
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Savvas Georgiades | + 2587 word(s) | 2587 | 2021-02-25 06:40:57 | | | |
| 2 | Camila Xu | Meta information modification | 2587 | 2021-03-03 08:46:04 | | |
Video Upload Options
G-quadruplexes, a family of (thermodynamically and kinetically stable) tetraplex helices, are non-canonical secondary structures derived from guanine (G)-rich sequences of nucleic acids. G-quadruplexes were found to occur in functionally-important regions of the human genome, including the telomere tandem sequences, several proto-oncogene promoters and other regulatory regions, ribosomal DNA (rDNA), as well as mRNA sequences encoding for proteins with roles in tumorigenesis, thus establishing a clear connection between G-quadruplexes and known hallmarks of cancer. Stabilization of G-quadruplexes belonging to the above categories, by means of small-molecule intervention, has been correlated with a range of anticancer effects, which has led to classifying G-quadruplexes as novel potential targets in anticancer research. The most common ways in which G-quadruplexes are now understood to serve in an anticancer capacity are presented herein.
1. Introduction
G-quadruplexes, a family of tetraplex helices, are non-canonical secondary structures derived from guanine (G)-rich sequences of nucleic acids and exhibiting remarkable thermodynamic and kinetic stability [1]. While G-quadruplexes form readily in vitro from single nucleic acid strands, their assembly and stabilization in vivo, where they may exist in equilibrium with a different type of structure (e.g., double-stranded DNA), has been suggested to require the function of protein chaperons [2].
The following organization is characteristic of a G-quadruplex assembly: Guanines from the participating sequence(s), in sets of four, are oriented in square planar quartets, driven by a network of Hoogsteen hydrogen bonds (Figure 1A); G-quartet stability is further enhanced by coordination of (monovalent) cations to guanine carbonyls (Figure 1A); and G-quartets accumulate atop each other due to π-π stacking, while interconnected by the sugar-phosphodiester backbone (Figure 1B,C) [3][4][5].
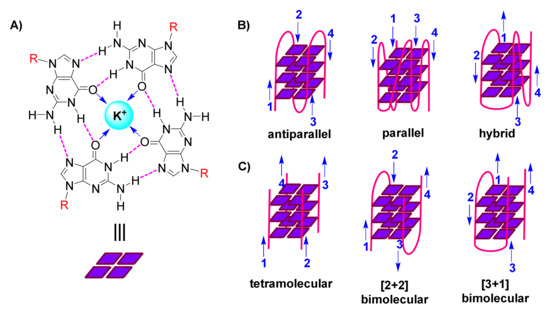
Figure 1. (A). Representation of a guanine(G)-quartet, highlighting the network of Hoogsteen hydrogen bonds (magenta), monovalent cation (cyan), dipole-cation interactions (blue arrows), and sites of connection to the sugar-phosphodiester backbone (R, red) (B). Cartoon representations of diverse unimolecular/intramolecular G-quadruplexes, with blue arrows indicating direction of each strand (numbered) (C). Cartoon representations of diverse intermolecular G-quadruplexes, with blue arrows indicating direction of each strand (numbered).
G-quadruplexes are polymorphic entities, as revealed by 3D structural studies, with their family comprising both unimolecular/intramolecular (Figure 1B) and intermolecular (Figure 1C) structures. These exhibit diversity in the lengths, sequences, folds and orientations of the loops that interconnect the participating strands, leading to classification of G-quadruplexes as parallel, antiparallel or hybrid (Figure 1B) [6][7][8].
Since the early days of this field, in attempting to answer the question whether G-quadruplexes are biologically relevant, algorithms have been devised and applied by various research teams, in order to predict possibility of occurrence of G-quadruplexes in the human and other genomes [9][10][11][12]. Genome-wide analyses have indicated a frequent occurrence of G-quadruplex-forming sequences in functional genomic regions, suggesting G-quadruplex association with telomere maintenance, replication, transcription and translation which, in turn, has led to suggestions of G-quadruplex-mediated regulatory mechanisms for these processes. The roles of G-quadruplexes in these processes are understood in much detail today [13].
Many of the >370,000 predicted G-quadruplex-forming sequences in humans [9][10] are traced in promoter regions of genes, close to transcription start sites [12]. Despite the fact that these predominantly exist in vivo in the form of double-stranded helices, their transient conversion to single-stranded is believed possible, in the course of replication, transcription and recombination. It can be achieved with the assistance of negative DNA supercoiling and conditions of molecular crowding, caused by protein binding, which favor folding into G-quadruplexes [14]. Moreover, the presence of tandem G-rich repeats in the human telomere [15][16], which is naturally single-stranded, energetically favors formation of multiple G-quadruplexes. On the other hand, RNAs containing G-quadruplex-forming motifs in their 5’-untranslated regions (5’-UTRs), estimated to be around 3000 in humans [17], are also single-stranded and readily fold into stable G-quadruplex structures.
Most G-quadruplex-related studies have been conducted ex vivo. However, accumulating experimental evidence is now providing proof of the in vivo occurrence of G-quadruplexes. An early study employing high-specificity antibodies against telomeric G-quadruplexes, raised by ribosome display, has achieved targeting of intermolecular antiparallel G-quadruplexes in the ciliate model organism Stylonychia [18]. More recent studies involving highly specific antibodies, have achieved visualization of G-quadruplexes in living human cancer cells [19][20] and tissues [21]. Also, over the last few years, there has been significant progress in the development of G-quadruplex-specific, small-molecule-based fluorescent probes and theranostics [22][23][24][25][26][27], which now find application as bioimaging agents to trace G-quadruplexes in a cellular context and expand our understanding on their functional roles in physiological processes, including those with consequences for cancer research.
The presence of G-quadruplex-forming motifs in key genomic DNA and RNA sequences, uniquely places them in position to regulate several cellular pathways. Importantly, many of these pathways are directly associated to well-established hallmarks of cancer [28]. Indicatively, G-quadruplexes have been correlated to chromosomal homeostasis, genome maintenance and integrity, apoptosis and survival, proto-oncogene and cancer protein expression and post-translational modifications [13]. G-quadruplex-forming sequences are often found amplified in certain cancers [29][30]. The realization of a strong link between G-quadruplexes and unprecedented anticancer mechanisms of action has leveraged G-quadruplex structures to therapeutic target status in oncology [31][32][33]. The physiological relevance and significance of G-quadruplexes in the context of cancer have been widely reviewed [34][35][36].
