| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Rafał Hrynkiewicz | + 2225 word(s) | 2225 | 2020-10-23 06:29:44 | | | |
| 2 | Bruce Ren | -68 word(s) | 2157 | 2021-01-20 03:02:25 | | | | |
| 3 | Bruce Ren | -68 word(s) | 2157 | 2021-01-20 03:05:16 | | |
Video Upload Options
Urinary bladder cancer is one of the most important cancers of the urinary-sex system, causing high morbidity and mortality in many patients. Various methods of treating this type of cancer have been developed over the years. The most popular is the highly effective method using Bacillus Calmette-Guerin, which has an effective effect on a large percentage of patients. However, due to the genetic instability of bladder cancer and the individual needs of patients, different methods of treatment are being sought. Immunological control points are molecules on the surface of the cell that affect the immune response and reduce the strength of the immune response. Among these checkpoints, PD-1 inhibitors (the protein that is programmed to kill cell-1) /PD-L1 (the ligand of the protein that is programmed to kill cell 1) are intended to block these molecules, resulting in activation of T cells, and bladder cancer have been described as the use of atezolizumab, awelumab, durwalumab, niwolumab and pembrolizumab. The inhibition of another key immunological control point, CTLA-4 (cytotoxic T-lymphocyte antigen), may result in mobilisation of the immune system against bladder cancer, and anti-CTLA-4 antibodies discussed the use of Ipilimumab and Tremelimumab. In addition, there are several different approaches to the effective treatment of bladder cancer, such as the use of ganciclovir kinase inhibitors and mTOR (mammalian rapamycin target), IL-12 (interleukin-12) and COX-2 (cyclooxygenase-2). Genetic therapy and disruption of different signal paths are currently being studied. Studies suggest that combining several methods increases the efficacy of treatment and positive results in the patient.
1. Entry
Urinary bladder cancer (BC) is the sixth most widely recognised cancer in men around the world and the tenth when considered together between men and women [1]. The age-standardised global maturity index (per 100 000 persons / years) is 9,6 for men and 2,4 for women [1]. In Europe, the general age standard is 20,2 for men and 4,3 for women. Greece has the highest standard of age for all European countries (40,4 for men and 4,5 for women) and Austria has the lowest (9,9 for men and 3,0 for women) [1]. In 2018 year around the world, around 550 000 new BC cases were diagnosed, with 200 000 deaths [1]. The incidence of BC has increased in many European countries, although mortality rates have fallen in more developed regions. As the population ages and grows, the absolute incidence of BC can continue to grow in European countries [2].
Smoking is the most important risk factor for BC. This is related to 50-65% male and 20-30% female cases. The incidence of BC is directly related to the length of smoking and the number of cigarettes smoked per day [2][3]. Professional factors are considered to be the second most important risk factor for BC [3]. Workers exposed to aromatic amines, polycyclic aromatic hydrocarbons, tobacco and tobacco smoke, combustion products and heavy metals are at increased risk [4].
Urinary bladder urotelial cancer is the most common histological type of cancer. In more than 70% cases, it is recognised at a non-invasive stage requiring only minimal invasive local treatment. Unfortunately, the disease has a high rate of relapse and repeated treatment may be necessary. Conversely, the stages of invasive and metastatic disease require multi-modal treatment strategies, including surgical treatment and chemotherapy under non-adjuvant, complementary or palliative conditions [5].
Cancer treatment methods that modify the immune status have a prominent place in oncology in recent years. Immunotherapy is usually used to complement conventional cancer treatments such as surgery, chemotherapy, and radiotherapy. For some cancers, immunotherapies are used as first-line treatment [6]. Immunotherapy in cancer treatment is a method that involves the patient's immune system to modify or increase defense mechanisms against a developing cancer [6][7]. The first clinical application of immunotherapy was recorded in the 1890s, when William Coley first used a bacterial preparation called Coley toxin. The effect of clinical trials was small. The toxin provided an early demonstration of the potential to produce an antitumor response by using the patient's immune system [6]. It was not until the mid-20th century that immunotherapies gained importance as part of standard cancer treatment, although they showed significant toxicity. Therapies were associated with the beginning of cell therapy with the development of bone marrow transplantation by Fritz Bach et al. in the 1960s and the production, testing, and approval in clinical trials of a high dose of IL-2 (interleukin 2) for metastatic renal and melanoma in the 1990s [8][9]. Currently, several types of immunotherapy are used to treat cancer, including immune checkpoint inhibitors, T-cell transfer therapy, monoclonal antibodies, treatment vaccines, and immune system modulators [7].
