
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Lucia Altucci | + 3047 word(s) | 3047 | 2021-01-06 03:57:40 | | | |
| 2 | Camila Xu | Meta information modification | 3047 | 2021-03-18 03:54:36 | | |
Video Upload Options
miRNAs role in hormone signaling pathways in CRC drug resistance and their potential as future targets for overcoming resistance to treatment.
1. The Role of Hormone Signaling in CRC Resistance
In receiving cells, hormone signaling comprises hormone binding to its receptor and signal transduction through different functional pathways. Several hormone signaling pathways are altered in CRC, often resulting in drug resistance. Tumor cells can avoid drug-mediated apoptosis at different levels of signaling pathways as these typically have multiple components. Most hormone signaling pathways exert their function through PI3K/Akt cascade: Upon hormone stimulation, PI3K is phosphorylated and its activity is mediated by RasandSrcfamilykinases that phosphorylate Akt, which, in turn, regulates the activation of proteins involved in apoptosis, metabolism, protein synthesis, and cell division [1]. Furthermore, the 3R3 subunit of PI3K is upregulated in CRC and controls the expression of TP via the NF-kB pathway, thus preventing the activation of drugs (5-FU, capecitabine) and resulting in chemoresistance [2]. PI3K/mTOR inhibition in CRC patient-derived cells has also been reported to reinstate cetuximab resistance [3]. PI3K action is regulated by the tumor suppressor phosphatase and tensin homolog (PTEN). Altered pathways responding to insulin, estrogen, and androgen involve PTEN [4], resulting in cetuximab and panitumumab resistance [5]. Thus, PI3K/Akt/mTOR signaling is effectively involved in many hormonal pathway-related resistances, though it is not the only one. Upon binding of ligands (such as insulin and growth factors) to their tyrosine kinase receptor [6], PTEN has also been involved in cetuximab resistance by altering Ras pathway activity [7]. MAPK/ERK pathway was found downstream of many growth factor receptors often overexpressed in CRC [8], such as insulin-like growth factor receptor (IGFR) [9]. MAPKs are involved in several cellular functions such as proliferation, differentiation, and apoptosis through three primary components: p38, ERK, and JNK. Hyperactivation of p38 was recently implicated in oxaliplatin resistance in CRC [10]. MAPKs exert their functions through NF-kB [4], activated in 60–80% of CRC patients [11], and involved in conferring chemoresistance through inhibition of apoptosis [12]. In other studies, CRC cells resistant to 5-FU showed high expression of TCF4 and β-catenin, indicating an altered Wnt pathway [13]. Since hormone signaling is triggered upon binding of the hormone to its receptor, expression of hormone receptors in recipient cells is important in modulating the impact of this interaction, as well as influencing downstream cascades.
1.1. Insulin/Insulin-Like Growth Factor Signaling Pathway in CRC
The insulin/IGF system plays a crucial role in CRC development [14]. Insulin/IGF bind to the insulin receptor (IR), which is converted into its activated phosphorylated form. High levels of pIR were observed in the transition phase from normal epithelium to adenocarcinoma, indicating the key role of insulin/IR in CRC progression [15]. IR substrates (IRS)-1 and -2 also contribute to disease development: IRS-1 overexpression is associated with metastasis [16], whereas IRS-2 is related to early-stage tumor formation. IRS-2 overexpression increases PI3K pathway activity, leading to Akt phosphorylation and reduced cell adhesion in colon cancer [17]. IGF-I and IGF-II are significantly overexpressed in CRC [18]. High IGF serum levels were detected in CRC patients and found to act through autocrine/paracrine as well as endocrine signaling [9]. IGF regulates cell survival, proliferation, and metabolism by binding to IGFR or IR. IGF-1R was found overexpressed in CRC [19] where it resulted in Akt activation and upregulation of the anti-apoptotic protein Bcl-xL [20]. IGF-1R is associated with drug resistance as it regulates the expression of multidrug-resistance-associated protein 2 (MRP2). For this purpose, Shen et al. silenced IGF-1R which subsequently drove the nuclear translocation of nuclear-factor-like2 and blockage of MRP2 gene expression [21]. Low MRP2 expression causes an increase in intracellular drug concentration and accumulation, suggesting how the IGF-1R signaling pathway may foster drug efflux and tumor cell survival despite therapy. Further analysis of these hormone receptors revealed that silencing of IGF-1R increases the sensitivity of CRC cells to 5 -FU, mitomycin C, oxaliplatin, and vincristine [21].
1.2. Thyroid Hormone Signaling Pathway in CRC
Animal studies and clinical data from CRC patients suggest that thyroid status may impact tumor formation, growth, metastasis, and drug sensitivity [22][23][24]. This physiological hormonal system comprises both triiodothyronine (T3) and thyroxine (T4) hormones. T4 is the primary inactive thyroid product, which is converted into active T3 by type 1 or 2 deiodinase enzymes (D1 and D2, respectively). In contrast, type 3 deiodinase (D3) transforms T4 and T3 into inactive metabolites [23][25]. Deiodinases represent the link between thyroid hormone signaling and β-catenin pathway. The β-catenin/TCF complex regulates D3 expression at the transcriptional level resulting in high expression of D3 in CRC. Indeed, using E-cadherin to sequester β-catenin, a reduction in D3 expression occurs.Low D3 levels lead to increased expression of T3, resulting in inhibited proliferation of CRC cells [26]. Furthermore, T3 may induce resistance to oxaliplatin, 5-FU, and their combination (FOX) by downregulating Akt/PI3K in CRC stem cells [27]. Basically, T3 binds to its receptors (TRα and TRβ), regulates the expression of target genes via thyroid hormone receptor response elements [28]. Thyroid hormone receptor (TR) β1 expression is downregulated in CRC, prompting cancer cells to proliferate and migrate via PI3K/Akt signaling modulation. Overexpression of TRβ1 induces a reduction of inactivated Akt, disrupting its pathway and suppressing CRC cell progression and migration [29]. In contrast, TRα1 overexpression and Wnt signaling pathway upregulation promote tumor formation in CRC [30] by contributing to escape from chemotherapy-induced apoptosis [31]. Indeed, further evidence report that T3 affects drug resistance targeting BMP4/Wnt pathway [28].
