| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Georgios Romanos | + 2693 word(s) | 2693 | 2021-01-26 05:04:45 | | | |
| 2 | Bruce Ren | -21 word(s) | 2672 | 2021-02-02 03:48:10 | | |
Video Upload Options
Titanium is considered high-ly biocompatible with low corrosion and toxicity, but recent studies indicate that this understand-ing may be misleading as the properties of the material change drastically when titanium nano-particles (NPs) are shed from implant surfaces. These NPs are immunogenic and are associated with a macrophage-mediated inflammatory response by the host.
1. Introduction
Dental implants have become a popular option for the replacement of one or more missing teeth in the oral cavity due to their long-term success rate [1]. Almost 11% of dental implants fail and are subsequently removed within the first 15 years of placement and osseointegration [2]. Regardless of the etiology, the point at which an implant becomes mobile is the point where it has become a failed implant. Bacterial challenge to the peri-implant tissues, in the form of plaque biofilms, is a well-known cause of local inflammation that may eventually lead to peri-implant mucositis, and in some patients, peri-implantitis [3]. Peri-implant biofilms differ in composition than those associated with teeth. Peri-implant biofilms contain some unique species as well as bacterial forms associated with periodontal pockets [4]. The peri-implant tissue is described as a zone approximately 1mm thick surrounding the implant body where interactions with peri-implant bone may occur [5][6]. Peri-implantitis is an inflammatory condition of the supporting bone around a dental implant. Peri-implant mucositis, which generally precedes peri-implantitis, is reversible inflammation of the soft tissue surrounding an implant [7][8]. The clinical signs of peri-implant conditions include bleeding on probing, suppuration, and increased probing depths. The diagnosis is confirmed by radiographic peri-implant bone loss [9].
Recently, studies on biomaterials have become an important part of dental implant research as investigators attempt to better understand the etiology of implant failures. Manufacturers have developed numerous different implant designs differing in proportions of metals and other materials as well as surface roughening treatments all in an effort to improve osseointegration and biocompatibility while decreasing the rate of implant failure [10]. Some of these implants include commercially pure (c.p.) titanium, Titanium Aluminum Vanadium (Ti-6Al-4V) alloys, and recently, titanium-zirconia alloys [11][12]. They are supposedly highly biocompatible and resistant to corrosion due to their ability to form a passive oxide film following exposure to oxygen [12].
Manufacturers have also modified the surface roughness of implants to improve the osseointegration by acid etching, sandblasting, and oxidation techniques [13]. Extensive research concluded that pure titanium and titanium alloys are both highly biocompatible with low toxicity and favorable properties for osseointegration [14]. Many of these studies, however, do not focus on the properties of dental implant materials when they are broken down to smaller particles. There has been growing concern over this issue as newer studies have shown that there are many phases of an implant’s life cycle during which there is wear of the implant that leads to the shedding of titanium particles which are then introduced to the local tissues . Once the titanium particles are shed from the implant surface, they can induce local inflammation. They may also be transported away from the oral cavity, after which the particles can be found, causing inflammation in distant tissues, with potential systemic involvement [14].
2. Titanium Wear at the Time of Implant Insertion
The study of titanium particles liberated from dental implant surfaces has become an important area of research in dental implantology. A 1993 study conducted by Schliephake et al. revealed the presence of titanium in the adjacent soft tissue surrounding titanium screws used to treat jaw fracture. The titanium was viewed 5-8 months post-operatively in transmission electron microscope images. The study suggested that the titanium particles localized extracellularly may be taken up by cells for lysosomal degradation, and the remnants may be left in place after the macrophages eventually clear out [15]. Subsequent research has found that titanium particles may be shed into the surrounding peri-implant tissues as early as the time of initial implant placement. In 2004, Franchi et al. found titanium granules separated from the implant surface in the peri-implant tissues (mucosa and bone) as early as 12 weeks after placement [16]. Their finding implies that masticatory forces, corrosion, and fretting are not necessary for titanium wear to occur and confirms that titanium wear occurs by the friction created between the implant and the bone during the implant insertion [16].
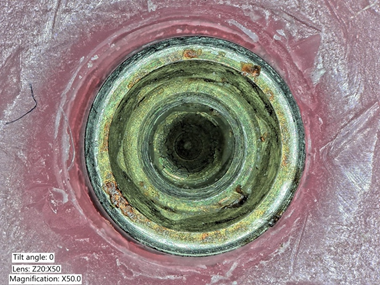
These results are further supported by a study done by Palazzo et al. [17]. Their histological analysis showed metal particles and ions released from osteosynthesis implants and concluded that metal microparticles could be released during the actual fixation of the implant into the bone (Figures 1–5), with the highest concentration of metal found close to the osteosynthesis or implant fixation and as far as 2.5cm away from the implants [17].
Figure 1. Metal particle apposition in bone block in vitro immediately after surgery.
Figure 2. Sandblasted and acid-etched surface demonstrating changes of the implant roughness after implant insertion, and titanium particles detachment from the implant surface (lighter areas).