The putative roles of G-quadruplexes in prevention of cancer pathogenesis have been, for years, a major inspiration and drive for research efforts by many teams, with implications from a pharmacological perspective, for the design of small-molecule ligands targeting G-quadruplexes and aiming to induce G-quadruplex-mediated anticancer effects. A vast number of scaffolds have been proposed and new compounds designed and synthesized to address the task at hand, namely the binding (with high affinity and selectivity) and stabilization of G-quadruplexes in nucleic acid sequences of cancer relevance [36][37][38][39][40][41][42]. Cellular responses upon treatment of cells with G-quadruplex-targeting ligands have been correlated with the perceived function of these G-quadruplexes. In parallel, several methodologies for ascertaining the anticancer potential of G-quadruplex-stabilizing ligands have been described [43].
2. G-Quadruplex-Mediated Anticancer Mechanisms
2.1. Interference with Chromosomal Homeostasis & Telomerase-Mediated Telomere Elongation
The telomere is a region of repetitive nucleotide sequences at chromosomal ends which, via complexation with various nucleoproteins, folds into higher-order secondary structures, that play the role of a ‘cap’, protecting the chromosome from deterioration or fusion with other chromosomes [48]. The type of ‘cap’ secondary structure and the participating proteins exhibit variability between different species [2][49]. The existence of an intact ‘cap’ also prevents improper activation of DNA damage-response pathways [50].
G-quadruplexes occur in high concentrations in telomeres [19][51], due to the high guanine content of the telomere tandem sequence (TTAGGG in vertebrates) and are, in fact, capable of protecting genome integrity in cases where normal telomeric ‘caps’ are compromised [52]. In vitro studies have shown telomeric G-quadruplexes to interact with human proteins TRF2, EWS and FUS, which can co-bind the long non-coding RNA TERRA [53][54][55]. The simultaneous binding of telomeric and TERRA G-quadruplexes causes recruitment of histone methyltransferases by FUS, thus providing an association with telomere heterochromatin maintenance [54].
Stabilization of G-quadruplexes in the telomere during DNA replication could generate problems. Loss of telomeric G-quadruplex-interacting proteins, such as the CST cluster [56] and RTEL1 helicase [57], results in telomere shortening and fragility, and affords altered rates of replication [58]. The addition of G-quadruplex-stabilizing ligands was shown to exacerbate this situation [56].
Importantly, telomerase, a reverse transcriptase that is over-expressed in about 85% of cancer cells [59], stem cells and germline cells, is responsible for providing genomic stability by elongating the protruding 3’ single-stranded G-rich overhang at the ends of telomeres. For this extension to be permitted, base pairing needs to take place between the G-rich overhang and a RNA template carried by telomerase to encode the telomeric repeat sequence [60]. Elongation, which counteracts telomere shortening, may be inhibited by the formation of G-quadruplexes in telomeric sequences [61]. This is a result of hindered access of telomerase to the telomere sequence, caused by formation of antiparallel intramolecular G-quadruplexes (Figure 2A). However, alternative intermolecular parallel G-quadruplexes may also form, which can be partially resolved by telomerase in vitro, allowing the extension to proceed [62]. Evidence from Saccharomyces cerevisiae indicates a co-localization of parallel G-quadruplexes in the telomere with telomerase [62][63]. On the other hand, the POT1-TPP1 protein complex, responsible for recruitment of telomerase to the telomeres, is capable of destabilizing G-quadruplexes [64][65]. Recent evidence shows the importance of G-quadruplex formation in a POT1-TPP1 mediated DNA synthesis [66]. Finally, telomerase activity may be affected by the 5’ end unfolding of its RNA component, caused by a small molecule [67].
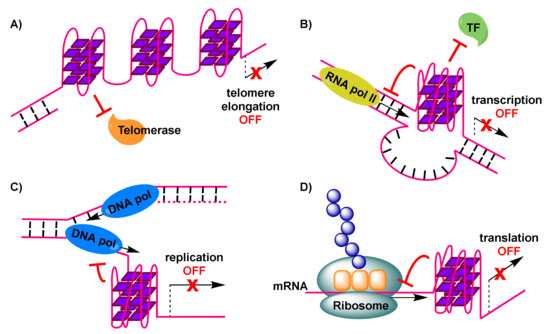
Figure 2. Formation of G-quadruplexes impacting physiological processes, with anticancer consequences: (A) G-quadruplexes in the telomere impose hindrance to telomerase, preventing elongation of the telomere and triggering DNA damage response signals. (B) G-quadruplex in oncogene-promoter region dislocates transcription factors and down-regulates RNA polymerase-mediated transcription of (onco)genes. (C) G-quadruplex in DNA undergoing replication stalls replication fork progression and leads to replicative stress, resulting in double strand breakpoints. (D) G-quadruplex in mRNA interferes with translation and formation of cancer proteins.
A vast number of ligands to stabilize telomeric G-quadruplexes in cancer cells have been described, despite the natural role of G-quadruplexes in telomerase-mediated telomere elongation not being fully elucidated. A resulting inhibition of telomerase activity upon addition of such ligands has been reported [68], while several ligands are able to displace members of the telomere protection complex shelterin, resulting in telomere damage and cell death [59][69]. While an alternative path of telomere elongation may be promoted upon G-quadruplex-imposed replication stress in certain cancer cells [70], presence of a G-quadruplex-stabilizing ligand may still result in cell death [71].
2.2. Transcriptional Regulation of Proto-Oncogene Promoters
The early detection, by means of applying computational predictive algorithms [12], of G-quadruplex-forming motifs in the promoter regions of several known proto-oncogenes [72], has indicated that G-quadruplexes are over-represented in these regions and may, in fact, possess regulatory roles with regard to the expression levels of oncogenes. Additional efforts have been successful in mapping G-quadruplex structures in chromatin to regulatory regions found adjacent to the transcription start sites of several of these genes in humans [30][73].
