
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Kurt Rosentrater | + 2642 word(s) | 2642 | 2021-04-09 12:23:07 | | | |
| 2 | Lily Guo | Meta information modification | 2642 | 2021-04-12 02:50:17 | | |
Video Upload Options
Aspartic acid, or “aspartate,” is a non-essential, four carbon amino acid produced and used by the body in two enantiomeric forms: L-aspartic acid and D-aspartic acid. The L-configuration of amino acids is the dominant form used in protein synthesis; thus, L-aspartic acid is by far the more common configuration. However, D-aspartic acid is one of only two known D-amino acids biosynthesized by eukaryotes. While L-aspartic acid is used in protein biosynthesis and neurotransmission, D-aspartic acid is associated with neurogenesis and the endocrine system. Aspartic acid production and use has been growing in recent years.
1. Industrial Utility
In addition to its biofunctionality, aspartic acid has wide application in the food, beverage, pharmaceutical, cosmetic, and agricultural industries [1]. L-aspartic acid is used as a nutritional supplement in both functional foods and beverages, but its primary use is in combination with the amino acid phenylalanine which together make aspartame, an artificial sweetener [2]. Aspartic acid is also used to bolster immune function and as a natural combatant to depression [1]. Its ability to aid in energy production, fatigue resistance, RNA and DNA synthesis, and liver detoxification give it broad clinical use [1]. Additionally, it is used as an intermediary substrate in the manufacture of pharmaceuticals and organic chemicals, serving as the building block molecule for active pharmaceutical ingredients [1]. Aspartic acid’s utility stretches further upon consideration of its derivatives including acetyl aspartic acid, used as an active ingredient in anti-aging cosmetics that target wrinkling, skin lifting, and loss of firmness [3]. It is also used to produce polyaspartic acid, a fertilizer synergist which increases both nitrogen absorption and crop yields [4]. Polyaspartic acid hydrogels are a type of biodegradable superabsorbent polymer which have exceptional water-holding abilities and are used in the production of many modern amenities including diapers, feminine products, and engineered tissue [5]. The range and depth of aspartic acid’s applicability, in particular the L-configuration, has placed it on the Department of Energy’s Top Value Added Chemicals from Biomass list [2].
2. Global Markets
The global aspartic acid market is a highly fractionated market meaning it consists of several small company players rather than large conglomerates, yet it is growing with significant potential for industrial relevance [6]. According to a 2015 report by Grand View Research, the global aspartic acid market is projected to reach $101 million with a market demand of 60.6 kilotons by 2022 which represents a compound annual growth rate of 5.6% [6]. As of 2014, the baseline year of said report, polyaspartic acid represented 22.6% of the total aspartic acid market volume making it the largest market segment, seconded by aspartame [6]. Both aspartic acid derivatives are anticipated to increase in demand as polyaspartic biodegradable polymers replace polyacrylic acid in agriculture, water treatment, and the petrochemical industries and as food and beverage trends shift towards added sugar labeling and health-conscious, convenience foods [6]. Of all aspartic acid market sectors, the medical sector is projected to grow the most as is attributed to the American healthcare system, which is housed in the largest regional market, accounting for 39.0% of total aspartic acid volume as of 2014 [6]. Internationally increased demand for aspartic acid is also expected to increase in the form of greater aspartame demand for carbonated beverages in Asia Pacific [6].
3. Production and Manufacturing
There are three main methods to produce aspartic acid: protein extraction, chemical synthesis, and enzymatic conversion [2]. The hydrolysis of protein for extraction methods produces an abundance of amino acids from which the L-aspartic acid must be separated. Chemical synthesis requires high temperature and pressure and results in a racemic mixture, producing both L- and D-isomers thereby requiring the additional processing steps of optical resolution and racemization to achieve the preferred L-isomer [1]. Thus, enzymatic conversion is the currently favored route of production. The enzymatic conversion process exists in two forms: simple enzyme-substrate interaction (hereafter referred to as “enzymatic conversion”) or whole-cell enzymatic conversion, i.e., fermentation. Table 1 summarizes various economic and technical aspects of the production of aspartic acid.