Figure 3. Anodized implant surface demonstrating areas of flattening and disappearance of the typical “crater” appearance, meaning particle detachment (lighter areas).
Figure 4. Sandblasted, large grit, acid-etched implant surface (SLA) demonstrating areas of particle detachment, and topographic changes after insertion (darker areas).
Figure 5. Sandblasted implant surface demonstrating areas of change after insertion.
Similar findings are seen in recent microscopical EDX analyses of the peri-implant tissues of immediately loaded implants placed in the maxilla and mandible. In an autopsy report conducted 7.5 months after implant placement and loading, analyses showed titanium particles in direct contact to the implant and in distant areas from the implant-bone interface [18].
3. Titanium Wear under Conditions of Loading/Forces of Mastication
One of the most important factors affecting the longevity of a dental implant restoration, is the fit of the implant with the abutment [19]. A less than ideal fit will lead to a larger micro gap between the implant and abutment, where microorganisms, oral fluids, and glycoproteins can accumulate and form stable biofilms leading to corrosion of the material (Figures 6–10) [19][20][21][22].
Figure 6. Explanted implant with a Trilobed connection. After disconnection of the implant crown, the evaluation of the implant connection shows severe wear, abrasion of the implant platform, and titanium particles.
Figure 7. Explanted implant with a Trilobed connection. Increased magnification allows to observe fracture lines, corrosion, and accumulation of titanium particles potentially from implant-abutment connection wear.
Figure 8. Explanted implant with a hexagonal connection. The implant crown was removed. The implant connection at microscope evaluation showed wear, signs of corrosion, and titanium particles.
Figure 9. Closer view of an explanted implant with a hexagonal connection. Corrosion, delamination, and abrasions of the connection surface can be appreciated.
Figure 10. Closer view of an explanted implant with a hexagonal connection. Corrosion, delamination, and abrasions of the connection surface can be appreciated.
A poor fit with a large micro gap will leave the implant vulnerable to structural damage in the form of fretting and micromovements at the connection when subjected to forces of mastication [19]. In this context, the biofilms act as a lubricant in the connection [19]. As the material at the interface is worn down, the micro gap becomes larger leading to further destabilization of the implant [23]. Morse taper connections have been found to provide a better fit than non-conical connections, thus decreasing the wear of the implant during mastication [24]. Multiple studies show bacteria accumulation in the peri-implant mucosa of implants with external connections, but recent clinical studies comparing morse-tapered with internal polygonal connections demonstrate the accumulation of bacteria deep within the internal (butt-joint) interface compared to the Morse tapered connections, where a bacterial sealing is found [25][26][27][28]. The micromovements of the abutment within the implant body under loading, which is known as “fretting”, leads to titanium nanoparticles in the surrounding implant-tissue interface and further metal corrosion. Studies have shown increased metal debris in the surrounding tissue of peri-implantitis sites compared to the tissue surrounding periodontitis sites. This indicates that metal debris in peri-implant hard and soft tissues may be an etiological factor for peri-implant disease [29]. Recent studies emphasized the importance of the stability of the implant-abutment connection when it comes to the health of peri-implant tissues. It has been found that conical implant-abutment connections minimize the micro gap at the connection than butt-join implant-abutment connections, and conical implant abutment connections allow less bacterial accumulation [30][31]. When the loading forces are increased, the contact wear becomes severe promoting microcracks and particle detachment at the titanium-bone interface especially in the cortical bone and areas with lower content of calcium and phosphate [32]. The significance of increased loading forces is also seen during both osteotomy and manual condensation, where bone density is altered. It has been found via histological analysis that functional loading forces further increase bone density [33][34][35][36].
4. Titanium Wear at Implant-Abutment Interface and Impact on the Peri-Implant Tissues
To understand the significance of the interactions at the implant-abutment interface, it is important to understand the phenomena of corrosion and fretting. Corrosion is a process by which a refined metal becomes a more stable oxide, hydroxide, or sulfide. Pure titanium becoming titanium dioxide NP in the peri-implant environment is an example of this conversion. Fretting is the process of wear at the contact points of uneven surfaces. This process occurs under loading forces and may also occur in the presence of corrosion [37]. Fretting refers to wear and sometimes corrosion damage at the asperities of contact surfaces. This damage is induced under load and in the presence of repeated relative surface motion, as induced for example by vibration [37].
There has recently been an observation of increased titanium dissolution products in biofilms isolated from peri-implantitis cases. These results suggest a possible modification in the structure and diversity of the microbiome due to the titanium dissolution [38]. These findings led to the investigation of the mechanism by which titanium disintegration occurs and factors that may play a role. Additionally, the role of titanium corrosion has become an area of interest [39]. Titanium implants are said to be resistant to corrosion; however, studies have shown that the stable oxide layer that is formed during the process of osseointegration is lost under certain conditions, particularly those seen in the oral cavity. Once this layer is lost, biodegradation and corrosion of the implant may occur [40][41]. Therefore, while titanium implants may be resistant to corrosion in vitro, this property should be explored more in depth in the oral environment [42]. Recent studies have shown that the acidic metabolic by-products of microorganisms and some substances found in fluoride solutions may increase corrosion (Figures 11–13), especially at the implant-abutment interface, where the micro gap exists [43][44][45][46][47][48]. In general, significant inflammatory conditions decrease titanium’s resistance to corrosion. This is a significant area of concern in dental implant research as titanium corrosion can contribute to peri-implantitis and eventual implant failure.