A number of in vitro studies applying small-molecule G-quadruplex-targeted ligands as agents inducing stabilization of G-quadruplexes in proto-oncogene promoter regions have demonstrated an ensuing reduction in oncogene transcription levels. Examples include transcriptional regulation of MYC, KRAS, KIT, BCL2 and VEGF [72][74][75][76][77]. However, explicit evidence of a link between G-quadruplexes and transcriptional control, coming from cellular studies, remains quite limited [78].
Indirect evidence of G-quadruplex impact on transcription of oncogenes is provided by the fact that certain transcription factors recognize G-quadruplex structures in vitro. Examples include recombinant nucleolin recognizing MYC [79], CNBP recognizing MYC [80] and SP1 recognizing ΚΙΤ [81]. This has led to the hypothesis that G-quadruplex-mediated mechanisms may be employed by nature for transcriptional regulation purposes.
To explain reduced expression levels of oncogenes, it has been suggested that G-quadruplex formation may impair initiation of transcription by preventing binding of RNA polymerase II and transcriptional machinery to the promoter transcription start site (Figure 2B) [74].
The formation of a G-quadruplex in the human telomerase reverse transcriptase gene (hTERT) has also been suggested to prevent binding of the gene repressor CCCTC binding factor, leading, in this case, to elevation of plasmid-encoded hTERT transcription [82].
2.3. Ribosomal DNA (rDNA) Transcription Inhibition
Ribosomal DNA (rDNA) is a GC-rich DNA sequence located in the nucleolus of cells, which encodes for ribosomal RNA. It contains more than 400 copies of the rRNA genes, organized in tandem arrays.
Ribosome biogenesis is under the control of multiple cellular signaling pathways, converging on the RNA polymerase I complex. RNA polymerase I is responsible for the transcription of rRNA genes and production of pre-rRNAs which, after maturation, will provide the main components for construction of the ribosome.
In cancer cells, proto-oncogene ‘gain-of-function’ and tumor-suppressor ‘loss-of-function’ mutations operate, leading to deregulated cellular signaling pathways, which in turn results in excessive ribosome biogenesis, required to support the rapid cell proliferation in tumors [83][84][85][86]. Given that the synthesis of rRNA by RNA polymerase I is considered the rate-limiting step in ribosome biogenesis [87], the interaction of rDNA with the RNA polymerase I protein complex could be a locus for anticancer intervention. Disruption of this interaction leads to arrest of ribosome biogenesis.
G-quadruplexes are believed to have a role in rDNA transcription. Specifically, G-quadruplexes may form transiently in the non-template strand in the course of rDNA transcription, and their occurrence prevents renaturation of the template DNA, assisting toward a dense arrangement of RNA polymerase I molecules on rRNA genes [88]. The formation of G-quadruplexes appears to be associated with their nanomolar-affinity interaction with nucleolin [89], an abundant nucleolar protein whose presence is essential for the progression of rDNA transcription [90]. Therefore, the disruption of G-quadruplex-nucleolin association, by means of interference with small-molecule ligands, is a way of inhibiting RNA polymerase I-mediated rDNA transcription, leading up to ribosome biogenesis suppression and eventually apoptosis of cancer cells.
2.4. Induction of Replication Stress Causing Genome Instability
Formation of G-quadruplexes in DNA sites during the transient opening of the double helix in the course of replication, has been implicated in increasing replication stress [91]. This is the result of obstruction caused to the progression of the replication forks (Figure 2C), leading to replication-fork collapse [92][93] and eventually the generation of double-strand breakpoints that cause genome instability and pose a threat to cell viability.
Via use of computational analyses of cancer databases, G-quadruplex formation has been associated with breakpoints in many cases in cancer cells, relevant to somatic copy-number alterations [94]. Stable G-quadruplexes were also found to be enriched in sites of somatic mutations, suggesting they may have roles as important determinants of mutagenesis [95]. G-quadruplex sequencing in the human genome has also revealed correlations of G-quadruplexes with gene amplifications, observed in cancer cells [29][30].
Evidence of genome instability due to G-quadruplex formation in the course of replication comes from elaborate studies in the model organisms Caenorhabditis elegans, Saccharomyces cerevisiae and Xenopus laevis, where the knock-out of a rescue system, namely helicases with the ability to resolve G-quadruplexes (such as DOG1, FANCJ and PIF1), renders the system prone to occurrence of DNA breakpoints [96][97][98][99][100][101]. These findings highlight the importance of helicases in cellular rescue mechanisms, as well as the relation between potential helicase ‘loss-of-function’ and genome instability.
2.5. Interference with Translation of Messenger RNA (mRNA) to Cancer Proteins
The bioinformatics discovery that G-quadruplex-forming motifs are prevalent in 5’-UTRs of RNAs [15], confirmed by spectroscopic studies on these sequences, has rendered such mRNA transcripts that encode for proteins with functional roles in cancer, attractive targets. The 5’-UTRs of mRNAs are located adjacent to translation initiation sites. Therefore, the formation of G-quadruplexes in 5’-UTRs of mRNA sequences (Figure 2D) may result in interference with mRNA translation [102] (e.g., potential formation of the ribosome at alternative, upstream start codons, thus preventing translation of the main open reading frame [103]), eventually depriving cancer cells of valuable proteins. An early prototype example, of interest to anticancer research, is the 5’-UTR of NRAS mRNA, where emergence of a G-quadruplex has been correlated with about 80% repression in protein levels in vitro, based on a luciferase reporter assay [17]. Many subsequent efforts, including studies in live cells, have identified additional G-quadruplex-forming sites in 5’-UTRs of the same and other mRNAs, which can be manipulated, by means of stabilization by appropriate small-molecule ligands, to achieve similar impact on translation.
G-rich sequences within mRNA coding regions are also encountered, however, at lower abundance compared to 5’-UTRs [104]. Upon G-quadruplex formation, they exhibit ability to stall translation, 6-7 nucleotides before the G-quadruplex [105].
The above findings, in addition to the identification of helicases capable of unwinding RNA G-quadruplexes [103], supports the notion that RNA G-quadruplexes may serve as a natural mechanism of regulating the expression levels of specific genes on a post-transcriptional level.