Research for best-tailored treatment for BC is ongoing, and immunotherapy seems to be the most promising prospect.
2. Bacillus Calmette–Guerin (BCG)
Bacillus Calmette–Guerin (BCG) is a weakened strain of Mycobacterium bovis. However, according to the European Association of Urology, there are currently 10 strains used for BCG therapy, but none of them has shown superiority over the others [10].
BCG use as cancer treatment was investigated in an animal model in 1974 [11], and in 1976 the first report on the successful use of BCG in BC was published [12]. In 1980 Lamm et al. reported that the use of BCG therapy following transurethral resection of bladder tumors (TURBT) reduces the chance of relapse compared to patients receiving only TURBT [13][14].
Currently, intravesical therapy with BCG is standard practice in the treatment of nonmuscle invasive BC (NMIBC), including in situ cancer, high-grade papillary tumors, and invasive plaque-proprious tumors [15]. Noninvasive tumors account for 70–80% of BC cases [16][17]. The standard treatment for this type of cancer is TURBT, followed by intravesical treatment with BCG or chemotherapy, as described by Lamm et al. years ago [14]. Whether BCG or chemotherapy is used depends on the progression and recurrence of the disease [18][19][20].
Although the BCG vaccine has been used to treat BC for decades, its mechanism of action is not yet fully understood [21]. BC cells themselves may play a role involving the attachment and internalization of BCG, the presentation of BCG and cancer antigens to cells of the immune system, and the mass release of cytokines and chemokines that occurs during BCG therapy [22][23]. What is certain is that BCG causes a strong innate immune response that leads to long-term adaptive immunity [24]. BCG therapy elicits an inflammatory reaction involving different immune cell subsets that kill cancer cells by direct cytotoxicity or by the secretion of toxic compounds, like the tumor necrosis factor-inducing ligand. The immune cell subsets that may be involved include CD4+ and CD8+ lymphocytes, NK (natural killer) cells, granulocytes, macrophages, and dendritic cells. Some cancer cells are also killed directly by the BCG.
T lymphocytes are present in the inflammatory infiltrate in the bladder of BCG-treated patients [25], and there is evidence that natural killer cells are cytotoxic against BCG-infected BC cells [26]. Granulocytes are also present in the inflammatory infiltrate in the bladder. In the mouse model, they were necessary for a proper immune response [27][28], along with CD4+ and CD8+ T cells [29]. Furthermore, BCG-exposed dendritic cells may stimulate the cytotoxicity of T lymphocytes against BCG-infected cancer cells [30]. Finally, macrophages are another component of the inflammatory infiltrate in the bladder of BCG-treated patients, and they are cytotoxic against cancer cells when stimulated by BCG [31][32].
BCG immunotherapy gives a high percentage of positive response, which is 55–65% for high-risk papillary tumors and 70–75% for carcinoma in situ (CIS) [33][34][35][36]. Unfortunately, as many as 25–45% of patients will not benefit from BCG therapy. In addition, about 40% of patients have relapse despite initial successes with BCG [21]. Despite the constant development of medicine and technology, the percentage of patients in whom BCG therapy does not have a positive effect remains similar to that reported in the early 1990s (30–35% of patients remain resistant to this method of treatment) [31][32]. Currently, patients can be divided into three groups: BCG resistant, BCG relapse, or BCG intolerance [37]. The differences between these groups remain extremely important because they can provide information on the response of individual patients to BCG therapy. Many studies are underway, including those showing promising early results in selected patients who do not respond to BCG but long-term results are still distant.
For this reason, understanding the immunobiology of BCG-induced tumor immunity is necessary to tailor BCG treatment to specific patients and to improve efficacy as well as to reduce intolerance to this therapy.
While some researchers are still trying to refine the BCG treatment method, some teams have focused on other, equally promising, immunotherapies against bladder cancer.
