
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Josko Bozic | + 1970 word(s) | 1970 | 2021-02-09 04:18:59 | | | |
| 2 | Catherine Yang | Meta information modification | 1970 | 2021-02-20 08:03:25 | | |
Video Upload Options
UA is an independent predictor of mortality in acute and chronic HF, making it a significant prognostic factor in both settings. High serum levels have been also associated with an increased incidence of HF, thus expanding the clinical utility of UA.
1. Introduction
With a worse prognosis than breast cancer in women and bladder cancer in men, heart failure (HF) represents a major global health problem [1][2]. This complex clinical entity, commonly defined as the inability of a heart to fulfil required metabolic demands and the perfusion of organs and tissues due to structural or functional cardiac abnormalities, is actually the most common cause of hospitalization after normal delivery, affecting more than 26 million people worldwide [3][4]. Major population studies reported stable incidence but an increase in HF prevalence and only a slight decrease in HF-related mortality in various populations, with a rather intriguing twist: a trend of a slight rise of HF-related mortality recently [5][6][7][8]. The observed increase in prevalence with stable incidence could be explained by the ageing population and improvements in HF treatment [2]. However, this rise will inevitably cause further increases in hospitalization rates and consequently, health care expenditures. Based on available data, experts agree that there is an urgent need for a cost-effective prognostic biomarker in HF. A significant number of biomarkers have already been investigated in the setting of HF [9].
2. Underlying Molecular Mechanisms of HF Development
Virtually any disease or defect that impairs heart structure or function can subsequently lead to HF development. Although most commonly caused by coronary artery disease (CAD), unregulated diabetes, and hypertension, a palette of other etiologic factors, both intra- and extracardiac, can induce HF development [10].
In multiple pathophysiological pathways that are operative in HF, such as myocardial necrosis, upregulation of the renin-angiotensin-aldosterone system (RAAS), overt activation of the sympathetic nervous system, and endothelial dysfunction, a recently recognized pathologic process of endothelial-to-mesenchymal transition (EndoMT) emerged as a potent pathobiological driver of pro-fibrotic signaling pathways in HF, thus leading to myocardial fibrosis and adverse ventricular remodeling [11][12][13][14][15][16][17][18]. EndoMT is a dynamic shift in endothelial cell phenotype toward mesenchymal cells such as myofibroblasts, smooth muscle cells, and osteoblasts [19]. EndoMT-mediated fibrosis seems to be driven mainly by TGF-β via SMAD-2/3/4 and the Slug signaling pathway [20][21]. Hence, it has been hypothesized that EndoMT represents an integrative pathophysiological crosstalk between inflammation and fibrosis, making it a potential therapeutic target in HF [22]. Apart from being implicated in cardiac fibrosis, recent evidence indicates that EndoMT plays a role in several cardiac pathologies, including pulmonary artery hypertension, atherosclerosis, endocardial fibroelastosis, and valvular heart disease [23][24][25][26][27].
In 2013, Paulus and Tschöpe elaborated a pathophysiological model of HF with preserved ejection fraction (HFpEF), which proposes that highly prevalent co-morbidities of HF such as ageing, diabetes mellitus, metabolic syndrome, salt-sensitive hypertension, atrial fibrillation (AF), anemia, chronic obstructive pulmonary disease, and especially obesity exert their detrimental effects on the heart via endothelium of coronary microcirculation, which, as hypothesized, acts as a sort of central processing unit and transfers damage to the heart [28]. According to this model, the mentioned comorbidities induce a systemic pro-inflammatory state and thereby stimulate endothelial cells on reactive oxygen species (ROS) production [29]. Consequently, ROS trigger cardiomyocyte autophagy, apoptosis, or necrosis and reduce nitric oxide (NO) bioavailability, leading to endothelial dysfunction [28]. Importantly, ROS-mediated impaired nitric oxide-cyclic guanosine monophosphate-protein kinase G (NO-cGMP-PKG) signaling also leads to a rise in the resting tension of cardiomyocytes (i.e., myocardial stiffness) via hypophosphorylation of titin [30][31][32][33].
