
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Steven Carter | + 3734 word(s) | 3734 | 2021-03-25 08:06:19 | | | |
| 2 | Conner Chen | Meta information modification | 3734 | 2021-04-01 15:43:31 | | | | |
| 3 | Conner Chen | Meta information modification | 3734 | 2021-04-06 08:40:29 | | |
Video Upload Options
Inflammatory diseases include a wide variety of highly prevalent conditions with high mortality rates in severe cases ranging from cardiovascular disease, to rheumatoid arthritis, to chronic obstructive pulmonary disease, to graft vs. host disease, to a number of gastrointestinal disorders. Many diseases that are not considered inflammatory per se are associated with varying levels of inflammation. Imaging of the immune system and inflammatory response is of interest as it can give insight into disease progression and severity. Clinical imaging technologies such as computed tomography (CT) and magnetic resonance imaging (MRI) are traditionally limited to the visualization of anatomical information; then, the presence or absence of an inflammatory state must be inferred from the structural abnormalities.
1. Cardiovascular Disease (CVD)
Cardiovascular disease (CVD) is the leading cause of death worldwide [1][2]. CVD is a broad term that encompasses many heart and circulatory system conditions, most of which develop gradually and are only diagnosed after the presentation of symptoms, which often result in fatality, mainly heart attack or stroke [3]. One person will die every 36 s from CVD in the United States alone, and with an increase in the number of smokers and growing obesity rates—two major risk factors for developing CVD—it is now more important than ever to focus on the development of early screening tools to identify the markers of CVD before it is too late [4][5][6].
Atherosclerosis occurs when plaque builds up inside the artery; over time, this plaque will harden and the artery will narrow, limiting blood flow, which often results in cardiovascular disease [7]. This plaque buildup is often only detected at the onset of symptoms, such as myocardial infarction or stroke, which are two of the most common causes of mortality in the United States and Europe [6]. At present, catheter-based X-ray angiography or intravascular ultrasound is used to identify coronary atherosclerosis, but this procedure is extremely invasive and only yields anatomical information about the degree of stenosis [1][8][9]. Non-invasive molecular imaging techniques must be utilized to characterize the plaque activity to determine which patients are extremely high-risk and require immediate intervention. Coronary CT angiography (CCTA) is a method for identifying the degree of stenosis and the plaque composition [10]. CCTA is able to score the degree of calcification of the coronary plaque, which is a strong predictor of a serious cardiovascular event [11][12]. While CCTA does provide functional information about CVD, it falls short of being a true molecular imaging technique, as it does not visualize changes on molecular level.
An increase in macrophage activity, reflective of inflammation, has been linked to a higher risk of plaque rupture; therefore, molecular imaging of macrophage activity in the arteries can help identify areas where plaque may be building [13][14][15]. 18F-Flourodeoxyglucose (FDG) PET imaging is commonly used to image the inflammatory component of atherosclerosis [16][17][18][19]. 18F-FDG is a radiolabeled glucose molecule, which is internalized by cells through the same mechanism in which glucose is metabolized. Both 18F-FDG and glucose are phosphorylated by hexokinase, where 18F-FDG becomes 18F-FDG-6-phosphate and glucose become glucose-6-phosphate. 18F-FDG-6-phosphate cannot be further metabolized by glucose-6-phosphate isomerase; therefore, it remains inside the cell for PET imaging [20]. In atherosclerosis, the accumulation of macrophages at locations of active plaque buildup requires a large amount of glucose, thus causing the upregulation of glucose transporters on the surface of these macrophages. Therefore, increased 18F-FDG uptake will be seen at locations of increased macrophage density, which is reflective of inflammation and active plaque buildup (Figure 1) [13][21]. It is unknown what the influence of 18F-FDG uptake from other cells, such as, neutrophils, endothelial cells, and lymphocytes, has on the observed signal [8][15]. Once the plaque cells have calcified, 18F-FDG uptake will subside substantially, making this type of PET imaging ineffective. PET imaging of atherosclerosis using 18F-FDG requires a circulation time of 2–3 h to allow for accumulation in the arterial wall and the decay or excretion of background levels of 18F-FDG [15]. 18F-FDG PET imaging in oncology typically needs 1 h of circulation time before imaging can begin.
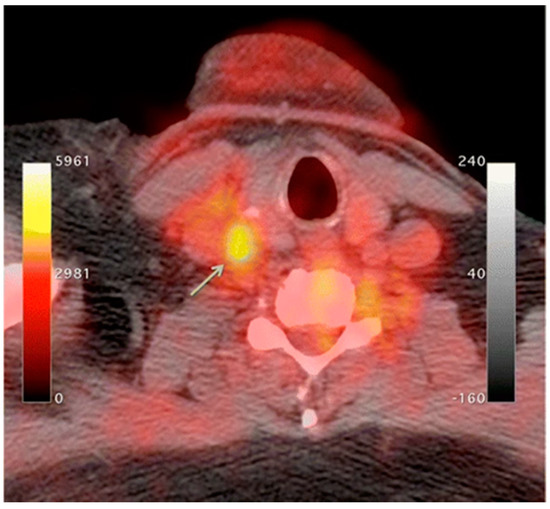
18F-FDG PET imaging is non-specific; therefore, it is complicated by highly metabolic neighboring tissues such as myocardial cells and neurons [13][22][23]. The suppression of myocardial 18F-FDG uptake can be achieved through dietary manipulation (high-fat, low-carb) to shift the body into beta-oxidation of fatty acids instead of metabolizing glucose as a primary energy source to try and limit this background activity [24][25]. Other radiotracers can be utilized that are specific to macrophages, limiting the effects from other highly metabolic cells. Translocator protein (TSPO)/peripheral benzodiazepine (PBR) receptors are overexpressed in activated macrophages, which is a great option for active targeting [26]. 11C-PK11195, (1-(2-Chlorophenyl)-N-(11C)methyl-N- (1-methylpropyl) -3-isoquinoline carboxamide) is a radiolabeled TSPO ligand that has been used as a PET tracer to visualize inflammatory plaque in atherosclerosis [26][27][28][29]. 11C-PK11195 uptake in patients with atherosclerosis was higher in patients who had a myocardial infarction or stroke compared to patients who were asymptomatic [28]. Other radiolabeled TSPO targeted ligands include 18F-GE-180, which showed a better signal-to-noise ratio and lower non-specific binding; more work must be done to validate this radiotracer [30].
68Ga-DOTATATE is another radiolabeled tracer that can be used to target inflammatory plaque in atherosclerosis by targeting the somatostatin receptor subtype 2 (SSR-2), which is also overexpressed on activated macrophages [31][32]. A copper radiolabel (64Cu) is frequently substituted for gallium because of the longer half-life and shorter positron range, which allow for better spatial resolution [22][33]. CXC-motif chemokine receptor 4 (CXCR-4) is also overexpressed on many immune cells, particularly monocytes and macrophages, making this receptor a good target for imaging inflammatory plaques of atherosclerosis [34]. Radiolabeled pentixafor, 68Ga-pentixafor, targets this CXCR-4 receptor for the quantification of arterial inflammation in atherosclerotic plaques [34][35][36].
As plaque builds up inside the artery, macrophages become active, and the region often becomes hypoxic due to the reduced oxygen diffusion efficiency from the thickening of the vessel wall. As active macrophages reflect sites of inflammation, it is suspected that macrophage activity is partially mediated by hypoxia as atherosclerotic plaques overexpress hypoxia-inducible factor 1-alpha (HIF-1α) [8][22][37]. There is ongoing research that focuses on the imaging of hypoxia as a surrogate biomarker of plaque inflammation and atherosclerosis. Radiolabeled ligands such as 18F-fluoromisonidazole (FMISO) or 18F-EF5 have been used to detect atherosclerotic plaques through PET imaging of hypoxia preclinically; more work must be done to advance these findings to the clinic [38][39][40].
2. Rheumatoid Arthritis
Rheumatoid arthritis (RA) is an autoimmune disorder that is characterized by chronic inflammation of the joints often causing degradation of the cartilage and bone, leading to a diminished quality of life due to musculoskeletal deficits and chronic pain [41]. For every 1000 adults, five will have RA, making it one of the most prevalent chronic inflammatory conditions worldwide [42]. RA etiology is not exactly known due to the synergistic effects of epigenetics [43] and environmental factors (smoking [44][45], obesity [46][47][48], and alcohol consumption [49][50][51]). Autoantibodies such as antibodies to citrullinated protein antigens (ACPAs) or rheumatoid factor (RF) have well-established roles in RA as accurate predictors of disease severity [52][53][54]. The current standard of care for the diagnosis of RA is through blood work to monitor the erythrocyte sedimentation rate (ESR), C-reactive protein levels (CRP), RF, and ACPAs [52][55] or anatomical imaging through MRI and ultrasound [56]. Power Doppler ultrasound (PDUS) is an US technique that is commonly used in the evaluation of RA, as it can visualize blood flow as well as anatomical information. The locations of active inflammation will have increased blood flow, making PDUS a good choice for not only diagnosing RA but also for assessing the severity and response to treatment [57][58].