3. Checkpoints’ Inhibitors Pathway
Responsiveness to checkpoint inhibitors (mainly PD-1/PD-L1 (programmed cell death protein-1/ programmed cell death protein ligand 1) and CTLA4 (cytotoxic T cell antigen)) is the key to successful cancer therapy, but still not every patient achieves clinical benefit [38]. Immune checkpoint efficacy is affected by various factors, among which tumor genomics, host germline genetics, PD-L1 levels, and gut microbiome may be enumerated [38]. Generally, in tumors, mutated or incorrectly expressed proteins are processed via the immunoproteasome into peptides that are usually loaded onto MHC (major histocompatibility complex) class I molecules, which further not always are able to elicit CD8+ T cell response [38]. This may lead to generating MHC-presented immunogenic neoepitopes [38]. It was shown, that once the intratumor heterogeneity rises, neoantigen-expressing clones became more homogenous with the differential expression of PD-L1 [39].
Also, mutations on several signaling pathways may influence the effectiveness of immune checkpoints inhibitors [39]. This was confirmed in bladder cancer for Janus kinase (JAK) signaling pathway, where negative regulator of JAK-SOCS3 (suppressor of cytokine signaling 3) was reduced, with simultaneous upregulation of miR-221 (micro RNA), leading to enhanced cell apoptosis, and attenuated cell proliferation [40]. Such dysregulation of JAK (Janus activated kinase)-STAT (signal transducer and activator of transcription) signaling pathway was also confirmed in patients with bladder cancer with high STAT3 expression [41].
Influence of the BC on cell cycle was also noticed, while some proteins like DEPDC1 (DEP domain containing 1) and MPHOSPH1 (M-phase phosphoprotein 1) are usually overexpressed in bladder cancer cells [42].
Overall, bladder cancer is a genetically heterogenous disease, with a high rate of somatic mutations, including genes involved in cell-cycle regulation, chromatin regulation, and signaling pathways [43]. In bladder cancer, according to The Cancer Genome Atlas (TCGA) Research Network, mutations of genes not significantly mutating in any other type of cancer were noticed [43]. Among most frequent mutations, one can enumerate aneuploidy, chromosomal instability, and fractional allelic losses [44]. Thus, those differences in the molecular features of BC, together with personal characteristic of patients, may seriously influence the efficacy of the use of the immune checkpoint inhibitors.
3.1. PD-1/PD-L1
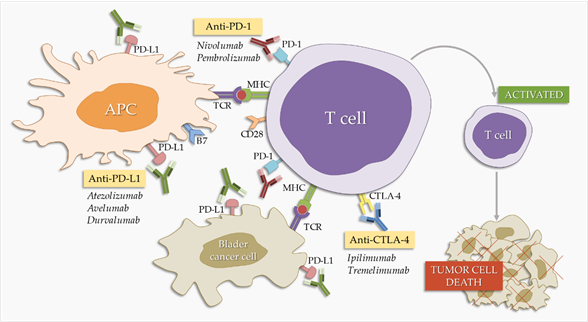
Programmed cell death protein 1 (PD-1) and its ligands, programmed death ligand 1 (PD-L1) and 2 (PD-L2), are part of an immune system checkpoint, which negatively regulates the immune system to weaken its response to antigens. PD-1 is expressed on the surface of activated T and B lymphocytes and macrophages (Figure 1) and PD-L1 is highly expressed by antigen-presenting cells [45]. PD-1 binding to PD-L1 blocks the activation of T cells, thereby reducing the production of IL-2 (interleukin-2) and interferon gamma [45]. This promotes self-tolerance by preventing the immune system from indiscriminately attacking the cells of the body, but it can also stop the immune system from attacking cancer cells that express PD-L1. PD-1/PD-L1 inhibitors are antibodies that block either of these two molecules, cancelling the checkpoint activity and thus resulting in T cell activation [46]. They were first introduced as second-line therapy in BC treatment, but are slowly establishing themselves as first-line therapy [47]. There are currently three PD-L1 inhibitors and two PD-1 inhibitors approved by the FDA (Food and Drug Administration) for BC treatment (Table 1).
Figure 1. Effect of checkpoint inhibitors in bladder cancer treatment. PD-1/PD-L1 and CTLA-4 blockers interfere with the immune system’s inhibitory checkpoint molecules, leading to T cell activation and tumor cell death. APC: antigen-presenting cells.