3. Molecular Mechanisms by Which UA Is Implicated in Pathophysiology of HF
UA is the end product of both dietary and endogenous purine metabolism in humans [34]. Due to the loss of uricase, an enzyme responsible for UA conversion into allantoin, humans are exposed to >50 times greater serum uric acid (SUA) concentrations than other mammals, making them susceptible to hyperuricemic repercussions [35]. Although from an evolutionary point of view the loss of uricase may have provided a survival advantage by amplifying the effects of fructose to enhance fat stores and by increasing blood pressure in response to salt, the absence of uricase gene expression, often referred to as “thrifty,” may have exhibited a range of detrimental effects on modern humans owing to the change in diet [36]. As a matter of fact, Neel et al. hypothesized that the loss of uricase could at least in part explain the current epidemic of obesity and diabetes [37]. Another evolutionary advantage of UA was proposed by Ames et al., who demonstrated that UA is a powerful scavenger of free radicals [38]. Figures suggest that UA contributes as much as 60% of free radical scavenging in human serum [39]. Moreover, systemic administration of UA increases plasma antioxidant capacity both at rest and after exercise in healthy volunteers [40][41].
Nevertheless, the biological effects of UA regarding oxidative stress are rather confounding. Unlike the antioxidant effects that UA exerts in extracellular, hydrophilic milieu, intracellularly it imposes detrimental effects, acting as a pro-oxidant [42]. Multiple experimental studies demonstrated that UA stimulated ROS creation in various cells, including endothelial cells, vascular smooth muscle cells (VSMCs), hepatocytes, and renal tubular cells, each with a set of repercussions. In endothelial cells it results in decreased NO bioavailability and inhibited cell migration and proliferation, whereas in hepatocytes it results in intracellular fat accumulation [43][44]. Furthermore, UA activates pro-inflammatory pathways and stimulates cell proliferation in VSMCs, stimulates EndoMT in renal tubular cells, and supports insulin resistance by generating oxidative stress in adipocytes [45][46][47]. Findings that implicate the pro-oxidative activity of UA are further substantiated by the protective effects of probenecid, an inhibitor of the organic anion transporter, which blocks the entry of UA into the cells and ameliorates oxidative stress [46]. Kang et al. tried to elucidate this peculiar dual role of UA in oxidative stress by the presence of an unrecognized molecular switch that controls the role of UA acting as a pro-oxidant or as an anti-oxidant [42]. Regarding the direct effects of UA on cardiomyocytes, multiple studies demonstrated that hyperuricemia inhibits myocardial cell activity by activating the extracellular signal-regulated kinase (ERK)/P38 signaling pathway through oxidative stress in vitro and induces cardiomyocyte apoptosis through the activation of calpain-1 and endoplasmic reticulum stress in rats [47][48][49][50]. Conversely, a study in healthy men showed that acute exposure to high levels of UA had no effect on hemodynamic variables, basal forearm blood flow, or nitric oxide-dependent endothelial function, implying that UA does not impair cardiovascular function [35]. Taken together, the direct effects of UA on the heart still remain quite ambiguous.
The last two steps of purine metabolism are catalyzed with xanthine oxidase (XO) [51]. The mentioned organic chemical reactions catalyzed by XO also generate free radicals as a byproduct [52]. Interestingly, XO was actually the first identified biological system to produce ROS and is in fact one of the strongest known sources of ROS production in human physiology [53]. In physiological milieu, XO-derived reactive oxygen species may have favorable effects, such as modulation of systemic redox balance and a line of defense against bacterial infections [54]. Conversely, overexpression of XO could have ROS-mediated detrimental effects such as endothelial function, inflammatory activation, mitochondrial damage, or impaired cardiac contractility, all of which are commonly seen in HF. Since the involvement of ROS in the development of HF has been well documented, multiple authors investigated the role of XO in HF pathophysiology in both animal and human studies [55]. XO upregulation in HF could be explained by commonly observed events in HF, such as hypoxia, increased catabolism, cell death, and insulin resistance, which lead to purine degradation and a subsequent increase in substrate supply [56][57]. In line with this, as demonstrated by multiple authors, a direct assessment of enzyme activity showed that XO activity was extremely upregulated (up to tenfold) in HF [58][59][60]. Studies suggest that XO is involved in HF development via endothelial dysfunction, myocyte apoptosis, and cardiac mechano-energetic coupling. Endothelial dysfunction is a direct result of an increase in the production of ROS as a consequence of XO upregulation in HF, as we noted in the previous section [61]. Additional evidence to support this notion is that the administration of allopurinol, a well-known XO inhibitor, improves endothelial dysfunction while reducing markers of oxidative stress among patients with HF [62]. In addition, Leyver et al. reported an inverse relationship between SUA and VO2 max and a positive correlation between UA levels and minute ventilation/carbon dioxide production (VE/VCO2), both of which suggest that increased SUA concentrations may reflect an impairment of the oxidative metabolism with consequent exercise intolerance in HF [63]. Apart from the abovementioned deleterious effects, XO upregulation is associated with increased filling pressures in systolic HF, diastolic dysfunction, and cachexia [64][65][66]. XO also emerged as a critical factor in upregulating myocardial apoptosis, a central feature in the progression of HF [67]. Finally, studies suggest that XO impedes the mechano-energetic uncoupling of the heart via crosstalk with cardiac NO signaling pathways. Mechano-energetic coupling is a phenomenon in a failing heart that implies that despite significantly impaired left ventricular LV work, the oxygen consumed for myocardial contraction remains relatively unchanged, resulting in a decrease in the mechanical efficiency of contractions [68]. It has been demonstrated that the administration of allopurinol in dogs with HF decreases oxygen consumption and increases myocardial contractility at both rest and exercise, as well as in response to the stimulation of dobutamine, all of the effects being limited to a failing heart [69][70]. In concordance with animal studies, Cappola et al. demonstrated that allopurinol administration can improve myocardial efficiency by decreasing oxygen consumption without simultaneous impairment in cardiac function [71]. Interestingly, these effects were abrogated by being blocked by N(G)-monomethyl L-arginine (L-NMMA), an NO synthase inhibitor, implicating the importance of NO signaling pathways in this process [70].
