
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Angelica Petrillo | + 3052 word(s) | 3052 | 2021-04-06 11:28:58 | | | |
| 2 | Vivi Li | Meta information modification | 3052 | 2021-04-14 07:59:47 | | |
Video Upload Options
Immune checkpoint inhibitors have revolutionized the treatment landscape for many solid tumors. Amongst gastric cancer subtypes, tumors with microsatellite instability and Epstein Barr Virus positive tumors provide the strongest rationale for responding to immunotherapy. Various predictive biomarkers such as mismatch repair status, programmed death ligand 1 expression, tumor mutational burden, assessment of tumor infiltrating lymphocytes and circulating biomarkers have been evaluated. However, results have been inconsistent due to different methodologies and thresholds used. Clinical implementation therefore remains a challenge. The role of immune checkpoint inhibitors in gastric cancer is emerging with data from monotherapy in the heavily pre-treated population already available and studies in earlier disease settings with different combinatorial approaches in progress. Immune checkpoint inhibitor combinations with chemotherapy (CT), anti-angiogenics, tyrosine kinase inhibitors, anti-Her2 directed therapy, poly (ADP-ribose) polymerase inhibitors or dual checkpoint inhibitor strategies are being explored.
1. Introduction
Overview of Gastric Cancer Classification and Relevance for Immunotherapy
Gastric cancer (GC) is a leading global cause of morbidity and mortality [1]. In 2020, over a million people were diagnosed with GC (representing almost 6% of all cancer diagnoses), and nearly 800,000 patients died due to this disease (representing 8.2% of all cancer deaths) [2]. Worldwide, GC is particularly prevalent in East Asia and central/Eastern Europe.
The Lauren classification, published in 1965, differentiates gastric adenocarcinoma into two distinct types, termed the intestinal and diffuse subtypes [3]. The intestinal type is most common, present in over half of the patients and characterized by microscopic glandular structures, with infiltrating capacity of the mesenchymal tissues [4]. The diffuse subtype accounts for a third of cases and is characterized by poor differentiation and poorly cohesive malignant cells with invasive capacity [5]. In general, the intestinal type is associated with exogenous risk factors such as Helicobacter pylori, while the diffuse subtype encompasses a hereditary familial pattern related to germline pathogenetic mutations of the E-cadherin (CDH1) and αE-catenin (CTNNA1) genes [6]. While these subtypes of GC are associated with different carcinogenesis mechanisms and disease biology, this classification, along with the subsequent World Health Organization classification of GC, has not translated into distinct subtype-driven treatment strategies [7][8]. More recently, following comprehensive molecular profiling, The Cancer Genome Atlas (TCGA) defined four distinct subtypes of gastric cancer: Epstein-Barr virus (EBV) positive, microsatellite unstable tumors (MSI), genomically stable tumors (GS) and tumors with chromosomal instability (CIN) [9]. Significant overlap was seen between the histologically determined Lauren’s diffuse variant and the molecular GS TCGA subtype [10]. Interestingly, certain molecular subtypes were most commonly detected in specific anatomic locations with EBV positive tumors more likely to be in the gastric fundus or body and CIN tumors in the cardia [9]. Although the molecular classification of gastric cancer has not directly changed clinical practice, it has provided an important platform to identify novel molecular targets and pave the way for innovative clinical trial design with the incorporation of biomarker enrichment stratification strategies. EBV-positive and MSI tumors are associated with signatures suggestive of an immune responsive profile [11]. A hyper-mutated DNA phenotype is defined as 20.5 mutations/Mb in GC and is a phenotype typical of most MSI tumors [12]. The MSI high (MSI-H) phenotype is most commonly related to epigenetic silencing of the mismatch repair gene, MLH1, rather than germline mutation (i.e., Lynch syndrome) [13]. The presence of a higher number of somatic mutations has been associated with a better prognosis [14] and an increased susceptibility to immune-activating antineoplastic treatments [15]. Currently, patients with MSI gastric cancer can benefit from established immunotherapy approaches with anti-programmed death-1 (anti-PD-1) immune-checkpoint inhibitors [16]. Rather than a hypermutated phenotype, EBV-positive tumors (accounting for 9% of GC) have a profile favoring immunotherapy in view of their high expression of membrane immune-checkpoint molecules such as programmed death ligand-1 (PD-L1) and 2. Key molecular features of EBV-positive tumors include the expression of virus-associated antigens (e.g., nuclear antigen 1, latent membrane protein 2A), the extensive methylation of viral and host genome and the epigenetic regulation of specific cytosine-phosphatidyl-guanosine (CpG) DNA islands through methylation mechanisms [17]. The pattern of DNA methylation of CpG has been associated with anti-tumor immune-activation, with predictive and prognostic significance [18][19]. Therefore, MSI and EBV-positive tumors have been proposed as chief candidates for immunotherapy trials, though not exclusively, for their intrinsic immune-mediated biology [11]. The advent of immunotherapy in oncology has in fact been embraced in most if not all tumor types and disease settings [20]. The identification of an immune-signature or predictive factors of immune-response in patients with GC have been identified as a research priority given that it is a tumor type associated with poor prognosis when diagnosed at an advanced stage and any benefit derived from chemotherapy (CT) is very limited [21]. While advancements in the development of pharmacotherapies have improved overall survival (OS) and quality of life, the low proportion of patients alive after two years from the diagnosis of metastatic disease remains a cause for concern [22][23].
The strategies implemented to enhance the immune response against tumors, including GC, aim to re-orient the immune-system response, by dampening the suppressive regulatory molecules and enhancing a stimulating milieu [24]. This strategy has been pursued by developing a number of immune-checkpoint inhibitors [e.g., PD-1, cytotoxic T-lymphocyte antigen-4 (CTLA-4)], a class of molecules capable of acting on several immune cells and (re-)activating an effective antineoplastic response [25]. This strategy is particularly beneficial in tumors exerting immune-activating signatures and/or recognized by the immune-system as foreign, and therefore regulated by the immune-response [26].