Table 1. List of approved checkpoint inhibitors used in bladder cancer treatment.
|
Compound |
Trade name |
Company |
Target |
Date of approval |
Clinical trial leading to approval |
|
Atezolizumab |
Tecentriq |
Genentech |
PD-L1 |
2016 |
IMVigor210 [48] |
|
Avelumab |
Bavencio |
Merck |
PD-L1 |
2017 |
JAVELIN [49] |
|
Durvalumab |
Imfinzi |
AstraZeneca |
PD-L1 |
2017 |
Study 1108 [50] |
|
Nivolumab |
Opdivo |
Bristol-Meyers Squibb |
PD-1 |
2017 |
CheckMate 275 [51] |
|
Pembrolizumab |
Keytruda |
Merck |
PD-1 |
2019 |
KEYNOTE-057 [52] |
|
Ipilimumab |
Yervoy |
Bristol-Meyers Squibb |
CTLA-4 |
2019 |
NCT01524991 [53] |
PD-L1: programmed death ligand 1; PD-1: programmed cell death protein 1; CTLA-4: Cytotoxic T lymphocyte-associated protein 4.
3.2. Anti-CTLA-4 Antibodies
Cytotoxic T lymphocyte-associated protein 4 (CTLA-4) is a surface molecule expressed by activated T cells. CTLA-4 binds B7.1 and B7.2 ligands, which are expressed on B lymphocytes, dendritic cells, and macrophages . CTLA-4 is a co-stimulant necessary for the activation of T lymphocytes [54][55]. It negatively regulates the immune response, but its mechanism of action is not yet fully understood. However, because it is structurally related to CD28, one suggestion is that it competes with CD28 in terms of ligand binding. Another suggestion involves the direct inhibition of CTLA-4 cytoplasmic tail signaling [56][57][58].
Zahamowanie CTLA-4 może zwiększyć regulację odpowiedzi immunologicznej na BC. Jest to hipoteza leżąca u podstaw trwających badań nad przeciwciałami anty-CTLA-4, które mają być stosowane jako pojedyncze środki w leczeniu BC (ryc. 1).
Szczepionkę S-288310 można zastosować alternatywnie. Działa poprzez aktywację cytotoksycznych limfocytów T. Badania kliniczne pokazują, że metoda ta jest wysoce skuteczna, a szczepionka była dobrze tolerowana przez pacjentów. Kryterium zastosowania szczepionki S-288310 była zwiększona ekspresja genu HLA-A 24:02 (ludzki antygen leukocytów) u pacjentów [59].
References
- Cancer Today (powered by GLOBOCAN 2018) IARC CancerBase No. 15; Ferlay J, Ervik M, Lam F, Colombet M, Mery L, Pińeros M, Znaor A, Soerjomataram I, B.F., Ed.; 2018; ISBN 978-92-832-0453-4.
- Kim, H.S.; Seo, H.K. Immune checkpoint inhibitors for urothelial carcinoma. Investig. Clin. Urol. 2018, 59, 285-296, doi: 10.4111/icu.2018.59.5.285.
- Saginala, K.; Barsouk, A.; Aluru, J.S.; Rawla, P.; Padala, S.A.; Barsouk, A. Epidemiology of Bladder Cancer. Med. Sci. 2020, 8, 15, doi: 10.3390/medsci8010015.
- Iliou, A.; Panagiotakis, A.; Giannopoulou, A.F.; Benaki, D.; Kosmopoulou, M.; Velentzas, A.D.; Tsitsilonis, O.E.; Papassideri, I.S.; Voutsinas, G.E.; Konstantakou, E.G.; Gikas, E.; Mikros, E.; Stravopodis, D.J. Malignancy Grade-Dependent Mapping of Metabolic Landscapes in Human Urothelial Bladder Cancer: Identification of Novel, Diagnostic, and Druggable Biomarkers. Int. J. Mol. Sci. 2020, 21, 1892, doi: 10.3390/ijms21051892.
- Chien, T.-M.; Chan, T.-C.; Huang, S. .-H.; Yeh, B.-W.; Li, W.-M.; Huang, C.-N.; Li, C.-C.; Wu, W.-J.; Li, C.-F. Role of Microtubule-Associated Protein 1b in Urothelial Carcinoma: Overexpression Predicts Poor Prognosis. Cancers 2020, 12, 630, doi:10.3390/cancers12030630
- Jagodinsky, J.C.; Harari, P.M.; Morris, Z.S. The Promise of Combining Radiation Therapy with Immunotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2020, doi: 10.1016/j.ijrobp.2020.04.023.