In the HF setting, elevated SUA levels are owed to at least two distinct mechanisms (Figure 1). The first is increased production and the latter is reduced excretion of UA. The former is caused by both a substantial increase in XO activity and increased oxidative stress, which arises from reduced tissue perfusion and altered metabolic state [51][67]. Conversely, multiple mechanisms lead to reduced kidney UA excretion. Functional renal impairment, as a part of cardiorenal syndrome, leads to decreased UA excretion in the kidney [72][73]. Furthermore, diuretics, which are widely prescribed to HF patients, lead to substantial loss of water and salt, thus stimulating proximal tubule reabsorption and a subsequent rise in SUA [74]. Other medications, such as noradrenaline and angiotensin II, can also promote hyperuricemia by stimulating UA tubular absorption [75]. In a state of impaired muscle perfusion and a consequent switch to an anaerobic metabolism such as HF, lactic acid plasma levels increase and lead to hyperuricemia by further mitigating UA renal excretion [76]. Ultimately, elevated UA itself can impair renal function, creating a positive feedback loop [77]. Of important note, patients with ischemic and non-ischemic HF show a similar distribution of SUA concentrations with respect to New York Heart Association (NYHA) classes [78]. This highlights the significant role of UA in HF independent of the presence of the metabolic syndrome, a common risk profile for ischemic heart disease, thus bringing further evidence that supports the notion that hyperuricemia is an intrinsic feature within the HF pathophysiology. Since SUA levels correlate with poor clinical outcomes of chronic HF (CHF) in a more evident manner among patients with impaired renal function, it seems that increased UA synthesis is a more significant contributor to the observed elevation of SUA in CHF than a reduction in UA excretion [79]. Conversely, Park et al. argued that cell death caused by ischemic insult or acute deterioration of renal function in acute heart failure (AHF) may be the dominant factor for hyperuricemia in that setting [80].
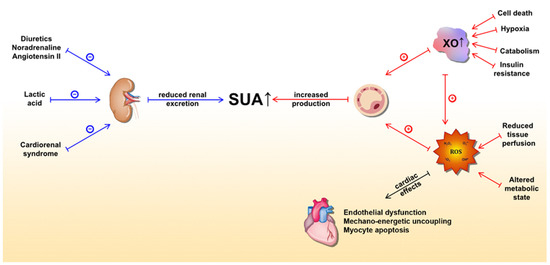
Figure 1. Underlying molecular mechanisms of serum uric acid (SUA) elevation in heart failure and detrimental effects of xanthine oxidase activity (mediated by ROS) on the heart. The blue lines represent mechanisms that lead to a reduction in UA excretion, whereas the red lines represent mechanisms leading to increased UA production. Abbreviations: SUA: serum uric acid; XO: xanthine oxidase; ROS: reactive oxygen species.
References
- Mamas, M.A.; Sperrin, M.; Watson, M.C.; Coutts, A.; Wilde, K.; Burton, C.; Kadam, U.T.; Kwok, C.S.; Clark, A.B.; Murchie, P. Do patients have worse outcomes in heart failure than in cancer? A primary care-based cohort study with 10-year follow-up in Scotland. Eur. J. Heart Fail. 2017, 19, 1095–1104.
- Savarese, G.; Lund, L.H. Global Public Health Burden of Heart Failure. Card. Fail. Rev. 2017, 3, 7–11.
