
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Chrysoula (Chrysa) Voidarou | -- | 3479 | 2024-03-18 09:19:27 | | | |
| 2 | Mona Zou | Meta information modification | 3479 | 2024-03-19 10:17:57 | | |
Video Upload Options
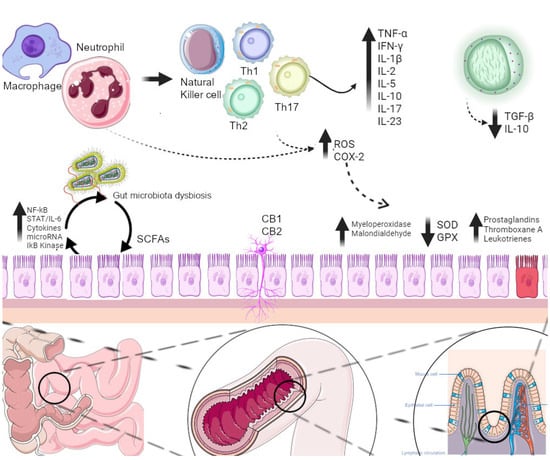
Inflammatory bowel disease (IBD) mainly comprises Crohn’s disease (CD) and ulcerative colitis (UC) and is a group of chronic relapsing disorders characterized by inflammation of the gastrointestinal tract with variable phenotypic expression. Multiple factors have been implicated in the etiology of IBD, including environmental, genetic, microbiological and immunological interactions. However, the exact reasons remain unclear, although substantial progress in elucidating the complexity of IBD manifestation has been made in the past decades. There is clinical overlap of symptoms of CD and UC, including bloody or watery diarrhea, recurrent abdominal pain, tenesmus as well as non-specific systemic symptoms such as fatigue, fever and weight loss. CD can affect various parts of the intestine, i.e., both small and large intestine, while UC is known to affect only the colon. IBD usually follows a lifelong pattern of remissions and flare-ups that impacts the quality of life of patients. The inflammation of the gastrointestinal tract during flare-ups is mediated by neutrophils that release cytokines, enzymes and reactive oxygen species (ROS) leading to damage and even ulceration of the mucosa.
1. Introduction
2. Phytochemicals
2.1. Classification of Compounds
2.2. Phytochemical Administration and Delivery in IBD Patients
3. IBD
3.1. Pathophysiological Pathways of Inflammation and Oxidative Stress
3.2. Current Practice for IBD Treatment
4. Antioxidant and Anti-Inflammatory Pathways Targeted by Phytochemicals for the Treatment of IBD
References
- Mahadevan, U.; Silverberg, M.S. Inflammatory Bowel Disease—Gastroenterology Diamond Jubilee Review. Gastroenterology 2018, 154, 1555–1558.
- Flynn, S.; Eisenstein, S. Inflammatory Bowel Disease Presentation and Diagnosis. Surg. Clin. N. Am. 2019, 99, 1051–1062.
- Guan, Q. A Comprehensive Review and Update on the Pathogenesis of Inflammatory Bowel Disease. J. Immunol. Res. 2019, 2019, 7247238.
- Imbrizi, M.; Magro, F.; Coy, C.S.R. Pharmacological Therapy in Inflammatory Bowel Diseases: A Narrative Review of the Past 90 Years. Pharmaceuticals 2023, 16, 1272.
- Kumar, A.; Nirmal, P.; Kumar, M.; Jose, A.; Tomer, V.; Oz, E.; Proestos, C.; Zeng, M.; Elobeid, T.; Sneha, K.; et al. Major Phytochemicals: Recent Advances in Health Benefits and Extraction Method. Molecules 2023, 28, 887.
- Lai, P.K.; Roy, J. Antimicrobial and Chemopreventive Properties of Herbs and Spices. Curr. Med. Chem. 2004, 11, 1451–1460.
- Campos-Vega, R.; Oomah, B.D. Chemistry and Classification of Phytochemicals. In Handbook of Plant Food Phytochemicals; Tiwari, B.K., Brunton, N.P., Brennan, C.S., Eds.; Wiley: Hoboken, NJ, USA, 2013; pp. 5–48. ISBN 978-1-4443-3810-2.
- Zhang, Y.-J.; Gan, R.-Y.; Li, S.; Zhou, Y.; Li, A.-N.; Xu, D.-P.; Li, H.-B. Antioxidant Phytochemicals for the Prevention and Treatment of Chronic Diseases. Molecules 2015, 20, 21138–21156.
- Tapsell, L.C.; Hemphill, I.; Cobiac, L.; Patch, C.S.; Sullivan, D.R.; Fenech, M.; Roodenrys, S.; Keogh, J.B.; Clifton, P.M.; Williams, P.G.; et al. Health Benefits of Herbs and Spices: The Past, the Present, the Future. Med. J. Aust. 2006, 185, S1–S24.