Figure 11. The evaluation of the removed crown abutment, also showed titanium particles and titanium delamination. Surface changes were also evident at the transmucosal portion of the abutment.
Figure 12. Corrosion of the abutment can occur and appears as pits, or color changes (dark, brown or yellow) distinguishable from the natural gray color of the implant.
Figure 13. Evaluation of the basal aspect of the implant crown, showing deformation of the hexagon, and particle delamination.
The corrosion of titanium is enhanced by the presence of H2O2 and albumin [49][50][51]. Inflammation is associated with production of highly reactive chemical molecules formed due to the electron acceptability of O₂, so called reactive oxygen species (ROS), such as H2O2 and superoxide ions, which further increase the corrosion of titanium metal. The inflammatory state is associated with increased neutrophils and macrophages which generate ROS via the respiratory burst pathway [52]. This phenomenon leads to higher porosity of the oxide films and a formation of a rougher, thicker implant surface. In general, the inflammatory state is associated with an acidic environment. This is due to increased oxygen and energy demands of inflammatory cells in the tissue. Glucose is metabolized at a greater rate via glycolysis; thus, more lactic acid is generated [53]. Metal degradation, in conjunction with the acidic environment of inflammation, concentrates the ROS species and up-regulates the corrosion of the implant body, thus promoting the accumulation of titanium ions and particles in the surrounding tissues with more bone resorption and loosening of the implant [54].
Surface analysis of retrieved titanium alloy hip implants from humans were investigated for evidence of localized or general corrosion in modular interfaces when mechanical abrasion of the oxide film (fretting) occurs. The study showed cracking, etching, pitting and delamination of the surface, as well as degradation in the crevice environment during fretting-crevice corrosion and hydrogen embrittlement [55]. Two surfaces under sliding contact induce formation of debris leading to more abrasion and creating a third body particles with impact on the inflammatory reaction in the peri-implant tissues as a process of bio-tribocorrosion [56][57][58][59][60][61].
Titanium accumulation of 13 ppm and above has been shown to induce epithelial cell necrosis and increase the sensitivity of epithelial cells of the peri-implant mucosa in microorganisms. This observation is due to ROS production and increased cytokine levels, leading to a reduction of cell viability and proliferation. Additionally, the induction of apoptosis and genotoxicity have been documented [62]. The biologic mechanism for cell necrosis induced by Ti-ions is not yet clear [63].
5. Titanium Wear from Instrumentation of Dental Implants for Reparative and Maintenance Procedures
There is no doubt that implant maintenance is required after implant therapy as a standard of care. Many clinicians practice instrumentation of dental implants routinely in order to control and manage peri-implant diseases [64][65][66]. Local antibiotics have also been utilized to treat peri-implantitis. Slow release chemotherapeutic devices with chlorhexidine also seem to have a positive impact on reduction of clinical parameters of peri-implant inflammation[67].
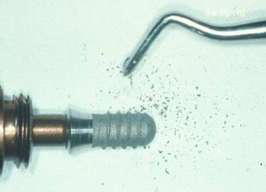
Recent studies have shown that mechanical alteration of implant surfaces may lead to negative consequences (Figure 14). The instrumentation of the implants may promote titanium wear, and the resulting debris may lead to local inflammation, increasing the patient’s risk of peri-implant complications, rather than preventing these complications as intended [64].
Figure 14. Scaling of the implant surface (Bonefit® implant with TPS-coating) leading to titanium particles removal and damage of the implant surface due to instrumentation. (Courtesy: Prof. Dr. G.-H. Nentwig, Frankfurt, Germany).
Peri-implantitis is a feared complication of dental implants as the inflammation and osteolysis surrounding the implant is nearly impossible to reverse [68]. Ultrasonic scaling has been used for subgingival debridement to prevent the progression of peri-implantitis. This progression is associated with an increased bioburden leading to an increased host immune response causing destruction of peri-implant tissues [64]. Ultrasonic scaling attempts may lead to further inflammation as opposed to reversing the progression of the condition. Harrel et al. conducted experiments where sandblasted, large grit, acid-etched implants (SLA) were exposed to ultrasonic scaling in vitro. The coolant water from the ultrasonic scaler was analyzed for titanium particles, and the implant surfaces were analyzed for damage to the SLA coating. They concluded that all implants subjected to ultrasonic scaling released titanium particles into the coolant water and all implants showed damage to the SLA surface pattern. The surface damage using ultrasonics has been previously documented in a variety of other studies as well [69][70][71][72][73][74]. These results suggest that the efficacy of ultrasonic scaling of dental implants should be further researched as the resulting titanium wear and implant surface damage may worsen implant health [75].