1.3. Additional Altered Hormone Signaling in Resistant CRC Cells
Other hormonal systems contribute to drug resistance in CRC cells. Leptin is an adipokine involved in regulating hyperphagia, glucose homeostasis, growth, immune response, and angiogenesis [32]. Leptin and its receptors (ObR) are highly expressed in CRC [33], indeed, leptin inhibition reduces tumor growth [34] and leads to an overexpression of ObR, which inhibits 5-FU-induced apoptosis [35]. Moreover, high levels of growth hormone (GH) were identified in CRC patients, where the hormone enhances cell proliferation, survival, and oncogenicity through autocrine signaling [36]. GH also plays a role in the acquisition of drug-induced apoptosis resistance by inhibiting the expression of PPARγ and BAX in CRC cells, allowing evasion from therapy-mediated DNA damage [37]. Corticotropin-releasing hormone (CRH) is a peptide hormone involved in stress response that exerts its function by binding to its specific receptors, including corticotropin-releasing hormone receptor2 (CRHR2), which exhibits a significantly reduced expression in CRC. Fas is one of the CRHR2/Ucn2 signaling targets in CRC and alternation in Fas expression prevents Fas-mediated apoptosis, thus conferring resistance to therapy [38]. The lower incidence of CRC in women than in men highlights the importance of estrogen as a biological protective effector in gastrointestinal disease [39][40]. Estrogens can bind to estrogen receptor α (ERα) orβ (ERβ). In normal colon regions, ERβ is implicated in epithelium maintenance and growth, and in immune system modulation, while decreased ERβ expression promotes the risk of CRC partly by influencing gut permeability [41]. Reduced ERβ expression in CRC was also correlated with increased proliferation and inhibition of apoptosis [42]. ERα downregulation in CRC [43] suggests that this receptor might contribute to drug resistance via regulatory effect in the expression of pro- or anti-apoptotic proteins, such as cyclin D1 [42]. Estrogen signaling might be indirectly involved in conferring resistance in CRC cells since a deficiency in ER signaling can be compensated for by other signaling pathways in these cells [44][45]. Estrogen-related receptor alpha (ERRα) is also involved in the same system. ERRα is an orphan nuclear receptor since its endogenous ligand has not yet been identified. Interestingly, it is constitutively active even without any ligand binding [46] and regulates the expression of many genes, most of which are also targeted by ERα [47]. ERRα is overexpressed in CRC [41] and its upregulation is responsible for resistance to trametinib [48], making ERRα a potential new target in CRC.
2. Role of miRNA in Hormone Signaling in Therapy-Resistant CRC
miRNAs are known as posttranscriptional regulators of gene expression, affecting about 60% of overall protein-coding genes [49], thereby modulating developmental, physiological, and pathological processes. Recent reports described their critical role in the regulation of hormone signaling, including expression modulation of hormone receptor-coding genes and genes related to hormonal intracellular signaling cascades [50][51][52]. Moreover, miRNAs can be modulated by hormones [53], thus suggesting a reciprocal flux which anyway revises cancer cell sensitivity to treatment. Several deregulated miRNAs are associated with drug resistance in CRC (Table 1), many of which are directly involved in hormone signaling. The following sections unfold the involvement of miRNAs into hormonal signaling pathways whose interactions are responsible for decreased therapy success by overcoming drug-mediated apoptosis and, thereby, inducing survival in CRC cells.
Table 1. Drug resistance-associated miRNAs in CRC.
| miRNA | Expression | Targets | Resistance | Reference |
|---|---|---|---|---|
| miR-21 | ↑ | PDCD4 TIAM1 PTEN TGFBR2 CDC25A |
5-FU | [54] |
| miR-143 | ↓ | NF-kB Bcl-2; IGF-1R |
5-FU | [52][55] |
| miR-365; miR-224 | ↓ | - | 5-FU | [56] |
| miR-34a | ↓ | SIRT1 | 5-FU | [57] |
| miR-494 | ↓ | DPYD | 5-FU | [58] |
| miR-302 | ↓ | IGF-1R | 5-FU | [59] |
| miR-10b | ↑ | BIM | 5-FU | [60] |
| miR-375-3p | ↓ | TYMS | 5-FU | [61] |
| miR-20a | ↑ | BNIP2 | 5-FU OxaliplatinTeniposide |
[62] |
| miR-320 | ↓ | FOXM1 | 5-FU Oxaliplatin |
[63] |
| miR-34a | ↑ | - | Oxaliplatin | [64] |
| let-7g | ↓ | Cyclin D Myc E2F |
S-1 | [65] |
2.1. miRNAs and Insulin-Like Growth Factor (IGF) Receptor
IGFs are implicated in tumorigenesis of different cancers, such as prostate, breast, and CRC [9]. As explained in sub-paragraph 2, both IGFs and IGF-R were found overexpressed in CRC, and such boosted IGF-I/IGF-IR signaling pathway enhances cell survival and resistance to chemotherapy [20], a process that apparently involves CIMP. The chromosomal segment 17p13.1 is frequently deleted in CRC, resulting in reduced expression of both miR-497 and miR-195, as their encoding sequences are located in this chromosomal region [66]. Guo et al. proposed miR-497 to have a role in inhibiting IGF-IR signaling pathway, indeed, in CRC, low miR-497 is associated with higher IGF-IR levels, which, in turn, modulates cell death. Restoration of miR-497 expression could be useful for inhibition of IGF-IR in CRC and resistance to apoptosis by blocking overactivation of survival signaling pathways [66]. Many other miRNAs are implicated in IGFR signaling modulation resulting in drug resistance. As a matter of fact, miR-143 targets IGF-1R resulting in oxaliplatin resistance [67] and, indeed, overexpression of miR-143 was reported to re-sensitize CRC cells to such therapeutic by caspases activation [67]. Interestingly, Xu et al. found that miR-143 is downregulated in blood as well, suggesting its potential as a prognostic biomarker in CRC. Besides miR-143, IGF-1R is also a target of miR-302, which downregulation in CRC may cause 5-FU resistance. Liu et al., actually found that miR-302 overexpression reduces 5-FU resistance as demonstrated by enhanced cell death, subsequently, therapeutic treatment. This re-sensitizing action is due to the direct repression of IGF-1R associated with consequent inhibition of Akt, frequently activated in chemo-resistant CRC cells [68]. Lastly, a further miRNA is able to modulate IGFRs expression skewing CRC therapeutic response. In this respect, miR-185 was found to be involved in radio-resistance in CRC cells by targeting IGF-1R and IGF-2R. In detail, miR-185 is downregulated in colon cancer, leading to increased expression of its targets and, thus, resulting in refractory to ionizing radiation, as it was confirmed by improved CRC cell radio-sensitivity upon restoration of the miRNA expression [69].