- Borges, A.; Saavedra, M.J.; Simões, M. Insights on Antimicrobial Resistance, Biofilms and the Use of Phytochemicals as New Antimicrobial Agents. Curr. Med. Chem. 2015, 22, 2590–2614.
- Shin, S.A.; Joo, B.J.; Lee, J.S.; Ryu, G.; Han, M.; Kim, W.Y.; Park, H.H.; Lee, J.H.; Lee, C.S. Phytochemicals as Anti-Inflammatory Agents in Animal Models of Prevalent Inflammatory Diseases. Molecules 2020, 25, 5932.
- Abubakar, A.R.; Haque, M. Preparation of Medicinal Plants: Basic Extraction and Fractionation Procedures for Experimental Purposes. J. Pharm. Bioallied Sci. 2020, 12, 1–10.
- Nahrstedt, A.; Butterweck, V. Lessons Learned from Herbal Medicinal Products: The Example of St. John’s Wort (Perpendicular). J. Nat. Prod. 2010, 73, 1015–1021.
- Okoli, E.C.; Umaru, I.J.; Olawale, O. Determination of Phytochemical Constituents, Antibacterial and Antioxidant Activities of Ethanolic Leaf Extracts of Pterocarpus Erinaceus. Biodiversitas 2023, 24, 2272–2277.
- Huang, Y.; Xiao, D.; Burton-Freeman, B.M.; Edirisinghe, I. Chemical Changes of Bioactive Phytochemicals during Thermal Processing. In Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2016; p. B9780081005965030559. ISBN 978-0-08-100596-5.
- Rowland, I. Optimal Nutrition: Fibre and Phytochemicals. Proc. Nutr. Soc. 1999, 58, 415–419.
- Yanai, H.; Levine, A.; Hirsch, A.; Boneh, R.S.; Kopylov, U.; Eran, H.B.; Cohen, N.A.; Ron, Y.; Goren, I.; Leibovitzh, H.; et al. The Crohn’s Disease Exclusion Diet for Induction and Maintenance of Remission in Adults with Mild-to-Moderate Crohn’s Disease (CDED-AD): An Open-Label, Pilot, Randomised Trial. Lancet Gastroenterol. Hepatol. 2022, 7, 49–59.
- Peng, Z.; Yi, J.; Liu, X. A Low-FODMAP Diet Provides Benefits for Functional Gastrointestinal Symptoms but Not for Improving Stool Consistency and Mucosal Inflammation in IBD: A Systematic Review and Meta-Analysis. Nutrients 2022, 14, 2072.
- Fitzpatrick, J.A.; Melton, S.L.; Yao, C.K.; Gibson, P.R.; Halmos, E.P. Dietary Management of Adults with IBD—The Emerging Role of Dietary Therapy. Nat. Rev. Gastroenterol. Hepatol. 2022, 19, 652–669.
- Sharma, R.; Padwad, Y. Plant-Polyphenols Based Second-Generation Synbiotics: Emerging Concepts, Challenges, and Opportunities. Nutrition 2020, 77, 110785.
- Glassner, K.L.; Abraham, B.P.; Quigley, E.M.M. The Microbiome and Inflammatory Bowel Disease. J. Allergy Clin. Immunol. 2020, 145, 16–27.
- Willing, B.P.; Dicksved, J.; Halfvarson, J.; Andersson, A.F.; Lucio, M.; Zheng, Z.; Järnerot, G.; Tysk, C.; Jansson, J.K.; Engstrand, L. A Pyrosequencing Study in Twins Shows That Gastrointestinal Microbial Profiles Vary with Inflammatory Bowel Disease Phenotypes. Gastroenterology 2010, 139, 1844–1854.e1.
- Aniwan, S.; Tremaine, W.J.; Raffals, L.E.; Kane, S.V.; Loftus, E.V. Antibiotic Use and New-Onset Inflammatory Bowel Disease in Olmsted County, Minnesota: A Population-Based Case-Control Study. J. Crohn’s Colitis 2018, 12, 137–144.
- Morgan, X.C.; Tickle, T.L.; Sokol, H.; Gevers, D.; Devaney, K.L.; Ward, D.V.; Reyes, J.A.; Shah, S.A.; LeLeiko, N.; Snapper, S.B.; et al. Dysfunction of the Intestinal Microbiome in Inflammatory Bowel Disease and Treatment. Genome Biol. 2012, 13, R79.