Another therapeutic approach is based on the bioengineering of immune-competent cells against specific tumor- associated antigens [27]. The principal expression of this approach is represented by the Chimeric Antigen Receptor T-cells (CAR-T) constructs. CAR-T are genetically engineered T-cells designed to direct the specific immune-response against tumor- antigens, thereby inducing an artificial acquired antineoplastic immune response, through cytotoxic activity. Though still widely experimental in solid neoplasms, the clinical implementation of CAR-T cells for hematological malignancies has paved a new way of cancer immunotherapy, due to the durable responses seen in some cases, the different patterns of response observed [28] as well as the specific safety profile which needs to be considered and the structural efforts required to build and deliver cell-based treatments [29].
2. Biomarkers of Response to Immunotherapy in Gastric Cancer
The characterization of immune-related biomarkers is becoming increasingly important in the multi-modality treatment of advanced GC (Figure 1) [30].
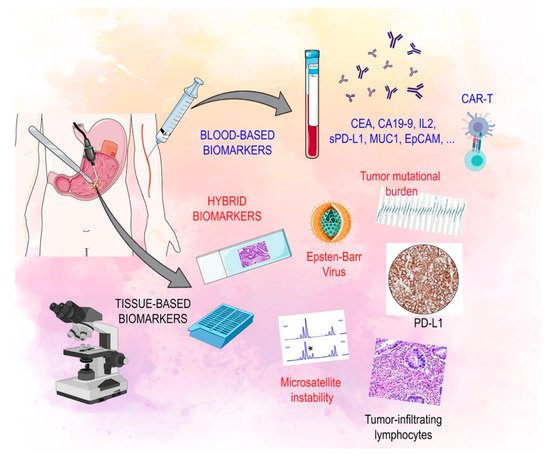
Figure 1. Biomarkers of response to immunotherapy: soluble, tissue based and hybrid. Legend: CAR-T: chimeric antigen receptor-T cell, CA 19.9: carbohydrate antigen 19.9, CEA: carcinoembryonic antigen, EPCAM: epithelial cell adhesion molecule, IL2: interleukin 2, MUC1: mucin 1, cell surface associated, PD-L1: programmed death-ligand 1, s PD-L1: serum programmed death-ligand.
2.1. Tissue Based Biomarkers
Currently, the most studied biomarkers include mismatch repair (MMR) status assessment, MSI identification, PD-L1 expression, tumor-infiltrating lymphocytes (TILs) assessment, and tumor mutational burden (TMB) quantification. However, there is currently a gap in knowledge regarding the reliability of these tests for clinical use in GC. MMR deficiency (dMMR) and/or MSI has been reported in approximately 14% and 22% of GCs, respectively [31][32]. The MMR system is able to identify and counteract unpaired DNA bases in order to preserve genome stability [33][34][35]. Alterations in this system, due to dMMR, are associated with the accumulation of alterations in microsatellite regions, resulting in variable degrees of MSI that are commonly defined as “low” (MSI-L) and “high” (MSI-H) [35]. MMR and MSI screening is recommended as a useful tool at all stages of GC to refine treatments and determine patient prognosis [36]. In GC, MSI is more common with older age, female sex, distal stomach location, lower number of lymph-node metastases and is associated with an overall better prognosis [36][37][38]. According to a meta-analysis which included 1556 resectable GC patients, MSI-H patients had longer five-year OS and disease-free survival (DFS) compared to patients with microsatellite stable tumors (OS, 77.5% vs. 59.3%; DFS, 71.8% vs. 52.3%) [14][39]. Evidence suggests that dMMR is more likely to activate an immune response and lead to the increased presence of TILs, and PD-L1 upregulation in GC [40][41][42][43]. In particular, PD-L1 is expressed on the surface of neoplastic cells in 15–70% of GC [37], with increased expression being associated with non-metastatic cancer tissue [44], well differentiated tumors [44] and improved OS (median OS not reached vs. 40 months; p = 0.008) [45] although its association with a favorable OS has not always been consistent [37][46]. The immunohistochemical expression of PD-L1 protein can be scored using the combined positive score (CPS), where CPS > 1 is considered positive [47]. There appears to be an association between PD-L1+ GC and MSI-H or EBV positive tumors [48]. The evaluation of PD-L1 CPS on formalin-fixed paraffin-embedded tumor tissue samples has been proposed as a method to select patients for immune-checkpoint inhibition [49]. High PD-L1 CPS score has been associated with a high density of CD3+/CD8+ TILs. Interestingly, PD-L1 negative tumors with high-density CD3+ and CD8+ cells had a good prognosis [46]. A meta-analysis on various TIL subtypes in GC has shown that high levels of CD8+, CD3+, and CD4+ T cell infiltration is associated with better OS. Additionally, a high density of forkhead box P3 (FOXP3) positive cells within the tumor does not appear to be a negative prognostic indicator [50]. TILs are gathering increasing importance as a prognostic biomarker in GC [51]. Regarding TIL assessment in GC, only the stromal count (= % area occupied by mononuclear inflammatory cells over the total stromal area)—stained by hematoxylin and eosin—has been suggested for evaluation, due to a lack of prognostic significance for intra-tumoral TILs [52]. However, this finding requires further validation [52][53].