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.; Coats, A.J.; Falk, V.; González-Juanatey, J.R.; Harjola, V.P.; Jankowska, E.A. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. J. Heart Fail. 2016, 18, 91–975.
- Braunwald, E. Heart failure. JACC Heart Fail. 2013, 1, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Mensah, G.A.; Wei, G.S.; Sorlie, P.D.; Fine, L.J.; Rosenberg, Y.; Kaufmann, P.G.; Mussolino, M.E.; Hsu, L.L.; Addou, E.; Engelgau, M.M. Decline in Cardiovascular Mortality: Possible Causes and Implications. Circ. Res. 2017, 120, 366–380.
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; Das, S.R.; de Ferranti, S.; Després, J.P.; Fullerton, H.J. Heart Disease and Stroke Statistics-2016 Update: A Report From the American Heart Association. American Heart Association Statistics Committee, Stroke Statistics Subcommittee. Circulation 2016, 133, e38–e360.
- Ohlmeier, C.; Mikolajczyk, R.; Frick, J.; Prütz, F.; Haverkamp, W.; Garbe, E. Incidence; prevalence and 1-year all-cause mortality of heart failure in Germany: A study based on electronic healthcare data of more than six million persons. Clin. Res. Cardiol. 2015, 104, 88–696.
- Sakata, Y.; Shimokawa, H. Epidemiology of heart failure in Asia. Circ. J. 2013, 77, 2209–2217.
- Spoletini, I.; Coats, A.J.S.; Senni, M.; Rosano, G.M.C. Monitoring of biomarkers in heart failure. Eur. Heart J. Suppl. 2019, 21, M5–M8.
- Ziaeian, B.; Fonarow, G.C. Epidemiology and aetiology of heart failure. Nat. Rev. Cardiol. 2016, 13, 368–378.
- Creemers, E.E.; Pinto, Y.M. Molecular mechanisms that control interstitial fibrosis in the pressure-overloaded heart. Cardiovasc. Res. 2011, 89, 265–272.
- Giannitsi, S.; Bougiakli, M.; Bechlioulis, A.; Naka, K. Endothelial dysfunction and heart failure: A review of the existing bibliography with emphasis on flow mediated dilation. JRSM Cardiovasc. Dis. 2019, 8.
- Gandhi, M.S.; Kamalov, G.; Shahbaz, A.U.; Bhattacharya, S.K.; Ahokas, R.A.; Sun, Y.; Gerling, I.C.; Weber, K.T. Cellular and molecular pathways to myocardial necrosis and replacement fibrosis. Heart Fail. Rev. 2011, 16, 23–34.
- Borovac, J.A.; D’Amario, D.; Bozic, J.; Glavas, D. Sympathetic nervous system activation and heart failure: Current state of evidence and the pathophysiology in the light of novel biomarkers. World J. Cardiol. 2020, 12, 373–408.
- Franssen, C.; Chen, S.; Unger, A.; Korkmaz, H.I.; De Keulenaer, G.W.; Tschöpe, C.; Leite-Moreira, A.F.; Musters, R.; Niessen, H.W.; Linke, W.A.; et al. Myocardial Microvascular Inflammatory Endothelial Activation in Heart Failure with Preserved Ejection Fraction. JACC Heart Fail. 2016, 4, 312–324.
- Willenheimer, R. Angiotensin receptor blockers in heart failure after the ELITE II trial. Curr. Control Trials Cardiovasc. Med. 2000, 1, 79–82.
- Erdmann, E. Indikationen für ACE-Hemmer bei der chronischen Herzinsuffizienz [Indications for ACE inhibitors in chronic heart failure]. Z Kardiol. 1994, 83, 75–79.
- Murdoch, C.E.; Chaubey, S.; Zeng, L.; Yu, B.; Ivetic, A.; Walker, S.J.; Vanhoutte, D.; Heymans, S.; Grieve, D.J.; Cave, A.C.; et al. Endothelial NADPH oxidase-2 promotes interstitial cardiac fibrosis and diastolic dysfunction through proinflammatory effects and endothelial-mesenchymal transition. J. Am. Coll. Cardiol. 2014, 63, 2734–2741.
- Kovacic, J.C.; Dimmeler, S.; Harvey, R.P.; Finkel, T.; Aikawa, E.; Krenning, G.; Baker, A.H. Endothelial to Mesenchymal Transition in Cardiovascular Disease: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2019, 73, 190–209.