2.2. miRNAs and IGF-R Downstream Components
Besides receptors, miRNAs impair hormone signaling also at the downstream level, resulting in ectopic pathways and then in drug resistance. Among such regulators, miR-1260b targets PDCD4, one of the effectors of the PI3K/Akt signaling pathway reported to regulate apoptosis [70] and the response to drug treatment [71]. miR-1260b expression was reported increased in CRC patients and linked to 5-FU therapy resistance. Accordingly, the use of a miR1260b inhibitor confirmed the involvement of such miRNA in 5-FU-resistance in CRC, further revealing to be mediated by inhibition of PI3K/Akt signaling pathway. Interestingly, the overexpression of IGF-I in CRC enhanced the activation of such pathway, indicating that miR-1260b may regulate drug sensitivity via IGF signal modulation [72]. IRS-1 is reported to be an oncogene involved in the regulation of angiogenesis, metastasis, and even chemosensitivity, thereby, IRS-1 increased levels due to the reduction of its regulatory miRNA expression may be responsible for lowering the therapy responsiveness in CRC. In the same manner, miR-145 exhibits a tumor suppressor activity in CRC by modulating Myc [73], STAT1 [74] and other genes including IRS-1 [75].In fact, miR-145 downregulation was shown to increase tumor survival [76], a feature sharply in contrast with the therapeutic success. Lastly amidst other miRNAs, miR-128 is downregulated in CRC, failing to restrain IRS-1 and resulting in inhibited apoptosis [77].
2.3. miR-155 and Adrenaline
Previous examples have exhibited a definite trend, where miRNAs were responsible for the refractory phenotype in CRC by skewing hormone signaling pathways. Nevertheless, there is evidence that a different course exists since hormones may affect miRNAs expression. The miR-155/adrenaline coupling is one example, as adrenaline was found to increase miR-155 expression, leading to drug resistance [78]. CRC cells significantly express adrenergic receptors [79] whose ligands are catecholamines, including adrenaline. Adrenaline was previously shown to induce 5-FU resistance in CRC in vitro by upregulating ABCB1 transporter [79].Indeed, mouse cancer models exposed to stressful stimuli to set up catecholamine signals, exhibited restrained response to anti-tumor therapy [80]. In this regard, adrenaline showed to activate NF-kB signaling in CRC cells, increasing the expression of miR-155, which, in turn, favored cell proliferation and reduced sensitivity to cisplatin [80], corroborating the potential of catecholamines in endowing resistance to CRC.
2.4. miR-7 and Corticotropin-Releasing Hormone Receptor
Nowadays, immune therapy has emerged as a rising approach to the conventional anti-tumor treatments since cancer cells were shown to express a large amount of inhibitory molecules, such as PD-L1 [81], CD80/CD86 [82], which undermine the immunological response through binding immune checkpoint receptors such as PD-1 and CTLA4 [83]. Another way exploited by cancer cells to deceive the immune-mediated cytotoxicity is the escape from Fas/FasL-mediated apoptosis [84][85][86]. Although cancer treatments, such as radiotherapy, exhibited a successful increase in Fas expression [85], immune escape is still a current challenge and the CRH system seems to be involved in it. Rodriguez et al. reported that CRHR2 expression is reduced in CRC cells supporting tumor survival, proliferation, EMT, metastasis, and resistance [87]. In vitro and in vivo studies showed that miR-7 expression is induced by CRHR2 signaling and its ectopic expression enhances apoptosis and cell cycle arrested by negatively regulating YY1 expression [88]. Thus, YY1 expression benefits from reduced CRHR signaling and its overexpression impairs Fas expression. Collectively, CRHR2/Ucn2 signaling sustains CRC cell resistance to Fas/FasL-mediated apoptosis by targeting miR-7/YY1/Fas circuit [38], as it was confirmed by increased CD-11 (Fas agonist)-mediated apoptosis after restoration of CRHR2 signaling.
3. Novel Therapeutic Agents in the Treatment of Drug Resistance in CRC
Many therapeutics acting on hormone signaling have been tested towards resistant CRC. In CRC, neurokinin receptor 1 (NK1R) antagonists reduced tumor growth through inhibition of Wnt/β-catenin and Akt/mTOR signaling pathways. The NKIR antagonist also inhibited cancer stem cells, which are thought to confer higher resistance to therapy [89]. Besides molecule antagonists, hormone analogs have been tested in CRC. Lee et al. exploited a T4 analog (named Tetrac) to block CRC progression competing with the endogenous hormone to bind and, thus, block the surface integrin receptor αvβ3. Its usage, in combination with cetuximab, was reported to inhibit cell proliferation and CRC xenografts [54]. Brigatinib, which inhibits IGF-1R [55], and Amiodarone, a Thyroid Hormone Receptor antagonist [56], were studied with encouraging results. Significant outcomes have also been obtained with natural products in resistant CRC cells. Gambogic acid is a natural compound extracted from the Garciniahanburyi plant [57]. It acts by interfering with MAPK signaling via JNK pathway, which, in turn, induces apoptosis and inhibits proliferation of 5-FU-resistant CRC cells [57]. The China Food and Drug Administration has approved its use in phase II clinical trials for the treatment of solid tumors. Piperlongumine, a natural constituent of the fruit of the long pepper (Piper longum), inhibits PI3K and Ras, reducing the activity of Akt/NF-kB, c-Myc, cyclin D1, blocking CRC cell growth, proliferation and survival. It also induces mitochondrial apoptosis, downregulating Bcl-2 [58]. In addition to the molecules that directly modulate pathway component activity and considering the emerging role of miRNAs in regulating the expression of genes involved in hormone signaling pathways, recent studies have been focused on strategies to regulate oncomiR expression, including antisense anti-miR oligonucleotides, locked nucleic acid anti-miRs, miRNA sponges and small molecule inhibitors of miRNAs (SMIRs) [65]. ASMIR against miR-21, named AC1MMYR2, was developed by Shi et al., who studied the three-dimensional structure of the Dicer binding site on pre-miR-21 to find a molecule able to block miR-21 maturation. AC1MMYR2 inhibits the expression of miR-21 and consequently increases the expression of its targets: PTEN (Figure 1), PDCD4 and RECK [90]. This axis is involved in 5-FU resistance (Table 1). miR-1260b inhibitor was tested by Zhao et al., who showed the reduction of PDCD4 expression and p-Ant and p-PI3K levels in IGF-R signaling pathway, thus, re-sensitizing CRC to 5-FU treatment [72]. One of the most used anti-miRs in CRC is anti-miR-135b [63][91]. Upregulation of miR-135b in CRC reduces apoptosis and increases cell growth by regulating the expression of TGF-βR2, DAPK1, APC and FIH by activating APC/β-catenin and Src-PI3K pathways. Anti-miR-135b was therefore used in a CRC mouse model, resulting in reduced proliferation and increased apoptosis [64] and drug sensitivity [91]. SD-208, a TGF-β receptor kinase inhibitor, downregulated miR-135b expression [63]. Among other targets, miR-135b affects FOXO1 and ERRα expression, which is associated with apoptosis modulation (Figure 2). FOXO1 is also involved in IR signaling cascade [92]. Thus, pre-treatment with SD-208 improved chemosensitivity in CRC cells resistant to 5-FU [63]. Melatonin was shown to exert a robust anti-tumor effect through modulation of cell cycle dynamics and apoptosis [93]. Recently, Sakatani et al. investigated the molecular mechanism of melatonin in 5-FU-resistant CRC cells [94], suggesting that melatonin strongly promotes apoptosis in 5-FU-resistant CRC cells by blocking thymidylate synthase (TYMS) activity, pointing to an inverse correlation between TYMS expression levels and 5-FU sensitivity in CRC. TYMS is one of the main downstream targets of miR-215-5p. Melatonin upregulates miR-125-5p expression, enhancing the suppressive effect of miR-125-5p on TYMS expression in 5-FU-resistant cells, thus, leading to inhibition of proliferation and increased apoptosis in these resistant cells [94].