Pettersson et al. supported the above conclusions with their study. In this study, both mucosa in periodontitis and peri-implantitis was analyzed for the presence of metal particles. Although there was metal debris in periodontitis mucosa, the presence of titanium was far more pronounced in peri-implantitis tissue biopsies. Although these results were not compared to the mucosa surrounding healthy implants, the substantial difference in the presence of titanium in the two inflammatory conditions indicates that titanium wear may worsen peri-implantitis inflammation and lead to a less favorable prognosis for implant survival.
Implant manufacturers practice various surface modification strategies in an attempt to increase surface roughness of the implants to increase osseointegration[76][77][78][79]. Atomic force microscopy experiments have determined that sandblasted/acid-etched titanium particles have been found to induce a greater inflammatory response and greater differentiation of osteoclasts than lipopolysaccharide from Gram-negative bacteria . In this case, the ability to promote osteoclast differentiation is used to compare inflammatory responses as this is the precursor to bone resorption. Machined discs produce a milder inflammatory response than sandblasted/acid-etched.
References
- Misch, C.E.; Perel, M.L.; Wang, H.-L.; Sammartino, G.; Galindo-Moreno, P.; Trisi, P.; Steigmann, M.; Rebaudi, A.; Palti, A.; Pikos, M.A.; et al. Implant Success, Survival, and Failure: The International Congress of Oral Implantologists (ICOI) Pisa Consensus Conference. Implant. Dent. 2008, 17, 5–15, doi:10.1097/id.0b013e3181676059.
- Apaza-Bedoya, K.; Bijukumar, D.; Benfatti, C.A.M.; Mathew, M.; Da Silva, J.; Souza, J. Adverse local and systemic effect of nanoparticles released from oral and cranio-maxillofacial implants. In Nanostructured Biomaterials for Cranio-Maxillofacial and Oral Applications; Elsevier: Amsterdam, The Netherlands, 2018.
- Schwarz, F.; Derks, J.; Monje, A.; Wang, H.-L. Peri-implantitis. J. Periodontol. 2018, 89, S267–S290, doi:10.1002/jper.16-0350.
- Daubert, D.M.; Weinstein, B.F. Biofilm as a risk factor in implant treatment. Periodontol. 2000 2019, 81, 29–40, doi:10.1111/prd.12280.
- Romanos, G.E.; Delgado-Ruiz, R.; Sculean, A. Concepts for prevention of complications in implant therapy. Periodontol. 2000 2019, 81, 7–17, doi:10.1111/prd.12278.
- Garetto, L.P.; Chen, J.; Parr, J.A.; Roberts, W.E.Remodeling dynamics of bone supporting rigidly fixed titanium implants: a histomorphometric comparison in four species including humans. Implant. Dent. 1995, 4, 235–243, doi:10.1097/00008505-199500440-00002.
- Jepsen, S.; Berglundh, T.; Genco, R.; Aass, A.M.; Demirel, K.; Derks, J.; Figuero, E.; Giovannoli, J.L.; Goldstein, M.; Lambert, F.; et al. Primary prevention of peri-implantitis: Managing peri-implant mucositis. J. Clin. Periodontol. 2015, 42, S152–S157, doi:10.1111/jcpe.12369.
- Lindhe, J.; Meyle, J.; on behalf of Group D of the European Workshop on Periodontology Peri-implant diseases: Consensus Report of the Sixth European Workshop on Periodontology. J. Clin. Periodontol. 2008, 35, 282–285, doi:10.1111/j.1600-051x.2008.01283.x.
- Berglundh, T.; Armitage, G.; Araujo, M.G.; Avila-Ortiz, G.; Blanco, J.; Camargo, P.M.; Chen, S.; Cochran, D.; Derks, J.; Fi-guero, E.; et al. Peri-implant diseases and conditions: Consensus report of workgroup 4 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Clin. Periodontol. 2018, 45, S286–S291, doi:10.1111/jcpe.12957.
- Duraccio, D.; Mussano, F.; Faga, M.G. Biomaterials for dental implants: Current and future trends. J. Mater. Sci. 2015, 50, 4779–4812, doi:10.1007/s10853-015-9056-3.
- Bosshardt, D.; Chappuis, V.; Buser, D. Osseointegration of titanium, titanium alloy and zirconia dental implants: Current knowledge and open questions. Periodontol. 2000 2017, 73, 22–40, doi:10.1111/prd.12179.
- Berbel, L.O.; Banczek, E.D.P.; Karousis, I.K.; Kotsakis, G.; Costa, I. Determinants of corrosion resistance of Ti-6Al-4V alloy dental implants in an In Vitro model of peri-implant inflammation. PLoS ONE 2019, 14, e0210530, doi:10.1371/journal.pone.0210530.
- Velasco-Ortega, E.; Ortiz-García, I.; Jiménez-Merchán, A.; Monsalve-Guil, L.; Muñoz-Guzón, F.; Perez, R.A.; Gil, F.J. Com-parison between Sandblasted Acid-Etched and Oxidized Titanium Dental Implants: In Vivo Study. Int. J. Mol. Sci. 2019, 20, 3267, doi:10.3390/ijms20133267.