Small-molecule-based tools that offer the ability to modulate the stability of G-quadruplexes of this type, in a dose- and time-dependent manner, can be pharmacologically useful, especially given the single-stranded nature of mRNAs, which makes them more susceptible to modulation compared to dsDNAs.
References
- Lane, A.N.; Chaires, J.B.; Gray, R.D.; Trent, J.O. Stability and kinetics of G-quadruplex structures. Nucleic Acids Res. 2008, 36, 5482–5515, doi:10.1093/nar/gkn517.
- Paeschke, K.; Simonsson, T.; Postberg, J.; Rhodes, D.; Lipps, H.J. Telomere end-binding proteins control the formation of G-quadruplex DNA structures in vivo. Nat. Struct. Mol. Biol. 2005, 12, 847–854, doi:10.1038/nsmb982.
- Sen, D.; Gilbert, W. Formation of parallel four-stranded complexes by guanine-rich motifs in DNA and its implications for meiosis. Nature 1988, 334, 364–366, doi:10.1038/334364a0.
- Sundquist, W.I.; Klug, A. Telomeric DNA dimerizes by formation of guanine tetrads between hairpin loops. Nature 1989, 342, 825–829, doi:10.1038/342825a0.
- Williamson, J.R.; Raghuraman, M.; Cech, T.R. Monovalent cation-induced structure of telomeric DNA: The G-quartet model. Cell 1989, 59, 871–880, doi:10.1016/0092-8674(89)90610-7.
- Patel, D.J.; Phan, A.T.; Kuryavyi, V. Human telomere, oncogenic promoter and 5′-UTR G-quadruplexes: Diverse higher or-der DNA and RNA targets for cancer therapeutics. Nucleic Acids Res. 2007, 35, 7429–7455, doi:10.1093/nar/gkm711.
- Burge, S.; Parkinson, G.N.; Hazel, P.; Todd, A.K.; Neidle, S. Quadruplex DNA: Sequence, topology and structure. Nucleic Ac-ids Res. 2006, 34, 5402–5415, doi:10.1093/nar/gkl655.
- Simonsson, T. G-quadruplex DNA structures variations on a theme. Biol. Chem. 2001, 382, 621–628, doi:10.1515/bc.2001.073.
- Huppert, J.L.; Balasubramanian, S. Prevalence of quadruplexes in the human genome. Nucleic Acids Res. 2005, 33, 2908–2916.
- Todd, A.K.; Johnston, M.; Neidle, S. Highly prevalent putative quadruplex sequence motifs in human DNA. Nucleic Acids Res. 2005, 33, 2901–2907, doi:10.1093/nar/gki553.
- Eddy, J.; Maizels, N. Gene function correlates with potential for G4 DNA formation in the human genome. Nucleic Acids Res. 2006, 34, 3887–3896, doi:10.1093/nar/gkl529.
- Huppert, J.L.; Balasubramanian, S. G-quadruplexes in promoters throughout the human genome. Nucleic Acids Res. 2006, 35, 406–413, doi:10.1093/nar/gkl1057.
- Varshney, D.; Spiegel, J.; Zyner, K.; Tannahill, D.; Balasubramanian, S. The regulation and functions of DNA and RNA G-quadruplexes. Nat. Rev. Mol. Cell Biol. 2020, 21, 459–474, doi:10.1038/s41580-020-0236-x.
- Lipps, H.J.; Rhodes, D. G-quadruplex structures: In vivo evidence and function. Trends Cell Biol. 2009, 19, 414–422, doi:10.1016/j.tcb.2009.05.002.
- Huppert, J. Hunting G-quadruplexes. Biochimie 2008, 90, 1140–1148, doi:10.1016/j.biochi.2008.01.014.
- Maizels, N. Dynamic roles for G4 DNA in the biology of eukaryotic cells. Nat. Struct. Mol. Biol. 2006, 13, 1055–1059, doi:10.1038/nsmb1171.
- Kumari, S.; Bugaut, A.; Huppert, J.L.; Balasubramanian, S. An RNA G-quadruplex in the 5′ UTR of the NRAS proto-oncogene modulates translation. Nat. Chem. Biol. 2007, 3, 218–221, doi:10.1038/nchembio864.
- Schaffitzel, C.; Berger, I.; Postberg, J.; Hanes, J.; Lipps, H.J.; Plückthun, A. In vitro generated antibodies specific for telomeric guanine-quadruplex DNA react with Stylonychia lemnae macronuclei. Proc. Natl. Acad. Sci. USA 2001, 98, 8572–8577, doi:10.1073/pnas.141229498.
- Biffi, G.; Tannahill, D.; McCafferty, J.; Balasubramanian, S. Quantitative visualization of DNA G-quadruplex structures in human cells. Nat. Chem. 2013, 5, 182–186, doi:10.1038/nchem.1548.
- Henderson, A.; Wu, Y.; Huang, Y.C.; Chavez, E.A.; Platt, J.; Johnson, F.B.; Brosh, R.M.; Sen, D.; Lansdorp, P.M. Detection of G-quadruplex DNA in mammalian cells. Nucleic Acids Res. 2014, 42, 860–869, doi:10.1093/nar/gkt957.
- Biffi, G.; Tannahill, D.; Miller, J.; Howat, W.J.; Balasubramanian, S. Elevated levels of G-quadruplex formation in human stomach and liver cancer tissues. PLoS ONE 2014, 9, e102711, doi:10.1371/journal.pone.0102711.
- Chilka, P.; Desai, N.; Datta, B. Small molecule fluorescent probes for G- quadruplex visualization as potential cancer theranostic agents. Molecules 2019, 24, 752, doi:10.3390/molecules24040752.
- Lu, Y.-J.; Hu, D.-P.; Zhang, K.; Wong, W.-L.; Chow, C.-F. New pyridinium-based fluorescent dyes: A comparison of sym-metry and side-group effects on G-Quadruplex DNA binding selectivity and application in live cell imaging. Biosens. Bioe-lectron. 2016, 81, 373–381, doi:10.1016/j.bios.2016.03.012.