Table 1. Summary of Aspartic Acid Supply Chain, Economic and Technological Considerations for Aspartic Acid Production.
| Category | Summary |
|---|---|
| Industrial importance and potential of biochemical | Aspartic acid is used in the food, beverage, pharmaceutical, cosmetic, and agricultural industries. The global aspartic acid market is projected to reach $101 million with a market demand of 60.6 kilotons by 2022 representing a compound annual growth rate of 5.6% [6]. |
| Industrial uses for biochemical | Aspartic acid is used in the production of: nutritional (amino acid) supplements; artificial sweetener (aspartame); polyaspartic acid hydrogels; and acetyl aspartic acid, the active ingredient in anti-aging cosmetics. |
| Substrates used for the production of biochemical | primary substrate: fumaric acid [7] cofactor: ammonia [7] enzyme: L-aspartate ammonia-lyase [7] |
| Microorganisms used for fermentation | Primary industrial species:E. coli and Cornybacterium glutamacium [8] Exploratory species [1]: Pseudomonas aeroginosa Pseudomonas fluorescens Candida hydrocarbofumarica Bacillus stearothermophilus Bacillus subtilis |
| What enzymes are needed to break down the substrate for fermentation | Fumaric acid used in aspartic acid production does not need to be broken down, rather, it is fermentatively produced from glucose or chemically produced from maleic anhydride [6]. |
| Fermentation conditions used: pH, substrate loading, temperatures, times, maximum yield, maximum fermentation rates | pH is initialized to 7.0 [9] substrate concentration: 1:1 or 1:2 ammonia to fumaric acid [9] time 2 to 10 days [9] temperature 27–40 °C [9] yield 77–95% (w/w of fumaric acid) depending on bacterial strain and fermentation conditions [9] |
| Separation equipment, conditions, efficiencies | batch fermentation: separation via anion exchange column and crystallization [9] continuous fermentation: separation via isoelectric point precipitation and crystallization [9] |
| Total energy used to produce this chemical | Data not currently published. |
| Estimated costs to produce this chemical | Cost as well as upstream and downstream raw materials and equipment analysis available in the global L-aspartic acid market report provided by Market Watch (2019), at https://www.researchreportsworld.com/purchase/14314090 (accessed on 21 February 2021) |
| Current aspartic acid manufacturers | The following companies are the top industrial producers of aspartic acid [10]; the corresponding links, when applicable, are to each respective company’s product information page. Ajinomoto Group https://www.ajiaminoacids.com/product/l-aspartic-acid (accessed on 21 February 2021) Evonik https://healthcare.evonik.com/product/health-care/en/products/pharmaceutical-amino-acids/REXIM/pages/parenteralnutrition.aspx?xd_co_f=M2Q2OWQ5N2ItYTZkOC00ZWZjLThjNmUtODFiYjQ3YmYwM2I2 (accessed on 21 February 2021) KYOWA http://www.kyowahakko-bio.co.jp/english/products/aminoacids/l_aspartic_acid/ (accessed on 21 February 2021) Jinghai Amino Acid http://en.chinaaminoacid.com/products/L-AsparticAcid.shtml (accessed on 21 February 2021) JIRONG PHARM Not currently available OR product catalogue not in English Siwei Amino AcidEnglish product description not available Zhangjiagangxingyu Technology http://www.zjgxykj.com/template/p13e.html (accessed on 21 February 2021) Hubei Bafeng Pharmaceutical Company page not accessible in English |
| Potential market segments, sales, etc. | The aspartic acid market is segmented into six market categories: Feed Supplements, Medicine, Polyaspartic Acid, Aspartame, L-Alanine, and “Others” [6]. The report summary states that polyaspartic acid represents 22.6% of the total market volume in 2014. Market volumes and revenue values available upon report purchase [10]. |
| Primary economic setbacks and challenges | Fermentative production competes economically with petroleum-derived production. Economic setbacks of aspartic acid include high fumaric substrate cost and the low yields currently achieved by switching to cheaper sugar-based feedstocks [9]. Crystallization utilized in downstream processing separations can be expensive and time-consuming [1]. |
| Technological setbacks and challenges | The fermentative production of aspartic acid from glucose or sugar-based feedstocks, both much cheaper and more available substrates than fumarate, currently generate much poorer yields, i.e., 95% versus 29% [11]. Thus, the main technological setback to more economical aspartic acid production is the ability to directly ferment sugar to L-aspartic acid. |