- Atarashi, K.; Tanoue, T.; Shima, T.; Imaoka, A.; Kuwahara, T.; Momose, Y.; Cheng, G.; Yamasaki, S.; Saito, T.; Ohba, Y.; et al. Induction of Colonic Regulatory T Cells by Indigenous Clostridium Species. Science 2011, 331, 337–341.
- Eslick, S.; Thompson, C.; Berthon, B.; Wood, L. Short-Chain Fatty Acids as Anti-Inflammatory Agents in Overweight and Obesity: A Systematic Review and Meta-Analysis. Nutr. Rev. 2022, 80, 838–856.
- Kabeerdoss, J.; Sankaran, V.; Pugazhendhi, S.; Ramakrishna, B.S. Clostridium Leptum Group Bacteria Abundance and Diversity in the Fecal Microbiota of Patients with Inflammatory Bowel Disease: A Case-Control Study in India. BMC Gastroenterol. 2013, 13, 20.
- Li, D.; Wang, P.; Wang, P.; Hu, X.; Chen, F. The Gut Microbiota: A Treasure for Human Health. Biotechnol. Adv. 2016, 34, 1210–1224.
- Sokol, H.; Pigneur, B.; Watterlot, L.; Lakhdari, O.; Bermúdez-Humarán, L.G.; Gratadoux, J.-J.; Blugeon, S.; Bridonneau, C.; Furet, J.-P.; Corthier, G.; et al. Faecalibacterium Prausnitzii Is an Anti-Inflammatory Commensal Bacterium Identified by Gut Microbiota Analysis of Crohn Disease Patients. Proc. Natl. Acad. Sci. USA 2008, 105, 16731–16736.
- Ardizzone, S.; Bianchi Porro, G. Biologic Therapy for Inflammatory Bowel Disease. Drugs 2005, 65, 2253–2286.
- Dolan, K.T.; Chang, E.B. Diet, Gut Microbes, and the Pathogenesis of Inflammatory Bowel Diseases. Mol. Nutr. Food Res. 2017, 61, 1600129.
- Hibi, T.; Ogata, H. Novel Pathophysiological Concepts of Inflammatory Bowel Disease. J. Gastroenterol. 2006, 41, 10–16.
- Kostovcikova, K.; Coufal, S.; Galanova, N.; Fajstova, A.; Hudcovic, T.; Kostovcik, M.; Prochazkova, P.; Jiraskova Zakostelska, Z.; Cermakova, M.; Sediva, B.; et al. Diet Rich in Animal Protein Promotes Pro-Inflammatory Macrophage Response and Exacerbates Colitis in Mice. Front. Immunol. 2019, 10, 919.
- Ho, S.-M.; Lewis, J.D.; Mayer, E.A.; Plevy, S.E.; Chuang, E.; Rappaport, S.M.; Croitoru, K.; Korzenik, J.R.; Krischer, J.; Hyams, J.S.; et al. Challenges in IBD Research: Environmental Triggers. Inflamm. Bowel Dis. 2019, 25, S13–S23.
- Korzenik, J.R.; Podolsky, D.K. Evolving Knowledge and Therapy of Inflammatory Bowel Disease. Nat. Rev. Drug Discov. 2006, 5, 197–209.
- Ince, M.N.; Elliott, D.E. Immunologic and Molecular Mechanisms in Inflammatory Bowel Disease. Surg. Clin. N. Am. 2007, 87, 681–696.
- Abraham, C.; Cho, J.H. Inflammatory Bowel Disease. N. Engl. J. Med. 2009, 361, 2066–2078.
- Gandhi, G.R.; Mohana, T.; Athesh, K.; Hillary, V.E.; Vasconcelos, A.B.S.; Farias De Franca, M.N.; Montalvão, M.M.; Ceasar, S.A.; Jothi, G.; Sridharan, G.; et al. Anti-Inflammatory Natural Products Modulate Interleukins and Their Related Signaling Markers in Inflammatory Bowel Disease: A Systematic Review. J. Pharm. Anal. 2023, 13, 1408–1428.
- Moura, F.A.; De Andrade, K.Q.; Dos Santos, J.C.F.; Araújo, O.R.P.; Goulart, M.O.F. Antioxidant Therapy for Treatment of Inflammatory Bowel Disease: Does It Work? Redox Biol. 2015, 6, 617–639.
- Goyette, P.; Labbé, C.; Trinh, T.T.; Xavier, R.J.; Rioux, J.D. Molecular Pathogenesis of Inflammatory Bowel Disease: Genotypes, Phenotypes and Personalized Medicine. Ann. Med. 2007, 39, 177–199.
- Bhattacharyya, A.; Chattopadhyay, R.; Mitra, S.; Crowe, S.E. Oxidative Stress: An Essential Factor in the Pathogenesis of Gastrointestinal Mucosal Diseases. Physiol. Rev. 2014, 94, 329–354.