Synovial membrane inflammation (synovitis) is a key characteristic of RA that involves the upregulation of both innate and adaptive immune cells and fibroblast-like synoviocytes (FLS) [59]. This immune response coupled with FLS results in inflammation and the activation of osteoclasts that leads to the degradation of cartilage [60][61]. The synovial fluid contains a variety of activated macrophages, B cells, and T cells, all of which are good targets for the molecular imaging of RA. The overexpression of inflammatory biomarkers can damage the existing vasculature, resulting in the enhanced permeability and retention (EPR) effect [62]. The newly permeable environment allows for the passive targeting of the immune cells of an inflammatory response. SPION-based contrast agents are small enough to penetrate the synovial fluid where they are phagocytized by active macrophages and can be visualized by T2-weighted MRI [63][64][65].
Activated macrophages can also be imaged using 18F-FDG PET imaging in the same manner described above [66][67][68]. While 18F-FDG PET imaging targets activated macrophages through elevated levels of glucose metabolism, there are more specific methods used to image active macrophages in RA. Folate receptor β (FRβ), a glycosylphosphatidyl plasma membrane anchored protein used to internalize folate needed for DNA synthesis and cell division, is overexpressed on activated macrophages in the synovial fluid, making it an attractive target for the molecular imaging of RA [69][70]. Radiolabeled folic acid can be imaged through scintigraphy or PET imaging for the detection of inflammation in the joints (Table 1) [69][71][72][73]. Spatial resolution of PET images is poor; a fluorescently labeled folate probe (NIR2-folate) can be visualized with NIR fluorescence imaging with greater spatial resolution, but this technique is limited by penetration depth due to light scattering in tissue [74]. Many other methods exist for targeting activated macrophages in RA [75].
Table 1. Summary of the molecular targets and tracers used to identify inflammatory disease that are discussed in this review.
| Disease | Target | Tracer | Inflammatory Component | Source |
|---|---|---|---|---|
| Cardiovascular Disease | Glucose Metabolism | 18F-Flourodeoxyglucose (FDG) | Activated macrophage accumulation | [16][17][18][19] |
| Translocator protein (TSPO) receptors | 11C- PK11195 18F-GE-180 |
Overexpressed on activated macrophages | [26][27][28][29] | |
| Somatostatin receptor subtype-2 (SSR-2) | 68Ga-DOTATATE/ 64Cu-DOTATATE |
Overexpressed on activated macrophages | [31][32] | |
| Chemokine receptor 4 | 68Ga-pentixafor | Overexpressed on activated macrophages | [34][35][36] | |
| Hypoxia | 18F-fluoromisonidazole (FMISO | Activated macrophage accumulation → inflammation and thickening of the vessel wall → decreased oxygen diffusion efficiency → Hypoxia | [39] | |
| 18F-EF5 | [40] | |||
| Rheumatoid Arthritis | Glucose metabolism | 18F-Flourodeoxyglucose (FDG) | Activated macrophage accumulation | [66][67][68] |
| Folate receptor β (FRβ) | 18F-Fluoro-PEG-folate 111In-folate conjugate |
Overexpressed on activated macrophages within the synovial fluid | [69][71][72][73] | |
| NIR2-Folate | [74] | |||
| E-selectin | 111In-labeled anti-E-selectin MAb | Overexpressed on endothelial cells due to TNFα | [76] | |
| DyLight 750/anti-E-selectin Mab probe | [62] | |||
| 99mTc-labelled anti-E-selectin FAb | [77] | |||
| MMPs | 18F-pyriminde-2,4,6,-triones | Elevated levels in synovial fluid correlate with inflammatory response | [78] | |
| NIR fluorescent MMP-3 specific chitosan nanoparticle | [79] | |||
| CD20 | 124I-Rituximab 89Zr-Rituximab |
Overexpressed on B lymphocytes as they accumulate in synovial fluid | [80][81] | |
| TNFα | 99mTc-Infliximab | Overexpressed in synovial fluid | [82][83] | |
| L-selectin/P-selectin | NIR Fluorescent Polyanionic dendritic polyglycerol sulfate (dPGS) | Movement of immune cells to the inflammatory location | [84][85] | |
| COPD | Pulmonary perfusion | 99mTc-labeled macroaggregated albumin | Ventilation/Perfusion (V/Q) scintigraphy to regional inflammatory/airflow differences | [86][87] |
| Pulmonary ventilation | 81mKr or 133Xe 99mTc-labeled DTPA 99mTc-labeled carbon particles (Technegas) |
[87] | ||
| Glucose metabolism | 18F-Flourodeoxyglucose (FDG) | Activated macrophage accumulation | [88][89][90][91] | |
| Translocator protein (TSPO) receptors | 11C-PK11195 | Overexpressed on activated macrophages | [92] | |
| MMPs | 18F-IPFP | Produced by active macrophages at the inflammatory location | [93] | |
| 99mTc-labeled RP805 | [94] | |||
| Gastrointestinal | Glucose metabolism | 18F-Flourodeoxyglucose (FDG) | Activated macrophage accumulation | [95][96] |
| CXCL8 receptor | 99mTc-CXCL8 | Overexpression on activated neutrophils | [97] | |
| Interleukin 1 β | 89Zr-lα-IL-1β | Secreted by immune cells indicating an inflammatory response | [98] | |
| CD11b | 89Zr-α-CD11b | Pan-myeloid innate immune marker | [98] | |
| CD4 | 89Zr-GK1.5 cys diabody (cDb) | CD4 positive T-Cells characterize IBD inflammatory response | [99] | |
| EGFR | 64Cu-Cetuximab fragment-DOTA | Overexpression in inflammatory cells | [100] |
Due to the abundance of immune cells in the synovial fluid, there is an overexpression of inflammatory cytokines that elicit certain cellular responses that can then be targeted for imaging. The presence of interleukin-1 and tumor necrosis factor alpha (TNF-α) stimulate the transient expression of surface protein E-selectin on vascular endothelial cells and the overexpression of matrix metalloproteases (MMPs) in the synovial fluid. Anti E-selectin antibodies and MMP-targeted probes can be either radiolabeled or conjugated to an NIR dye and visualized through scintillation/PET or NIR fluorescence imaging [76][101][77][79][78]. Biologicals used as therapeutics for RA can also be radiolabeled and used to image RA. Rituximab, a monoclonal antibody that targets CD20, a cell surface marker that is expressed on most B cells, can be radiolabeled and used as a probe for the in vivo molecular imaging of RA based on B lymphocyte accumulation in the synovial fluid (Figure 2) [102][80][81]. Infliximab, a monoclonal antibody that targets tumor necrosis factor alpha (TNFα), has also been radiolabeled with 99mTc, which demonstrated a superior sensitivity to inflammation than MRI and clinical examinations in patients with RA [82][103][83].
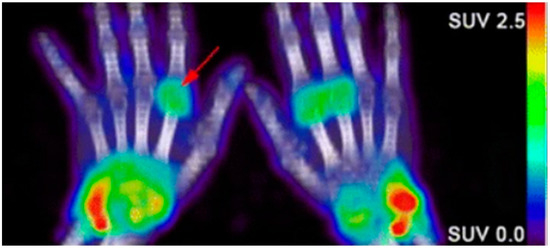
Figure 2. Confirmation of rheumatoid arthritis (RA) in the wrists/hands of patients using 89Zr-rituximab PET imaging to target B-cell accumulation [80].
Carbohydrate-binding proteins, L-selection and P-selection, are involved in the movement of immune cells before and during the inflammatory response [104]. Polyanionic dendritic polyglycerol sulfate (dPGS) targets inflammation through binding with these selectins. Conjugation with indocyanine green (ICG), an NIR fluorescent dye, allowed for the in vivo differentiation of RA-positive joints from RA negative joints in a preclinical rat arthritis model as seen by a 3.5-fold greater fluorescence imaging signal [84]. As the clinical translation of NIR fluorescence is limited by low penetration depth, multispectral optoacoustic tomography (MSOT) can overcome those limitations. MSOT imaging is based on a light-in, sound-out approach, having all the benefits of optical imaging but allowing for increased depth penetration, since photon scattering is irrelevant to acoustic waves [105]. Then, NIR-labeled dPGS can be imaged at much greater depths using MSOT [85].