- Zhang, S.; Sun, H.; Wang, L.; Liu, Y.; Chen, H.; Li, Q.; Guan, A.; Liu, M.; Tang, Y. Real-time monitoring of DNA G-quadruplexes in living cells with a small-molecule fluorescent probe. Nucleic Acids Res. 2018, 46, 7522–7532, doi:10.1093/nar/gky665.
- Laguerre, A.; Desbois, N.; Stefan, L.; Richard, P.; Gros, C.P.; Monchaud, D. Porphyrin-based design of bioinspired multitar-get quadruplex ligands. ChemMedChem 2014, 9, 2035–2039, doi:10.1002/cmdc.201300526.
- Laguerre, A.; Hukezalie, K.; Winckler, P.; Katranji, F.; Chanteloup, G.; Pirrotta, M.; Perrier-Cornet, J.-M.; Wong, J.M.Y.; Monchaud, D. Visualization of RNA-quadruplexes in live cells. J. Am. Chem. Soc. 2015, 137, 8521–8525, doi:10.1021/jacs.5b03413.
- Shivalingam, A.; Izquierdo, M.A.; Le Marois, A.; Vyšniauskas, A.; Suhling, K.; Kuimova, M.K.; Vilar, R. The interactions between a small molecule and G-quadruplexes are visualized by fluorescence lifetime imaging microscopy. Nat. Commun. 2015, 6, 8178, doi:10.1038/ncomms9178.
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674, doi:10.1016/j.cell.2011.02.013.
- Chambers, V.S.; Marsico, G.; Boutell, J.M.; Di Antonio, M.; Smith, G.P.; Balasubramanian, S. High-throughput sequencing of DNA G-quadruplex structures in the human genome. Nat. Biotechnol. 2015, 33, 877–881, doi:10.1038/nbt.3295.
- Hänsel-Hertsch, R.; Beraldi, D.; Lensing, S.V.; Marsico, G.; Zyner, K.; Parry, A.; Di Antonio, M.; Pike, J.; Kimura, H.; Narita, M.; et al. G-quadruplex structures mark human regulatory chromatin. Nat. Genet. 2016, 48, 1267–1272, doi:10.1038/ng.3662.
- Han, H.; Hurley, L. G-quadruplex DNA: A potential target for anti-cancer drug design. Trends Pharmacol. Sci. 2000, 21, 136–142, doi:10.1016/s0165-6147(00)01457-7.
- Murat, P.; Balasubramanian, S. Existence and consequences of G-quadruplex structures in DNA. Curr. Opin. Genet. Dev. 2014, 25, 22–29, doi:10.1016/j.gde.2013.10.012.
- Cimino-Reale, G.; Zaffaroni, N.; Folini, M. Emerging role of G-quadruplex DNA as target in anticancer therapy. Curr. Pharm. Des. 2017, 22, 6612–6624, doi:10.2174/1381612822666160831101031.
- Ou, T.-M.; Lu, Y.-J.; Tan, J.-H.; Huang, Z.-S.; Wong, K.-Y.; Gu, L.-Q. G-quadruplexes: Targets in anticancer drug design. ChemMedChem 2008, 3, 690–713, doi:10.1002/cmdc.200700300.
- Balasubramanian, S.; Hurley, L.H.; Neidle, S. Targeting G-quadruplexes in gene promoters: A novel anticancer strategy? Nat. Rev. Drug Discov. 2011, 10, 261–275, doi:10.1038/nrd3428.
- Carvalho, J.; Mergny, J.-L.; Salgado, G.F.; Queiroz, J.A.; Cruz, C. G-quadruplex, friend or foe? The role of G-quartet in anti-cancer strategies. Trends Mol. Med. 2020, 26, 848–861.
- Monchaud, D.; Teulade-Fichou, M.-P. A hitchhiker’s guide to G-quadruplex ligands. Org. Biomol. Chem. 2007, 6, 627–636, doi:10.1039/b714772b.
- Monchaud, D.; Granzhan, A.; Saettel, N.; Guédin, A.; Mergny, J.; Teulade-Fichou, M.-P. “One ring to bind them all”—Part I: The efficiency of the macrocyclic scaffold for G-quadruplex DNA recognition. J. Nucleic Acids 2010, 2010, 1–19, doi:10.4061/2010/525862.
- Georgiades, S.N.; Karim, N.H.A.; Suntharalingam, K.; Vilar, R. Interaction of metal complexes with G-quadruplex DNA. Angew. Chem. Int. Ed. 2010, 49, 4020–4034, doi:10.1002/anie.200906363.
- Sun, Z.-Y.; Wang, X.-N.; Cheng, S.-Q.; Su, X.-X.; Ou, T.-M. Developing novel G-quadruplex ligands: From interaction with nucleic acids to interfering with nucleic acid–protein interaction. Molecules 2019, 24, 396, doi:10.3390/molecules24030396.
- Asamitsu, S.; Obata, S.; Yu, Z.; Bando, T.; Sugiyama, H. Recent progress of targeted G-quadruplex-preferred ligands toward cancer therapy. Molecules 2019, 24, 429, doi:10.3390/molecules24030429.
- Asamitsu, S.; Bando, T.; Sugiyama, H. Ligand design to acquire specificity to intended G-quadruplex structures. Chem. Eur. J. 2019, 25, 417–430, doi:10.1002/chem.201802691.
- Dakal, T.C. Methodologies for ascertaining the anti-cancer potential of DNA G-quadruplex stabilizers. J. Bioinform. Genom. Proteom. 2018, 3, 1028.
- Arachchilage, G.M.; Kharel, P.; Reid, J.; Basu, S. Targeting of G-quadruplex harboring pre-miRNA 92b by LNA rescues PTEN expression in NSCL cancer cells. ACS Chem. Biol. 2018, 13, 909–914.
- Rosenberg, J.E.; Bambury, R.; Drabkin, H.A.; Lara, P.N., Jr.; Harzstark, A.L.; Figlin, R.A.; Smith, G.W.; Choueiri, T.; Erlands-son, F.; Laber, D.A. A phase II trial of the nucleolin-targeted DNA aptamer AS1411 in metastatic refractory renal cell carci-noma. Invest. New Drugs 2014, 32, 178–187.