- National Cancer Institute. Available online: https://www.cancer.gov/aboutcancer/treatment/types/immun
- otherapy#what-are-the-types-of-immunotherapy (accessed on 28.04.2020).
- Fyfe, G.A.; Fisher, R.I.; Rosenberg, S.A.; Sznol, M.; Parkinson, D.R.; Louie, A.C. Long-term response data for 255 patients with metastatic renal cell carcinoma treated with high-dose recombinant interleukin-2 therapy. J. Clin. Oncol. 1996, 14, 2410-2411, doi: 10.1200/JCO.1996.14.8.2410.
- Atkins, M.B.; Lotze, M.T.; Dutcher, J.P.; Fisher, R.I. Weiss, G.; et al. High-dose recombinant interleukin 2 therapy for patients with metastatic melanoma: analysis of 270 patients treated between 1985 and 1993. J. Clin. Oncol. 1999, 17, 2105-2116. doi: 10.1200/JCO.1999.17.7.2105.
- EAU Guidelines on Non-muscle-invasive Bladder Cancer (TaT1 and CIS) 2019. European Association of Urology. Available online: https://uroweb.org/wp-content/uploads/05-Non-muscle-InvasiveBC_TaT1_201
- 9v2-1.pdf (Accessed on 28 April 2020).
- Farina, M.S.; Lundgren, K.T.; Bellmunt, J. Immunotherapy in Urothelial Cancer: Recent Results and Future Perspectives. Drugs 2017, 77, 1077–1089, doi: 10.1007/s40265-017-0748-7.
- Morales, A.; Eidinger, D.; Bruce, A.W. Intracavitary Bacillus Calmette-guerin in the Treatment of Superficial Bladder Tumors. J. Urol. 1976, 116, 180–182, doi: 10.1016/s0022-5347(17)58737-6
- Song, D.; Powles, T.; Shi, L.; Zhang, L.; Ingersoll, M.A.; Lu, Y.J. Bladder cancer, a unique model to understand cancer immunity and develop immunotherapy approaches. J. Pathol. 2019, 249, 151–165, doi: 10.1002/path.5306.
- Lamm, D.L.; Thor, D.E.; Harris, S.C.; Reyna, J.A.; Stogdill, V.D.; Radwin, H.M. Bacillus Calmette-Guerin immunotherapy of superficial bladder cancer. J. Urol. 1980, 124, 38-40, doi: 10.1016/s0022-5347(17)55282-9.
- Zhang, C.; Berndt-Paetz, M.; Neuhaus, J. Identification of Key Biomarkers in Bladder Cancer: Evidence from a Bioinformatics Analysis. Diagnostics 2020, 10, 66, doi: 10.3390/diagnostics10020066.
- Abugomaa, A.; Elbadawy, M.; Yamawaki, H.; Usui, T.; Sasaki, K. Emerging Roles of Cancer Stem Cells in Bladder Cancer Progression, Tumorigenesis, and Resistance to Chemotherapy: A Potential Therapeutic Target for Bladder Cancer. Cells 2020, 9, 235, doi: 10.3390/cells9010235.
- Batista, R.; Vinagre, N.; Meireles, S.; Vinagre, J.; Prazeres, H.; Leão, R.; Máximo, V.; Soares, P. Biomarkers for Bladder Cancer Diagnosis and Surveillance: A Comprehensive Review. Diagnostics 2020, 10, 39, doi: 10.3390/diagnostics10010039.
- Degeorge, K.; Holt, H.; Hodges, S. Bladder Cancer: Diagnosis and Treatment. Am. Fam. Physician 2017, 96, 507–514.
- Bellmunt, J.; Orsola, A.; Leow, J.J.; Wiegel, T.; De Santis, M.; Horwich, A. Bladder cancer: ESMO Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2014, 25, iii40-iii48, doi: 10.1093/annonc/mdu223.
- Borkowska, E.M.; Konecki, T.; Pietrusiński, M.; Borowiec, M.; Jabłonowski, Z. MicroRNAs Which Can Prognosticate Aggressiveness of Bladder Cancer. Cancers 2019, 11, 1551, doi: 10.3390/cancers11101551.