2.3. Chronic Obstructive Pulmonary Disease (COPD)
Chronic obstructive pulmonary disease (COPD) is a preventable, but underdiagnosed inflammatory disease with an extremely high morbidity and mortality rate [106]. Approximately 90% of all COPD cases are related to smoking, yet only 20% of smokers will develop COPD, suggesting that other environmental and genetic factors must also play a role [107][108]. COPD is characterized by airway obstruction due to chronic inflammation and tissue damage caused by a decrease in alveolar elasticity and gas exchange, which ultimately leads to an irreversible decrease in lung function [109]. Pulmonary function testing (PFT) to measure airflow coupled with conventional imaging modalities, CT or MRI, to visualize morphological changes in the airway, is the current standard for diagnosing COPD [110]. Since COPD is an inflammatory disease, these imaging modalities must infer about the inflammatory state through surrogate biomarkers such as airway thickness and airway wall area [109]. Emphysema and chronic bronchitis are two subtypes of COPD that have very distinct molecular characteristics. Emphysema is an irreversible condition induced by smoking or inhaling irritants that destroys the alveoli; this leads to a decrease in the surface area of the lungs, making it difficult to obtain oxygen, causing inflammation of the lung parenchyma [111][112]. Chronic bronchitis is the persistent inflammation of the bronchial tubes due to a chronic cough, which leads to sputum build up in the airways, restricting airflow [113][114]. Early identification of COPD and proper differentiation of different phenotypes is imperative for the development of a proper treatment plan.
Molecular imaging techniques have been developed to target the inflammatory response of COPD. As the airways become inflamed, there will be changes in the pulmonary blood flow as well as airflow. These changes often precede morphological changes that can be detected by CT. Perfusion scintigraphy through the injection of 99mTc-labeled macroaggregated albumin coupled with ventilation scintigraphy through the inhalation of either an inert radioactive gas (81mKr or 133Xe), an aerosol-based 99mTc-labeled DTPA, or Technegas (99mTc-labeled carbon particles) will uncover aspects of the heterogeneity of the disease that cannot be seen using PFT or CT [86]. A great comparative study of these radiolabeled tracers in ventilation scintigraphy is found here [115]. The Ventilation to Perfusion (V/Q) ratio obtained will yield important information about regional differences in airflow and inflammation, where larger V/Q values indicate emphysema and lower values reflect chronic bronchitis [87][116]. Similarly, MRI using hyperpolarized noble gas (3He or 129Xe) can also be used to assess the ventilation status through imaging of the airspaces of the lungs rather than the tissue [117]. Apparent diffusion coefficient (ADC) maps of the hyperpolarized gas can be obtained on a voxel-wise basis using diffusion-weighted MRI (DWI-MRI), where high ADC values reflect areas of severe disease [118][119]. While hyperpolarized MRI is able to visualize the ventilation deficiencies associated with COPD, it is limited by spatial resolution and the ability of the patient to hold their breath.
As with other inflammatory diseases, COPD can also be visualized through immune cells. 18F-FDG PET/CT imaging is commonly used to monitor the metabolic activity of immune cells to diagnose and identify disease severity [88][89][90][91]. Since 18F-FDG is a non-specific biomarker of immune activity, the addition of 11C-PK11195, a macrophage-targeted radiotracer, allows for the non-specific visualization of neutrophil activity as well as the more specific visualization of macrophage accumulation. A study involving six patients with COPD and five control subjects saw a greater accumulation of 18F-FDG in all COPD patients compared to control, and greater 11C-PK11195 accumulation in four of six COPD patients compared to control [92]. Macrophages will secrete matrix metalloproteases (MMPs) and many other cytokines, which are all attractive options for the molecular imaging of COPD. Using a mouse model of COPD, a radiofluorinated probe, 18F-IPFP, was developed and tested to target MMP-9 and MMP-12; the accumulation of 18F-IPFP was 4× higher in the lungs of COPD mice than in normal mice [93]. 99mTc-labeled RP805 is another MMP targeted radiotracer that saw significantly greater accumulation in IL-13 transgenic mice than control mice using SPECT/CT (Figure 3) [94].
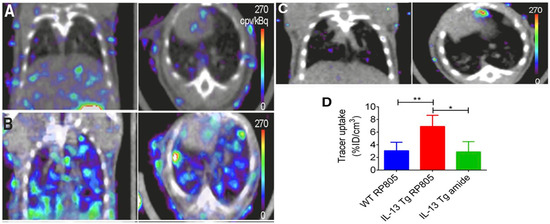
Figure 3. Coronal (left) and transversal (right) SPECT/CT imaging of matrix metalloproteases (MMPs). (A) Wild-type mice injected with 99mTc-labeled RP805 (B) IL-13 transgenic mice injected with 99mTc-labeled RP805 (C) IL-13 transgenic mice injected with an amide analog tracer as a control. (D) Quantification of uptake in SPECT images. * p < 0.01 ** p < 0.001 [94].
3. Gastrointestinal
Different gastrointestinal (GI) diseases can present with common, non-specific symptoms such as diarrhea and abdominal pain, making accurate diagnosis challenging without molecular information in addition to history and physical exam. [120][121]. Globally, the prevalence of inflammatory GI conditions such as inflammatory bowel disease (IBD) has increased significantly over time [122], particularly in developing countries [123]. Several causes, including genetic factors, diet, and infection, can result in inflammation of the GI tract. Identification of GI inflammation can aid in monitoring response to interventions. Subsequently, appropriate treatment can be administered to relieve symptoms or prevent disease progression. This can be especially critical in lowering patient risk for colorectal cancers [124].
Historically, tests using blood, stool, or biopsied tissue samples have been paired with invasive imaging techniques, such as endoscopy, to diagnose and assess patient GI disease [124]. Currently available invasive and non-invasive imaging techniques such as endoscopy, CT, MRI, and US, can show the macroscopic structural abnormalities associated with inflammatory bowel disease such as bowel wall thickening, abscesses, or fistulas to identify the scope of disease [125]. When combining multiple standard imaging modalities, the presence of inter-clinician reader variability and the lack of molecular information contained in the images (Figure 4) often requires a biopsy for an accurate diagnosis. In the context of Figure 4, the numerous lesions within the colon result in a higher potential of a biopsy sampling error and the possibility to miss areas of early-stage colon cancer.
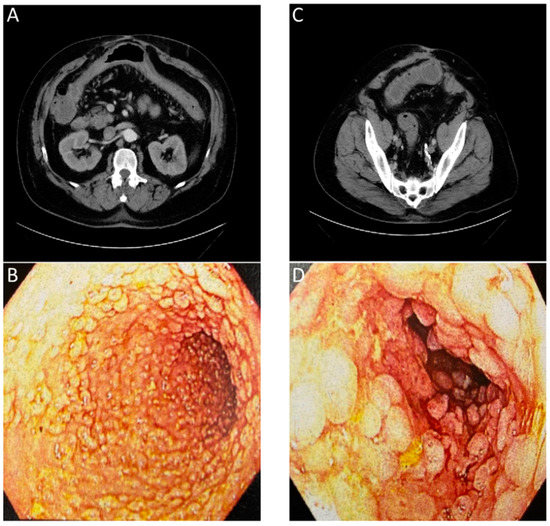
Figure 4. Images of a patient with history of chronic diarrhea that is occasionally bloody. CT ordered for unrelated reasons incidentally showed non-specific inflammation. Correlation with endoscopy showed substantial chronic inflammation. (A) CT with arrows showing inflammation of transverse colon. (B) Endoscopic images of transverse colon with diffuse pseudopolyps. (C) CT with arrows showing inflammation of sigmoid colon. (D) Endoscopic images of sigmoid colon with diffuse pseudopolyps. The lack of an inflammation or cancer specific contrast agent for the CT or endoscopic evaluation required a biopsy to confirm a lack of neoplasia.
PET imaging is currently the only clinically approved molecular imaging approach for GI inflammation [95][96]. Specifically, 18F-FDG PET is used to measure the extent and magnitude of GI inflammation, indicating areas of low or high inflammation based on metabolic differences throughout the GI tract. The high metabolic need of inflamed tissue alongside the increased presence and activity of immune cells, such as leukocytes, results in increased glucose metabolism at sites of inflammation [126]. Differences in 18F-FDG consumption highlight areas of increased inflammation while contrasting against normal healthy tissue. PET alone offers limited spatial resolution despite its potential for high contrast imaging. Additionally, the uptake of 18F-FDG occurs in off-target sites, resulting in high background signal. As such, PET is frequently paired with either CT or MRI imaging to better monitor disease status and accurately assess disease location, as shown in (Figure 5) [127][128][129].