In the KEYNOTE 059 study, PD-L1 expression as a potential biomarker of response to pembrolizumab in advanced and refractory GC patients was evaluated and demonstrated a higher overall response rate (ORR) in PD-L1+ compared to the PD-L1 negative tumors (15.5%, CPS ≥1 vs. 6.4%, CPS < 1). However, PD-L1 negative tumors displayed greater complete response rate (CR) (2.8% vs. 2.0%) [39][54]. Such findings prompted the need for defining and utilizing further biomarkers of response to immunotherapy. Tumor mutational burden (TMB), dMMR/MSI, TILs and EBV have been broadly explored as the main molecular determinants of immunotherapy response in GC. Of note, TMB (i.e., the number of somatic mutations derived from next-generation sequencing techniques) has been correlated with higher levels of neoantigen expression, and subsequently increased immune responses [55]. High TMB has been suggested in 3% and 5% of patients with esophageal and stomach cancer, respectively (>20 mut/Mb) [30]. Recently, the food and drug administration (FDA) approved the FoundationOneCDx assay (Foundation Medicine, Inc., Cambridge, MA, USA) as a companion diagnostic (CDx) for treatment with pembrolizumab in unresectable or metastatic TMB-high solid tumors (≥10 mut/Mb) [56]. Findings in GC also show an improved OS in TMB-High tumors treated with immunotherapy, compared to those with lower TMB levels: 80% 2-year survival for TMB-high vs. 12% for TMB-low, p = 0.03 [57] and median OS = 16.8 vs. 6.62 months, p = 0.058 [58]. Yet, TMB as a potential biomarker of response to immunotherapy is challenged by the lack of harmonized sequencing panels as well as lack of clearly defined cut-offs for implementation in clinical practice [36].
MSI-H tumors have also been associated with a good response to immunotherapy [15][59][60]. Based on these findings, the FDA approved pembrolizumab for the treatment of MSI-H tumors that had progressed following prior treatment, irrespective of tumor site [61]. In GC, studies have also demonstrated that dMMR and MSI-H tumors generally have a favorable response to immune-checkpoint blockade [36][59]. A multi cohort study of pembrolizumab monotherapy in advanced GC showed that MSI-H tumors had greater ORR (57.1%) compared to MSS patients (9%) and also a significant disease control rate (DCR) of 71.4% was achieved [30][36]. These results were supported by findings from the phase III, KEYNOTE-062 clinical trial which are discussed in the clinical trial section of this review [62]. MMR status is commonly assessed by immunohistochemistry although the lack of CDx tests and tumor-specific guidelines is seen as a disadvantage [35]. However, unlike in other types of tumors, a high correlation between MMR immunohistochemistry and MSI testing in gastro-esophageal cancer is generally observed [36]. Hence, the PCR-based Bethesda panel, consisting of two mononucleotide repeats and three dinucleotide repeats, and NGS are employed in MSI evaluation [63].
EBV+ tumors are associated with a response to immunotherapy and this appears to be independent of TMB and MSI status [64]. According to a multi-factorial genomic biomarker analysis in GC patients administered pembrolizumab, EBV positivity, MSI and PD-L1 expression are associated with improved ORR (100%, 85.7%, and 50%, respectively) [65]. EBV+ GC appears to be relatively immunogenic. This results in increased infiltration with immune cells and also increased PD-L1 and PD-L2 gene expression [65]. Further investigation into biomarkers of response to immunotherapy in advanced GC/gastro-esophageal cancer is warranted.
2.2. Circulating Biomarkers
Circulating molecules and their role in predicting response to immunotherapy is a topic of great interest and is an area that still has not been studied in depth. These soluble factors can be released from both tumor cells and immune cells and may provide a simple method to evaluate the dynamic behavior of the immune system in cancer patients during treatment and avoid the need for invasive procedures [66]. Much effort is being spent on identifying primary responders to immunotherapy at a relatively early treatment timepoint. Circulating biomarkers, some of which are currently used in clinical practice, such as pepsinogen, carcinoembryonic antigen (CEA), carbohydrate antigen 19-9 (CA19-9) and soluble IL-2, are not accurate enough to predict prognosis in GC [67][68][69].
Recent evidence has shown that patients with GC have higher serum soluble PD-L1 (sPD-L1) concentrations than healthy controls. Moreover, both elevated tissue PD-L1 and serum sPD-L1 were independent prognostic factors for poor OS and poor DFS in GC patients who underwent surgery [70][71][72]. Lymphocyte activation gene 3 (LAG3) is a checkpoint receptor localized on activated T cell surfaces and NK cells. The soluble variant, in turn, can have a regulatory function on immune cells [73][74]. Its role has been investigated in GC patients. High sLAG-3 expression is associated with a better prognosis in GC and its expression was positively correlated with IL-12 and IFN-γ production in GC patients. In a recent in vivo experiment sLAG3 was shown to be able to promote the activation of CD8 T cells and the production of INF and IL-12, resulting in tumor growth inhibition [75].
While the prognostic value of soluble checkpoints is under investigation in several solid tumors, the question that remains to be answered is whether soluble checkpoints can predict response to treatment. Given that the immune system is a key factor involved in the response to treatments such as immunotherapy and CT, there is a clear rationale to suggest that such soluble markers could be biomarkers of response to treatment [76]. A study including 11 patients with NSCLC and 9 patients with GC treated with an anti-PD1 agent showed that pre-treatment levels of sPD-L1 were not associated with OS in these patients. However, reduction in plasma sPD-L1 levels was significantly associated with tumor response after four cycles of treatment [77]. A study including 68 patients with metastatic GC eligible for first line CT analyzed baseline level of sPDL1 and the dynamic changes during therapy. Patients with low levels of sPD-L1 at diagnosis showed a better OS and PFS than patients with a high sPDL1. Patients whose sPDL1 increased after the first cycle of CT showed worse PFS and OS. This result suggests that soluble checkpoints may be the ideal method of studying the immune system as an extremely dynamic entity allowing real-time, non-invasive monitoring during cancer treatment [78]. Takahashi et al. confirmed in their study that high serum levels of sPD-L1 correlated with worse OS in patients with metastatic GC treated with first-line CT [79]. These data suggest the possibility of individualizing the therapeutic choice based on the immunological profile, thereby leading to promising new combination strategies in the near future.