- Fraga, C.G.; Oteiza, P.I. Iron Toxicity and Antioxidant Nutrients. Toxicology 2002, 180, 23–32.
- Bergmark, E.; Calleman, C.J.; He, F.; Costa, L.G. Determination of Hemoglobin Adducts in Humans Occupationally Exposed to Acrylamide. Toxicol. Appl. Pharmacol. 1993, 120, 45–54.
- Deledda, A.; Annunziata, G.; Tenore, G.C.; Palmas, V.; Manzin, A.; Velluzzi, F. Diet-Derived Antioxidants and Their Role in Inflammation, Obesity and Gut Microbiota Modulation. Antioxidants 2021, 10, 708.
- Saleh, H.A.; Yousef, M.H.; Abdelnaser, A. The Anti-Inflammatory Properties of Phytochemicals and Their Effects on Epigenetic Mechanisms Involved in TLR4/NF-κB-Mediated Inflammation. Front. Immunol. 2021, 12, 606069.
- Direito, R.; Barbalho, S.M.; Figueira, M.E.; Minniti, G.; de Carvalho, G.M.; de Oliveira Zanuso, B.; de Oliveira Dos Santos, A.R.; de Góes Corrêa, N.; Rodrigues, V.D.; de Alvares Goulart, R.; et al. Medicinal Plants, Phytochemicals and Regulation of the NLRP3 Inflammasome in Inflammatory Bowel Diseases: A Comprehensive Review. Metabolites 2023, 13, 728.
- Hossen, I.; Hua, W.; Ting, L.; Mehmood, A.; Jingyi, S.; Duoxia, X.; Yanping, C.; Hongqing, W.; Zhipeng, G.; Kaiqi, Z.; et al. Phytochemicals and Inflammatory Bowel Disease: A Review. Crit. Rev. Food Sci. Nutr. 2020, 60, 1321–1345.
- Somani, S.J.; Modi, K.P.; Majumdar, A.S.; Sadarani, B.N. Phytochemicals and Their Potential Usefulness in Inflammatory Bowel Disease. Phytother. Res. 2015, 29, 339–350.
- Riaz Rajoka, M.S.; Thirumdas, R.; Mehwish, H.M.; Umair, M.; Khurshid, M.; Hayat, H.F.; Phimolsiripol, Y.; Pallarés, N.; Martí-Quijal, F.J.; Barba, F.J. Role of Food Antioxidants in Modulating Gut Microbial Communities: Novel Understandings in Intestinal Oxidative Stress Damage and Their Impact on Host Health. Antioxidants 2021, 10, 1563.
- Li, L.; Peng, P.; Ding, N.; Jia, W.; Huang, C.; Tang, Y. Oxidative Stress, Inflammation, Gut Dysbiosis: What Can Polyphenols Do in Inflammatory Bowel Disease? Antioxidants 2023, 12, 967.
- Punchard, N.A.; Greenfield, S.M.; Thompson, R.P. Mechanism of Action of 5-Arninosalicylic Acid. Mediat. Inflamm. 1992, 1, 151–165.
- Creed, T.J.; Probert, C.S.J. Review Article: Steroid Resistance in Inflammatory Bowel Disease—Mechanisms and Therapeutic Strategies. Aliment. Pharmacol. Ther. 2007, 25, 111–122.
- Cai, Z.; Wang, S.; Li, J. Treatment of Inflammatory Bowel Disease: A Comprehensive Review. Front. Med. 2021, 8, 765474.
- Sandborn, W.J.; Rutgeerts, P.; Feagan, B.G.; Reinisch, W.; Olson, A.; Johanns, J.; Lu, J.; Horgan, K.; Rachmilewitz, D.; Hanauer, S.B.; et al. Colectomy Rate Comparison after Treatment of Ulcerative Colitis with Placebo or Infliximab. Gastroenterology 2009, 137, 1250–1260.
- M’Koma, A.E. Inflammatory Bowel Disease: Clinical Diagnosis and Surgical Treatment-Overview. Medicina 2022, 58, 567.
- Chao, Y.-S.; Loshak, H. Biologics Versus Immunomodulators for the Treatment of Ulcerative Colitis: A Review of Comparative Clinical Effectiveness and Cost-Effectiveness; CADTH Rapid Response Reports; Canadian Agency for Drugs and Technologies in Health: Ottawa, ON, Canada, 2019.
- Kotla, N.G.; Rochev, Y. IBD Disease-Modifying Therapies: Insights from Emerging Therapeutics. Trends. Mol. Med. 2023, 29, 241–253.