- Cooley, B.C.; Nevado, J.; Mellad, J.; Yang, D.; St Hilaire, C.; Negro, A.; Fang, F.; Chen, G.; San, H.; Walts, A.D.; et al. TGF-β signaling mediates endothelial-to-mesenchymal transition (EndMT) during vein graft remodeling. Sci. Transl. Med. 2014, 6, 227ra34.
- Jiang, Y.; Zhou, X.; Hu, R.; Dai, A. TGF-β1-induced SMAD2/3/4 activation promotes RELM-β transcription to modulate the endothelium-mesenchymal transition in human endothelial cells. Int. J. Biochem. Cell Biol. 2018, 105, 52–60.
- Cho, J.G.; Lee, A.; Chang, W.; Lee, M.S.; Kim, J. Endothelial to Mesenchymal Transition Represents a Key Link in the Interaction between Inflammation and Endothelial Dysfunction. Front. Immunol. 2018, 9, 294.
- Good, R.B.; Gilbane, A.J.; Trinder, S.L.; Denton, C.P.; Coghlan, G.; Abraham, D.J.; Holmes, A.M. Endothelial to Mesenchymal Transition Contributes to Endothelial Dysfunction in Pulmonary Arterial Hypertension. Am. J. Pathol. 2015, 185, 1850–1858.
- Evrard, S.M.; Lecce, L.; Michelis, K.C.; Nomura-Kitabayashi, A.; Pandey, G.; Purushothaman, K.R.; d’Escamard, V.; Li, J.R.; Hadri, L.; Fujitani, K. Endothelial to mesenchymal transition is common in atherosclerotic lesions and is associated with plaque instability. Nat. Commun. 2016, 7, 11853.
- Xu, X.; Friehs, I.; Zhong Hu, T.; Melnychenko, I.; Tampe, B.; Alnour, F.; Iascone, M.; Kalluri, R.; Zeisberg, M.; Del Nido, P.J. Endocardial fibroelastosis is caused by aberrant endothelial to mesenchymal transition. Circ. Res. 2015, 116, 857–866.
- Hulshoff, M.S.; Xu, X.; Krenning, G.; Zeisberg, E.M. Epigenetic Regulation of Endothelial-to-Mesenchymal Transition in Chronic Heart Disease. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 1986–1996.
- Wirrig, E.E.; Yutzey, K.E. Conserved transcriptional regulatory mechanisms in aortic valve development and disease. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 737–741.
- Paulus, W.J.; Tschöpe, C. A novel paradigm for heart failure with preserved ejection fraction: Comorbidities drive myocardial dysfunction and remodeling through coronary microvascular endothelial inflammation. J. Am. Coll. Cardiol. 2013, 62, 263–271.
- Griendling, K.K.; Sorescu, D.; Ushio-Fukai, M. NAD(P)H oxidase: Role in cardiovascular biology and disease. Circ. Res. 2000, 86, 494–501.
- van Heerebeek, L.; Hamdani, N.; Handoko, M.L.; Falcao-Pires, I.; Musters, R.J.; Kupreishvili, K.; Ijsselmuiden, A.J.; Schalkwijk, C.G.; Bronzwaer, J.G.; Diamant, M. Diastolic stiffness of the failing diabetic heart: Importance of fibrosis, advanced glycation end products, and myocyte resting tension. Circulation 2008, 117, 43–51.
- Krüger, M.; Kötter, S.; Grützner, A.; Lang, P.; Andresen, C.; Redfield, M.M.; Butt, E.; dos Remedios, C.G.; Linke, W.A. Protein kinase G modulates human myocardial passive stiffness by phosphorylation of the titin springs. Circ. Res. 2009, 104, 87–94.
- Borbély, A.; Falcao-Pires, I.; van Heerebeek, L.; Hamdani, N.; Edes, I.; Gavina, C.; Leite-Moreira, A.F.; Bronzwaer, J.G.; Papp, Z.; van der Velden, J. Hypophosphorylation of the Stiff N2B titin isoform raises cardiomyocyte resting tension in failing human myocardium. Circ. Res. 2009, 104, 780–786.
- Bishu, K.; Hamdani, N.; Mohammed, S.F.; Kruger, M.; Ohtani, T.; Ogut, O.; Brozovich, F.V.; Burnett, J.C., Jr.; Linke, W.A.; Redfield, M.M. Sildenafil and B-type natriuretic peptide acutely phosphorylate titin and improve diastolic distensibility in vivo. Circulation 2011, 124, 2882–2891.
- Maiuolo, J.; Oppedisano, F.; Gratteri, S.; Muscoli, C.; Mollace, V. Regulation of uric acid metabolism and excretion. Int. J. Cardiol. 2016, 213, 8–14.