Immunotherapeutics in solid tumors is constantly evolving due to the introduction of new technologies to manipulate the patient’s immune system to attack cancer cells. Tumor antigen vaccines are currently being studied in several solid tumors. They are created from cancer cells’ pure tumor antigens [80]. The antitumor activity of tumor peptide vaccines, such as G17DT, vascular endothelial growth factor receptor (VEGFR) and OTSGC-A24, have been investigated in GC patients. G17DT is a vaccine able to promote an immune response against gastrin, a hormone involved in carcinogenesis and progression in GC [81][82][83]. A phase II/III study (NCT00042510) reported that G17DT is able to induce efficient anti-gastrin antibody production and is able to inhibit tumor proliferation and progression [84]. A multi-center study showed that the combination of G17DT and platinum-5FU CT prolonged the median time-to-progression and median survival time for patients with unresectable cancer of the stomach or gastroesophageal junction, compared to platinum-5FU CT alone. Therefore, the FDA approved the fast track designation for the vaccine G17DT in February 2003 [85]. Another peptide vaccine involving the use of VEGFR 1 and 2, receptors of the VEGF angiogenic factor, has been investigated. In a phase I/II study, the administration of the VEGFR1/2 peptide vaccine in combination with CT induced a cytotoxic T cell response. In the 82% of patients with a cytotoxic T lymphocyte response to VEGFR2-169 peptide, time to progression and OS were significantly prolonged compared to those without such a response [86]. Such findings are encouraging, although it should be noted that only 22 patients were included. A phase I/Ib study (NCT01227772) evaluated OTSGC-A24, which is thought to be able to target several specific tumor antigens, such as forkhead box M1, DEP domain containing 1, kinesin family member 20A, URLC10 and VEGFR1. Although the treatment was well tolerated, no radiological responses were observed [83].
An innovative immunotherapeutic strategy uses adoptive T cell therapy to overcome the immune-evasion mechanisms mediated by cancer cells. T lymphocytes are removed from patients and modified in vitro in order to activate specific immune cells. Then, the modified activated T cells are administered to patients, thereby eliciting a tumor response against cancer [87]. Chimeric antigen receptor-T (CAR-T) cell therapy was shown to be effective in hematologic disease and it is actually under investigation in several solid tumors [88][89].
In GC, several antigens, including human epidermal growth factor receptor 2 (HER2), carcinoembryonic antigen (CEA), mucin 1 (MUC1) and epithelial cell adhesion molecule (EpCAM), have been used as targets for CAR-T. The anti-HER2 CAR-modified T cell was evaluated in many pre-clinical studies [90]. Clinical studies are now evaluating it in GC patients (NCT02713984, NCT01935843, NCT00889954). CEA-specific CAR-T cells were confirmed to be active in pre-clinical studies in mice with GC. Since then, a clinical trial is ongoing (NCT02349724) to define the correct dose and safety profile [91][92]. MUC1 and EpCAM are transmembrane glycoproteins expressed in different solid tumors, but in GC they are markers of aggressive disease. Clinical Phase I/II trials (NCT02617134, NCT02725125) are evaluating EpCAM and MUC1 modified CAR-T in solid tumors expressing these targets [93].
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424.
- Global Cancer Observatory: Gastric Cancer. Available online: (accessed on 23 December 2020).
- Laurén, P. The two histological main types of gastric carcinoma: Diffuse and so-called intestinal-type carcinoma. Acta Pathol. Microbiol. Scand. 1965, 64, 31–49.
- Marqués-Lespier, J.M.; González-Pons, M.; Cruz-Correa, M. Current Perspectives on Gastric Cancer. Gastroenterol. Clin. North Am. 2016, 45, 413–428.
- Ma, J.; Shen, H.; Kapesa, L.; Zeng, S. Lauren classification and individualized chemotherapy in gastric cancer. Oncol. Lett. 2016, 11, 2959–2964.
- Blair, V.R.; McLeod, M.; Carneiro, F.; Coit, D.G.; D’Addario, J.L.; van Dieren, J.M.; Harris, K.L.; Hoogerbrugge, N.; Oliveira, C.; van der Post, R.S.; et al. Hereditary diffuse gastric cancer: Updated clinical practice guidelines. Lancet Oncol. 2020, 21, e386–e397.
- Berlth, F. Pathohistological classification systems in gastric cancer: Diagnostic relevance and prognostic value. World, J. Gastroenterol. 2014, 20, 5679.
- Bosman, F.T.; Carneiro, F.; Hruban, R.H.; Theise, N.D. WHO Classification of Tumours of the Digestive System; World Health Organization: Geneva, Switzerland, 2010; ISBN 9789283224327.
- Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature 2014, 513, 202–209.
- Ling, Y.; Watanabe, Y.; Nagahashi, M.; Shimada, Y.; Ichikawa, H.; Wakai, T.; Okuda, S. Genetic profiling for diffuse type and genomically stable subtypes in gastric cancer. Comput. Struct. Biotechnol. J. 2020, 18, 3301–3308.
- Rodriquenz, M.G.; Roviello, G.; D’Angelo, A.; Lavacchi, D.; Roviello, F.; Polom, K. MSI and EBV Positive Gastric Cancer’s Subgroups and Their Link with Novel Immunotherapy. J. Clin. Med. 2020, 9, 1427.
- Li, X.; Wu, W.K.K.; Xing, R.; Wong, S.H.; Liu, Y.; Fang, X.; Zhang, Y.; Wang, M.; Wang, J.; Li, L.; et al. Distinct Subtypes of Gastric Cancer Defined by Molecular Characterization Include Novel Mutational Signatures with Prognostic Capability. Cancer Res. 2016, 76, 1724–1732.
- Leung, S.Y.; Yuen, S.T.; Chung, L.P.; Chu, K.M.; Chan, A.S.Y.; Ho, J.C.I. hMLH1 Promoter Methylation and Lack of hMLH1 Expression in Sporadic Gastric Carcinomas with High-Frequency Microsatellite Instability. Cancer Res. 1999, 59, 159–164.
- Pietrantonio, F.; Miceli, R.; Raimondi, A.; Kim, Y.W.; Kang, W.K.; Langley, R.E.; Choi, Y.Y.; Kim, K.-M.; Nankivell, M.G.; Morano, F.; et al. Individual Patient Data Meta-Analysis of the Value of Microsatellite Instability As a Biomarker in Gastric Cancer. J. Clin. Oncol. 2019, 37, 3392–3400.