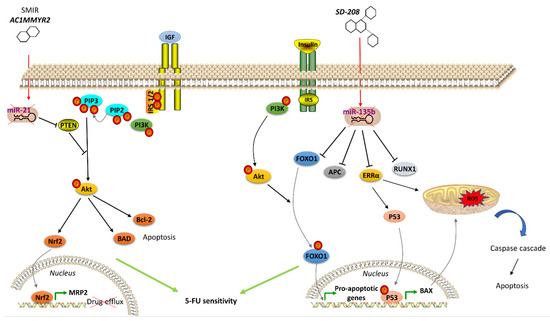
Figure 1. Agents acting on hormone signaling to revert drug resistance in CRC. Left, the small molecular inhibitor of specific miRNA (SMIR) AC1MMYR2 represses miR-21, which regulates PTEN expression, thus modulating IGFR signaling pathway and resulting in inhibited drug efflux and induced apoptosis in resistant CRC cells. Right, SD-208 inhibits miR-135b, which controls expression of proteins (including FOXO1 in IR signaling pathway) involved in apoptosis regulation, thus resulting in apoptosis of resistant CRC cells. (↑ = miRNA overexpression) (↓ = miRNA downexpression)
References
- Aksamitiene, E.; Kiyatkin, A.; Kholodenko, B.N. Cross-talk between mitogenicRas/MAPK and survival PI3K/Akt pathways: A fine balance. Biochem. Soc. Trans. 2012, 40, 139–146.
- Ibrahim, S.; Li, G.; Hu, F.; Hou, Z.; Chen, Q.; Li, G.; Luo, X.; Hu, J.; Feng, Y. PIK3R3 promotes chemotherapeutic sensitivity of colorectal cancer through PIK3R3/NF-kB/TP pathway. Cancer Biol. Ther. 2018, 19, 222–229.
- Bray, S.M.; Lee, J.; Kim, S.T.; Hur, J.Y.; Ebert, P.J.; Calley, J.N.; Wulur, I.H.; Gopalappa, T.; Wong, S.S.; Qian, H.R.; et al. Genomic characterization of intrinsic and acquired resistance to cetuximab in colorectal cancer patients. Sci. Rep. 2019, 9, 15365.
- Slattery, M.L.; Wolff, R.K.; Lundgreen, A. A pathway approach to evaluating the association between the CHIEF pathway and risk of colorectal cancer. Carcinogenesis 2015, 36, 49–59.
- Salvatore, L.; Calegari, M.A.; Loupakis, F.; Fassan, M.; Di Stefano, B.; Bensi, M.; Bria, E.; Tortora, G. PTEN in Colorectal Cancer: Shedding Light on Its Role as Predictor and Target. Cancers 2019, 11, 1765.
- Chalhoub, N.; Baker, S.J. PTEN and the PI3-kinase pathway in cancer. Annu. Rev. Pathol. 2009, 4, 127–150.
- Negri, F.V.; Bozzetti, C.; Lagrasta, C.A.; Crafa, P.; Bonasoni, M.P.; Camisa, R.; Pedrazzi, G.; Ardizzoni, A. PTEN status in advanced colorectal cancer treated with cetuximab. Br. J. Cancer 2010, 102, 162–164.
- Fang, J.Y.; Richardson, B.C. The MAPK signalling pathways and colorectal cancer. Lancet Oncol. 2005, 6, 322–327.
- Denduluri, S.K.; Idowu, O.; Wang, Z.; Liao, Z.; Yan, Z.; Mohammed, M.K.; Ye, J.; Wei, Q.; Wang, J.; Zhao, L.; et al. Insulin-like growth factor (IGF) signaling in tumorigenesis and the development of cancer drug resistance. Genes Dis. 2015, 2, 13–25.
- Chocry, M.; Leloup, L.; Kovacic, H. Reversion of resistance to oxaliplatin by inhibition of p38 MAPK in colorectal cancer cell lines: Involvement of the calpain/Nox1 pathway. Oncotarget 2017, 8, 103710–103730.
- Slattery, M.L.; Mullany, L.E.; Sakoda, L.; Samowitz, W.S.; Wolff, R.K.; Stevens, J.R.; Herrick, J.S. The NF-kappaBsignalling pathway in colorectal cancer: Associations between dysregulated gene and miRNA expression. J. Cancer Res. Clin. Oncol. 2018, 144, 269–283.
- McCool, K.W.; Miyamoto, S. DNA damage-dependent NF-kappaB activation: NEMO turns nuclear signaling inside out. Immunol. Rev. 2012, 246, 311–326.
- He, L.; Zhu, H.; Zhou, S.; Wu, T.; Wu, H.; Yang, H.; Mao, H.; SekharKathera, C.; Janardhan, A.; Edick, A.M.; et al. Wnt pathway is involved in 5-FU drug resistance of colorectal cancer cells. Exp. Mol. Med. 2018, 50, 101.