- Tuan, R.S.; Lee, F.Y.-I.; Konttinen, Y.T.; Wilkinson, M.J.; Smith, R.L. What are the local and systemic biologic reactions and mediators to wear debris, and what host factors determine or modulate the biologic response to wear particles? J. Am. Acad. Orthop. Surg. 2008, 16, S42–S48, doi:10.5435/00124635-200800001-00010.
- Schlilephake, H.; Lehmann, H.; Kunz, U.; Schmelzeilsen, H. Ultrastructural findings in soft tissues adjacent to titanium plates used in jaw fracture treatment. Int. J. Oral Maxillofac. Surg. 1993, 22, 20–25, doi:10.1016/s0901-5027(05)80350-9.
- Franchi, M.; Bacchelli, B.; Martini, D.; De Pasquale, V.; Orsini, E.; Ottani, V.; Fini, M.; Giavaresi, G.; Giardino, R.; Ruggeri, A. Early detachment of titanium particles from various different surfaces of endosseous dental implants. Biomater. 2004, 25, 2239–2246, doi:10.1016/j.biomaterials.2003.09.017.
- Palazzo, E.; Andreola, S.; Battistini, A.; Gentile, G.; Zoja, R. Release of metals from osteosynthesis implants as a method for identification: Post-autopsy histopathological and ultrastructural forensic study. Int. J. Leg. Med. 2009, 125, 21–26, doi:10.1007/s00414-009-0394-z.
- Romanos, G.E.; Zamparini, F.; Spinelli, A.G.M.; Gandolfi, M.G.; Prati, C. Esem-Edx Microanalysis at Bone-Implant Interface on Postmortem Retrieved Immediately Loaded Implants. Int. J. Oral Maxillofac. Implants (Submitted) 2020.
- Apaza-Bedoya, K.; Tarce, M.; Benfatti, C.A.M.; Henriques, B.; Mathew, M.T.; Teughels, W.; Souza, J.C.M. Synergistic interac-tions between corrosion and wear at titanium-based dental implant connections: A scoping review. J. Periodontal Res. 2017, 52, 946–954, doi:10.1111/jre.12469.
- Quirynen, M.; Van Steenberghe, D. Bacterial colonization of the internal part of two-stage implants. An in vivo study . Clin. Oral Implant. Res. 1993, 4, 158–161, doi:10.1034/j.1600-0501.1993.040307.x.
- Dibart, S.; Warbington, M.; Su, M.F.; Skobe, Z. In vitro evaluation of the implant-abutment bacterial seal: The locking taper system. Int. J. Oral Maxillofac. Implant. 2005, 20, 732–737.
- Prado, A.M.; Pereira, J.; Henriques, B.; Benfatti, C.A.; Magini, R.S.; López-López, J.; Souza, J.C.M. Biofilm Affecting the Me-chanical Integrity of Implant-Abutment Joints. Int. J. Prosthodont. 2016, 29, 381–383, doi:10.11607/ijp.4759.
- Schwarz, M.S. Mechanical complications of dental implants. Clin. Oral Implant. Res. 2000, 11, 156–158, doi:10.1034/j.1600-0501.2000.011s1156.x.
- Schmitt, C.M.; Nogueira-Filho, G.; Tenenbaum, H.C.; Lai, J.Y.; Brito, C.; Doring, H.; Nonhoff, J. Performance of conical abutment (Morse Taper) connection implants: A systematic review. J. Biomed. Mater. Res. Part A 2014, 102, 552–574.
- Romanos, G.E.; Biltucci, M.T.; Kokaras, A.; Paster, B.J. Bacterial Composition at the Implant-Abutment Connection under Loading in vivo. Clin. Implant. Dent. Relat. Res. 2014, 18, 138–145, doi:10.1111/cid.12270.
- Ericsson, I.; Persson, L.G.; Berglundh, T.; Marinello, C.P.; Lindhe, J.; Klinge, B. Different types of inflammatory reactions in peri-implant soft tissues. J. Clin. Periodontol. 2005, 22, 255–261, doi:10.1111/j.1600-051x.1995.tb00143.x.
- Koutouzis, T.; Mesia, R.; Calderon, N.; Wong, F.; Wallet, S. The Effect of Dynamic Loading on Bacterial Colonization of the Dental Implant Fixture–Abutment Interface: An In Vitro Study. J. Oral Implant. 2014, 40, 432–437, doi:10.1563/aaid-joi-d-11-00207.
- Liu, Y.; Wang, J. Influences of microgap and micromotion of implant–abutment interface on marginal bone loss around im-plant neck. Arch. Oral Biol. 2017, 83, 153–160, doi:10.1016/j.archoralbio.2017.07.022.
- Pettersson, M.; Pettersson, J.; Johansson, A.; Thorén, M.M. Titanium release in peri-implantitis. J. Oral Rehabilitation 2019, 46, 179–188, doi:10.1111/joor.12735.