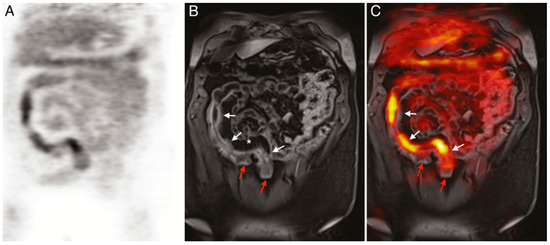
Figure 5. (A) 18F-FDG PET image of human patient with Crohn’s disease. (B) T1-weighted magnetic resonance imaging (MRI) image of the same patient. (C) Merged PET/MRI. White arrows indicate locations of acute inflammation while red arrows highlight damage resulting from earlier disease action. The asterisk (*) shows a site of proliferation of fibrofatty compounds in the mesentery. SUVmax of 18F-FDG 5.6–9.2 vs. SUVmax of background bowel 1.5–2.8 [128].
Current molecular imaging techniques prove mostly effective for verifying the extent and magnitude of GI inflammation. Preclinically, there has been investigation into the manipulation of contrast agents for the molecular imaging of GI inflammation. Wang et al. quantified inflammation in acute colitis mouse models using ultrasound with a P- and E-selectin targeted contrast agent and 18F-FDG-PET/CT. Similar results were obtained with both modalities [130]. P- and E-selectin are overexpressed on endothelial cells at sites of active inflammation, suggesting the future utility of this work in inflammatory GI disorders. While not practiced in the clinic at this time, immuno-PET techniques use radiolabeled proteins to target the upregulated immune cell presence or biochemical activity around inflamed tissues [97][98]. For example, antibody fragments targeting mouse CD4 cells, which are increasingly present at sites of GI inflammation, indicated the location and intensity of colorectal inflammation in mouse models [99]. Another modality undergoing preclinical assessment for the imaging of inflammation is multispectral optoacoustic tomography (MSOT). MSOT permits accurate, non-invasive imaging of the molecular characteristics of the disease through the visualization of exogenous or endogenous contrast agents [131][132]. Preclinical MSOT analysis has been shown to accurately detect in vivo colitis through measuring hypervascularity, which is common in inflamed tissue, and oxyhemoglobin levels in inoculated mouse models [133]. Alongside imaging modalities, new molecular targets are being investigated for improved diagnostic capabilities. α4β7 integrin is currently under investigation to determine whether it has the potential to increase the accuracy of IBD imaging. This is based on the increased presence of α4β7 integrin on the activated lymphocytes found in inflamed tissue [134][135]. Endothelial growth factor receptor (EGFR) may be another target for imaging given its overexpression in inflamed and malignant cells. One study demonstrated the ability of radiolabeled anti-EGFR antibody fragments to successfully detect sites of IBD in mouse models, presenting greater target specificity and signal intensity relative to 18F-FDG [100]. As new markers, probes, and imaging modalities are developed or found, accuracy in imaging diagnoses and tracking of GI inflammation is sure to improve.
References
- Tarkin, J.M.; Dweck, M.R.; Evans, N.R.; Takx, R.A.P.; Brown, A.J.; Tawakol, A.; Fayad, Z.A.; Rudd, J.H.F. Imaging Atherosclerosis. Circ. Res. 2016, 118, 750–769.
- Van Camp, G. Cardiovascular Disease Prevention. Acta Clin/Belg. 2014, 69, 407–411.
- Francula-Zaninovic, S.; Nola, I.A. Management of Measurable Variable Cardiovascular Disease’ Risk Factors. Curr. Cardiol. Rev. 2018, 14, 153–163.
- Jokinen, E. Obesity and Cardiovascular Disease. Minerva Pediatr. 2015, 67, 25–32.
- Kondo, T.; Nakano, Y.; Adachi, S.; Murohara, T. Effects of Tobacco Smoking on Cardiovascular Disease. Circ. J. 2019, 83, 1980–1985.
- Virani, S.S.; Alonso, A.; Benjamin, E.J.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Delling, F.N.; et al. Heart Disease and Stroke Statistics—2020 Update: A Report from the American Heart Association. AHA/ASA J. 2020.
- Frostegård, J. Immunity, Atherosclerosis and Cardiovascular Disease. BMC Med. 2013, 11, 117.
- Rosenbaum, D.; Millon, A.; Fayad, Z.A. Molecular Imaging in Atherosclerosis: FDG PET. Curr. Atheroscler. Rep. 2012, 14, 429–437.
- Syed, M.B.; Fletcher, A.J.; Forsythe, R.O.; Kaczynski, J.; Newby, D.E.; Dweck, M.R.; van Beek, E.J. Emerging Techniques in Atherosclerosis Imaging. Br. J. Radiol. 2019, 92, 20180309.
- Sandfort, V.; Lima Joao, A.C.; Bluemke, D.A. Noninvasive Imaging of Atherosclerotic Plaque Progression. Circ. Cardiovasc. Imaging 2015, 8, e003316.
- Detrano, R.; Guerci, A.D.; Carr, J.J.; Bild, D.E.; Burke, G.; Folsom, A.R.; Liu, K.; Shea, S.; Szklo, M.; Bluemke, D.A. Coronary Calcium as a Predictor of Coronary Events in Four Racial or Ethnic Groups. N. Engl. J. Med. 2008, 358, 1336–1345.
- Park, H.-B.; Heo, R.; Hartaigh, B.Ó.; Cho, I.; Gransar, H.; Nakazato, R.; Leipsic, J.; Mancini, G.B.J.; Koo, B.-K.; Otake, H.; et al. Atherosclerotic Plaque Characteristics by CT Angiography Identify Coronary Lesions That Cause Ischemia: A Direct Comparison to Fractional Flow Reserve. JACC Cardiovasc. Imaging 2015, 8, 1–10.
- Dunphy, M.P.; Freiman, A.; Larson, S.M.; Strauss, H.W. Association of Vascular 18F-FDG Uptake with Vascular Calcification. J. Nucl. Med. 2005, 46, 1278–1284.
- Moore, K.J.; Tabas, I. Macrophages in the Pathogenesis of Atherosclerosis. Cell 2011, 145, 341–355.
- Tarkin, J.M.; Joshi, F.R.; Rudd, J.H.F. PET Imaging of Inflammation in Atherosclerosis. Nat. Rev. Cardiol. 2014, 11, 443–457.
- Lederman, R.; Raylman, R.; Fisher, S.; Kison, P.; San, H.; Nabel, E.; Wahl, R. Detection of Atherosclerosis Using a Novel Positron-Sensitive Probe and 18-fluorodeoxyglucose (FDG). Nucl. Med. Commun. 2001, 22, 747–753.
- Skagen, K.; Johnsrud, K.; Evensen, K.; Scott, H.; Krohg-Sørensen, K.; Reier-Nilsen, F.; Revheim, M.E.; Fjeld, J.G.; Skjelland, M.; Russell, D. Carotid Plaque Inflammation Assessed with (18)F-FDG PET/CT Is Higher in Symptomatic Compared with Asymptomatic Patients. Int. J. Stroke 2015, 10, 730–736.
- Vallabhajosula, S.; Machac, J.; Knesaurek, K.; Telsey, J.; Lipszyc, H.; Bastidas, D.; Zhao, Q.; Buchsbaum, M. Imaging Atherosclerotic Macrophage Density by Positron Emission Tomography Using F-18-fluorodeoxyglucose (FDG). J. Nucl. Med. 1996, 37, 144.
- Yun, M.; Yeh, D.; Araujo, L.I.; Jang, S.; Newberg, A.; Alavi, A. F-18 FDG Uptake in the Large Arteries: A New Observation. Clin. Nucl. Med. 2001, 26, 314–319.
- Buck, A.K.; Reske, S.N. Cellular Origin and Molecular Mechanisms of 18F-FDG Uptake: Is There a Contribution of the Endothelium? J. Nucl. Med. 2004, 45, 461–463.
- Rudd, J.H.; Warburton, E.; Fryer, T.D.; Jones, H.; Clark, J.; Antoun, N.; Johnstrom, P.; Davenport, A.P.; Kirkpatrick, P.J.; Arch, B.N. Imaging Atherosclerotic Plaque Inflammation with [18F]-fluorodeoxyglucose Positron Emission Tomography. Circulation 2002, 105, 2708–2711.
- Evans, N.R.; Tarkin, J.M.; Chowdhury, M.M.; Warburton, E.A.; Rudd, J.H.F. PET Imaging of Atherosclerotic Disease: Advancing Plaque Assessment from Anatomy to Pathophysiology. Curr. Atheroscler. Rep. 2016, 18, 30.
- Mazurek, T.; Kobylecka, M.; Zielenkiewicz, M.; Kurek, A.; Kochman, J.; Filipiak, K.J.; Mazurek, K.; Huczek, Z.; Królicki, L.; Opolski, G. PET/CT Evaluation of 18 F-FDG Uptake in Pericoronary Adipose Tissue in Patients with Stable Coronary Artery Disease: Independent Predictor of Atherosclerotic Lesions’ Formation? J. Nucl. Cardiol. 2017, 24, 1075–1084.