- García-Recio, E.M.; Pinto-Díez, C.; Pérez-Morgado, M.I.; García-Hernández, M.; Fernández, G.; Martín, M.E.; González, V.M. Characterization of MNK1b DNA aptamers that inhibit proliferation in MDA-MB231 breast cancer cells. Mol. Ther. Nucleic Acids 2016, 5, e275, doi:10.1038/mtna.2015.50.
- Pecoraro, A.; Virgilio, A.; Esposito, V.; Galeone, A.; Russo, G.; Russo, A. uL3 mediated nucleolar stress pathway as a new mechanism of action of antiproliferative G-quadruplex TBA derivatives in colon cancer cells. Biomolecules 2020, 10, 583, doi:10.3390/biom10040583.
- Shay, J.W.; Wright, W.E. Telomeres and telomerase: Three decades of progress. Nat. Rev. Genet. 2019, 20, 299–309, doi:10.1038/s41576-019-0099-1.
- De Lange, T. T-loops and the origin of telomeres. Nat. Rev. Mol. Cell Biol. 2004, 5, 323–329.
- De Lange, T. Shelterin: The protein complex that shapes and safeguards human telomeres. Genes Dev. 2005, 19, 2100–2110.
- Liu, H.-Y.; Zhao, Q.; Zhang, T.-P.; Wu, Y.; Xiong, Y.-X.; Wang, S.-K.; Ge, Y.-L.; He, J.-H.; Lv, P.; Ou, T.-M.; et al. Conformation selective antibody enables genome profiling and leads to discovery of parallel G-quadruplex in human telomeres. Cell Chem. Biol. 2016, 23, 1261–1270, doi:10.1016/j.chembiol.2016.08.013.
- Smith, J.S.; Chen, Q.; Yatsunyk, L.A.; Nicoludis, J.M.; Garcia, M.S.; Kranaster, R.; Balasubramanian, S.; Monchaud, D.; Teu-lade-Fichou, M.-P.; Abramowitz, L.; et al. Rudimentary G-quadruplex- based telomere capping in Saccharomyces cerevisiae. Nat. Struct. Mol. Biol. 2011, 18, 478–485.
- Biffi, G.; Tannahill, D.; Balasubramanian, S. An intramolecular G-quadruplex structure is required for binding of telomeric repeat-containing RNA to the telomeric protein TRF2. J. Am. Chem. Soc. 2012, 134, 11974–11976, doi:10.1021/ja305734x.
- Takahama, K.; Takada, A.; Tada, S.; Shimizu, M.; Sayama, K.; Kurokawa, R.; Oyoshi, T. Regulation of telomere length by G-quadruplex telomere DNA- and TERRA-binding protein TLS/FUS. Chem. Biol. 2013, 20, 341–350, doi:10.1016/j.chembiol.2013.02.013.
- Takahama, K.; Kino, K.; Arai, S.; Kurokawa, R.; Oyoshi, T. Identification of Ewing’s sarcoma protein as a G-quadruplex DNA- and RNA-binding protein. FEBS J. 2011, 278, 988–998, doi:10.1111/j.1742-4658.2011.08020.x.
- Zhang, M.; Wang, B.; Li, T.; Liu, R.; Xiao, Y.; Geng, X.; Li, G.; Liu, Q.; Price, C.M.; Liu, Y.; et al. Mammalian CST averts repli-cation failure by preventing G-quadruplex accumulation. Nucleic Acids Res. 2019, 47, 5243–5259, doi:10.1093/nar/gkz264.
- Vannier, J.-B.; Pavicic-Kaltenbrunner, V.; Petalcorin, M.I.; Ding, H.; Boulton, S.J. RTEL1 dismantles T Loops and counteracts telomeric G4-DNA to maintain telomere integrity. Cell 2012, 149, 795–806, doi:10.1016/j.cell.2012.03.030.
- Sfeir, A.; Kosiyatrakul, S.T.; Hockemeyer, D.; MacRae, S.L.; Karlseder, J.; Schildkraut, C.L.; de Lange, T. Mammalian telo-meres resemble fragile sites and require TRF1 for efficient replication. Cell 2009, 138, 90–103.
- Neidle, S. Quadruplex nucleic acids as novel therapeutic targets. J. Med. Chem. 2016, 59, 5987–6011, doi:10.1021/acs.jmedchem.5b01835.
- Cristofari, G.; Lingner, J.; de Lange, T.; Lundblad, V.; Blackburn, E. (Eds.) Cold Spring Harbor Laboratory Press: New York, NY, USA, 2006.
- Zahler, A.M.; Williamson, J.R.; Cech, T.R.; Prescott, D.M. Inhibition of telomerase by G-quartet DNA structures. Nature 1991, 350, 718–720.
- Moye, A.L.; Porter, K.C.; Cohen, S.B.; Phan, T.; Zyner, K.G.; Sasaki, N.; Lovrecz, G.O.; Beck, J.L.; Bryan, T.M. Telomeric G-quadruplexes are a substrate and site of localization for human telomerase. Nat. Commun. 2015, 6, 7643, doi:10.1038/ncomms8643.
- Zhang, M.-L.; Tong, X.-J.; Fu, X.-H.; Zhou, B.O.; Wang, J.; Liao, X.-H.; Li, Q.-J.; Shen, N.; Ding, J.; Zhou, J.-Q. Yeast telomerase subunit Est1p has guanine quadruplex-promoting activity that is required for telomere elongation. Nat. Struct. Mol. Biol. 2010, 17, 202–209, doi:10.1038/nsmb.1760.
- Zaug, A.J.; Podell, E.R.; Cech, T.R. Human POT1 disrupts telomeric G-quadruplexes allowing telomerase extension in vitro. Proc. Natl. Acad. Sci. USA 2005, 102, 10864–10869, doi:10.1073/pnas.0504744102.
- Hwang, H.; Buncher, N.; Opresko, P.L.; Myong, S. POT1-TPP1 regulates telomeric overhang structural dynamics. Structure 2012, 20, 1872–1880, doi:10.1016/j.str.2012.08.018.