- Waring, W.S.; Adwani, S.H.; Breukels, O.; Webb, D.J.; Maxwell, S.R. Hyperuricaemia does not impair cardiovascular function in healthy adults. Heart 2004, 90, 155–159.
- Kratzer, J.T.; Lanaspa, M.A.; Murphy, M.N.; Cicerchi, C.; Graves, C.L.; Tipton, P.A.; Ortlund, E.A.; Johnson, R.J.; Gaucher, E.A. Evolutionary history and metabolic insights of ancient mammalian uricases. Proc. Natl. Acad. Sci. USA 2014, 111, 3763–3768.
- Neel, J.V. Diabetes mellitus: A “thrifty” genotype rendered detrimental by “progress”? Am. J. Hum. Genet. 1962, 14, 353–362.
- Ames, B.N.; Cathcart, R.; Schwiers, E.; Hochstein, P. Uric acid provides an antioxidant defence in humans against oxidant- and radical-caused aging and cancer: A hypothesis. Proc. Natl. Acad. Sci. USA 1981, 78, 6858–6862.
- Maxwell, S.R.; Thomason, H.; Sandler, D.; Leguen, C.; Baxter, M.A.; Thorpe, G.H.; Jones, A.F.; Barnett, A.H. Antioxidant status in patients with uncomplicated insulin-dependent and non-insulin-dependent diabetes mellitus. Eur. J. Clin. Investig. 1997, 27, 484–490.
- Waring, W.S.; Webb, D.J.; Maxwell, S.R. Systemic uric acid administration increases serum antioxidant capacity in healthy volunteers. J. Cardiovasc. Pharmacol. 2001, 38, 365–371.
- Waring, W.S.; Convery, A.; Mishra, V.; Shenkin, A.; Webb, D.J.; Maxwell, S.R. Uric acid reduces exercise-induced oxidative stress in healthy adults. Clin. Sci. (Lond.) 2003, 105, 425–430.
- Kang, D.H.; Ha, S.K. Uric Acid Puzzle: Dual Role as Anti-oxidant and Pro-oxidant. Electrolyte Blood Press. 2014, 12, 1–6.
- Yu, M.A.; Sanchez-Lozada, L.G.; Johnson, R.J.; Kang, D.H. Oxidative stress with an activation of the renin-angiotensin system in human vascular endothelial cells as a novel mechanism of uric acid-induced endothelial dysfunction. J. Hypertens. 2010, 28, 1234–1242.
- Lanaspa, M.A.; Sanchez-Lozada, L.G.; Choi, Y.J.; Cicerchi, C.; Kanbay, M.; Roncal-Jimenez, C.A.; Ishimoto, T.; Li, N.; Marek, G.; Duranay, M. Uric acid induces Hepatic Steatosis by Generation of Mitochondrial Oxidative Stress: Potential Role in Fructose-Dependent and- Independent Fatty Liver. J. Biol. Chem. 2012, 287, 40732–40744.
- Sánchez-Lozada, L.G.; Soto, V.; Tapia, E.; Avila-Casado, C.; Sautin, Y.Y.; Nakagawa, T.; Franco, M.; Rodríguez-Iturbe, B.; Johnson, R.J. Role of oxidative stress in the renal abnormalities induced by experimental hyperuricemia. Am. J. Physiol. 2008, 295, F1134–F1141.
- Ryu, E.S.; Kim, M.J.; Shin, H.S.; Jang, Y.H.; Choi, H.S.; Jo, I.; Johnson, R.J.; Kang, D.H. Uric acid-induced phenotypic transition of renal tubular cells as a novel mechanism of chronic kidney disease. Am. J. Physiol. Renal Physiol. 2013, 304, F471–F480.
- Sautin, Y.Y.; Nakagawa, T.; Zharikov, S.; Johnson, R.J. Adverse effects of the classic antioxidant uric acid in adipocytes: NADPH oxidase-mediated oxidative/nitrosative stress. Am. J. Physiol. Cell Physiol. 2007, 293, C584–C596.
- Li, Z.; Shen, Y.; Chen, Y.; Zhang, G.; Cheng, J.; Wang, W. High Uric Acid Inhibits Cardiomyocyte Viability Through the ERK/P38 Pathway via Oxidative Stress. Cell Physiol. Biochem. 2018, 45, 1156–1164.
