles of medicinal plants for the treatment of viral diseases focusing on COVID-19: A review. Phytother. Res. 2020.
- Liu, Z.M.; Ho, S.C.; Woo, J.; Chen, Y.M.; Wong, C. Randomized controlled trial of whole soy and isoflavone daidzein on menopausal symptoms in equol-producing Chinese postmenopausal women. Menopause 2014, 21, 653–660.
- Lamartiniere, C.A.; Wang, J.; Smith-Johnson, M.; Eltoum, I.E. Daidzein: Bioavailability, potential for reproductive toxicity, and breast cancer chemoprevention in female rats. Toxicol. Sci. 2002, 65, 228–238.
- Kaludjerovic, J.; Chen, J.; Ward, W.E. Early life exposure to genistein and daidzein disrupts structural development of reproductive organs in female mice. J. Toxicol. Environ. Health A 2012, 75, 649–660.
- Kouki, T.; Kishitake, M.; Okamoto, M.; Oosuka, I.; Takebe, M.; Yamanouchi, K. Effects of neonatal treatment with phytoestrogens, genistein and daidzein, on sex difference in female rat brain function: Estrous cycle and lordosis. Horm. Behav. 2003, 44, 140–145.
- Kouki, T.; Okamoto, M.; Wada, S.; Kishitake, M.; Yamanouchi, K. Suppressive effect of neonatal treatment with a phytoestrogen, coumestrol, on lordosis and estrous cycle in female rats. Brain Res. Bull. 2005, 64, 449–454.
- Talsness, C.; Grote, K.; Kuriyama, S.; Presibella, K.; Sterner-Kock, A.; Poça, K.; Chahoud, I. Prenatal exposure to the phytoestrogen daidzein resulted in persistent changes in ovarian surface epithelial cell height, folliculogenesis, and estrus phase length in adult Sprague-Dawley rat offspring. J. Toxicol. Environ. Health A 2015, 78, 635–644.
- Mahalingam, S.; Gao, L.; Gonnering, M.; Helferich, W.; Flaws, J.A. Equol inhibits growth, induces atresia, and inhibits steroidogenesis of mouse antral follicles in vitro. Toxicol. Appl. Pharmacol. 2016, 295, 47–55.
- Liu, H.; Zhang, C.; Zeng, W. Estrogenic and antioxidant effects of a phytoestrogen daidzein on ovarian germ cells in embryonic chickens. Domest. Anim. Endocrinol. 2006, 31, 258–268.
- Liu, H.Y.; Zhang, C.Q. Effects of daidzein on messenger ribonucleic acid expression of gonadotropin receptors in chicken ovarian follicles. Poult. Sci. 2008, 87, 541–545.
- Medigović, I.M.; Živanović, J.B.; Ajdžanović, V.Z.; Nikolić-Kokić, A.L.; Stanković, S.D.; Trifunović, S.L.; Milošević, V.L.; Nestorović, N.M. Effects of soy phytoestrogens on pituitary-ovarian function in middle-aged female rats. Endocrine 2015, 50, 764–776.
- Dorward, A.M.; Shultz, K.L.; Beamer, W.G. LH analog and dietary isoflavones support ovarian granulosa cell tumor development in a spontaneous mouse model. Endocr. Relat. Cancer 2007, 14, 369–379.
- Takaoka, O.; Mori, T.; Ito, F.; Okimura, H.; Kataoka, H.; Tanaka, Y.; Koshiba, A.; Kusuki, I.; Shigehiro, S.; Amami, T.; et al. Daidzein-rich isoflavone aglycones inhibit cell growth and inflammation in endometriosis. J. Steroid Biochem. Mol. Biol. 2018, 181, 125–132.
- Mlynarczuk, J.; Wrobel, M.H.; Kotwica, J. The adverse effect of phytoestrogens on the synthesis and secretion of ovarian oxytocin in cattle. Reprod. Domest. Anim. 2011, 46, 21–28.
- Sirotkin, A.; Záhoranska, Z.; Tarko, A.; Popovska-Percinic, F.; Alwasel, S.; Harrath, A.H. Plant isoflavones can prevent adverse effects of benzene on porcine ovarian activity: An in vitro study. Environ. Sci. Pollut. Res. Int. 2020, 27, 29589–29598.
- Sirotkin, A.; Záhoranska, Z.; Tarko, A.; Fabova, Z.; Alwasel, S.; Halim Harrath, A. Plant polyphenols can directly affect ovarian cell functions and modify toluene effects. J. Anim Physiol. Anim. Nutr. 2020.
- Somjen, D.; Katzburg, S.; Nevo, N.; Gayer, B.; Hodge, R.P.; Renevey, M.D.; Kalchenko, V.; Meshorer, A.; Stern, N.; Kohen, F. A daidzein-daunomycin conjugate improves the therapeutic response in an animal model of ovarian carcinoma. J. Steroid Biochem. Mol. Biol. 2008, 110, 144–149.
- Green, J.M.; Alvero, A.B.; Kohen, F.; Mor, G. 7-(O)-Carboxymethyl daidzein conjugated to N-t-Boc-hexylenediamine: A novel compound capable of inducing cell death in epithelial ovarian cancer stem cells. Cancer Biol. Ther. 2009, 8, 1747–1753.
- Sirotkin, A.V. Regulators of Ovarian Functions, 2nd ed.; Nova Science Publishers, Inc.: New York, NY, USA, 2014; p. 194. ISBN 978-1-62948-574-4.
- Nynca, A.; Jablonska, O.; Slomczynska, M.; Petroff, B.K.; Ciereszko, R.E. Effects of phytoestrogen daidzein and estradiol on steroidogenesis and expression of estrogen receptors in porcine luteinized granulosa cells from large follicles. J. Physiol. Pharmacol. 2009, 60, 95–105.
- Nynca, A.; Słonina, D.; Jablońska, O.; Kamińska, B.; Ciereszko, R.E. Daidzein affects steroidogenesis and oestrogen receptor expression in medium ovarian follicles of pigs. Acta Vet. Hung. 2013, 61, 85–98.
- Otokozawa, S.; Tanaka, R.; Akasaka, H.; Ito, E.; Asakura, S.; Ohnishi, H.; Saito, S.; Miura, T.; Saito, T.; Mori, M. Associations of Serum Isoflavone, Adiponectin and Insulin Levels with Risk for Epithelial Ovarian Cancer: Results of a Case-control Study. Asian Pac. J. Cancer Prev. 2015, 16, 4987–4991.
- Chan, K.K.L.; Siu, M.K.Y.; Jiang, Y.X.; Wang, J.J.; Leung, T.H.Y.; Ngan, H.Y.S. Estrogen receptor modulators genistein, daidzein and ERB-041 inhibit cell migration, invasion, proliferation and sphere formation via modulation of FAK and PI3K/AKT signaling in ovarian cancer. Cancer Cell Int. 2018, 18, 65.