| Side products, byproducts, waste products and associated cost | Organisms whose genomes also code fumarase (e.g., C. glutamicum, E. coli) produce malic acid from fumarate as a byproduct in effect utilizing substrate and decreasing aspartic acid yield. Without heat treatments Tajima et al. (2015) lost 25% of the fumaric acid substrate to malic acid production which translates to significant yield losses [7]. |
| Downstream processing operations | L-aspartic acid can be separated from the culture broth or eluate in batch systems via ion exchange resins utilizing an anion exchange column followed by crystallization of the eluate [1]. Continuous systems can extract the L-aspartic acid via isoelectric point precipitation (adjust broth pH to 2.8) followed by crystallization [9]. |
| New technologies, strains, equipment developments | Membrane reactor systems (MRS), as they are currently being developed, utilize growth-arrested cells eliminating the need for cell or enzyme immobilization [11]. The MRS system employed by Yukawa et al. (2009) overcomes the low mass transfer rates and low volumetric productivity issues associated with immobilization systems and simplifies the overall production process, allowing for easier separation of cells from the reaction mixture and generating high yield and productivity during long periods of operation [11]. Genetic modification of the metabolic pathways and feedback regulators within E. coli and C. glutamicum, the two major strains involved in industrial amino acid synthesis, are the next steps in improving L-aspartic acid production via the development of new, high-producing strains. |
Stereospecific, industrial production of L-aspartic acid currently utilizes a one-step reaction of fumaric acid, in the presence of high concentrations of ammonia, to L-aspartic acid via L- aspartate ammonia-lyase, an enzyme also referred to as “L-aspartase” (Figure 1) [7]. L-aspartase can be purified and immobilized in a gel matrix for continuous production or overexpressed in bacterial cells bound to polyurethane carriers [7]. Production via immobilized enzymatic conversion or bacterial fermentation utilizes the same enzyme and substrate; however, enzymatic production is favored for its high product concentration, productivity, minimal byproducts, and the ease of downstream processing [2]. Yet, fermentative production, albeit less productive, has been around since the 1950s when research and development into the production of aspartic acid began [11].
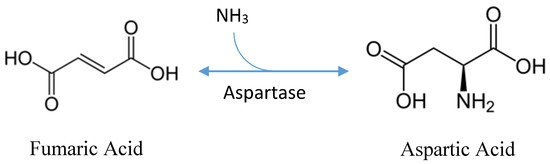
While several species of bacteria including select Pseudomonas, Bacillus, and Proteus have been identified as producers of aspartic acid, E. coli and Cornybacterium glutamacium are nearly exclusively used by industry [1][8]. Fumaric acid is the primary substrate in L-aspartic acid production; however, maleate, a less expensive feedstock, can be used in a two-step reaction which uses maleate isomerase to convert maleate to fumaric acid which is then converted via L-aspartase to L-aspartic acid in the presence of ammonium ions [11].
The basic fermentation process, as developed and patented in the 1960s, utilizes a sugar-free medium wherein fumaric acid is the sole source of carbon subsequently minimizing the production of unwanted byproducts [9]. Ammonia, while required for catalysis, also serves as the nitrogen source and is formulated at a 1:1 or 1:2 fumaric acid-ammonia ratio [9]. Prior to inoculation, the broth pH is initialized to 7.0 and left unregulated as it will naturally increase to a 8.4–9.6 range in the initial stages of the fermentation, allowing for the production of acid [9].
Fermentation can be conducted with or without agitation for 2 to 10 days at 27–40 °C [9]. The L-aspartic acid will be extracellularly secreted and accumulate in the culture broth [9]. Several methods of downstream processing are available to separate L-aspartic acid from the culture broth or eluate. In the case of batch fermentation, ion exchange resins can be used to separate and purify the L-aspartic acid on an anion exchange column followed by crystallization of the eluate (Figure 2). For continuous fermentation, L-aspartic acid can be separated by adjusting the broth to 90 °C and a pH of 2.8 with sulfuric acid [9]. Adjusting the pH to 2.8, i.e., the isoelectric point, will cause L-aspartic acid to precipitate out of solution where it is then subjected to a two hour incubation period at 15 °C to induce protein crystallization [9]. Under these conditions, 95% of the theoretical yield of L-aspartic acid was achieved by Masahiro et al. (1965) which aligns with the 77–95% yield range achieved similar fermentation processes utilizing various bacterial strains [1][9]. Table 2 provides commonly used L-aspartic acid fermentation parameters.
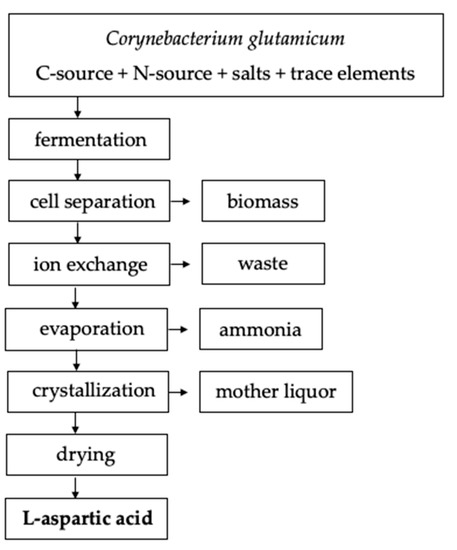
Figure 2. Example amino acid fermentation and downstream process flowchart adapted from Leuchtenberger et al. (2005) [12].