- Le, D.T.; Durham, J.N.; Smith, K.N.; Wang, H.; Bartlett, B.R.; Aulakh, L.K.; Lu, S.; Kemberling, H.; Wilt, C.; Luber, B.S.; et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science 2017, 357, 409–413.
- Le, D.T.; Uram, J.N.; Wang, H.; Bartlett, B.R.; Kemberling, H.; Eyring, A.D.; Skora, A.D.; Luber, B.S.; Azad, N.S.; Laheru, D.; et al. PD-1 Blockade in Tumors with Mismatch-Repair Deficiency. N. Engl. J. Med. 2015, 372, 2509–2520.
- Eichelberg, M.R.; Welch, R.; Guidry, J.T.; Ali, A.; Ohashi, M.; Makielski, K.R.; McChesney, K.; Van Sciver, N.; Lambert, P.F.; Keleș, S.; et al. Epstein-Barr Virus Infection Promotes Epithelial Cell Growth by Attenuating Differentiation-Dependent Exit from the Cell Cycle. MBio 2019, 10.
- Jung, H.; Kim, H.S.; Kim, J.Y.; Sun, J.-M.; Ahn, J.S.; Ahn, M.-J.; Park, K.; Esteller, M.; Lee, S.-H.; Choi, J.K. DNA methylation loss promotes immune evasion of tumours with high mutation and copy number load. Nat. Commun. 2019, 10, 4278.
- Geddert, H.; Zur Hausen, A.; Gabbert, H.E.; Sarbia, M. EBV-infection in cardiac and non-cardiac gastric adenocarcinomas is associated with promoter methylation of p16, p14 and APC, but not hMLH1. Anal. Cell. Pathol. (Amst.) 2010, 33, 143–149.
- Xin Yu, J.; Hubbard-Lucey, V.M.; Tang, J. Immuno-oncology drug development goes global. Nat. Rev. Drug Discov. 2019, 18, 899–900.
- Yang, L.; Wang, Y.; Wang, H. Use of immunotherapy in the treatment of gastric cancer (Review). Oncol. Lett. 2019, 18, 5681–5690.
- Zhao, L.; Li, J.; Bai, C.; Nie, Y.; Lin, G. Multi-Modality Treatment for Patients With Metastatic Gastric Cancer: A Real-World Study in China. Front. Oncol. 2019, 9, 1155.
- Ebinger, S.M.; Warschkow, R.; Tarantino, I.; Schmied, B.M.; Güller, U.; Schiesser, M. Modest overall survival improvements from 1998 to 2009 in metastatic gastric cancer patients: A population-based SEER analysis. Gastric Cancer 2016, 19, 723–734.
- Robert, C. A decade of immune-checkpoint inhibitors in cancer therapy. Nat. Commun. 2020, 11, 3801.
- Schumacher, T.N.; Scheper, W.; Kvistborg, P. Cancer Neoantigens. Annu. Rev. Immunol. 2019, 37, 173–200.
- Hendrickx, W.; Simeone, I.; Anjum, S.; Mokrab, Y.; Bertucci, F.; Finetti, P.; Curigliano, G.; Seliger, B.; Cerulo, L.; Tomei, S.; et al. Identification of genetic determinants of breast cancer immune phenotypes by integrative genome-scale analysis. Oncoimmunology 2017, 6, e1253654.
- Wagner, J.; Wickman, E.; DeRenzo, C.; Gottschalk, S. CAR T Cell Therapy for Solid Tumors: Bright Future or Dark Reality? Mol. Ther. 2020, 28, 2320–2339.
- Huang, J.; Rong, L.; Wang, E.; Fang, Y. Pseudoprogression of extramedullary disease in relapsed acute lymphoblastic leukemia after CAR T-cell therapy. Immunotherapy 2021, 13, 5–10.
- Ou, Z. PCN198 Global regulatory challenges of CAR T-Cell therapies: Approval, pricing, and access. Value Health 2019, 22, S93.
- Lin, E.M.; Gong, J.; Klempner, S.J.; Chao, J. Advances in immuno-oncology biomarkers for gastroesophageal cancer: Programmed death ligand 1, microsatellite instability, and beyond. World J. Gastroenterol. 2018, 24, 2686–2697.
- Ratti, M.; Lampis, A.; Hahne, J.C.; Passalacqua, R.; Valeri, N. Microsatellite instability in gastric cancer: Molecular bases, clinical perspectives, and new treatment approaches. Cell. Mol. Life Sci. 2018, 75, 4151–4162.
- Kim, J.-Y.; Shin, N.R.; Kim, A.; Lee, H.-J.; Park, W.; Kim, J.-Y.; Lee, C.-H.; Huh, G.-Y.; Park, D.Y. Microsatellite Instability Status in Gastric Cancer: A Reappraisal of Its Clinical Significance and Relationship with Mucin Phenotypes. Korean J. Pathol. 2013, 47, 28.
- Trenner, A.; Sartori, A.A. Harnessing DNA Double-Strand Break Repair for Cancer Treatment. Front. Oncol. 2019, 9, 1388.
- Motegi, A.; Masutani, M.; Yoshioka, K.; Bessho, T. Aberrations in DNA repair pathways in cancer and therapeutic significances. Semin. Cancer Biol. 2019, 58, 29–46.
- Corti, C.; Sajjadi, E.; Fusco, N. Determination of Mismatch Repair Status in Human Cancer and Its Clinical Significance. Adv. Anat. Pathol. 2019, 26, 270–279.
- Lopez, G.; Venetis, K.; Sajjadi, E.; Fusco, N. Mismatch Repair System Genomic Scars in Gastroesophageal Cancers: Biology and Clinical Testing. Gastrointest. Disord. 2020, 2, 341–352.
- Matsuoka, T.; Yashiro, M. Biomarkers of gastric cancer: Current topics and future perspective. World J. Gastroenterol. 2018, 24, 2818–2832.
- Dhakras, P.; Uboha, N.; Horner, V.; Reinig, E.; Matkowskyj, K.A. Gastrointestinal cancers: Current biomarkers in esophageal and gastric adenocarcinoma. Transl. Gastroenterol. Hepatol. 2020, 5, 55.