- Gullberg, G.T.; Shrestha, U.M.; Seo, Y. PET Imaging of Glucose and Fatty Acid Metabolism for NAFLD Patients. J. Nucl. Cardiol. 2020, 27, 1689–1697.
- Wykrzykowska, J.; Lehman, S.; Williams, G.; Parker, J.A.; Palmer, M.R.; Varkey, S.; Kolodny, G.; Laham, R. Imaging of Inflamed and Vulnerable Plaque in Coronary Arteries with 18F-FDG PET/CT in Patients with Suppression of Myocardial Uptake Using a Low-Carbohydrate, High-Fat Preparation. J. Nucl. Med. 2009, 50, 563–568.
- Bird, J.L.E.; Izquierdo-Garcia, D.; Davies, J.R.; Rudd, J.H.F.; Probst, K.C.; Figg, N.; Clark, J.C.; Weissberg, P.L.; Davenport, A.P.; Warburton, E.A. Evaluation of Translocator Protein Quantification as a Tool for Characterising Macrophage Burden in Human Carotid Atherosclerosis. Atherosclerosis 2010, 210, 388–391.
- Fujimura, Y.; Hwang, P.M.; Iii, H.T.; Kozloff, L.; Imaizumi, M.; Innis, R.B.; Fujita, M. Increased Peripheral Benzodiazepine Receptors in Arterial Plaque of Patients with Atherosclerosis: An Autoradiographic Study with [(3)H]PK 11195. Atherosclerosis 2008, 201, 108–111.
- Gaemperli, O.; Shalhoub, J.; Owen, D.R.; Lamare, F.; Johansson, S.; Fouladi, N.; Davies, A.H.; Rimoldi, O.E.; Camici, P.G. Imaging Intraplaque Inflammation in Carotid Atherosclerosis with 11C-PK11195 Positron Emission Tomography/Computed Tomography. Eur. Heart J. 2012, 33, 1902–1910.
- Pugliese, F.; Gaemperli, O.; Kinderlerer, A.R.; Lamare, F.; Shalhoub, J.; Davies, A.H.; Rimoldi, O.E.; Mason, J.C.; Camici, P.G. Imaging of Vascular Inflammation With [11C]-PK11195 and Positron Emission Tomography/Computed Tomography Angiography. J. Am. Coll. Cardiol. 2010, 56, 653–661.
- Boutin, H.; Murray, K.; Pradillo, J.; Maroy, R.; Smigova, A.; Gerhard, A.; Jones, P.A.; Trigg, W. 18F-GE-180: A Novel TSPO Radiotracer Compared to 11C-R-PK11195 in a Preclinical Model of Stroke. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 503–511.
- Rinne, P.; Hellberg, S.; Kiugel, M.; Virta, J.; Li, X.-G.; Käkelä, M.; Helariutta, K.; Luoto, P.; Liljenbäck, H.; Hakovirta, H. Comparison of Somatostatin Receptor 2-targeting PET Tracers in the Detection of Mouse Atherosclerotic Plaques. Mol. Imaging Biol. 2016, 18, 99–108.
- Tarkin, J.M.; Joshi, F.R.; Evans, N.R.; Chowdhury, M.M.; Figg, N.L.; Shah, A.V.; Starks, L.T.; Martin-Garrido, A.; Manavaki, R.; Yu, E. Detection of Atherosclerotic Inflammation by 68Ga-DOTATATE PET Compared to [18F] FDG PET Imaging. J. Am. Coll. Cardiol. 2017, 69, 1774–1791.
- Pérez-Medina, C.; Fayad, Z.A.; Mulder, W.J.M. Atherosclerosis Immunoimaging by Positron Emission Tomography. Arter. Thromb. Vasc. Biol. 2020, 40, 865–873.
- Hyafil, F.; Pelisek, J.; Laitinen, I.; Schottelius, M.; Mohring, M.; Döring, Y.; van der Vorst, E.P.; Kallmayer, M.; Steiger, K.; Poschenrieder, A. Imaging the Cytokine Receptor CXCR4 in Atherosclerotic Plaques with the Radiotracer 68Ga-pentixafor for PET. J. Nucl. Med. 2017, 58, 499–506.
- Li, X.; Heber, D.; Leike, T.; Beitzke, D.; Lu, X.; Zhang, X.; Wei, Y.; Mitterhauser, M.; Wadsak, W.; Kropf, S.; et al. [68Ga]Pentixafor-PET/MRI for the Detection of Chemokine Receptor 4 Expression in Atherosclerotic Plaques. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 558–566.
- Weiberg, D.; Thackeray, J.T.; Daum, G.; Sohns, J.M.; Kropf, S.; Wester, H.-J.; Ross, T.L.; Bengel, F.M.; Derlin, T. Clinical Molecular Imaging of Chemokine Receptor CXCR4 Expression in Atherosclerotic Plaque Using 68Ga-pentixafor PET: Correlation with Cardiovascular Risk Factors and Calcified Plaque Burden. J. Nucl. Med. 2018, 59, 266–272.
- Marsch, E.; Sluimer, J.C.; Daemen, M.J. Hypoxia in Atherosclerosis and Inflammation. Curr. Opin. Lipidol. 2013, 24, 393–400.
- Joshi, F.R.; Manavaki, R.; Fryer, T.D.; Figg, N.L.; Sluimer, J.C.; Aigbirhio, F.I.; Davenport, A.P.; Kirkpatrick, P.J.; Warburton, E.A.; Rudd, J.H.F. Vascular Imaging with (18)F-Fluorodeoxyglucose Positron Emission Tomography Is Influenced by Hypoxia. J. Am. Coll. Cardiol. 2017, 69, 1873–1874.
- Mateo, J.; Izquierdo-Garcia, D.; Badimon, J.J.; Fayad, Z.A.; Fuster, V. Noninvasive Assessment of Hypoxia in Rabbit Advanced Atherosclerosis Using 18F-fluoromisonidazole Positron Emission Tomographic Imaging. Circ. Cardiovasc. Imaging 2014, 7, 312–320.
- Silvola, J.M.; Saraste, A.; Forsback, S.; Laine, V.J.; Saukko, P.; Heinonen, S.E.; Ylä-Herttuala, S.; Roivainen, A.; Knuuti, J. Detection of Hypoxia by [18F]EF5 in Atherosclerotic Plaques in Mice. Arter. Thromb. Vasc. Biol. 2011, 31, 1011–1015.
- Smolen, J.S.; Aletaha, D.; McInnes, I.B. Rheumatoid Arthritis. Lancet 2016, 388, 2023–2038.
- Aletaha, D.; Smolen, J.S. Diagnosis and Management of Rheumatoid Arthritis: A Review. JAMA 2018, 320, 1360–1372.
- Verheul, M.; Fearon, U.; Trouw, L.; Veale, D. Biomarkers for Rheumatoid and Psoriatic Arthritis. Clin. Immunol. 2015, 161, 2–10.
- Chang, K.; Yang, S.M.; Kim, S.H.; Han, K.H.; Park, S.J.; Shin, J.I. Smoking and Rheumatoid Arthritis. Int. J. Mol. Sci. 2014, 15, 22279–22295.
- Cheng, L.; Qian, L.; Xu, Z.-Z.; Tan, Y.; Luo, C.-Y. Aromatic Hydrocarbon Receptor Provides a Link between Smoking and Rheumatoid Arthritis in Peripheral Blood Mononuclear Cells. Clin. Exp. Rheumatol. 2019, 37, 445–449.
- Dar, L.; Tiosano, S.; Watad, A.; Bragazzi, N.L.; Zisman, D.; Comaneshter, D.; Cohen, A.; Amital, H. Are Obesity and Rheumatoid Arthritis Interrelated? Int. J. Clin. Pract. 2018, 72, e13045.
- Nikiphorou, E.; Norton, S.; Young, A.; Dixey, J.; Walsh, D.; Helliwell, H.; Kiely, P. Early Rheumatoid Arthritis Study and the Early Rheumatoid Arthritis Network. The Association of Obesity with Disease Activity, Functional Ability and Quality of Life in Early Rheumatoid Arthritis: Data from the Early Rheumatoid Arthritis Study/Early Rheumatoid Arthritis Network UK Prospective Cohorts. Rheumatology 2018, 57, 1194–1202.
- Tolusso, B.; Gigante, M.R.; Alivernini, S.; Petricca, L.; Fedele, A.L.; Di Mario, C.; Aquilanti, B.; Magurano, M.R.; Ferraccioli, G.; Gremese, E. Chemerin and PEDF Are Metaflammation-Related Biomarkers of Disease Activity and Obesity in Rheumatoid Arthritis. Front. Med. 2018, 5.