- Zipprich, H.; Miatke, S.; Hmaidouch, R.; Lauer, H.-C. A New Experimental Design for Bacterial Microleakage Investigation at the Implant-Abutment Interface: An In Vitro Study. Int. J. Oral Maxillofac. Implant. 2016, 31, 37–44, doi:10.11607/jomi.3713.
- Zipprich, H.; Weigl, P.; Ratka, C.; Lange, B.; Lauer, H.-C. The micromechanical behavior of implant-abutment connections under a dynamic load protocol. Clin. Implant. Dent. Relat. Res. 2018, 20, 814–823, doi:10.1111/cid.12651.
- Batten, R. Bone repair and fracture healing in man. Inj. 1982, 13, 532–533, doi:10.1016/0020-1383(82)90200-5.
- Piattelli, A.; Corigliano, M.; Scarano, A.; Costigliola, G.; Paolantonio, M. Immediate Loading of Titanium Plasma-Sprayed Implants: An Histologic Analysis in Monkeys. J. Periodontol. 1998, 69, 321–327, doi:10.1902/jop.1998.69.3.321.
- Romanos, G.E.; Toh, C.G.; Siar, C.H.; Wicht, H.; Yacoob, H.; Nentwig, G.-H. Bone-Implant Interface Around Titanium Im-plants Under Different Loading Conditions: A Histomorphometrical Analysis in the Macaca fascicularis Monkey. J. Periodon-tol. 2003, 74, 1483–1490, doi:10.1902/jop.2003.74.10.1483.
- Romanos, G.E.; Toh, C.G.; Siar, C.H.; Swaminathan, D.; Ong, A.H.; Donath, K.; Yaacob, H.; Nentwig, G.-H. Peri-Implant Bone Reactions to Immediately Loaded Implants. An Experimental Study in Monkeys. J. Periodontol. 2001, 72, 506–511, doi:10.1902/jop.2001.72.4.506.
- Romanos, G.E. Wound healing in immediately loaded implants. Periodontol. 2000 2015, 68, 153–167, doi:10.1111/prd.12058.
- Korb, J.L.; Olson, L.D. ASM Metals Handbook; ASM: Materials Park, OH, USA, 2005; Volume 13—Corrosion.
- Daubert, D.M.; Pozhitkov, A.; McLean, J.; Kotsakis, G. Titanium as a modifier of the peri-implant microbiome structure. Clin. Implant. Dent. Relat. Res. 2018, 20, 945–953, doi:10.1111/cid.12676.
- Safioti, L.M.; Kotsakis, G.A.; Pozhitkov, A.E.; Chung, W.O.; Daubert, D.M. Increased Levels of Dissolved Titanium Are Asso-ciated With Peri-Implantitis—A Cross-Sectional Study. J. Periodontol. 2017, 88, 436–442, doi:10.1902/jop.2016.160524.
- Black, J. Does corrosion matter? J. Bone Jt. Surgery. Br. Vol. 1988, 517–520, doi:10.1302/0301-620x.70b4.3403590.
- Jonas, G.F.L.; Henkel, C.R.K.-O.; Holzhuter, G.; Mathieu, H.J. Biodegradation of Titanium Implants After Long-Time Inser-tion Used for the Treatment of Fractured Upper and Lower Jaws Through Osteosynthesis: Element Analysis by Electron Mi-croscopy and EDX or EELS. Ultrastruct. Pathol. 2001, 25, 375–383, doi:10.1080/019131201317101252.
- Shah, R.; Penmetsa, D.S.L.; Thomas, R.; Mehta, D.S. Titanium Corrosion: Implications For Dental Implants. Eur. J. Prostho-dont. Restor. Dent. 2016, 24, 171–180.
- Nikolopoulou, F. Saliva and Dental Implants. Implant. Dent. 2006, 15, 372–376, doi:10.1097/01.id.0000239320.57403.8d.
- Juanito, G.; Morsch, C.; Benfatti, C.; Fredel, M.; Magini, R.; Effect of Fluoride and Bleaching Agents on the Degradation of Titanium: Literature Review. Dentistry 2015, doi:10.4172/2161-1122.100027.
- Souza, J.C.M.; Ponthiaux, P.; Henriques, M.; Oliveira, R.; Teughels, W.; Celis, J.-P.; Rocha, L.A. Corrosion behaviour of tita-nium in the presence of Streptococcus mutans. J. Dent. 2013, 41, 528–534, doi:10.1016/j.jdent.2013.03.008.
- Souza, J.C.M.; Barbosa, S.; Ariza, E.; Celis, J.-P.; Rocha, L. Simultaneous degradation by corrosion and wear of titanium in artificial saliva containing fluorides. Wear 2012, 82–88, doi:10.1016/j.wear.2012.05.030.
- Mathew, M.T.; Abbey, S.; Hallab, N.J.; Hall, D.J.; Sukotjo, C.; Wimmer, M.A. Influence of pH on the tribocorrosion behavior of CpTi in the oral environment: Synergistic interactions of wear and corrosion. J. Biomed. Mater. Res. Part B: Appl. Biomater. 2012, 100, 1662–1671, doi:10.1002/jbm.b.32735.