- Pagni, F.; Guerini-Rocco, E.; Schultheis, A.M.; Grazia, G.; Rijavec, E.; Ghidini, M.; Lopez, G.; Venetis, K.; Croci, G.A.; Malapelle, U.; et al. Targeting Immune-Related Biological Processes in Solid Tumors: We do Need Biomarkers. Int. J. Mol. Sci. 2019, 20, 5452.
- Sajjadi, E.; Venetis, K.; Scatena, C.; Fusco, N. Biomarkers for precision immunotherapy in the metastatic setting: Hope or reality? Ecancermedicalscience 2020, 14.
- Polom, K.; Marano, L.; Marrelli, D.; De Luca, R.; Roviello, G.; Savelli, V.; Tan, P.; Roviello, F. Meta-analysis of microsatellite instability in relation to clinicopathological characteristics and overall survival in gastric cancer. Br. J. Surg. 2018, 105, 159–167.
- Giampieri, R.; Maccaroni, E.; Mandolesi, A.; Del Prete, M.; Andrikou, K.; Faloppi, L.; Bittoni, A.; Bianconi, M.; Scarpelli, M.; Bracci, R.; et al. Mismatch repair deficiency may affect clinical outcome through immune response activation in metastatic gastric cancer patients receiving first-line chemotherapy. Gastric Cancer 2017, 20, 156–163.
- Yuza, K.; Nagahashi, M.; Watanabe, S.; Takabe, K.; Wakai, T. Hypermutation and microsatellite instability in gastrointestinal cancers. Oncotarget 2017, 8, 112103–112115.
- He, P.; Ma, Z.; Han, H.; Zhang, X.; Niu, S.; Du, L.; Zheng, Y.; Liu, H. Expression of programmed death ligand 1 (PD-L1) is associated with metastasis and differentiation in gastric cancer. Life Sci. 2020, 242, 117247.
- Liu, X.; Choi, M.G.; Kim, K.; Kim, K.-M.; Kim, S.T.; Park, S.H.; Cristescu, R.; Peter, S.; Lee, J. High PD-L1 expression in gastric cancer (GC) patients and correlation with molecular features. Pathol. Res. Pract. 2020, 216, 152881.
- Ju, X.; Shen, R.; Huang, P.; Zhai, J.; Qian, X.; Wang, Q.; Chen, M. Predictive relevance of PD-L1 expression with pre-existing TILs in gastric cancer. Oncotarget 2017, 8, 99372–99381.
- Fassan, M.; Brignola, S.; Pennelli, G.; Alberti, G.; Angerilli, V.; Bressan, A.; Pellino, A.; Lanza, C.; Salmaso, R.; Lonardi, S.; et al. PD-L1 expression in gastroesophageal dysplastic lesions. Virchows Arch. 2020, 477, 151–156.
- Ma, C.; Patel, K.; Singhi, A.D.; Ren, B.; Zhu, B.; Shaikh, F.; Sun, W. Programmed Death-Ligand 1 Expression Is Common in Gastric Cancer Associated With Epstein-Barr Virus or Microsatellite Instability. Am. J. Surg. Pathol. 2016, 40, 1496–1506.
- Ye, D.; Xu, G.; Ma, W.; Li, Y.; Luo, W.; Xiao, Y.; Liu, Y.; Zhang, Z. Significant function and research progress of biomarkers in gastric cancer (Review). Oncol. Lett. 2019, 19, 17–29.
- Zhang, N.; Cao, M.; Duan, Y.; Bai, H.; Li, X.; Wang, Y. Prognostic role of tumor-infiltrating lymphocytes in gastric cancer: A meta-analysis and experimental validation. Arch. Med. Sci. 2020, 16, 1092–1103.
- Lee, J.S.; Won, H.S.; Sun, D.S.; Hong, J.H.; Ko, Y.H. Prognostic role of tumor-infiltrating lymphocytes in gastric cancer. Medicine (Baltimore) 2018, 97, e11769.
- Hendry, S.; Salgado, R.; Gevaert, T.; Russell, P.A.; John, T.; Thapa, B.; Christie, M.; van de Vijver, K.; Estrada, M.V.; Gonzalez-Ericsson, P.I.; et al. Assessing Tumor-Infiltrating Lymphocytes in Solid Tumors. Adv. Anat. Pathol. 2017, 24, 311–335.
- Liu, J.; Xu, Y.; Yu, M.; Liu, Z.; Xu, Y.; Ma, G.; Zhou, W.; Kong, P.; Ling, L.; Wang, S.; et al. Increased Stromal Infiltrating Lymphocytes are Associated with Circulating Tumor Cells and Metastatic Relapse in Breast Cancer Patients After Neoadjuvant Chemotherapy. Cancer Manag. Res. 2019, 11, 10791–10800.
- Fuchs, C.S.; Doi, T.; Jang, R.W.; Muro, K.; Satoh, T.; Machado, M.; Sun, W.; Jalal, S.I.; Shah, M.A.; Metges, J.-P.; et al. Safety and Efficacy of Pembrolizumab Monotherapy in Patients With Previously Treated Advanced Gastric and Gastroesophageal Junction Cancer. JAMA Oncol. 2018, 4, e180013.
- Sholl, L.M.; Hirsch, F.R.; Hwang, D.; Botling, J.; Lopez-Rios, F.; Bubendorf, L.; Mino-Kenudson, M.; Roden, A.C.; Beasley, M.B.; Borczuk, A.; et al. The Promises and Challenges of Tumor Mutation Burden as an Immunotherapy Biomarker: A Perspective from the International Association for the Study of Lung Cancer Pathology Committee. J. Thorac. Oncol. 2020, 15, 1409–1424.
- U.S. Food and Drug Administration. FDA Approves Pembrolizumab for Adults and Children with TMB-H Solid Tumors. Available online: (accessed on 19 December 2020).