- Grygielska, J.; Raciborski, F.; Kłak, A.; Owoc, J. The Impact of Nutrition and Generally Available Products Such as Nicotine and Alcohol on Rheumatoid Arthritis—Review of the Literature. Reumatologia 2018, 56, 121.
- Hu, Y.; Sparks, J.A.; Malspeis, S.; Costenbader, K.H.; Hu, F.B.; Karlson, E.W.; Lu, B. Long-Term Dietary Quality and Risk of Developing Rheumatoid Arthritis in Women. Ann. Rheum. Dis. 2017, 76, 1357–1364.
- Lu, B.; Solomon, D.H.; Costenbader, K.H.; Karlson, E.W. Alcohol Consumption and Risk of Incident Rheumatoid Arthritis in Women: A Prospective Study. Arthritis Rheumatol. 2014, 66, 1998–2005.
- De Brito Rocha, S.; Baldo, D.C.; Andrade, L.E.C. Clinical and Pathophysiologic Relevance of Autoantibodies in Rheumatoid Arthritis. Adv. Rheumatol. 2019, 59, 2.
- Demoruelle, M.K.; Deane, K. Antibodies to Citrullinated Protein Antigens (ACPAs): Clinical and Pathophysiologic Significance. Curr. Rheumatol. Rep. 2011, 13, 421–430.
- Moeez, S.; John, P.; Bhatti, A. Anti-Citrullinated Protein Antibodies: Role in Pathogenesis of RA and Potential as a Diagnostic Tool. Rheumatol. Int. 2013, 33, 1669–1673.
- Ingegnoli, F.; Castelli, R.; Gualtierotti, R. Rheumatoid Factors: Clinical Applications. Dis. Markers 2013, 35, 726598.
- Aletaha, D.; Neogi, T.; Silman, A.J.; Funovits, J.; Felson, D.T.; Bingham, C.O., III; Birnbaum, N.S.; Burmester, G.R.; Bykerk, V.P.; Cohen, M.D.; et al. 2010 Rheumatoid Arthritis Classification Criteria: An American College of Rheumatology/European League Against Rheumatism Collaborative Initiative. Arthritis Rheum. 2010, 62, 2569–2581.
- Joshua, F.; Edmonds, J.; Lassere, M. Power Doppler Ultrasound in Musculoskeletal Disease: A Systematic Review. Semin. Arthritis Rheum. 2006, 36, 99–108.
- Shaloo Bhasin, P.P.C. The Role of Power Doppler Ultrasonography as Disease Activity Marker in Rheumatoid Arthritis. Dis. Markers 2015, 2015.
- Yamanishi, Y.; Firestein, G.S. Pathogenesis of Rheumatoid Arthritis: The Role of Synoviocytes. Rheum. Dis. Clin. N. Am. 2001, 27, 355–371.
- Ostrowska, M.; Maśliński, W.; Prochorec-Sobieszek, M.; Nieciecki, M.; Sudoł-Szopińska, I. Cartilage and Bone Damage in Rheumatoid Arthritis. Reumatologia 2018, 56, 111–120.
- Yoshitomi, H. Regulation of Immune Responses and Chronic Inflammation by Fibroblast-Like Synoviocytes. Front. Immunol. 2019, 10.
- Nehoff, H.; Parayath, N.N.; Domanovitch, L.; Taurin, S.; Greish, K. Nanomedicine for Drug Targeting: Strategies beyond the Enhanced Permeability and Retention Effect. Int. J. Nanomed. 2014, 9, 2539–2555.
- Neuwelt, A.; Sidhu, N.; Hu, C.-A.A.; Mlady, G.; Eberhardt, S.C.; Sillerud, L.O. Iron-Based Superparamagnetic Nanoparticle Contrast Agents for MRI of Infection and Inflammation. Am. J. Roentgenol. 2015, 204, W302–W313.
- Butoescu, N.; Seemayer, C.A.; Foti, M.; Jordan, O.; Doelker, E. Dexamethasone-Containing PLGA Superparamagnetic Microparticles as Carriers for the Local Treatment of Arthritis. Biomaterials 2009, 30, 1772–1780.
- Wu, L.; Shen, S. What Potential do Magnetic Iron Oxide Nanoparticles Have for the Treatment of Rheumatoid Arthritis? Nanomedicine 2019, 14, 927–930.
- Beckers, C.; Ribbens, C.; André, B.; Marcelis, S.; Kaye, O.; Mathy, L.; Kaiser, M.J.; Hustinx, R.; Foidart, J.; Malaise, M.G. Assessment of Disease Activity in Rheumatoid Arthritis with (18)F-FDG PET. J. Nucl. Med. 2004, 45, 956–964.
- Chaudhari, A.J.; Ferrero, A.; Godinez, F.; Yang, K.; Shelton, D.K.; Hunter, J.C.; Naguwa, S.M.; Boone, J.M.; Raychaudhuri, S.P.; Badawi, R.D. High-Resolution 18F-FDG PET/CT for Assessing Disease Activity in Rheumatoid and Psoriatic Arthritis: Findings of a Prospective Pilot Study. Br. J. Radiol. 2016, 89, 20160138.
- Kubota, K.; Yamashita, H.; Mimori, A. Clinical Value of FDG-PET/CT for the Evaluation of Rheumatic Diseases: Rheumatoid Arthritis, Polymyalgia Rheumatica, and Relapsing Polychondritis. Semin. Nucl. Med. 2017, 47, 408–424.
- Chandrupatla, D.; Molthoff, C.F.M.; Lammertsma, A.A.; van der Laken, C.J.; Jansen, G. The Folate Receptor β as a Macrophage-Mediated Imaging and Therapeutic Target in Rheumatoid Arthritis. Drug Deliv. Transl. Res. 2019, 9, 366–378.
- Xia, W.; Hilgenbrink, A.R.; Matteson, E.L.; Lockwood, M.B.; Cheng, J.-X.; Low, P.S. A Functional Folate Receptor Is Induced during Macrophage Activation and Can Be Used to Target Drugs to Activated Macrophages. Blood J. Am. Soc. Hematol. 2009, 113, 438–446.
- De Visser, H.M.; Korthagen, N.M.; Müller, C.; Ramakers, R.M.; Krijger, G.C.; Lafeber, F.P.J.G.; Beekman, F.J.; Mastbergen, S.C.; Weinans, H. Imaging of Folate Receptor Expressing Macrophages in the Rat Groove Model of Osteoarthritis: Using a New DOTA-Folate Conjugate. Cartilage 2018, 9, 183–191.
- Van Der Heijden, J.W.; Oerlemans, R.; Dijkmans, B.A.; Qi, H.; Laken, C.J.V.D.; Lems, W.F.; Jackman, A.L.; Kraan, M.C.; Tak, P.P.; Ratnam, M. Folate Receptor β as a Potential Delivery Route for Novel Folate Antagonists to Macrophages in the Synovial Tissue of Rheumatoid Arthritis Patients. Arthritis Rheum. 2009, 60, 12–21.
- Verweij, N.J.F.; Yaqub, M.; Bruijnen, S.T.G.; Pieplenbosch, S.; Ter Wee, M.M.; Jansen, G.; Chen, Q.; Low, P.S.; Windhorst, A.D.; Lammertsma, A.A.; et al. First in Man Study of [(18)F]fluoro-PEG-folate PET: A Novel Macrophage Imaging Technique to Visualize Rheumatoid Arthritis. Sci. Rep. 2020, 10, 1047.
- Chen, W.-T.; Mahmood, U.; Weissleder, R.; Tung, C.-H. Arthritis Imaging Using a Near-Infrared Fluorescence Folate-Targeted Probe. Arthritis Res. Ther. 2005, 7, R310–R317.
- Put, S.; Westhovens, R.; Lahoutte, T.; Matthys, P. Molecular Imaging of Rheumatoid Arthritis: Emerging Markers, Tools, and Techniques. Arthritis Res. Ther. 2014, 16, 208.
- Chapman, P.T.; Jamar, F.; Keelan, E.T.; Peters, A.M.; Haskard, D.O. Use of a Radiolabeled Monoclonal Antibody against E-Selectin for Imaging of Endothelial Activation in Rheumatoid Arthritis. Arthritis Rheum. 1996, 39, 1371–1375.
- Jamar, F.; Houssiau, F.; Devogelaer, J.P.; Chapman, P.; Haskard, D.; Beaujean, V.; Beckers, C.; Manicourt, D.H.; Peters, A. Scintigraphy Using a Technetium 99m-Labelled Anti-E-Selectin Fab Fragment in Rheumatoid Arthritis. Rheumatology 2002, 41, 53–61.
- Schrigten, D.; Breyholz, H.-J.; Wagner, S.; Hermann, S.; Schober, O.; Schäfers, M.; Haufe, G.; Kopka, K. A New Generation of Radiofluorinated Pyrimidine-2,4,6-triones as MMP-Targeted Radiotracers for Positron Emission Tomography. J. Med. Chem. 2012, 55, 223–232.