- Vigneri, P.G.; Tirro, E.; Pennisi, M.S.; Massimino, M.; Stella, S.; Romano, C.; Manzella, L. The Insulin/IGF System in Colorectal Cancer Development and Resistance to Therapy. Front. Oncol. 2015, 5, 230.
- Abbruzzese, C.; Diodoro, M.G.; Sperduti, I.; Mileo, A.M.; Pattaro, G.; De Salvo, L.; Cosimelli, M.; Perrotti, N.; Paggi, M.G. Detection of phosphorylated insulin receptor in colorectal adenoma and adenocarcinoma: Implications for prognosis and clinical outcome. J. Cell. Physiol. 2015, 230, 562–567.
- Esposito, D.L.; Aru, F.; Lattanzio, R.; Morgano, A.; Abbondanza, M.; Malekzadeh, R.; Bishehsari, F.; Valanzano, R.; Russo, A.; Piantelli, M.; et al. The insulin receptor substrate 1 (IRS1) in intestinal epithelial differentiation and in colorectal cancer. PLoS ONE 2012, 7, e36190.
- Day, E.; Poulogiannis, G.; McCaughan, F.; Mulholland, S.; Arends, M.J.; Ibrahim, A.E.; Dear, P.H. IRS2 is a candidate driver oncogene on 13q34 in colorectal cancer. Int. J. Exp. Pathol. 2013, 94, 203–211.
- Weber, M.M.; Fottner, C.; Liu, S.B.; Jung, M.C.; Engelhardt, D.; Baretton, G.B. Overexpression of the insulin-like growth factor I receptor in human colon carcinomas. Cancer 2002, 95, 2086–2095.
- Lahm, H.; Amstad, P.; Wyniger, J.; Yilmaz, A.; Fischer, J.R.; Schreyer, M.; Givel, J.C. Blockade of the insulin-like growth-factor-I receptor inhibits growth of human colorectal cancer cells: Evidence of a functional IGF-II-mediated autocrine loop. Int. J. Cancer 1994, 58, 452–459.
- Sekharam, M.; Zhao, H.; Sun, M.; Fang, Q.; Zhang, Q.; Yuan, Z.; Dan, H.C.; Boulware, D.; Cheng, J.Q.; Coppola, D. Insulin-like growth factor 1 receptor enhances invasion and induces resistance to apoptosis of colon cancer cells through the Akt/Bcl-x(L) pathway. Cancer Res. 2003, 63, 7708–7716.
- Shen, K.; Cui, D.; Sun, L.; Lu, Y.; Han, M.; Liu, J. Inhibition of IGF-IR increases chemosensitivity in human colorectal cancer cells through MRP-2 promoter suppression. J. Cell. Biochem. 2012, 113, 2086–2097.
- L’Heureux, A.; Wieland, D.R.; Weng, C.H.; Chen, Y.H.; Lin, C.H.; Lin, T.H.; Weng, C.H. Association Between Thyroid Disorders and Colorectal Cancer Risk in Adult Patients in Taiwan. JAMA Netw. Open 2019, 2, e193755.
- Dentice, M.; Antonini, D.; Salvatore, D. Type 3 deiodinase and solid tumors: An intriguing pair. Expert Opin. Ther. Targets 2013, 17, 1369–1379.
- Krashin, E.; Piekielko-Witkowska, A.; Ellis, M.; Ashur-Fabian, O. Thyroid Hormones and Cancer: A Comprehensive Review of Preclinical and Clinical Studies. Front. Endocrinol. 2019, 10, 59.
- Dentice, M.; Marsili, A.; Zavacki, A.; Larsen, P.R.; Salvatore, D. The deiodinases and the control of intracellular thyroid hormone signaling during cellular differentiation. Biochim. Biophys. Acta 2013, 1830, 3937–3945.
- Dentice, M.; Luongo, C.; Ambrosio, R.; Sibilio, A.; Casillo, A.; Iaccarino, A.; Troncone, G.; Fenzi, G.; Larsen, P.R.; Salvatore, D. beta-Cateninregulatesdeiodinaselevels and thyroidhormonesignaling in colon cancer cells. Gastroenterology 2012, 143, 1037–1047.
- Catalano, V.; Dentice, M.; Ambrosio, R.; Luongo, C.; Carollo, R.; Benfante, A.; Todaro, M.; Stassi, G.; Salvatore, D. Activated Thyroid Hormone Promotes Differentiation and Chemotherapeutic Sensitization of Colorectal Cancer Stem Cells by Regulating Wnt and BMP4 Signaling. Cancer Res. 2016, 76, 1237–1244.
- Yen, P.M. Physiological and molecular basis of thyroid hormone action. Physiol. Rev. 2001, 81, 1097–1142.
- Zhu, L.; Tian, G.; Yang, Q.; De, G.; Zhang, Z.; Wang, Y.; Nie, H.; Zhang, Y.; Yang, X.; Li, J. Thyroid hormone receptor beta1 suppresses proliferation and migration by inhibiting PI3K/Akt signaling in human colorectal cancer cells. Oncol. Rep. 2016, 36, 1419–1426.
- Uchuya-Castillo, J.; Aznar, N.; Frau, C.; Martinez, P.; Le Neve, C.; Marisa, L.; Penalva, L.O.F.; Laurent-Puig, P.; Puisieux, A.; Scoazec, J.Y.; et al. Increased expression of the thyroid hormone nuclear receptor TRalpha1 characterizes intestinal tumors with high Wnt activity. Oncotarget 2018, 9, 30979–30996.
- Yuan, S.; Tao, F.; Zhang, X.; Zhang, Y.; Sun, X.; Wu, D. Role of Wnt/beta-Catenin Signaling in the Chemoresistance Modulation of Colorectal Cancer. BioMed Res. Int. 2020, 2020, 9390878.
- Kawashima, K.; Maeda, K.; Saigo, C.; Kito, Y.; Yoshida, K.; Takeuchi, T. Adiponectin and Intelectin-1: Important Adipokine Players in Obesity-Related Colorectal Carcinogenesis. Int. J. Mol. Sci. 2017, 18, 866.
- Al-Shibli, S.M.; Harun, N.; Ashour, A.E.; MohdKasmuri, M.H.B.; Mizan, S. Expression of leptin and leptin receptors in colorectal cancer-an immunohistochemical study. PeerJ 2019, 7, e7624.