- Ku, G.Y.; Sanchez-Vega, F.; Chatila, W.; Margolis, M.; Fein, C.; Ilson, D.H.; Hechtman, J.F.; Tuvy, Y.; Bouvier, N.; Kundra, R.; et al. Correlation of benefit from immune checkpoint inhibitors with next gen sequencing (NGS) profiles in esophagogastric cancer (EGC) patients. J. Clin. Oncol. 2017, 35, 4025.
- Janjigian, Y.Y.; Sanchez-Vega, F.; Jonsson, P.; Chatila, W.K.; Hechtman, J.F.; Ku, G.Y.; Riches, J.C.; Tuvy, Y.; Kundra, R.; Bouvier, N.; et al. Genetic Predictors of Response to Systemic Therapy in Esophagogastric Cancer. Cancer Discov. 2018, 8, 49–58.
- Sundar, R.; Smyth, E.C.; Peng, S.; Yeong, J.P.S.; Tan, P. Predictive Biomarkers of Immune Checkpoint Inhibition in Gastroesophageal Cancers. Front. Oncol. 2020, 10, 763.
- Zhao, P.; Li, L.; Jiang, X.; Li, Q. Mismatch repair deficiency/microsatellite instability-high as a predictor for anti-PD-1/PD-L1 immunotherapy efficacy. J. Hematol. Oncol. 2019, 12, 54.
- Marcus, L.; Lemery, S.J.; Keegan, P.; Pazdur, R. FDA Approval Summary: Pembrolizumab for the Treatment of Microsatellite Instability-High Solid Tumors. Clin. Cancer Res. 2019, 25, 3753–3758.
- Shitara, K.; Van Cutsem, E.; Bang, Y.-J.; Fuchs, C.; Wyrwicz, L.; Lee, K.-W.; Kudaba, I.; Garrido, M.; Chung, H.C.; Lee, J.; et al. Efficacy and Safety of Pembrolizumab or Pembrolizumab Plus Chemotherapy vs Chemotherapy Alone for Patients With First-line, Advanced Gastric Cancer. JAMA Oncol. 2020, 6, 1571.
- Cho, J.; Kang, S.Y.; Kim, K.-M. MMR protein immunohistochemistry and microsatellite instability in gastric cancers. Pathology 2019, 51, 110–113.
- Luchini, C.; Bibeau, F.; Ligtenberg, M.J.L.; Singh, N.; Nottegar, A.; Bosse, T.; Miller, R.; Riaz, N.; Douillard, J.-Y.; Andre, F.; et al. ESMO recommendations on microsatellite instability testing for immunotherapy in cancer, and its relationship with PD-1/PD-L1 expression and tumour mutational burden: A systematic review-based approach. Ann. Oncol. 2019, 30, 1232–1243.
- Kim, S.T.; Cristescu, R.; Bass, A.J.; Kim, K.-M.; Odegaard, J.I.; Kim, K.; Liu, X.Q.; Sher, X.; Jung, H.; Lee, M.; et al. Comprehensive molecular characterization of clinical responses to PD-1 inhibition in metastatic gastric cancer. Nat. Med. 2018, 24, 1449–1458.
- Chen, Y.; Wang, Q.; Shi, B.; Xu, P.; Hu, Z.; Bai, L.; Zhang, X. Development of a sandwich ELISA for evaluating soluble PD-L1 (CD274) in human sera of different ages as well as supernatants of PD-L1+ cell lines. Cytokine 2011, 56, 231–238.
- Duraker, N.; Naci Çelik, A.; Gençler, N. The prognostic significance of gastric juice CA 19–9 and CEA levels in gastric carcinoma patients. Eur. J. Surg. Oncol. 2002, 28, 844–849.
- Miki, K.; Morita, M.; Sasajima, M.; Hoshina, R.; Kanda, E.; Urita, Y. Usefulness of gastric cancer screening using the serum pepsinogen test method. Am. J. Gastroenterol. 2003, 98, 735–739.
- Miki, K. Gastric cancer screening using the serum pepsinogen test method. Gastric Cancer 2006, 9, 245–253.
- Zheng, Z.; Bu, Z.; Liu, X.; Zhang, L.; Li, Z.; Wu, A.; Wu, X.; Cheng, X.; Xing, X.; Du, H.; et al. Level of circulating PD-L1 expression in patients with advanced gastric cancer and its clinical implications. Chin. J. Cancer Res. 2014, 26, 104–111.
- Shigemori, T.; Toiyama, Y.; Okugawa, Y.; Yamamoto, A.; Yin, C.; Narumi, A.; Ichikawa, T.; Ide, S.; Shimura, T.; Fujikawa, H.; et al. Soluble PD-L1 Expression in Circulation as a Predictive Marker for Recurrence and Prognosis in Gastric Cancer: Direct Comparison of the Clinical Burden Between Tissue and Serum PD-L1 Expression. Ann. Surg. Oncol. 2019, 26, 876–883.
- Gershtein, E.S.; Ognerubov, N.A.; Chang, V.L.; Delektorskaya, V.V.; Korotkova, E.A.; Sokolov, N.Y.; Polikarpova, S.B.; Stilidi, I.S.; Kushlinskii, N.E. [The content of the soluble forms PD-1 and PD-L1 in blood serum of patients with gastric cancer and their relationship with clinical and morphological characteristics of the disease.]. Klin. Lab. Diagn. 2020, 65, 347–352.
- Shapiro, M.; Herishanu, Y.; Katz, B.-Z.; Dezorella, N.; Sun, C.; Kay, S.; Polliack, A.; Avivi, I.; Wiestner, A.; Perry, C. Lymphocyte activation gene 3: A novel therapeutic target in chronic lymphocytic leukemia. Haematologica 2017, 102, 874–882.
- Okamura, T.; Fujio, K.; Sumitomo, S.; Yamamoto, K. Roles of LAG3 and EGR2 in regulatory T cells. Ann. Rheum. Dis. 2012, 71, i96–i100.
- Li, N.; Jilisihan, B.; Wang, W.; Tang, Y.; Keyoumu, S. Soluble LAG3 acts as a potential prognostic marker of gastric cancer and its positive correlation with CD8+T cell frequency and secretion of IL-12 and INF-γ in peripheral blood. Cancer Biomarkers 2018, 23, 341–351.