Table 2. Specific Production Parameters for the Fermentative Synthesis of Aspartic Acid.
| Yukawa et al. (2009) [11] | Tajima et al. (2015) [7] | Chibata et al. (1986) [13] | Szymanska et al. (2011) [14] | Papierz et al. (2007) [15] | |
|---|---|---|---|---|---|
| Pretreatments and Conditions Used | Genetically modify C. glutamicum to overproduce maleate isomerase and aspartase | E. coli DH5 used to produce plasmid containing aspA gene which is inserted into S. livingstonensis; sonicate then heat treat cells | Entrap E. coli in polyacrylamide gel via polymerization reaction then break gel in 3–4 mm diameter granules; wash granules in water | Immobilize cells in chitosan gel; culture in FF medium for biomass cultivation (or other chemically defined media as outlined on pg. 2) | Cell membrane permeabilization activates cells prior to aspartic acid production; perfomed in activation medium (chemically defined pg. 2) at 37 °C for 48 h |
| Substrate Used | Maleate ammonium | fumarate-NH3 | 1 M ammonium fumarate used for aspartic acid production by immobilized aspartase but no mention if substrate changed in subsequent trials | ammonium fumarate | fumaric acid |
| Substrate Loadings | Specifics not published | 860 mM fumarate-NH3 solution (pH 9) | 417 mM/h ammonium fumarate used for aspartic acid production by immobilized aspartase no mention if substrate loading changed in subsequent trials | 150.0 g/L ammonium fumarate | 100g/L fumaric acid 1 g biomass into 10 mL production media |
| Enzymes Used | maleate isomerase, aspartase | Enzymes are generated intracellularly | intracellular aspartase | intracellular aspartase | intracellular aspartase |
| Enzyme Loadings | Specifics not published | Not applicable | Not applicable | Not applicable | Not applicable |
| Reaction Times | production is continuous | 1–2 h | enzyme activity observed after 24–48 h found in production media; however, production can expands weeks in a continuous reactor | >603 h (production can be continuous) | 18–30 h |
| Bioreactor Conditions | pH 8.5 Temp 30 °C |
Heat treatment prior to fermentation performed in water bath; optimal conditions were 50 °C for 15 min | intended for continuous production; pack cells in a column reactor | biocatalyst bed height to volume ratio = 3:1; liquid hour space velocity value was 5.2 (i.e., volume of feeding substrate passed per volume of catalyst in bioreactor per one hour) |
100 mL shake flasks |
| Microorganisms Used | C. glutamicum | S. livingstonensis | E. coli ATCC 11303 | Escherichia coli mutant strains B-715 and P1 | Escherichia coli mutant strain B-715 |
| Fermentation Conditions Used (Temp, pH, etc.) | pH 8.5 Temp 30 °C |
whole cell production set at 37 °C for 3 h | Temp 37 °C half-life of column was 120 days |
initial media pH 8.5 Temp 40 °C |
initial media pH 8.5 Temp 37 °C |
| Separation Technologies Used | ultrafiltration | centrifugation, supernatant separated by HPLC with RI detector and ion exclusion column | Specifics not published | HPLC | HPLC |
| Separation Conditions Used | Specifics not published | Eluate at 60 °C using 0.1% (v/v) phosphoric acid for mobile phase with 0.7 mL/ min. flow rate; quantify via derivatization with DNFB | Specifics not published | HPLC with 250–4 LichrospherTM 100RP-18 (Merck) column and Waters fluorescence detector | Deproteinize with methanol, centrifuge, then run on HPLC column set to a flow-rate of 1 mL/min, 22 °C, and 2100 PSI with mobile phase of 200 mL methanol and 800 mL 0.05 M sodium phosphate buffer |
| Biochemical Yields Achieved | “High yield and productivity” hints that it should be within >95% range as achieved by immobilized cell methods; however, specifics not published | 95.2–99.3% | Immobilized aspartase had 29% activity yield but this “activity yield and the stability of the immobilized enzyme were not satisfactory for industrial purposes” thus the need to increase yield from this starting point in subsequent trials; for the set of conditions listed here, the results only mention that activity was notably increased | 99.8% conversion rate 6 g L-aspartic acid/g of cells /hour |
0.19–0.35 g L-aspartic acid/g of dry biomass/min during 1 h of biosynthesis |
| Inhibitory Compounds Observed, Developments and Impacts | L-malic acid is a byproduct (reduces yield) which can be avoided by inactivating fumarase via incubation at 45 °C for 5 h | L-malic acid also major byproduct (reduces yield) | Increased membrane permeability to substrate (“activation”) and later product increases enzyme activity and is the result of autolysis of the cells in the gel; Tween 80 required for E. coli P1 | Better immobilization and aspartic acid production with added surfactants for cell activation and a media 2-fold lower in yeast extract (found to be an inhibitory ingredient for biomass production) | Improved production following incubation in the activation medium containing 5 g/L ammonium fumarate |
| Notes | Incredibly limited in method detail and results | Exact methodology published, even greater detail in literature since multiple fermentation conditions were tested. | Review is very dated (1986); however, it covers several additional methods utilizing different gels and optimized parameters of base method. It appears to contain foundational work from which the popular immobilization technique of aspartic acid production was developed. | Highly detailed methodology | L-aspartic acid production was used to determine best aspartase-active mutant strain, i.e., conditions may not reflect requirements for scaled-up industrial production |
References
- Kumar, P.; Nika, B.; Mangala, D. Production of Aspartic Acid—A Short Review. Int. J. Eng. Trends Technol. 2017, 45, 254–257.