- Crispen, P.L.; Kusmartsev, S. Mechanisms of immune evasion in bladder cancer. Cancer Immunol. Immunother. 2019, 69, 3-14, doi: 10.1007/s00262-019-02443-4.
- De Boer, E.C.; De Jong, W.H.; Steerenberg, P.A.; Aarden, L.A.; Tetteroo, E.; De Groot, E.R.; Van der Meijden, A.P.M.; Vegt, P.D.J.; Debruyne, F.M.J.; Ruitenberg, E.J. Induction of urinary interleukin-1 (IL-1), IL-2, IL-6, and tumour necrosis factor during intravesical immunotherapy with bacillus Calmette-Guérin in superficial bladder cancer. Cancer Immunol. Immunother. 1992, 34, 306–312.
- Luo, Y.; Chen, X.; O’Donnell, M.A. Mycobacterium bovis bacillus Calmette-Guérin (BCG) induces human CC- and CXC-chemokines in vitro and in vivo. Clin. Exp. Immunol. 2006, 147, 370–378, doi: 10.1111/j.1365-2249.2006.03288.x.
- Kawai, K.; Miyazaki, J.; Joraku, A.; Nishiyama, H.; Akaza, H. Bacillus Calmette-Guerin (BCG) immunotherapy for bladder cancer: current understanding and perspectives on engineered BCG vaccine. Cancer Sci. 2013, 104, 22-27, doi: 10.1111/cas.12075.
- Biot, C.; Rentsch, C.A.; Gsponer, J.R.; Birkhauser, F.D.; Jusforgues-Saklani, H.; Lemaitre, F.; Auriau, C.; Bachmann, A.; Bousso, P.; Demangel, C.; et al. Preexisting BCG-Specific T Cells Improve Intravesical Immunotherapy for Bladder Cancer. Sci. Transl. Med. 2012, 4, 137ra72, doi: 10.1126/scitranslmed.3003586.
- Brandau, S.; Riemensberger, J.; Jacobsen, M.; Kemp, D.; Zhao, W.; Zhao, X.; Jocham, D.; Ratliff, T.L.; Böhle, A. NK cells are essential for effective BCG immunotherapy. Int. J. Cancer 2001, 92, 697–702, doi: 10.1002/1097-0215(20010601)92:5<697::aid-ijc1245>3.0.co;2-z.
- Siracusano, S.; Vita, F.; Abbate, R.; Ciciliato, S.; Borelli, V.; Bernabei, M.; Zabucchi, G. The Role of Granulocytes Following Intravesical BCG Prophylaxis. Eur. Urol. 2007, 51, 1589–1599, doi: 10.1016/j.eururo.2006.11.045.
- Suttmann, H.; Riemensberger, J.; Bentien, G.; Schmaltz, D.; Stöckle, M.; Jocham, D.; Böhle, A.; Brandau, S. Neutrophil Granulocytes Are Required for Effective Bacillus Calmette-Guérin Immunotherapy of Bladder Cancer and Orchestrate Local Immune Responses. Cancer Res. 2006, 66, 8250–8257, doi: 10.1158/0008-5472.CAN-06-1416.
- Ratliff, T.L.; Ritchey, J.K.; Yuan, J.J.J.; Andriole, G.L.; Catalona, W.J. T-Cell Subsets Required for Intravesical BCG Immunotherapy for Bladder Cancer. J. Urol. 1993, 150, 1018–1023, doi: 10.1016/s0022-5347(17)35678-1.
- Batista, R.; Lima, L.; Vinagre, J.; Pinto, V.; Lyra, J.; Máximo, V.; Santos, L.; Soares, P. TERT Promoter Mutation as a Potential Predictive Biomarker in BCG-Treated Bladder Cancer Patients. Int. J. Mol. Sci. 2020, 21, 947, doi: 10.3390/ijms21030947.
- De Boer, E.C.; De Jong, W.H.; Van Der Meijden, A.P.M.; Steerenberg, P.A.; Witjes, J.A.; Vegt, P.D.J.; Debruyne, F.M.J.; Ruitenberg, E.J. Presence of activated lymphocytes in the urine of patients with superficial bladder cancer after intravesical immunotherapy with bacillus Calmette-Guérin. Cancer Immunol. Immunother. 1991, 33, 411–416, doi: 10.1007/BF01741603.