- Fernández-Bañares, F.; Hinojosa, J.; Sánchez-Lombraña, J.L.; Navarro, E.; Martínez-Salmerón, J.F.; García-Pugés, A.; González-Huix, F.; Riera, J.; González-Lara, V.; Domínguez-Abascal, F.; et al. Randomized Clinical Trial of Plantago Ovata Seeds (Dietary Fiber) as Compared with Mesalamine in Maintaining Remission in Ulcerative Colitis. Spanish Group for the Study of Crohn’s Disease and Ulcerative Colitis (GETECCU). Am. J. Gastroenterol. 1999, 94, 427–433.
- Roediger, W.E. The Colonic Epithelium in Ulcerative Colitis: An Energy-Deficiency Disease? Lancet 1980, 2, 712–715.
- Segain, J.P.; Raingeard de la Blétière, D.; Bourreille, A.; Leray, V.; Gervois, N.; Rosales, C.; Ferrier, L.; Bonnet, C.; Blottière, H.M.; Galmiche, J.P. Butyrate Inhibits Inflammatory Responses through NFkappaB Inhibition: Implications for Crohn’s Disease. Gut 2000, 47, 397–403.
- McIntyre, A.; Gibson, P.R.; Young, G.P. Butyrate Production from Dietary Fibre and Protection against Large Bowel Cancer in a Rat Model. Gut 1993, 34, 386–391.
- Facchin, S.; Vitulo, N.; Calgaro, M.; Buda, A.; Romualdi, C.; Pohl, D.; Perini, B.; Lorenzon, G.; Marinelli, C.; D’Incà, R.; et al. Microbiota Changes Induced by Microencapsulated Sodium Butyrate in Patients with Inflammatory Bowel Disease. Neurogastroenterol. Motil. 2020, 32, e13914.
- Jamka, M.; Kokot, M.; Kaczmarek, N.; Bermagambetova, S.; Nowak, J.K.; Walkowiak, J. The Effect of Sodium Butyrate Enemas Compared with Placebo on Disease Activity, Endoscopic Scores, and Histological and Inflammatory Parameters in Inflammatory Bowel Diseases: A Systematic Review of Randomised Controlled Trials. Complement. Med. Res. 2021, 28, 344–356.
- Pietrzak, A.; Banasiuk, M.; Szczepanik, M.; Borys-Iwanicka, A.; Pytrus, T.; Walkowiak, J.; Banaszkiewicz, A. Sodium Butyrate Effectiveness in Children and Adolescents with Newly Diagnosed Inflammatory Bowel Diseases-Randomized Placebo-Controlled Multicenter Trial. Nutrients 2022, 14, 3283.
- Greenfield, S.M.; Green, A.T.; Teare, J.P.; Jenkins, A.P.; Punchard, N.A.; Ainley, C.C.; Thompson, R.P. A Randomized Controlled Study of Evening Primrose Oil and Fish Oil in Ulcerative Colitis. Aliment. Pharmacol. Ther. 1993, 7, 159–166.
- Nishida, T.; Miwa, H.; Shigematsu, A.; Yamamoto, M.; Iida, M.; Fujishima, M. Increased Arachidonic Acid Composition of Phospholipids in Colonic Mucosa from Patients with Active Ulcerative Colitis. Gut 1987, 28, 1002–1007.
- Kirtland, S.J. Prostaglandin E1: A Review. Prostaglandins Leukot. Essent. Fat. Acids 1988, 32, 165–174.
- González-Domínguez, Á.; Belmonte, T.; González-Domínguez, R. Childhood Obesity, Metabolic Syndrome, and Oxidative Stress: MicroRNAs Go on Stage. Rev. Endocr. Metab. Disord. 2023, 24, 1147–1164.
- Boesch-Saadatmandi, C.; Loboda, A.; Wagner, A.E.; Stachurska, A.; Jozkowicz, A.; Dulak, J.; Döring, F.; Wolffram, S.; Rimbach, G. Effect of Quercetin and Its Metabolites Isorhamnetin and Quercetin-3-Glucuronide on Inflammatory Gene Expression: Role of miR-155. J. Nutr. Biochem. 2011, 22, 293–299.
- Papada, E.; Kaliora, A.C. Antioxidant and Anti-Inflammatory Properties of Mastiha: A Review of Preclinical and Clinical Studies. Antioxidants 2019, 8, 208.
- Amerikanou, C.; Papada, E.; Gioxari, A.; Smyrnioudis, I.; Kleftaki, S.-A.; Valsamidou, E.; Bruns, V.; Banerjee, R.; Trivella, M.G.; Milic, N.; et al. Mastiha Has Efficacy in Immune-Mediated Inflammatory Diseases through a microRNA-155 Th17 Dependent Action. Pharmacol. Res. 2021, 171, 105753.