- Ryu, J.H.; Lee, A.; Chu, J.U.; Koo, H.; Ko, C.Y.; Kim, H.S.; Yoon, S.Y.; Kim, B.S.; Choi, K.; Kwon, I.C.; et al. Early Diagnosis of Arthritis in Mice with Collagen-Induced Arthritis, Using a Fluorogenic Matrix Metalloproteinase 3-Specific Polymeric Probe. Arthritis Rheum. 2011, 63, 3824–3832.
- Bruijnen, S.; Tsang-A-Sjoe, M.; Raterman, H.; Ramwadhdoebe, T.; Vugts, D.; van Dongen, G.; Huisman, M.; Hoekstra, O.; Tak, P.-P.; Voskuyl, A.; et al. B-Cell Imaging with Zirconium-89 Labelled Rituximab PET-CT at Baseline Is Associated with Therapeutic Response 24 Weeks after Initiation of Rituximab Treatment in Rheumatoid Arthritis Patients. Arthritis Res. Ther. 2016, 18, 266.
- Tran, L.; Huitema, A.D.; van Rijswijk, M.H.; Dinant, H.J.; Baars, J.W.; Beijnen, J.H.; Vogel, W.V. CD20 Antigen Imaging with 124I-rituximab PET/CT in Patients with Rheumatoid Arthritis. Hum. Antibodies 2011, 20, 29–35.
- Barrera, P.; Oyen, W.; Boerman, O.; Van Riel, P. Scintigraphic Detection of Tumour Necrosis Factor in Patients with Rheumatoid Arthritis. Ann. Rheum. Dis. 2003, 62, 825–828.
- Roimicher, L.; Lopes, F.P.; de Souza, S.A.; Mendes, L.F.; Domingues, R.C.; da Fonseca, L.M.; Gutfilen, B. 99mTc-anti-TNF-α Scintigraphy in RA: A Comparison Pilot Study with MRI and Clinical Examination. Rheumatology 2011, 50, 2044–2050.
- Licha, K.; Welker, P.; Weinhart, M.; Wegner, N.; Kern, S.; Reichert, S.; Gemeinhardt, I.; Weissbach, C.; Ebert, B.; Haag, R.; et al. Fluorescence Imaging with Multifunctional Polyglycerol Sulfates: Novel Polymeric near-IR Probes Targeting Inflammation. Bioconjug. Chem. 2011, 22, 2453–2460.
- Beziere, N.; von Schacky, C.; Kosanke, Y.; Kimm, M.; Nunes, A.; Licha, K.; Aichler, M.; Walch, A.; Rummeny, E.J.; Ntziachristos, V.; et al. Optoacoustic Imaging and Staging of Inflammation in a Murine Model of Arthritis. Arthritis Rheumatol. 2014, 66, 2071–2078.
- Mortensen, J.; Berg, R.M.G. Lung Scintigraphy in COPD. Semin. Nucl. Med. 2019, 49, 16–21.
- Brudin, L.H.; Rhodes, C.G.; Valind, S.O.; Buckingham, P.D.; Jones, T.; Hughes, J.M. Regional Structure-Function Correlations in Chronic Obstructive Lung Disease Measured with Positron Emission Tomography. Thorax 1992, 47, 914–921.
- Coello, C.; Fisk, M.; Mohan, D.; Wilson, F.J.; Brown, A.P.; Polkey, M.I.; Wilkinson, I.; Tal-Singer, R.; Murphy, P.S.; Cheriyan, J.; et al. Quantitative Analysis of Dynamic (18)F-FDG PET/CT for Measurement of Lung Inflammation. EJNMMI Res. 2017, 7, 47.
- Kothekar, E.; Borja, A.J.; Gerke, O.; Werner, T.J.; Alavi, A.; Revheim, M.-E. Assessing Respitatory Muscle Activity with (18)F-FDG-PET/CT in Patients with COPD. Am. J. Nucl. Med. Mol. Imaging 2019, 9, 309–315.
- Pournazari, K.; Jahangiri, P.; Al-zaghal, A.; Arani, L.; Mehdizadeh Seraj, S.; Werner, T.; Alavi, A.; Torigian, D. Feasibility of Using Global Lung FDG Uptake in COPD Patients on PET/CT to Assess the Degree of Pulmonary Inflammation in Relation to Emphysema Severity. J. Nucl. Med. 2018, 59, 517.
- Subramanian, D.R.; Jenkins, L.; Edgar, R.; Quraishi, N.; Stockley, R.A.; Parr, D.G. Assessment of Pulmonary Neutrophilic Inflammation in Emphysema by Quantitative Positron Emission Tomography. Am. J. Respir. Crit. Care Med. 2012, 186, 1125–1132.
- Jones, H.A.; Marino, P.S.; Shakur, B.H.; Morrell, N.W. In Vivo Assessment of Lung Inflammatory Cell Activity in Patients with COPD and Asthma. Eur. Respir. J. 2003, 21, 567–573.
- Kondo, N.; Temma, T.; Aita, K.; Shimochi, S.; Koshino, K.; Senda, M.; Iida, H. Development of Matrix Metalloproteinase-Targeted Probes for Lung Inflammation Detection with Positron Emission Tomography. Sci. Rep. 2018, 8, 1347.
- Golestani, R.; Razavian, M.; Ye, Y.; Zhang, J.; Jung, J.-J.; Toczek, J.; Gona, K.; Kim, H.-Y.; Elias, J.A.; Lee, C.G.; et al. Matrix Metalloproteinase-Targeted Imaging of Lung Inflammation and Remodeling. J. Nucl. Med. 2017, 58, 138–143.
- Brewer, S.; McPherson, M.; Fujiwara, D.; Turovskaya, O.; Ziring, D.; Chen, L.; Takedatsu, H.; Targan, S.R.; Wei, B.; Braun, J. Molecular Imaging of Murine Intestinal Inflammation with 2-deoxy-2-[18F]fluoro-D-glucose and Positron Emission Tomography. Gastroenterology 2008, 135, 744–755.
- Pio, B.S.; Byrne, F.R.; Aranda, R.; Boulay, G.; Spicher, K.; Song, M.H.; Birnbaumer, L.; Phelps, M.E.; Czernin, J.; Silverman, D.H. Noninvasive Quantification of Bowel Inflammation Through Positron Emission Tomography Imaging of 2-deoxy-2-[18F]fluoro-D-glucose-labeled White Blood Cells. Mol. Imaging Biol. 2003, 5, 271–277.
- Aarntzen, E.H.; Hermsen, R.; Drenth, J.P.; Boerman, O.C.; Oyen, W.J. 99mTc-CXCL8 SPECT to Monitor Disease Activity in Inflammatory Bowel Disease. J. Nucl. Med. 2016, 57, 398–403.
- Dmochowska, N.; Tieu, W.; Keller, M.D.; Wardill, H.R.; Mavrangelos, C.; Campaniello, M.A.; Takhar, P.; Hughes, P.A. Immuno-PET of Innate Immune Markers CD11b and IL-1beta Detects Inflammation in Murine Colitis. J. Nucl. Med. 2019, 60, 858–863.
- Freise, A.C.; Zettlitz, K.A.; Salazar, F.B.; Tavare, R.; Tsai, W.K.; Chatziioannou, A.F.; Rozengurt, N.; Braun, J.; Wu, A.M. Immuno-PET in Inflammatory Bowel Disease: Imaging CD4-Positive T Cells in a Murine Model of Colitis. J. Nucl. Med. 2018, 59, 980–985.
- Turker, N.S.; Heidari, P.; Kucherlapati, R.; Kucherlapati, M.; Mahmood, U. An EGFR Targeted PET Imaging Probe for the Detection of Colonic Adenocarcinomas in the Setting of Colitis. Theranostics 2014, 4, 893–903.
- Gompels, L.L.; Madden, L.; Lim, N.H.; Inglis, J.J.; McConnell, E.; Vincent, T.L.; Haskard, D.O.; Paleolog, E.M. In Vivo Fluorescence Imaging of E-Selectin: Quantitative Detection of Endothelial Activation in a Mouse Model of Arthritis. Arthritis Rheum. 2011, 63, 107–117.
- Malviya, G.; Anzola, K.L.; Podestà, E.; Laganà, B.; Del Mastro, C.; Dierckx, R.A.; Scopinaro, F.; Signore, A. (99m)Tc-Labeled Rituximab for Imaging B Lymphocyte Infiltration in Inflammatory Autoimmune Disease Patients. Mol. Imaging Biol. 2012, 14, 637–646.