- Zhou, W.; Tian, Y.; Gong, H.; Guo, S.; Luo, C. Oncogenic role and therapeutic target of leptin signaling in colorectal cancer. Expert Opin. Ther. Targets 2014, 18, 961–971.
- Bartucci, M.; Svensson, S.; Ricci-Vitiani, L.; Dattilo, R.; Biffoni, M.; Signore, M.; Ferla, R.; De Maria, R.; Surmacz, E. Obesity hormone leptin induces growth and interferes with the cytotoxic effects of 5-fluorouracil in colorectal tumor stem cells. Endocr.-Relat. Cancer 2010, 17, 823–833.
- Wang, J.J.; Chong, Q.Y.; Sun, X.B.; You, M.L.; Pandey, V.; Chen, Y.J.; Zhuang, Q.S.; Liu, D.X.; Ma, L.; Wu, Z.S.; et al. AutocrinehGH stimulates oncogenicity, epithelial-mesenchymal transition and cancer stem cell-like behavior in human colorectal carcinoma. Oncotarget 2017, 8, 103900–103918.
- Bogazzi, F.; Ultimieri, F.; Raggi, F.; Russo, D.; Vanacore, R.; Guida, C.; Brogioni, S.; Cosci, C.; Gasperi, M.; Bartalena, L.; et al. Growth hormone inhibits apoptosis in human colonic cancer cell lines: Antagonistic effects of peroxisome proliferator activated receptor-gamma ligands. Endocrinology 2004, 145, 3353–3362.
- Pothoulakis, C.; Torre-Rojas, M.; Duran-Padilla, M.A.; Gevorkian, J.; Zoras, O.; Chrysos, E.; Chalkiadakis, G.; Baritaki, S. CRHR2/Ucn2 signaling is a novel regulator of miR-7/YY1/Fas circuitry contributing to reversal of colorectal cancer cell resistance to Fas-mediated apoptosis. Int. J. Cancer 2018, 142, 334–346.
- Williams, C.; DiLeo, A.; Niv, Y.; Gustafsson, J.A. Estrogen receptor beta as target for colorectal cancer prevention. Cancer Lett. 2016, 372, 48–56.
- Jacenik, D.; Beswick, E.J.; Krajewska, W.M.; Prossnitz, E.R. G protein-coupled estrogen receptor in colon function, immune regulation and carcinogenesis. World J. Gastroenterol. 2019, 25, 4092–4104.
- Pierdominici, M.; Maselli, A.; Varano, B.; Barbati, C.; Cesaro, P.; Spada, C.; Zullo, A.; Lorenzetti, R.; Rosati, M.; Rainaldi, G.; et al. Linking estrogen receptor beta expression with inflammatory bowel disease activity. Oncotarget 2015, 6, 40443–40451.
- Caiazza, F.; Ryan, E.J.; Doherty, G.; Winter, D.C.; Sheahan, K. Estrogen receptors and their implications in colorectal carcinogenesis. Front. Oncol. 2015, 5, 19.
- Elbanna, H.G.; Ebrahim, M.A.; Abbas, A.M.; Zalata, K.; Hashim, M.A. Potential value of estrogen receptor beta expression in colorectal carcinoma: Interaction with apoptotic index. J. Gastrointest. Cancer 2012, 43, 56–62.
- Ariazi, E.A.; Kraus, R.J.; Farrell, M.L.; Jordan, V.C.; Mertz, J.E. Estrogen-related receptor alpha1 transcriptional activities are regulated in part via the ErbB2/HER2 signaling pathway. Mol. Cancer Res. 2007, 5, 71–85.
- Chang, C.Y.; Kazmin, D.; Jasper, J.S.; Kunder, R.; Zuercher, W.J.; McDonnell, D.P. The metabolic regulator ERRalpha, a downstream target of HER2/IGF-1R, as a therapeutic target in breast cancer. Cancer Cell 2011, 20, 500–510.
- Kallen, J.; Schlaeppi, J.M.; Bitsch, F.; Filipuzzi, I.; Schilb, A.; Riou, V.; Graham, A.; Strauss, A.; Geiser, M.; Fournier, B. Evidence for ligand-independent transcriptional activation of the human estrogen-related receptor alpha (ERRalpha): Crystal structure of ERRalpha ligand binding domain in complex with peroxisome proliferator-activated receptor coactivator-1alpha. J. Biol. Chem. 2004, 279, 49330–49337.
- Vanacker, J.M.; Pettersson, K.; Gustafsson, J.A.; Laudet, V. Transcriptional targets shared by estrogen receptor- related receptors (ERRs) and estrogen receptor (ER) alpha, but not by ERbeta. EMBO J. 1999, 18, 4270–4279.
- Zhou, S.; Xia, H.; Xu, H.; Tang, Q.; Nie, Y.; Gong, Q.Y.; Bi, F. ERRα suppression enhances the cytotoxicity of the MEK inhibitor trametinib against colon cancer cells. J. Exp. Clin. Cancer Res. 2018, 37, 1–14.
- Catalanotto, C.; Cogoni, C.; Zardo, G. MicroRNA in Control of Gene Expression: An Overview of Nuclear Functions. Int. J. Mol. Sci. 2016, 17, 1712.
- Peng, C.; Wang, Y.L. Editorial: MicroRNAs as New Players in Endocrinology. Front. Endocrinol. 2018, 9, 459.
- Luo, L.; Ye, G.; Nadeem, L.; Fu, G.; Yang, B.B.; Honarparvar, E.; Dunk, C.; Lye, S.; Peng, C. MicroRNA-378a-5p promotes trophoblast cell survival, migration and invasion by targeting Nodal. J. Cell Sci. 2012, 125, 3124–3132.
- Gao, W.L.; Liu, M.; Yang, Y.; Yang, H.; Liao, Q.; Bai, Y.; Li, Y.X.; Li, D.; Peng, C.; Wang, Y.L. The imprinted H19 gene regulates human placental trophoblast cell proliferation via encoding miR-675 that targets Nodal Modulator 1 (NOMO1). RNA Biol. 2012, 9, 1002–1010.
- Gonzalez-Duarte, R.J.; Cazares-Ordonez, V.; Avila-Chavez, E. The microRNA biogenesis machinery: Regulation by steroid hormones and alterations in cancer. Rev. Investig. Clin. 2014, 66, 460–464.