- Jiang, Q.; Liu, P.; Wu, X.; Liu, W.; Shen, X.; Lan, T.; Xu, S.; Peng, J.; Xie, X.; Huang, H. Berberine Attenuates Lipopolysaccharide-Induced Extracelluar Matrix Accumulation and Inflammation in Rat Mesangial Cells: Involvement of NF-κB Signaling Pathway. Mol. Cell Endocrinol. 2011, 331, 34–40.
- Vuddanda, P.R.; Chakraborty, S.; Singh, S. Berberine: A Potential Phytochemical with Multispectrum Therapeutic Activities. Expert Opin. Investig. Drugs 2010, 19, 1297–1307.
- Xu, L.; Zhang, Y.; Xue, X.; Liu, J.; Li, Z.-S.; Yang, G.-Y.; Song, Y.; Pan, Y.; Ma, Y.; Hu, S.; et al. A Phase I Trial of Berberine in Chinese with Ulcerative Colitis. Cancer Prev. Res. 2020, 13, 117–126.
- Wu, L.-H.; Xu, Z.-L.; Dong, D.; He, S.-A.; Yu, H. Protective Effect of Anthocyanins Extract from Blueberry on TNBS-Induced IBD Model of Mice. Evid. Based Complement. Altern. Med. 2011, 2011, 525462.
- Osman, N.; Adawi, D.; Ahrné, S.; Jeppsson, B.; Molin, G. Probiotics and Blueberry Attenuate the Severity of Dextran Sulfate Sodium (DSS)-Induced Colitis. Dig. Dis. Sci. 2008, 53, 2464–2473.
- Montrose, D.C.; Horelik, N.A.; Madigan, J.P.; Stoner, G.D.; Wang, L.-S.; Bruno, R.S.; Park, H.J.; Giardina, C.; Rosenberg, D.W. Anti-Inflammatory Effects of Freeze-Dried Black Raspberry Powder in Ulcerative Colitis. Carcinogenesis 2011, 32, 343–350.
- Balmus, I.M.; Ciobica, A.; Trifan, A.; Stanciu, C. The Implications of Oxidative Stress and Antioxidant Therapies in Inflammatory Bowel Disease: Clinical Aspects and Animal Models. Saudi J. Gastroenterol. 2016, 22, 3–17.
- Young Hong, M. Effects of Resveratrol on Inflammatory Bowel Disease: A Review. J. Nutr. Health Food Sci. 2014, 2, 1–6.
- Yao, J.; Wang, J.-Y.; Liu, L.; Li, Y.-X.; Xun, A.-Y.; Zeng, W.-S.; Jia, C.-H.; Wei, X.-X.; Feng, J.-L.; Zhao, L.; et al. Anti-Oxidant Effects of Resveratrol on Mice with DSS-Induced Ulcerative Colitis. Arch. Med. Res. 2010, 41, 288–294.
- Brasnyó, P.; Molnár, G.A.; Mohás, M.; Markó, L.; Laczy, B.; Cseh, J.; Mikolás, E.; Szijártó, I.A.; Mérei, A.; Halmai, R.; et al. Resveratrol Improves Insulin Sensitivity, Reduces Oxidative Stress and Activates the Akt Pathway in Type 2 Diabetic Patients. Br. J. Nutr. 2011, 106, 383–389.
- Singh, R.; Akhtar, N.; Haqqi, T.M. Green Tea Polyphenol Epigallocatechin-3-Gallate: Inflammation and Arthritis. Life Sci. 2010, 86, 907–918.
- Khan, N.; Mukhtar, H. Multitargeted Therapy of Cancer by Green Tea Polyphenols. Cancer Lett. 2008, 269, 269–280.
- Grimm, T.; Chovanová, Z.; Muchová, J.; Sumegová, K.; Liptáková, A.; Duracková, Z.; Högger, P. Inhibition of NF-kappaB Activation and MMP-9 Secretion by Plasma of Human Volunteers after Ingestion of Maritime Pine Bark Extract (Pycnogenol). J. Inflamm. 2006, 3, 1.
- Rohdewald, P. A Review of the French Maritime Pine Bark Extract (Pycnogenol), a Herbal Medication with a Diverse Clinical Pharmacology. Int. J. Clin. Pharmacol. Ther. 2002, 40, 158–168.
- Schäfer, A.; Chovanová, Z.; Muchová, J.; Sumegová, K.; Liptáková, A.; Duracková, Z.; Högger, P. Inhibition of COX-1 and COX-2 Activity by Plasma of Human Volunteers after Ingestion of French Maritime Pine Bark Extract (Pycnogenol). Biomed. Pharmacother. 2006, 60, 5–9.