- Werpy, T.; Petersen, G. Top Value Added Chemicals from Biomass: Volume I—Results of Screening for Potential Candidates from Sugars and Synthesis Gas; US Department of Energy: Washington, DC, USA, 2004.
- Mavon, A. Acetyl Aspartic Acid, a Novel Active Ingredient, Demonstrates Potential to Improve Signs of Skin Ageing: From Consumer Need to Clinical Proof. Int. J. Cosmet. Sci. 2015, 37, 1–2.
- Kinnersley, A.M.; Koskan, L.P.; Strom, D.J.; Meah, A.R.Y. Kingenta. Polyaspartic Acid. US Patent US5593947A, 14 January 1997. Available online: (accessed on 14 February 2020).
- Meng, H.; Zhang, X.; Chen, Q.; Wei, J.; Wang, Y.; Dong, A.; Yang, H.; Tan, T.; Cao, H. Preparation of Poly(Aspartic Acid) Superabsorbent Hydrogels by Solvent-free Processes. J. Polym. Eng. 2015, 35, 647–655.
- Global Aspartic Acid Market by Application (Feed Supplements, Medicine, Polyaspartic Acid, Aspartame, L-Alanine) Expected to Reach USD 101.0 Million by 2022: Grand View Research, Inc. Available online: (accessed on 14 February 2020).
- Tajima, T.; Hamada, M.; Nakashimada, Y.; Kato, J. Efficient Aspartic Acid Production by a Psychrophile-based Simple Biocatalyst. J. Ind. Microbiol. Biotechnol. 2015, 42, 1319–1324.
- Li, Y.; Wei, H.; Wang, T.; Xu, Q.; Zhang, C.; Fan, X.; Ma, Q.; Chen, N.; Xie, X. Current Status on Metabolic Engineering for the Production of l-aspartate Family Amino Acids and Derivatives. Bioresour. Technol. 2017, 245, 1588–1602.
- Masahiro, T.; Yasoji, M.; Shinji, O.; Moriyoshi, I.; Tetsuo, H. Process for Producing l-aspartic Acid. U.S. Patent US2953499A, 1965.
- L-aspartic Acid Market 2019 Global Industry Demand, Recent Trends, Size and Share Estimation by 2025 with Top Players—ResearchReportsWorld.com. Available online: (accessed on 18 June 2019).
- Yukawa, H.; Ookino, S.; Inui, M. L-Aspartic Acid, Production Processes. In Encyclopedia of Industrial Biotechnology; Wiley: Hoboken, NJ, USA, 2010; pp. 1–3.
- Leuchtenberger, W.; Huthmacher, K.; Drauz, K. Biotechnological Production of Amino Acids and Derivatives: Current Status and Prospects. Appl. Microbiol. Biotechnol. 2005, 69, 1–8.
- Chibata, I.; Tosa, T.; Sato, T. Continuous Production of L-aspartic Acid. Appl. Biochem. Biotechnol. 1986, 13, 231–240.
- Szymańska, G.; Sobierajski, B.; Chmiel, A. Immobilized Cells of Recombinant Escherichia coli Strain for Continuous Production of L-aspartic Acid. Pol. J. Microbiol. 2011, 60, 105–112.
- Papierz, M.; Gadomska, G.; Sobierajski, B.; Chmiel, A. Selection and Activation of Escherichia coli Strains for L-aspartic Acid Biosynthesis. Pol. J. Microbiol. 2007, 56, 71.