- Lee, Y.S.; Chin, Y.T.; Yang, Y.S.H.; Wei, P.L.; Wu, H.C.; Shih, A.; Lu, Y.T.; Pedersen, J.Z.; Incerpi, S.; Liu, L.F.; et al. The combination of tetraiodothyroacetic acid and cetuximab inhibits cell proliferation in colorectal cancers with different K-ras status. Steroids 2016, 111, 63–70.
- Garcia-Aranda, M.; Redondo, M. Targeting Receptor Kinases in Colorectal Cancer. Cancers 2019, 11, 433.
- Chang, Y.L.; Liu, S.T.; Wang, Y.W.; Lin, W.S.; Huang, S.M. Amiodarone promotes cancer cell death through elevated truncated SRSF3 and downregulation of miR-224. Oncotarget 2018, 9, 13390–13406.
- Wen, C.; Huang, L.; Chen, J.; Lin, M.; Li, W.; Lu, B.; Rutnam, Z.J.; Iwamoto, A.; Wang, Z.; Yang, X.; et al. Gambogic acid inhibits growth, induces apoptosis, and overcomes drug resistance in human colorectal cancer cells. Int. J. Oncol. 2015, 47, 1663–1671.
- Kumar, S.; Agnihotri, N. Piperlongumine, a piper alkaloid targets Ras/PI3K/Akt/mTOR signaling axis to inhibit tumor cell growth and proliferation in DMH/DSS induced experimental colon cancer. Biomed. Pharmacother. 2019, 109, 1462–1477.
- Bayraktar, R.; Van Roosbroeck, K.; Calin, G.A. Cell-to-cell communication: microRNAs as hormones. Mol. Oncol. 2017, 11, 1673–1686.
- Wang, Y.; Li, Y.J.; Huang, X.H.; Zheng, C.C.; Yin, X.F.; Li, B.; He, Q.Y. Liensinine perchlorate inhibits colorectal cancer tumorigenesis by inducing mitochondrial dysfunction and apoptosis. Food Funct. 2018, 9, 5536–5546.
- Zheng, Z.; Xu, L.; Zhang, S.; Li, W.; Tou, F.; He, Q.; Rao, J.; Shen, Q. Peiminine inhibits colorectal cancer cell proliferation by inducing apoptosis and autophagy and modulating key metabolic pathways. Oncotarget 2017, 8, 47619–47631.
- Zheng, X.; Shi, C.; Shujun, F.; Yukun, L.; Juan, Z.; Hui, L.; Ying, Z.; Xi, Z. Function and mechanism of microRNA-20a in colorectal cancer. Exp. Ther. Med. 2020, 19, 1605–1616.
- Akbari, A.; Ghahremani, M.H.; Mobini, G.R.; Abastabar, M.; Akhtari, J.; Bolhassani, M.; Heidari, M. Down-regulation of miR-135b in colon adenocarcinoma induced by a TGF-beta receptor I kinase inhibitor (SD-208). Iran. J. Basic Med. Sci. 2015, 18, 856–861.
- Valeri, N.; Braconi, C.; Gasparini, P.; Murgia, C.; Lampis, A.; Paulus-Hock, V.; Hart, J.R.; Ueno, L.; Grivennikov, S.I.; Lovat, F.; et al. MicroRNA-135b promotes cancer progression by acting as a downstream effector of oncogenic pathways in colon cancer. Cancer Cell 2014, 25, 469–483.
- Shah, M.Y.; Ferrajoli, A.; Sood, A.K.; Lopez-Berestein, G.; Calin, G.A. microRNA Therapeutics in Cancer—An Emerging Concept. EBioMedicine 2016, 12, 34–42.
- Guo, S.T.; Jiang, C.C.; Wang, G.P.; Li, Y.P.; Wang, C.Y.; Guo, X.Y.; Yang, R.H.; Feng, Y.; Wang, F.H.; Tseng, H.Y.; et al. MicroRNA-497 targets insulin-like growth factor 1 receptor and has a tumour suppressive role in human colorectal cancer. Oncogene 2013, 32, 1910–1920.
- Qian, X.; Yu, J.; Yin, Y.; He, J.; Wang, L.; Li, Q.; Zhang, L.Q.; Li, C.Y.; Shi, Z.M.; Xu, Q.; et al. MicroRNA-143 inhibits tumor growth and angiogenesis and sensitizes chemosensitivity to oxaliplatin in colorectal cancers. Cell Cycle 2013, 12, 1385–1394.
- Liu, N.; Li, J.; Zhao, Z.; Han, J.; Jiang, T.; Chen, Y.; Hou, N.; Huang, C. MicroRNA-302a enhances 5-fluorouracil-induced cell death in human colon cancer cells. Oncol. Rep. 2017, 37, 631–639.
- Afshar, S.; Najafi, R.; SedighiPashaki, A.; Sharifi, M.; Nikzad, S.; Gholami, M.H.; Khoshghadam, A.; Amini, R.; Karimi, J.; Saidijam, M. MiR-185 enhances radiosensitivity of colorectal cancer cells by targeting IGF1R and IGF2. Biomed. Pharmacother. 2018, 106, 763–769.
- Liwak, U.; Thakor, N.; Jordan, L.E.; Roy, R.; Lewis, S.M.; Pardo, O.E.; Seckl, M.; Holcik, M. Tumor suppressor PDCD4 represses internal ribosome entry site-mediated translation of antiapoptotic proteins and is regulated by S6 kinase 2. Mol. Cell. Biol. 2012, 32, 1818–1829.
- Jansen, A.P.; Camalier, C.E.; Stark, C.; Colburn, N.H. Characterization of programmed cell death 4 in multiple human cancers reveals a novel enhancer of drug sensitivity. Mol. Cancer Ther. 2004, 3, 103–110.
- Zhao, J.; Cao, J.; Zhou, L.; Du, Y.; Zhang, X.; Yang, B.; Gao, Y.; Wang, Y.; Ma, N.; Yang, W. MiR-1260b inhibitor enhances the chemosensitivity of colorectal cancer cells to fluorouracil by targeting PDCD4/IGF1. Oncol. Lett. 2018, 16, 5131–5139.
- Sachdeva, M.; Zhu, S.; Wu, F.; Wu, H.; Walia, V.; Kumar, S.; Elble, R.; Watabe, K.; Mo, Y.Y. p53 represses c-Myc through induction of the tumor suppressor miR-145. Proc. Natl. Acad. Sci. USA 2009, 106, 3207–3212.