- Koláček, M.; Muchová, J.; Dvořáková, M.; Paduchová, Z.; Žitňanová, I.; Čierna, I.; Országhová, Z.; Székyová, D.; Jajcaiová-Zedníčková, N.; Kovács, L.; et al. Effect of Natural Polyphenols (Pycnogenol) on Oxidative Stress Markers in Children Suffering from Crohn’s Disease—A Pilot Study. Free Radic. Res. 2013, 47, 624–634.
- Esmaily, H.; Hosseini-Tabatabaei, A.; Rahimian, R.; Khorasani, R.; Baeeri, M.; Barazesh-Morgani, A.; Yasa, N.; Khademi, Y.; Abdollahi, M. On the Benefits of Silymarin in Murine Colitis by Improving Balance of Destructive Cytokines and Reduction of Toxic Stress in the Bowel Cells. Open Life Sci. 2009, 4, 204–213.
- Miroliaee, A.E.; Esmaily, H.; Vaziri-Bami, A.; Baeeri, M.; Shahverdi, A.R.; Abdollahi, M. Amelioration of Experimental Colitis by a Novel Nanoselenium-Silymarin Mixture. Toxicol. Mech. Methods 2011, 21, 200–208.
- Esmaily, H.; Vaziri-Bami, A.; Miroliaee, A.E.; Baeeri, M.; Abdollahi, M. The Correlation between NF-κB Inhibition and Disease Activity by Coadministration of Silibinin and Ursodeoxycholic Acid in Experimental Colitis. Fundam. Clin. Pharmacol. 2011, 25, 723–733.
- Huber, R.; Ditfurth, A.V.; Amann, F.; Güthlin, C.; Rostock, M.; Trittler, R.; Kümmerer, K.; Merfort, I. Tormentil for Active Ulcerative Colitis: An Open-Label, Dose-Escalating Study. J. Clin. Gastroenterol. 2007, 41, 834–838.
- Wei, X.; Gong, J.; Zhu, J.; Wang, P.; Li, N.; Zhu, W.; Li, J. The Suppressive Effect of Triptolide on Chronic Colitis and TNF-Alpha/TNFR2 Signal Pathway in Interleukin-10 Deficient Mice. Clin. Immunol. 2008, 129, 211–218.
- Yu, C.; Shan, T.; Feng, A.; Li, Y.; Zhu, W.; Xie, Y.; Li, N.; Li, J. Triptolide Ameliorates Crohn’s Colitis Is Associated with Inhibition of TLRs/NF-κB Signaling Pathway. Fitoterapia 2011, 82, 709–715.
- Li, Y.; Yu, C.; Zhu, W.-M.; Xie, Y.; Qi, X.; Li, N.; Li, J.-S. Triptolide Ameliorates IL-10-Deficient Mice Colitis by Mechanisms Involving Suppression of IL-6/STAT3 Signaling Pathway and down-Regulation of IL-17. Mol. Immunol. 2010, 47, 2467–2474.
- Li, G.; Ren, J.; Wang, G.; Gu, G.; Hu, D.; Ren, H.; Hong, Z.; Wu, X.; Liu, S.; Li, J. T2 Enhances in Situ Level of Foxp3+ Regulatory Cells and Modulates Inflammatory Cytokines in Crohn’s Disease. Int. Immunopharmacol. 2014, 18, 244–248.
- Zhang, C.; Sun, P.-P.; Guo, H.-T.; Liu, Y.; Li, J.; He, X.-J.; Lu, A.-P. Safety Profiles of Tripterygium Wilfordii Hook F: A Systematic Review and Meta-Analysis. Front. Pharmacol. 2016, 7, 402.
- Ito, Y.; Sugawara, Y.; Takaiti, O.; Nakamura, S. Metabolic Fate of a New Anti-Ulcer Drug (+)-(1R,4aS,10aR)-1,2,3,4,4a,9,10,10a- Octahydro-1,4a-Dimethyl-7-(1-Methylethyl)-6-Sulfo-1- Phenanthrenecarboxylic Acid 6-Sodium Salt Pentahydrate (TA-2711). II. Distribution in the Rat Stomach. J. Pharmacobiodyn. 1991, 14, 547–554.
- Kinoshita, M.; Kume, E.; Tamaki, H. Roles of Prostaglandins, Nitric Oxide and the Capsaicin-Sensitive Sensory Nerves in Gastroprotection Produced by Ecabet Sodium. J. Pharmacol. Exp. Ther. 1995, 275, 494–501.
- Kinoshita, M.; Tamaki, H. Possible Mechanism of Increase in Gastric Mucosal PGE2 and PGI2 Generation Induced by Ecabet Sodium, a Novel Gastroprotective Agent. Dig. Dis. Sci. 1997, 42, 83–90.