- Kim, K.T.; Eo, M.Y.; Nguyen, T.T.H.; Kim, S.M. General review of titanium toxicity. Int. J. Implant. Dent. 2019, 5, 1–12, doi:10.1186/s40729-019-0162-x.
- Tengvall, P.; Lundström, I. Physico-chemical considerations of titanium as a biomaterial. Clin. Mater. 1992, 9, 115–134, doi:10.1016/0267-6605(92)90056-y.
- Takemoto, S.; Hattori, M.; Yoshinari, M.; Kawada, E.; Oda, Y. Discoloration of titanium alloy in acidic saline solutions with peroxide. Dent. Mater. J. 2013, 32, 19–24, doi:10.4012/dmj.2012-194.
- Zhang, Y.; Addison, O.; Yu, F.; Troconis, B.C.R.; Scully, J.R.; Davenport, A.J. Time-dependent Enhanced Corrosion of Ti6Al4V in the Presence of H2O2 and Albumin. Sci. Rep. 2018, 8, 1–11, doi:10.1038/s41598-018-21332-x.
- Fantone, J.C.; Ward, P.A. Role of oxygen-derived free radicals and metabolites in leukocyte-dependent inflammatory reac-tions. Am. J. Pathol. 1982, 107, 395–418.
- Rajamäki, K.; Nordström, T.; Nurmi, K.; Åkerman, K.E.O.; Kovanen, P.T.; Öörni, K.; Eklund, K.K. Extracellular Acidosis Is a Novel Danger Signal Alerting Innate Immunity via the NLRP3 Inflammasome. J. Biol. Chem. 2013, 288, 13410–13419, doi:10.1074/jbc.m112.426254.
- Rodrigues, D.C.; Valderrama, P.; Jr, T.G.W.; Palmer, K.L.; Thomas, A.; Sridhar, S.; Adapalli, A.; Burbano, M.; Wadhwani, C. Titanium Corrosion Mechanisms in the Oral Environment: A Retrieval Study. Mater. 2013, 6, 5258–5274, doi:10.3390/ma6115258.
- Rodrigues, D.C.; Urban, R.M.; Jacobs, J.J.; Gilbert, J.L. In vivosevere corrosion and hydrogen embrittlement of retrieved mod-ular body titanium alloy hip-implants. J. Biomed. Mater. Res. Part B: Appl. Biomater. 2008, 88, 206–219, doi:10.1002/jbm.b.31171.
- Buly, R.L.; Huo, M.H.; Salvati, E.; Brien, W.; Bansal, M. Titanium wear debris in failed cemented total hip arthroplasty. J. Arthroplast. 1992, 7, 315–323, doi:10.1016/0883-5403(92)90056-v.
- Diomidis, N.; Ponthiaux, P.; Wenger, F.; Celis, J.-P. A methodology for the assessment of the tribocorrosion of passivating metallic materials. Lubr. Sci. 2009, 21, 53–67, doi:10.1002/ls.73.
- Cruz, H.V.; Souza, J.C.M.; Henriques, M.; Rocha, L.A. Tribocorrosion and Bio-Tribocorrosion in the Oral Environment: The Case of Dental Implants. In Biomedical Tribology; Nova Science Publishers: Hauppauge, NY, USA, 2011.
- Nuevo-Ordoñez, Y.; Montes-Bayón, M.; González, E.B.; Sanz-Medel, A. Titanium preferential binding sites in human serum transferrin at physiological concentrations. Metallomics 2011, 3, 1297–1303, doi:10.1039/c1mt00109d.
- Matusiewicz, H. Potential release of in vivo trace metals from metallic medical implants in the human body: From ions to nanoparticles—A systematic analytical review. Acta Biomater. 2014, 10, 2379–2403, doi:10.1016/j.actbio.2014.02.027.
- Oliveira, M.N.; Schunemann, W.V.H.; Mathew, M.T.; Henriques, B.; Magini, R.S.; Teughels, W.; Souza, J.C.M. Can degrada-tion products released from dental implants affect peri-implant tissues? J. Periodontal Res. 2018, 53, 1–11, doi:10.1111/jre.12479.
- Iavicoli, I.; Leso, V.; Fontana, L.; Bergamaschi, A. Toxicological effects of titanium dioxide nanoparticles: A review of in vitro mammalian studies. Eur. Rev. Med Pharmacol. Sci. 2011, 15.
- Makihira, S.; Mine, Y.; Nikawa, H.; Shuto, T.; Iwata, S.; Hosokawa, R.; Kamoi, K.; Okazaki, S.; Yamaguchi, Y. Titanium ion induces necrosis and sensitivity to lipopolysaccharide in gingival epithelial-like cells. Toxicol. Vitr. 2010, 24, 1905–1910, doi:10.1016/j.tiv.2010.07.023.