- Gregersen, L.H.; Jacobsen, A.B.; Frankel, L.B.; Wen, J.; Krogh, A.; Lund, A.H. MicroRNA-145 targets YES and STAT1 in colon cancer cells. PLoS ONE 2010, 5, e8836.
- Shi, B.; Sepp-Lorenzino, L.; Prisco, M.; Linsley, P.; de Angelis, T.; Baserga, R. Micro RNA 145 targets the insulin receptor substrate-1 and inhibits the growth of colon cancer cells. J. Biol. Chem. 2007, 282, 32582–32590.
- Schetter, A.J.; Okayama, H.; Harris, C.C. The role of microRNAs in colorectal cancer. Cancer J. 2012, 18, 244–252.
- Wu, L.; Shi, B.; Huang, K.; Fan, G. MicroRNA-128 suppresses cell growth and metastasis in colorectal carcinoma by targeting IRS1. Oncol. Rep. 2015, 34, 2797–2805.
- Pu, J.; Bai, D.; Yang, X.; Lu, X.; Xu, L.; Lu, J. Adrenaline promotes cell proliferation and increases chemoresistance in colon cancer HT29 cells through induction of miR-155. Biochem. Biophys. Res. Commun. 2012, 428, 210–215.
- Yao, H.; Duan, Z.; Wang, M.; Awonuga, A.O.; Rappolee, D.; Xie, Y. Adrenaline induces chemoresistance in HT-29 colon adenocarcinoma cells. Cancer Genet. Cytogenet. 2009, 190, 81–87.
- Kerr, L.R.; Hundal, R.; Silva, W.A.; Emerman, J.T.; Weinberg, J. Effects of social housing condition on chemotherapeutic efficacy in a Shionogi carcinoma (SC115) mouse tumor model: Influences of temporal factors, tumor size, and tumor growth rate. Psychosom. Med. 2001, 63, 973–984.
- Kim, J.M.; Chen, D.S. Immune escape to PD-L1/PD-1 blockade: Seven steps to success (or failure). Ann. Oncol. 2016, 27, 1492–1504.
- Melichar, B.; Nash, M.A.; Lenzi, R.; Platsoucas, C.D.; Freedman, R.S. Expression of costimulatory molecules CD80 and CD86 and their receptors CD28, CTLA-4 on malignant ascites CD3+ tumour-infiltrating lymphocytes (TIL) from patients with ovarian and other types of peritoneal carcinomatosis. Clin. Exp. Immunol. 2000, 119, 19–27.
- Intlekofer, A.M.; Thompson, C.B. At the bench: Preclinical rationale for CTLA-4 and PD-1 blockade as cancer immunotherapy. J. Leukoc. Biol. 2013, 94, 25–39.
- Strater, J.; Hinz, U.; Hasel, C.; Bhanot, U.; Mechtersheimer, G.; Lehnert, T.; Moller, P. Impaired CD95 expression predisposes for recurrence in curatively resected colon carcinoma: Clinical evidence for immunoselection and CD95L mediated control of minimal residual disease. Gut 2005, 54, 661–665.
- Liu, K. Role of apoptosis resistance in immune evasion and metastasis of colorectal cancer. World J. Gastrointest. Oncol. 2010, 2, 399–406.
- Cheng, W.F.; Lee, C.N.; Chang, M.C.; Su, Y.N.; Chen, C.A.; Hsieh, C.Y. Antigen-specific CD8+ T lymphocytes generated from a DNA vaccine control tumors through the Fas-FasL pathway. Mol. Ther. 2005, 12, 960–968.
- Rodriguez, J.A.; Huerta-Yepez, S.; Law, I.K.; Baay-Guzman, G.J.; Tirado-Rodriguez, B.; Hoffman, J.M.; Iliopoulos, D.; Hommes, D.W.; Verspaget, H.W.; Chang, L.; et al. Diminished expression of CRHR2 in human colon cancer promotes tumor growth and EMT via persistent IL-6/Stat3 signaling. Cell. Mol. Gastroenterol. Hepatol. 2015, 1, 610–630.
- Zhang, N.; Li, X.; Wu, C.W.; Dong, Y.; Cai, M.; Mok, M.T.; Wang, H.; Chen, J.; Ng, S.S.; Chen, M.; et al. microRNA-7 is a novel inhibitor of YY1 contributing to colorectal tumorigenesis. Oncogene 2013, 32, 5078–5088.
- Garnier, A.; Vykoukal, J.; Hubertus, J.; Alt, E.; von Schweinitz, D.; Kappler, R.; Berger, M.; Ilmer, M. Targeting the neurokinin-1 receptor inhibits growth of human colon cancer cells. Int. J. Oncol. 2015, 47, 151–160.
- Shi, Z.; Zhang, J.; Qian, X.; Han, L.; Zhang, K.; Chen, L.; Liu, J.; Ren, Y.; Yang, M.; Zhang, A.; et al. AC1MMYR2, an inhibitor of dicer-mediated biogenesis of Oncomir miR-21, reverses epithelial-mesenchymal transition and suppresses tumor growth and progression. Cancer Res. 2013, 73, 5519–5531.
- Qin, Y.; Li, L.; Wang, F.; Zhou, X.; Liu, Y.; Yin, Y.; Qi, X. Knockdown of Mir-135b Sensitizes Colorectal Cancer Cells to Oxaliplatin-Induced Apoptosis Through Increase of FOXO1. Cell. Physiol. Biochem. 2018, 48, 1628–1637.
- Ostermann, A.L.; Wunderlich, C.M.; Schneiders, L.; Vogt, M.C.; Woeste, M.A.; Belgardt, B.F.; Niessen, C.M.; Martiny, B.; Schauss, A.C.; Frommolt, P.; et al. Intestinal insulin/IGF1 signalling through FoxO1 regulates epithelial integrity and susceptibility to colon cancer. Nat. Metab. 2019, 1, 371–389.
- Xin, Z.; Jiang, S.; Jiang, P.; Yan, X.; Fan, C.; Di, S.; Wu, G.; Yang, Y.; Reiter, R.J.; Ji, G. Melatonin as a treatment for gastrointestinal cancer: A review. J. Pineal Res. 2015, 58, 375–387.
- Sakatani, A.; Sonohara, F.; Goel, A. Melatonin-mediated downregulation of thymidylate synthase as a novel mechanism for overcoming 5-fluorouracil associated chemoresistance in colorectal cancer cells. Carcinogenesis 2019, 40, 422–431.