- Yan, M.; Chen, K.; He, L.; Li, S.; Huang, D.; Li, J. Uric Acid Induces Cardiomyocyte Apoptosis via Activation of Calpain-1 and Endoplasmic Reticulum Stress. Cell Physiol. Biochem. 2018, 45, 2122–2135.
- Liu, D.; Gao, K.; Xie, Y.; Li, Z. The effect of high uric acid on the activity of cardiomyocytes and its related mechanism. Tianjin Med. J. 2020, 48, 931–936.
- Meneshian, A.; Bulkley, G.B. The physiology of endothelial xanthine oxidase: From urate catabolism to reperfusion injury to inflammatory signal transduction. Microcirculation 2002, 9, 161–175.
- Berry, C.E.; Hare, J.M. Xanthine oxidoreductase and cardiovascular disease: Molecular mechanisms and pathophysiological implications. J. Physiol. 2004, 555, 589–606.
- McCord, J.M.; Fridovich, I. The reduction of cytochrome c by milk xanthine oxidase. J. Biol. Chem. 1968, 243, 5753–5760.
- Glantzounis, G.K.; Tsimoyannis, E.C.; Kappas, A.M.; Galaris, D.A. Uric acid and oxidative stress. Curr. Pharm. Des. 2005, 11, 4145–4151.
- Tsutsui, H.; Kinugawa, S.; Matsushima, S. Oxidative stress and heart failure. Am. J. Physiol. Heart Circ. Physiol. 2011, 301, H2181–H2190.
- Leyva, F.; Chua, T.P.; Anker, S.D.; Coats, A.J. Uric acid in chronic heart failure: A measure of the anaerobic threshold. Metabolism 1998, 47, 1156–1159.
- Leyva, F.; Wingrove, C.S.; Godsland, I.F.; Stevenson, J.C. The glycolytic pathway to coronary heart disease: A hypothesis. Metabolism 1998, 47, 657–662.
- Landmesser, U.; Spiekermann, S.; Dikalov, S.; Tatge, H.; Wilke, R.; Kohler, C.; Harrison, D.G.; Hornig, B.; Drexler, H. Vascular oxidative stress and endothelial dysfunction in patients with chronic heart failure: Role of xanthine-oxidase and extracellular superoxide dismutase. Circulation 2002, 106, 3073–3078.
- Doehner, W.; Tarpey, M.T.; Pavitt, D.V.; Bolger, A.P.; Wensel, R.; von Haehling, S.; Reaveley, D.A.; Anker, S.D. Elevated plasma xanthine oxidase activity in chronic heart failure: Source of increased oxygen radical load and effect of allopurinol in a placebo controlled, double blinded treatment study. J. Am. Coll. Cardiol. 2003, 41, 207.
- de Jong, J.W.; Schoemaker, R.G.; de Jonge, R.; Bernocchi, P.; Keijzer, E.; Harrison, R.; Sharma, H.S.; Ceconi, C. Enhanced expression and activity of xanthine oxidoreductase in the failing heart. J. Mol. Cell Cardiol. 2000, 32, 2083–2089.
- Maxwell, A.J.; Bruinsma, K.A. Uric acid is closely linked to vascular nitric oxide activity. Evidence for mechanism of association with cardiovascular disease. J. Am. Coll. Cardiol. 2001, 38, 1850–1858.
- Farquharson, C.A.; Butler, R.; Hill, A.; Belch, J.J.; Struthers, A.D. Allopurinol improves endothelial dysfunction in chronic heart failure. Circulation 2002, 106, 221–226.
- Leyva, F.; Anker, S.; Swan, J.W.; Godsland, I.F.; Wingrove, C.S.; Chua, T.P.; Stevenson, J.C.; Coats, A.J. Serum uric acid as an index of impaired oxidative metabolism in chronic heart failure. Eur. Heart J. 1997, 18, 858–865.
- Doehner, W.; Rauchhaus, M.; Florea, V.G.; Sharma, R.; Bolger, A.P.; Davos, C.H.; Coats, A.J.; Anker, S.D. Uric acid in cachectic and noncachectic patients with chronic heart failure: Relationship to leg vascular resistance. Am. Heart J. 2001, 141, 792–799.
- Amin, A.; Vakilian, F.; Maleki, M. Serum uric acid levels correlate with filling pressures in systolic heart failure. Congest. Heart Fail. 2011, 17, 80–84.
- Cicoira, M.; Zanolla, L.; Rossi, A.; Golia, G.; Franceschini, L.; Brighetti, G.; Zeni, P.; Zardini, P. Elevated serum uric acid levels are associated with diastolic dysfunction in patients with dilated cardiomyopathy. Am. Heart J. 2002, 143, 1107–1111.