- Maini, R.N.; Feldmann, M. How Does Infliximab Work in Rheumatoid Arthritis? Arthritis Res. Ther. 2002, 4, S22–S28.
- Barthel, S.R.; Gavino, J.D.; Descheny, L.; Dimitroff, C.J. Targeting Selectins and Selectin Ligands in Inflammation and Cancer. Expert Opin. Ther. Targets 2007, 11, 1473–1491.
- MacCuaig, W.M.; Jones, M.A.; Abeyakoon, O.; McNally, L.R. Development of Multispectral Optoacoustic Tomography as a Clinically Translatable Modality for Cancer Imaging. Radiol. Imaging Cancer 2020, 2, e200066.
- Criner, R.N.; Han, M.K. COPD Care in the 21st Century: A Public Health Priority. Respir. Care 2018, 63, 591–600.
- Athanazio, R. Airway Disease: Similarities and Differences between Asthma, COPD and Bronchiectasis. Clinics 2012, 67, 1335–1343.
- Rovina, N.; Koutsoukou, A.; Koulouris, N.G. Inflammation and Immune Response in COPD: Where Do We Stand? Mediat. Inflamm. 2013, 2013, 413735.
- Myc, L.A.; Shim, Y.M.; Laubach, V.E.; Dimastromatteo, J. Role of Medical and Molecular Imaging in COPD. Clin. Transl. Med. 2019, 8, 12.
- Sverzellati, N.; Molinari, F.; Pirronti, T.; Bonomo, L.; Spagnolo, P.; Zompatori, M. New Insights on COPD Imaging via CT and MRI. Int. J. Chron. Obstruct. Pulmon. Dis. 2007, 2, 301–312.
- Shah, P.L.; Herth, F.J.; van Geffen, W.H.; Deslee, G.; Slebos, D.-J. Lung Volume Reduction for Emphysema. Lancet Respir. Med. 2017, 5, 147–156.
- Sharafkhaneh, A.; Hanania, N.A.; Kim, V. Pathogenesis of Emphysema: From the Bench to the Bedside. Proc. Am. Thorac. Soc. 2008, 5, 475–477.
- Izquierdo-Alonso, J.L.; Rodriguez-Gonzálezmoro, J.M.; de Lucas-Ramos, P.; Unzueta, I.; Ribera, X.; Antón, E.; Martín, A. Prevalence and Characteristics of Three Clinical phenotypes of Chronic Obstructive Pulmonary Disease (COPD). Respir. Med. 2013, 107, 724–731.
- Lahousse, L.; Seys, L.J.M.; Joos, G.F.; Franco, O.H.; Stricker, B.H.; Brusselle, G.G. Epidemiology and Impact of Chronic Bronchitis in Chronic obstructive Pulmonary Disease. Eur. Respir. J. 2017, 50, 1602470.
- Magnant, J.; Vecellio, L.; de Monte, M.; Grimbert, D.; Valat, C.; Boissinot, E.; Guilloteau, D.; Lemarié, E.; Diot, P. Comparative Analysis of Different Scintigraphic Approaches to Assess Pulmonary Ventilation. J. Aerosol Med. 2006, 19, 148–159.
- Milne, S.; King, G.G. Advanced Imaging in COPD: Insights into Pulmonary Pathophysiology. J. Thorac. Dis. 2014, 6, 1570–1585.
- Roos, J.E.; McAdams, H.P.; Kaushik, S.S.; Driehuys, B. Hyperpolarized Gas MR Imaging: Technique and Applications. Magn. Reson. Imaging Clin. N. Am. 2015, 23, 217–229.
- Fain, S.B.; Panth, S.R.; Evans, M.D.; Wentland, A.L.; Holmes, J.H.; Korosec, F.R.; O’Brien, M.J.; Fountaine, H.; Grist, T.M. Early Emphysematous Changes in Asymptomatic Smokers: Detection with 3He MR Imaging. Radiology 2006, 239, 875–883.
- Tafti, S.; Garrison, W.J.; Mugler, J.P.; Shim, Y.M.; Altes, T.A.; Mata, J.F.; de Lange, E.E.; Cates, G.D.; Ropp, A.M.; Wang, C.; et al. Emphysema Index Based on Hyperpolarized 3He or 129Xe Diffusion MRI: Performance and Comparison with Quantitative CT and Pulmonary Function Tests. Radiology 2020, 297, 201–210.
- Ady, J.; Fong, Y. Imaging for Infection: From Visualization of Inflammation to Visualization of Microbes. Surg. Infect. (Larchmt) 2014, 15, 700–707.
- Frickenstein, A.N.; Jones, M.A.; Behkam, B.; McNally, L.R. Imaging Inflammation and Infection in the Gastrointestinal Tract. Int. J. Mol. Sci. 2019, 21, 243.
- Alatab, S.; Sepanlou, S.G.; Ikuta, K.; Vahedi, H.; Bisignano, C.; Safiri, S.; Sadeghi, A.; Nixon, M.R.; Abdoli, A.; Abolhassani, H.; et al. The Global, Regional, and National Burden of Inflammatory Bowel Disease in 195 Countries and Territories, 1990–2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 2020, 5, 17–30.
- Molodecky, N.A.; Kaplan, G.G. Environmental Risk Factors for Inflammatory Bowel Disease. Gastroenterol. Hepatol. (NY) 2010, 6, 339–346.
- Mattar, M.C.; Lough, D.; Pishvaian, M.J.; Charabaty, A. Current Management of Inflammatory Bowel Disease and Colorectal Cancer. Gastrointest. Cancer Res. 2011, 4, 53–61.
- Kilcoyne, A.; Kaplan, J.L.; Gee, M.S. Inflammatory Bowel Disease Imaging: Current Practice and Future Directions. World J. Gastroenterol. 2016, 22, 917–932.
- Gotthardt, M.; Bleeker-Rovers, C.P.; Boerman, O.C.; Oyen, W.J. Imaging of Inflammation by PET, Conventional Scintigraphy, and Other Imaging Techniques. J. Nucl. Med. 2010, 51, 1937–1949.
- Catalano, O.A.; Wu, V.; Mahmood, U.; Signore, A.; Vangel, M.; Soricelli, A.; Salvatore, M.; Gervais, D.; Rosen, B.R. Diagnostic Performance of PET/MR in the Evaluation of Active Inflammation in Crohn Disease. Am. J. Nucl. Med. Mol. Imaging 2018, 8, 62–69.
- Le Fur, M.; Zhou, I.Y.; Catalano, O.; Caravan, P. Toward Molecular Imaging of Intestinal Pathology. Inflamm. Bowel Dis. 2020, 26, 1470–1484.
- Perlman, S.B.; Hall, B.S.; Reichelderfer, M. PET/CT Imaging of Inflammatory Bowel Disease. Semin. Nucl. Med. 2013, 43, 420–426.
- Wang, H.; Machtaler, S.; Bettinger, T.; Lutz, A.M.; Luong, R.; Bussat, P.; Gambhir, S.S.; Tranquart, F.; Tian, L.; Willmann, J.K. Molecular Imaging of Inflammation in Inflammatory Bowel Disease with a Clinically Translatable Dual-Selectin-Targeted US Contrast Agent: Comparison with FDG PET/CT in a Mouse Model. Radiology 2013, 267, 818–829.
- Beziere, N.; Ntziachristos, V. Optoacoustic Imaging: An Emerging Modality for the Gastrointestinal Tract. Gastroenterology 2011, 141, 1979–1985.
- McNally, L.R.; Mezera, M.; Morgan, D.E.; Frederick, P.J.; Yang, E.S.; Eltoum, I.E.; Grizzle, W.E. Current and Emerging Clinical Applications of Multispectral Optoacoustic Tomography (MSOT) in Oncology. Clin. Cancer Res. 2016, 22, 3432–3439.
- Bhutiani, N.; Grizzle, W.E.; Galandiuk, S.; Otali, D.; Dryden, G.W.; Egilmez, N.K.; McNally, L.R. Noninvasive Imaging of Colitis Using Multispectral Optoacoustic Tomography. J. Nucl. Med. 2017, 58, 1009–1012.
- Dearling, J.L.; Park, E.J.; Dunning, P.; Baker, A.; Fahey, F.; Treves, S.T.; Soriano, S.G.; Shimaoka, M.; Packard, A.B.; Peer, D. Detection of Intestinal Inflammation by MicroPET Imaging Using a (64)Cu-Labeled Anti-beta(7) Integrin Antibody. Inflamm. Bowel Dis. 2010, 16, 1458–1466.
- Rath, T.; Bojarski, C.; Neurath, M.F.; Atreya, R. Molecular Imaging of Mucosal α4β7 Integrin Expression with the Fluorescent Anti-Adhesion Antibody Vedolizumab in Crohn’s Disease. Gastrointest. Endosc. 2017, 86, 406–408.