- Eger, M.; Sterer, N.; Liron, T.; Kohavi, D.; Gabet, Y. Scaling of titanium implants entrains inflammation-induced osteolysis. Sci. Rep. 2017, 7, 39612, doi:10.1038/srep39612.
- A Heitz-Mayfield, L.J.; Mombelli, A. The Therapy of Peri-implantitis: A Systematic Review. Int. J. Oral Maxillofac. Implant. 2014, 29, 325–345, doi:10.11607/jomi.2014suppl.g5.3.
- A Heitz-Mayfield, L.J.; Needleman, I.; E Salvi, G.; E Pjetursson, B. Consensus Statements and Clinical Recommendations for Prevention and Management of Biologic and Technical Implant Complications. Int. J. Oral Maxillofac. Implant. 2014, 29, 346–350, doi:10.11607/jomi.2013.g5.
- Machtei, E.E.; Romanos, G.; Kang, P.; Travan, S.; Schmidt, S.; Papathanasiou, E.; Tatarakis, N.; Tandlich, M.; Liberman, L.H.; Horwitz, J.; et al. Repeated delivery of chlorhexidine chips for the treatment of periimplantitis: A multicenter, randomized, comparative clinical trial. J. Periodontol. 2020, doi:10.1002/jper.20-0353.
- Esposito, M.; Grusovin, M.G.; Worthington, H.V. Interventions for replacing missing teeth: Treatment of peri-implantitis. Cochrane Database Syst. Rev. 2012, 1, CD004970, doi:10.1002/14651858.cd004970.pub5.
- Meschenmoser, A.; D’Hoedt, B.; Meyle, J.; Elßner, G.; Korn, D.; Hämmerle, H.; Schulte, W. Effects of Various Hygiene Pro-cedures on the Surface Characteristics of Titanium Abutments. J. Periodontol. 1996, 67, 229–235, doi:10.1902/jop.1996.67.3.229.
- Augthun, M.; Tinschert, J.; Huber, A. In Vitro Studies on the Effect of Cleaning Methods on Different Implant Surfaces. J. Periodontol. 1998, 69, 857–864, doi:10.1902/jop.1998.69.8.857.
- Hallmon, W.; Waldrop, T.; Meffert, R.; Wade, B. A comparative study of the effects of metallic, nonmetallic, and sonic in-strumentation on titanium abutment surfaces. Implant. Dent. 1996, 5, 296–100, doi:10.1097/00008505-199600540-00030.
- Kocher, T.; Langenbeck, M.; Rühling, A.; Plagmann, H.-C. Subgingival polishing with a teflon-coated sonic scaler insert in comparison to conventional instruments as assessed on extracted teeth. J. Clin. Periodontol. 2000, 27, 243–249, doi:10.1034/j.1600-051x.2000.027004243.x.
- Mengel, R.; E Buns, C.; Mengel, C.; Flores-De-Jacoby, L. An in vitro study of the treatment of implant surfaces with different instruments. Int. J. Oral Maxillofac. Implant. 1998, 13, 91–6.
- Rapley, J.W.; Swan, R.H.; Hallmon, W.W.; Mills, M.P. The surface characteristics produced by various oral hygiene instru-ments and materials on titanium implant abutments. Int. J. Oral Maxillofac. Implant. 1990, 5, 47–52.
- Harrel, S.K.; Jr, T.G.W.; Pandya, M.; Diekwisch, T.G.H. Titanium particles generated during ultrasonic scaling of implants. J. Periodontol. 2019, 90, 241–246, doi:10.1002/jper.18-0230.
- Schuler, M.; Owen, G.R.; Hamilton, D.W.; De Wild, M.; Textor, M.; Brunette, D.M.; Tosatti, S.G. Biomimetic modification of titanium dental implant model surfaces using the RGDSP-peptide sequence: A cell morphology study. Biomater. 2006, 27, 4003–4015, doi:10.1016/j.biomaterials.2006.03.009.
- Schwartz, Z.; Martin, J.Y.; Dean, D.D.; Simpson, J.; Cochran, D.L.; Boyan, B.D. Effect of titanium surface roughness on chondrocyte proliferation, matrix production, and differentiation depends on the state of cell maturation. J. Biomed. Mater. Res. 1996, doi:10.1002/(SICI)1097-4636(199602)30:23.0.CO;2-R.
- Zreiqat, H.; Valenzuela, S.M.; Ben Nissan, B.; Roest, R.; Knabe, C.; Radlanski, R.J.; Renz, H.; Evans, P.J. The effect of surface chemistry modification of titanium alloy on signalling pathways in human osteoblasts. Biomater. 2005, 26, 7579–7586, doi:10.1016/j.biomaterials.2005.05.024.
- Le Guillou-Buffello, D.; Bareille, R.; Gindre, M.; Sewing, A.; Laugier, P.; Amédée, J. Additive Effect of RGD Coating to Func-tionalized Titanium Surfaces on Human Osteoprogenitor Cell Adhesion and Spreading. Tissue Eng. Part A 2008, 14, 1445–1455, doi:10.1089/ten.tea.2007.0292.