- Bergamini, C.; Cicoira, M.; Rossi, A.; Vassanelli, C. Oxidative stress and hyperuricaemia: Pathophysiology, clinical relevance, and therapeutic implications in chronic heart failure. Eur. J. Heart Fail. 2009, 11, 444–452.
- Ekelund, U.E.; Harrison, R.W.; Shokek, O.; Thakkar, R.N.; Tunin, R.S.; Senzaki, H.; Kass, D.A.; Marbán, E.; Hare, J.M. Intravenous allopurinol decreases myocardial oxygen consumption and increases mechanical efficiency in dogs with pacing-induced heart failure. Circ. Res. 1999, 85, 437–445.
- Ukai, T.; Cheng, C.P.; Tachibana, H.; Thakkar, R.N.; Tunin, R.S.; Senzaki, H.; Kass, D.A.; Marbán, E.; Hare, J.M. Allopurinol enhances the contractile response to dobutamine and exercise in dogs with pacing-induced heart failure. Circulation 2001, 103, 750–755.
- Saavedra, W.F.; Paolocci, N.; St John, M.E.; Skaf, M.W.; Stewart, G.C.; Xie, J.S.; Harrison, R.W.; Zeichner, J.; Mudrick, D.; Marbán, E. Imbalance between xanthine oxidase and nitric oxide synthase signal-ing pathways underlies mechanoenergetic uncoupling in the failing heart. Circ. Res. 2002, 90, 297–304.
- Cappola, T.P.; Kass, D.A.; Nelson, G.S.; Berger, R.D.; Rosas, G.O.; Kobeissi, Z.A.; Marbán, E.; Hare, J.M. Allopurinol improves myocardial efficiency in patients with idiopathic dilated cardiomyopathy. Circulation 2001, 104, 2407–2411
- Johnson, R.J.; Nakagawa, T.; Jalal, D.; Sánchez-Lozada, L.G.; Kang, D.H.; Ritz, E. Uric acid and chronic kidney disease: Which is chasing which? Nephrol. Dial Transplant. 2013, 28, 2221–2228.
- Hahn, K.; Kanbay, M.; Lanaspa, M.A.; Johnson, R.J.; Ejaz, A.A. Serum uric acid and acute kidney injury: A mini review. J. Adv. Res. 2017, 8, 529–536.
- Ranieri, L.; Contero, C.; Peral, M.L.; Calabuig, I.; Zapater, P.; Andres, M. Impact of diuretics on the urate lowering therapy in patients with gout: Analysis of an inception cohort. Arthritis Res. Ther. 2018, 20, 53.
- Pascual-Figal, D.A.; Hurtado-Martínez, J.A.; Redondo, B.; Antolinos, M.J.; Ruiperez, J.A.; Valdes, M. Hyperuricaemia and long-term outcome after hospital discharge in acute heart failure patients. Eur. J. Heart Fail. 2007, 9, 518–524.
- Herrmann, R.; Sandek, A.; von Haehling, S.; Doehner, W.; Schmidt, H.B.; Anker, S.D.; Rauchhaus, M. Risk stratification in patients with chronic heart failure based on metabolic-immunological, functional and haemodynamic parameters. Int. J. Cardiol. 2012, 156, 62–68.
- Ohno, I. Relationship between hyperuricemia and chronic kidney disease. Nucleosides Nucleotides Nucleic Acids 2011, 30, 1039–1044.
- Khan, A.; Shah, M.H.; Khan, S.; Shamim, U.; Arshad, S. Serum Uric Acid level in the severity of Congestive Heart Failure (CHF). Pak. J. Med. Sci. 2017, 33, 330–334.
- Filippatos, G.S.; Ahmed, M.I.; Gladden, J.D.; Mujib, M.; Aban, I.B.; Love, T.E.; Sanders, P.W.; Pitt, B.; Anker, S.D.; Ahmed, A. Hyperuricaemia, chronic kidney disease, and outcomes in heart failure: Potential mechanistic insights from epidemiological data. Eur. Heart J. 2011, 32, 712–720.
- Park, H.S.; Kim, H.; Sohn, J.H.; Shin, H.W.; Cho, Y.K.; Yoon, H.J.; Nam, C.W.; Hur, S.H.; Kim, Y.N.; Kim, K.B.; et al. Combination of uric acid and NT-ProBNP: A more useful prognostic marker for short-term clinical outcomes in patients with acute heart failure. Korean J. Intern. Med. 2010, 25, 253–259.