- Ichikawa, T.; Ishihara, K.; Hayashida, H.; Hiruma, H.; Saigenji, K.; Hotta, K. Effects of Ecabet Sodium, a Novel Gastroprotective Agent, on Mucin Metabolism in Rat Gastric Mucosa. Dig. Dis. Sci. 2000, 45, 606–613.
- Langhorst, J.; Varnhagen, I.; Schneider, S.B.; Albrecht, U.; Rueffer, A.; Stange, R.; Michalsen, A.; Dobos, G.J. Randomised Clinical Trial: A Herbal Preparation of Myrrh, Chamomile and Coffee Charcoal Compared with Mesalazine in Maintaining Remission in Ulcerative Colitis—A Double-Blind, Double-Dummy Study. Aliment. Pharmacol. Ther. 2013, 38, 490–500.
- Bonini, S.A.; Premoli, M.; Tambaro, S.; Kumar, A.; Maccarinelli, G.; Memo, M.; Mastinu, A. Cannabis Sativa: A Comprehensive Ethnopharmacological Review of a Medicinal Plant with a Long History. J. Ethnopharmacol. 2018, 227, 300–315.
- Patil, N.; Chandel, V.; Rana, A.; Jain, M.; Kaushik, P. Investigation of Cannabis Sativa Phytochemicals as Anti-Alzheimer’s Agents: An In Silico Study. Plants 2023, 12, 510.
- Pesce, M.; D’Alessandro, A.; Borrelli, O.; Gigli, S.; Seguella, L.; Cuomo, R.; Esposito, G.; Sarnelli, G. Endocannabinoid-Related Compounds in Gastrointestinal Diseases. J. Cell Mol. Med. 2018, 22, 706–715.
- Couch, D.G.; Maudslay, H.; Doleman, B.; Lund, J.N.; O’Sullivan, S.E. The Use of Cannabinoids in Colitis: A Systematic Review and Meta-Analysis. Inflamm. Bowel Dis. 2018, 24, 680–697.
- Marquéz, L.; Suárez, J.; Iglesias, M.; Bermudez-Silva, F.J.; Rodríguez de Fonseca, F.; Andreu, M. Ulcerative Colitis Induces Changes on the Expression of the Endocannabinoid System in the Human Colonic Tissue. PLoS ONE 2009, 4, e6893.
- Macfarlane, G.T.; Cummings, J.H. Probiotics and Prebiotics: Can Regulating the Activities of Intestinal Bacteria Benefit Health? BMJ 1999, 318, 999–1003.
- Steed, H.; Macfarlane, G.T.; Macfarlane, S. Prebiotics, Synbiotics and Inflammatory Bowel Disease. Mol. Nutr. Food Res. 2008, 52, 898–905.
- Satoskar, R.R.; Shah, S.J.; Shenoy, S.G. Evaluation of Anti-Inflammatory Property of Curcumin (Diferuloyl Methane) in Patients with Postoperative Inflammation. Int. J. Clin. Pharmacol. Ther. Toxicol. 1986, 24, 651–654.
- Surh, Y.-J. Anti-Tumor Promoting Potential of Selected Spice Ingredients with Antioxidative and Anti-Inflammatory Activities: A Short Review. Food Chem. Toxicol. 2002, 40, 1091–1097.
- Toda, S.; Miyase, T.; Arichi, H.; Tanizawa, H.; Takino, Y. Natural Antioxidants. III. Antioxidative Components Isolated from Rhizome of Curcuma Longa L. Chem. Pharm. Bull. 1985, 33, 1725–1728.
- Plummer, S.M.; Holloway, K.A.; Manson, M.M.; Munks, R.J.; Kaptein, A.; Farrow, S.; Howells, L. Inhibition of Cyclo-Oxygenase 2 Expression in Colon Cells by the Chemopreventive Agent Curcumin Involves Inhibition of NF-kappaB Activation via the NIK/IKK Signalling Complex. Oncogene 1999, 18, 6013–6020.
- Sugimoto, K.; Hanai, H.; Tozawa, K.; Aoshi, T.; Uchijima, M.; Nagata, T.; Koide, Y. Curcumin Prevents and Ameliorates Trinitrobenzene Sulfonic Acid-Induced Colitis in Mice. Gastroenterology 2002, 123, 1912–1922.
- Salh, B.; Assi, K.; Templeman, V.; Parhar, K.; Owen, D.; Gómez-Muñoz, A.; Jacobson, K. Curcumin Attenuates DNB-Induced Murine Colitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2003, 285, G235–G243.