- Jansson, L.I.; Hentschel, J.; Parks, J.W.; Chang, T.R.; Lu, C.; Baral, R.; Bagshaw, C.R.; Stone, M.D. Telomere DNA G-quadruplex folding within actively extending human telomerase. Proc. Natl. Acad. Sci. USA 2019, 116, 9350–9359, doi:10.1073/pnas.1814777116.
- Booy, E.P.; Meier, M.; Okun, N.; Novakowski, S.K.; Xiong, S.; Stetefeld, J.; McKenna, S.A. The RNA helicase RHAU (DHX36) unwinds a G4-quadruplex in human telomerase RNA and promotes the formation of the P1 helix template boundary. Nu-cleic Acids Res. 2012, 40, 4110–4124, doi:10.1093/nar/gkr1306.
- Sun, D.; Thompson, B.; Cathers, B.E.; Salazar, M.; Kerwin, S.M.; Trent, J.O.; Jenkins, T.C.; Neidle, S.; Hurley, L.H. Inhibition of human telomerase by a G-quadruplex-interactive compound. J. Med. Chem. 1997, 40, 2113–2116, doi:10.1021/jm970199z.
- Neidle, S. Human telomeric G-quadruplex: The current status of telomeric G-quadruplexes as therapeutic targets in human cancer. FEBS J. 2009, 277, 1118–1125, doi:10.1111/j.1742-4658.2009.07463.x.
- Clynes, D.; Jelinska, C.; Xella, B.; Ayyub, H.; Scott, C.; Mitson, M.; Taylor, S.S.; Higgs, D.R.; Gibbons, R.J. Suppression of the alternative lengthening of telomere pathway by the chromatin remodelling factor ATRX. Nat. Commun. 2015, 6, 7538, doi:10.1038/ncomms8538.
- Gowan, S.M.; Heald, R.; Stevens, M.F.G.; Kelland, L.R. Potent inhibition of telomerase by small-molecule pentacyclic acri-dines capable of interacting with G-quadruplexes. Mol. Pharmacol. 2001, 60, 981–988, doi:10.1124/mol.60.5.981.
- Simonsson, T.; Kubista, M.; Pecinka, P. DNA tetraplex formation in the control region of c-myc. Nucleic Acids Res. 1998, 26, 1167–1172, doi:10.1093/nar/26.5.1167.
- Kouzine, F.; Wojtowicz, D.; Baranello, L.; Yamane, A.; Nelson, S.; Resch, W.; Kieffer-Kwon, K.-R.; Benham, C.J.; Casellas, R.; Przytycka, T.M.; et al. Permanganate/S1 nuclease footprinting reveals Non-B DNA structures with regulatory potential across a mammalian genome. Cell Syst. 2017, 4, 344–356.e7, doi:10.1016/j.cels.2017.01.013.
- Rigo, R.; Palumbo, M.; Sissi, C. G-quadruplexes in human promoters: A challenge for therapeutic applications. Biochim. Bio-phys. Acta Gen. Subj. 2017, 1861, 1399–1413, doi:10.1016/j.bbagen.2016.12.024.
- Siddiqui-Jain, A.; Grand, C.L.; Bearss, D.J.; Hurley, L.H. Direct evidence for a G-quadruplex in a promoter region and its targeting with a small molecule to repress c-MYC transcription. Proc. Natl. Acad. Sci. USA 2002, 99, 11593–11598, doi:10.1073/pnas.182256799.
- Cogoi, S.; Xodo, L.E. G-quadruplex formation within the promoter of the KRAS proto-oncogene and its effect on transcrip-tion. Nucleic Acids Res. 2006, 34, 2536–2549, doi:10.1093/nar/gkl286.
- Bejugam, M.; Sewitz, S.; Shirude, P.S.; Rodriguez, R.; Shahid, R.; Balasubramanian, S. Trisubstituted isoalloxazines as a new class of G-quadruplex binding ligands: Small molecule regulation of c-kit oncogene expression. J. Am. Chem. Soc. 2007, 129, 12926–12927, doi:10.1021/ja075881p.
- Wu, G.; Xing, Z.; Tran, E.J.; Yang, D. DDX5 helicase resolves G-quadruplex and is involved in MYC gene transcriptional ac-tivation. Proc. Natl. Acad. Sci. USA 2019, 116, 20453–20461, doi:10.1073/pnas.1909047116.
- González, V.; Guo, K.; Hurley, L.; Sun, D. Identification and characterization of nucleolin as a c-myc G-quadruplex-binding protein. J. Biol. Chem. 2009, 284, 23622–23635, doi:10.1074/jbc.m109.018028.
- Borgognone, M.; Armas, P.; Calcaterra, N.B. Cellular nucleic-acid-binding protein, a transcriptional enhancer of c-Myc, promotes the formation of parallel G-quadruplexes. Biochem. J. 2010, 428, 491–498, doi:10.1042/bj20100038.
- Raiber, E.-A.; Kranaster, R.; Lam, E.; Nikan, M.; Balasubramanian, S. A non-canonical DNA structure is a binding motif for the transcription factor SP1 in vitro. Nucleic Acids Res. 2011, 40, 1499–1508, doi:10.1093/nar/gkr882.
- Li, P.-T.; Wang, Z.-F.; Chu, I.-T.; Kuan, Y.-M.; Li, M.-H.; Huang, M.-C.; Chiang, P.-C.; Chang, T.-C.; Chen, C.-T. Expression of the human telomerase reverse transcriptase gene is modulated by quadruplex formation in its first exon due to DNA meth-ylation. J. Biol. Chem. 2017, 292, 20859–20870, doi:10.1074/jbc.m117.808022.
- Arabi, A.; Wu, S.; Ridderstråle, K.; Bierhoff, H.; Shiue, C.; Fatyol, K.; Fahlén, S.; Hydbring, P.; Söderberg, O.; Grummt, I.; et al. c-Myc associates with ribosomal DNA and activates RNA polymerase I transcription. Nat. Cell Biol. 2005, 7, 303–310, doi:10.1038/ncb1225.
- Zhai, W.; Comai, L. Repression of RNA polymerase I transcription by the tumor suppressor p53. Mol. Cell. Biol. 2000, 20, 5930–5938, doi:10.1128/MCB.20.16.5930-5938.2000.