- Pryor, K.; Goddard, J.; Goldstein, D.; Stricker, P.; Russell, P.; Golovsky, D.; Penny, R. Bacillus Calmette-Guerin (BCG) enhances monocyte- and lymphocyte-mediated bladder tumour cell killing. Br. J. Cancer 1995, 71, 801–807, doi: 10.1038/bjc.1995.155.
- Pettanati, C.; Ingersol, M.A. Mechanisms of BCG immunotherapy and its outlook for bladder cancer. Nat. Rev. Urol. 2018, 15, 615-625, doi: 10.1038/s41585-018-0055-4.
- Lamm, D.L.; Blumenstein, B.A.; Crissman, J.D.; Montie, J.E.; Gottesman, J.E.; Lowe, B.A.; Sarosdy, M.F.; Bohl, R.D.; Grossman, H.B.; Beck, T.M.; Leimert, J.T.; Crawford, E.D. Maintenance bacillus Calmette-Guerin immunotherapy for recurrent TA, T1 and carcinoma in situ transitional cell carcinoma of the bladder: a randomized Southwest Oncology Group Study. J. Urol. 2000, 163, 1124-1129.
- Askeland, E.J.; Newton, M.R.; O’Donnell, M.A.; Luo, Y. Bladder cancer immunotherapy: BCG and beyond. Adv. Urol. 2012, 2012, 181987, doi: 10.1155/2012/181987.
- Sylvester, R.J.; van der Meijden, A.P.; Witjes, J.A.; Kurth, K. Bacillus calmette-guerin versus chemotherapy for the intravesical treatment of patients with carcinoma in situ of the bladder: a meta-analysis of the published results of randomized clinical trials. J. Urol. 2005, 174, 86–91.
- Kamat, A.M.; Flaig, T.W.; Grossman, H.B.; Konety, B.; Lamm. D.; O’Donnell, M.A.; Uchio, E.; Efstathiou, J.A.; Taylor, J.A.3rd. Expert consensus document: Consensus statement on best practice management regarding the use of intravesical immunotherapy with BCG for bladder cancer. Net. Rev. Urol. 2015, 12, 225-235, doi: 10.1186/s40425-017-0271-0.
- Havel, J.J.; Chowell, D.; Chan, T.A. The evolving landscape of biomarkers for checkpoint inhibitor immunotherapy. Nat. Rev. Cancer. 2019, 19, 133-150, doi: 10.1038/s41568-019-0116-x.
- Darvin, P.; Toor, S.M.; Sasidharan, Nair, V.; Elkord, E. Immune checkpoint inhibitors: recent progress and potential biomarkers. Exp. Mol. Med. 2018, 50, 1-11, doi: 10.1038/s12276-018-0191-1.
- Liu, H., Chang, J.K., Hou, J.Q., Zhao, Z.H., Zhang, L.D. Inhibition of miR-221 influences bladder cancer cell proliferation and apoptosis. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 3193-3199.
- Hindupur, S.V.; Schmid, S.C.; Koch, J.A.; Youssef, A.; Baur, E.M.; Wang, D.; Horn, T.; Slotta-Huspenina, J.; Gschwend, J.E.; Holm, P.S.; Nawroth, R. STAT3/5 inhibitors suppress proliferation in bladder cancer and enhance oncolytic adenovirus therapy. Int. J. Mol. Sci. 2020, 21, E1106, doi: 10.3390/ijms21031106.
- Tsuruta, M.; Ueda, S.; Yew, P.Y.; Fukuda, I.; Yoshimura, S.; Kishi, H.; Hamana, H.; Hirayama, M.; Yatsuda, J.; Irie, A.; Sensju, S.; Yuba, E.; Kamba, T.; Eto, M.; Nakayama, H.; Nishimura, Y. Bladder cancer-associated cancer-testis antigen-derived long peptides encompassing both CTL and promiscuous HLA class-II-restricted Th cell epitopes induced CD4+ T cells expressing converged T-cell receptor genes in vitro. Oncoimmunology 2018, 7, e1415687, doi: 10.1080/2162402X.2017.1415687.
- Rink, M. The landscape of genetics and biomarkers in bladder cancer. Transl. Androl. Urol. 2017, 6, 1027-1030, doi: 10.21037/tau.2017.11.12.