- Botticelli, A.; Mezi, S.; Pomati, G.; Cerbelli, B.; Di Rocco, C.; Amirhassankhani, S.; Sirgiovanni, G.; Occhipinti, M.; Napoli, V.; Emiliani, A.; et al. The 5-Ws of immunotherapy in head and neck cancer. Crit. Rev. Oncol. Hematol. 2020, 153, 103041.
- Ando, K.; Hamada, K.; Watanabe, M.; Ohkuma, R.; Shida, M.; Onoue, R.; Kubota, Y.; Matsui, H.; Ishiguro, T.; Hirasawa, Y.; et al. Plasma Levels of Soluble PD-L1 Correlate With Tumor Regression in Patients With Lung and Gastric Cancer Treated With Immune Checkpoint Inhibitors. Anticancer Res. 2019, 39, 5195–5201.
- Park, W.; Bang, J.-H.; Nam, A.-R.; Jin, M.H.; Seo, H.; Kim, J.-M.; Oh, K.S.; Kim, T.-Y.; Oh, D.-Y. Prognostic Value of Serum Soluble Programmed Death-Ligand 1 and Dynamics During Chemotherapy in Advanced Gastric Cancer Patients. Cancer Res. Treat. 2021, 53, 199–206.
- Takahashi, N.; Iwasa, S.; Sasaki, Y.; Shoji, H.; Honma, Y.; Takashima, A.; Okita, N.T.; Kato, K.; Hamaguchi, T.; Yamada, Y. Serum levels of soluble programmed cell death ligand 1 as a prognostic factor on the first-line treatment of metastatic or recurrent gastric cancer. J. Cancer Res. Clin. Oncol. 2016, 142, 1727–1738.
- Ribas, A.; Butterfield, L.H.; Glaspy, J.A.; Economou, J.S. Current developments in cancer vaccines and cellular immunotherapy. J. Clin. Oncol. 2003, 21, 2415–2432.
- Gilliam, A.D.; Watson, S.A. G17DT: An antigastrin immunogen for the treatment of gastrointestinal malignancy. Expert Opin. Biol. Ther. 2007, 7, 397–404.
- Park, D.J.; Thomas, N.J.; Yoon, C.; Yoon, S.S. Vascular Endothelial Growth Factor A Inhibition in Gastric Cancer. Gastric Cancer 2015, 18, 33–42.
- Sundar, R.; Rha, S.Y.; Yamaue, H.; Katsuda, M.; Kono, K.; Kim, H.S.; Kim, C.; Mimura, K.; Kua, L.-F.; Yong, W.P. A phase I/Ib study of OTSGC-A24 combined peptide vaccine in advanced gastric cancer. BMC Cancer 2018, 18, 332.
- Gilliam, A.; Watson, S.; Henwood, M.; McKenzie, A.; Humphreys, J.; Elder, J.; Iftikhar, S.; Welch, N.; Fielding, J.; Broome, P.; et al. A phase II study of G17DT in gastric carcinoma. Eur. J. Surg. Oncol. 2004, 30, 536–543.
- Ajani, J.A.; Randolph Hecht, J.; Ho, L.; Baker, J.; Oortgiesen, M.; Eduljee, A.; Michaeli, D. An open-label, multinational, multicenter study of G17DT vaccination combined with cisplatin and 5-fluorouracil in patients with untreated, advanced gastric or gastroesophageal cancer: The GC4 study. Cancer 2006, 106, 1908–1916.
- Masuzawa, T.; Yoshiyuki, F.; Okada, K.; Nakamura, A.; Takiguchi, S.; Nakajima, K.; Miyata, H.; Yamasaki, M.; Kurokawa, Y.; Osawa, R.; et al. Phase I/II study of S-1 plus cisplatin combined with peptide vaccines for human vascular endothelial growth factor receptor 1 and 2 in patients with advanced gastric cancer. Int. J. Oncol. 2012, 41, 1297–1304.
- Stauss, H.J.; Morris, E.C.; Abken, H. Cancer gene therapy with T cell receptors and chimeric antigen receptors. Curr. Opin. Pharmacol. 2015, 24, 113–118.
- Li, J.; Li, W.; Huang, K.; Zhang, Y.; Kupfer, G.; Zhao, Q. Chimeric antigen receptor T cell (CAR-T) immunotherapy for solid tumors: Lessons learned and strategies for moving forward. J. Hematol. Oncol. 2018, 11, 22.
- Gill, S.; Maus, M.V.; Porter, D.L. Chimeric antigen receptor T cell therapy: 25years in the making. Blood Rev. 2016, 30, 157–167.
- Whilding, L.M.; Maher, J. ErbB-targeted CAR T-cell immunotherapy of cancer. Immunotherapy 2015, 7, 229–241.
- Wang, L.; Ma, N.; Okamoto, S.; Amaishi, Y.; Sato, E.; Seo, N.; Mineno, J.; Takesako, K.; Kato, T.; Shiku, H. Efficient tumor regression by adoptively transferred CEA-specific CAR-T cells associated with symptoms of mild cytokine release syndrome. Oncoimmunology 2016, 5, e1211218.
- Guest, R.D.; Kirillova, N.; Mowbray, S.; Gornall, H.; Rothwell, D.G.; Cheadle, E.J.; Austin, E.; Smith, K.; Watt, S.M.; Kühlcke, K.; et al. Definition and application of good manufacturing process-compliant production of CEA-specific chimeric antigen receptor expressing T-cells for phase I/II clinical trial. Cancer Immunol. Immunother. 2014, 63, 133–145.
- Warneke, V.S.; Behrens, H.-M.; Haag, J.; Krüger, S.; Simon, E.; Mathiak, M.; Ebert, M.P.A.; Röcken, C. Members of the EpCAM signalling pathway are expressed in gastric cancer tissue and are correlated with patient prognosis. Br. J. Cancer 2013, 109, 2217–2227.