- Hannan, K.M.; Hannan, R.D.; Smith, S.D.; Jefferson, L.S.; Lun, M.; Rothblum, L.I. Rb and p130 regulate RNA polymerase I transcription: Rb disrupts the interaction between UBF and SL-1. Oncogene 2000, 19, 4988–4999, doi:10.1038/sj.onc.1203875.
- Zhang, C.; Comai, L.; Johnson, D.L. PTEN represses RNA polymerase I transcription by disrupting the SL1 complex. Mol. Cell. Biol. 2005, 25, 6899–6911, doi:10.1128/mcb.25.16.6899-6911.2005.
- Chédin, S.; Laferté, A.; Hoang, T.; Lafontaine, D.L.J.; Riva, M.; Carles, C. Is ribosome synthesis controlled by pol I transcrip-tion? Cell Cycle 2007, 6, 11–15.
- French, S.L.; Osheim, Y.N.; Cioci, F.; Nomura, M.; Beyer, A.L. In exponentially growing Saccharomyces cerevisiae cells, rRNA synthesis is determined by the summed RNA polymerase I loading rate rather than by the number of active genes. Mol. Cell. Biol. 2003, 23, 1558–1568, doi:10.1128/mcb.23.5.1558-1568.2003.
- Hanakahi, L.A.; Sun, H.; Maizels, N. High affinity interactions of nucleolin with G-G-paired rDNA. J. Biol. Chem. 1999, 274, 15908–15912, doi:10.1074/jbc.274.22.15908.
- Rickards, B.; Flint, S.J.; Cole, M.D.; Leroy, G. Nucleolin is required for RNA polymerase I transcription in vivo. Mol. Cell. Biol. 2006, 27, 937–948, doi:10.1128/MCB.01584-06.
- Valton, A.-L.; Prioleau, M.-N. G-quadruplexes in DNA replication: A problem or a necessity? Trends Genet. 2016, 32, 697–706, doi:10.1016/j.tig.2016.09.004.
- Puget, N.; Miller, K.M.; Legube, G. Non-canonical DNA/RNA structures during transcription-coupled double-strand break repair: Roadblocks or Bona fide repair intermediates? DNA Repair 2019, 81, 102661, doi:10.1016/j.dnarep.2019.102661.
- Técher, H.; Koundrioukoff, S.; Nicolas, A.; Debatisse, M. The impact of replication stress on replication dynamics and DNA damage in vertebrate cells. Nat. Rev. Genet. 2017, 18, 535–550, doi:10.1038/nrg.2017.46.
- De, S.; Michor, F. DNA secondary structures and epigenetic determinants of cancer genome evolution. Nat. Struct. Mol. Biol. 2011, 18, 950–955, doi:10.1038/nsmb.2089.
- Georgakopoulos-Soares, I.; Morganella, S.; Jain, N.; Hemberg, M.; Nik-Zainal, S. Non-canonical secondary structures arising from non-B-DNA motifs are determinants of mutagenesis. Genome Res. 2018, 28, 1264–1271.
- Kruisselbrink, E.; Guryev, V.; Brouwer, K.; Pontier, D.B.; Cuppen, E.; Tijsterman, M. Mutagenic capacity of endogenous G4 DNA underlies genome instability in FANCJ-defective C. elegans. Curr. Biol. 2008, 18, 900–905, doi:10.1016/j.cub.2008.05.013.
- London, T.B.C.; Barber, L.J.; Mosedale, G.; Kelly, G.P.; Balasubramanian, S.; Hickson, I.D.; Boulton, S.J.; Hiom, K. FANCJ is a structure-specific DNA helicase associated with the maintenance of genomic G/C tracts. J. Biol. Chem. 2008, 283, 36132–36139, doi:10.1074/jbc.m808152200.
- Paeschke, K.; Capra, J.A.; Zakian, V.A. DNA replication through G-quadruplex motifs is promoted by the Saccharomyces cerevisiae Pif1 DNA helicase. Cell 2011, 145, 678–691, doi:10.1016/j.cell.2011.04.015.
- Lopes, J.; Piazza, A.; Bermejo, R.; Kriegsman, B.; Colosio, A.; Teulade-Fichou, M.-P.; Foiani, M.; Nicolas, A. G-quadruplex-induced instability during leading-strand replication. EMBO J. 2011, 30, 4033–4046, doi:10.1038/emboj.2011.316.
- Paeschke, K.; Bochman, M.L.; Garcia, P.D.; Cejka, P.; Friedman, K.L.; Kowalczykowski, S.C.; Zakian, V.A. Pif1 family hel-icases suppress genome instability at G-quadruplex motifs. Nature 2013, 497, 458–462.
- Lemmens, B.; van Schendel, R.; Tijsterman, M. Mutagenic consequences of a single G-quadruplex demonstrate mitotic in-heritance of DNA replication fork barriers. Nat. Commun. 2015, 6, 8909, doi:10.1038/ncomms9909.
- Kharel, P.; Balaratnam, S.; Beals, N.; Basu, S. The role of RNA G-quadruplexes in human diseases and therapeutic strategies. Wiley Interdiscip. Rev. RNA 2020, 11, e1568, doi:10.1002/wrna.1568.
- Murat, P.; Marsico, G.; Herdy, B.; Ghanbarian, A.T.; Portella, G.; Balasubramanian, S. RNA G-quadruplexes at upstream open reading frames cause DHX36- and DHX9-dependent translation of human mRNAs. Genome Biol. 2018, 19, 1–24, doi:10.1186/s13059-018-1602-2.
- Huppert, J.; Bugaut, A.; Kumari, S.; Balasubramanian, S. G-quadruplexes: The beginning and end of UTRs. Nucleic Acids Res. 2008, 36, 6260–6268, doi:10.1093/nar/gkn511.
- Endoh, T.; Kawasaki, Y.; Sugimoto, N. Suppression of gene expression by G-quadruplexes in open reading frames depends on G-quadruplex stability. Angew. Chem. Int. Ed. 2013, 52, 5522–5526, doi:10.1002/anie.201300058.