- Tsao, J.L.; Yatabe, Y., Markl, I.D.C., Hajyan, K., Jones, P.A., Shibata, D. Bladder cancer genotype stability during clinical progression. Genes Chromosom. Cancer 2000, 29, 26-32, doi: 10.1002/1098-2264(2000)9999:9999<::AID-GCC1002>3.0.CO;2-X.
- Rundo, F.; Spampinato, C.; Banna, G.L.; Conoci, S. Advanced Deep Learning Embedded Motion Radiomics Pipeline for Predicting Anti-PD-1/PD-L1 Immunotherapy Response in the Treatment of Bladder Cancer: Preliminary Results. Electronics 2019, 8, 1134, doi: 10.3390/electronics8101134.
- Alsaab, H.O.; Sau, S.; Alzhrani, R.; Tatiparti, K.; Bhise, K.; Kashaw, S.K.; Iyer, A.K. PD-1 and PD-L1 Checkpoint Signaling Inhibition for Cancer Immunotherapy: Mechanism, Combinations, and Clinical Outcome. Front. Pharmacol. 2017, 8, 561, doi: 10.3389/fphar.2017.00561.
- Kim, T.J.; Cho, K.S.; Koo, K.C. Current Status and Future Perspectives of Immunotherapy for Locally Advanced or Metastatic Urothelial Carcinoma: A Comprehensive Review. Cancers 2020, 12, 192, doi: 10.3390/cancers12010192.
- (FDA) FDA approves new, targeted treatment for bladder cancer. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-new-targeted-treatment-bladder-cancer (accessed on 26 March 2020).
- Tucker, N. Avelumab Induces OS Benefit in Locally Advanced or Metastatic Urothelial Cancer. Available online: https://www.targetedonc.com/news/avelumab-induces-os-benefit-in-locally-advanced-or-metastatic-urothelial-cancer (accessed on 26 March 2020).
- (FDA) Durvalumab (Imfinzi) PATIENTS. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/durvalumab-imfinzi (accessed on 26 March 2020).
- Sharma, P.; Retz, M.; Siefker-Radtke, A.; Baron, A.; Necchi, A.; Bedke, J.; Plimack, E.R.; Vaena, D.; Grimm, M.-O.; Bracarda, S.; et al. Nivolumab in metastatic urothelial carcinoma after platinum therapy (CheckMate 275): a multicentre, single-arm, phase 2 trial. Lancet Oncol. 2017, 18, 312–322, doi: 10.1016/S1470-2045(17)30065-7.
- Ishii, A.; Yokoyama, M.; Tsuji, H.; Fujii, Y.; Tamaoka, A. Pembrolizumab treatment of metastatic urothelial cancer without exacerbating myasthenia gravis. eNeurologicalSci 2020, 19, doi:10.1016/j.ensci.2020.100236.
- Pollak, M.N.; Foulkes, W.D.; Mancuso, J.G. Cancer immunoprevention: A case report raising the possibility of “immuno-interception.” Cancer Prev. Res. 2020, 13, 351–356, doi:10.1158/1940-6207.CAPR-19-0528.
- Hojeij, R.; Domingos-Pereira, S.; Nkosi, M.; Gharbi, D.; Derré, L.; Schiller, J.; Jichlinski, P.; Nardelli-Haefliger, D. Immunogenic Human Papillomavirus Pseudovirus-Mediated Suicide-Gene Therapy for Bladder Cancer. Int. J. Mol. Sci. 2016, 17, 1125, doi: 10.3390/ijms17071125.
- Schulz, W.A.; Sørensen, K.D. Epigenetics of Urological Cancers. Int. J. Mol. Sci. 2019, 20, 4775, doi: 10.3390/ijms20194775
- Peggs, K.S.; Quezada, S.A.; Chambers, C.A.; Korman, A.J.; Allison, J.P. Blockade of CTLA-4 on both effector and regulatory T cell compartments contributes to the antitumor activity of anti–CTLA-4 antibodies. J. Exp. Med. 2009, 206, 1717–1725, doi: 10.1084/jem.20082492.
- van der Merwe, P.A.; Davis, S.J. Molecular interactions mediating T cell antigen recognition. Annu. Rev. Immunol. 2003, 21, 659–684, doi: 10.1146/annurev.immunol.21.120601.141036.