
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Huanyu Xiong | -- | 2723 | 2024-03-14 12:53:38 | | | |
| 2 | Lindsay Dong | Meta information modification | 2723 | 2024-03-15 07:41:10 | | |
Video Upload Options
Chronic pain is sustained, in part, through the intricate process of central sensitization (CS), marked by maladaptive neuroplasticity and neuronal hyperexcitability within central pain pathways. Accumulating evidence suggests that CS is also driven by neuroinflammation in the peripheral and central nervous system. In any chronic disease, the search for perpetuating factors is crucial in identifying therapeutic targets and developing primary preventive strategies. The brain-derived neurotrophic factor (BDNF) emerges as a critical regulator of synaptic plasticity, serving as both a neurotransmitter and neuromodulator. Mounting evidence supports BDNF’s pro-nociceptive role, spanning from its pain-sensitizing capacity across multiple levels of nociceptive pathways to its intricate involvement in CS and neuroinflammation.
1. Introduction
2. The Physiological Role of BDNF
3. The Role of BDNF in Patients with Chronic Pain
3.1. The Role of BDNF in Central Sensitization
3.2. Neuroinflammation Drives Chronic Pain via Glial-Derived BDNF and CS
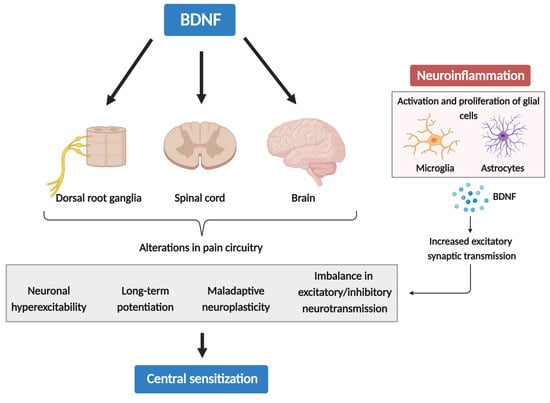
3.3. Pro-Nociceptive and Anti-Nociceptive Role of BDNF
3.4. Genetics and BDNF in Chronic Pain
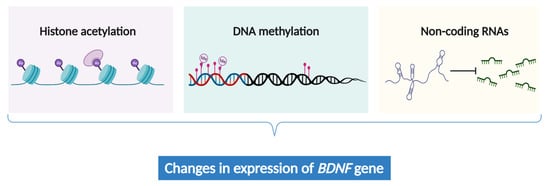
3.5. The Epigenetic Regulation of BDNF Expression in Chronic Pain
4. Clinical and Methodological Implications
4.1. BDNF Treatment for Chronic Pain in a Broader Picture
4.2. Using BDNF as an Objective Biomarker
References
- GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858.
- GBD 2021 Low Back Pain Collaborators. Global, regional, and national burden of low back pain, 1990–2020, its attributable risk factors, and projections to 2050: A systematic analysis of the Global Burden of Disease Study 2021. Lancet Rheumatol. 2023, 5, e316–e329.
- Cohen, S.P.; Vase, L.; Hooten, W.M. Chronic pain: An update on burden, best practices, and new advances. Lancet 2021, 397, 2082–2097.
- Mills, S.E.E.; Nicolson, K.P.; Smith, B.H. Chronic pain: A review of its epidemiology and associated factors in population-based studies. Br. J. Anaesth. 2019, 123, e273–e283.
- Bannister, K.; Sachau, J.; Baron, R.; Dickenson, A.H. Neuropathic Pain: Mechanism-Based Therapeutics. Annu. Rev. Pharmacol. Toxicol. 2020, 60, 257–274.
- Taneja, A.; Della Pasqua, O.; Danhof, M. Challenges in translational drug research in neuropathic and inflammatory pain: The prerequisites for a new paradigm. Eur. J. Clin. Pharmacol. 2017, 73, 1219–1236.
- Maher, D.P.; Wong, C.H.; Siah, K.W.; Lo, A.W. Estimates of Probabilities of Successful Development of Pain Medications: An Analysis of Pharmaceutical Clinical Development Programs from 2000 to 2020. Anesthesiology 2022, 137, 243–251.
- Basbaum, A.I.; Braz, J.M. Cell transplants to treat the “disease” of neuropathic pain and itch. Pain 2016, 157 (Suppl. 1), S42–S47.
- Moayedi, M.; Weissman-Fogel, I.; Salomons, T.V.; Crawley, A.P.; Goldberg, M.B.; Freeman, B.V.; Tenenbaum, H.C.; Davis, K.D. Abnormal gray matter aging in chronic pain patients. Brain Res. 2012, 1456, 82–93.
- Nijs, J.; George, S.Z.; Clauw, D.J.; Fernández-de-las-Peñas, C.; Kosek, E.; Ickmans, K.; Fernández-Carnero, J.; Polli, A.; Kapreli, E.; Huysmans, E.; et al. Central sensitisation in chronic pain conditions: Latest discoveries and their potential for precision medicine. Lancet Rheumatol. 2021, 3, e383–e392.
- Kregel, J.; Meeus, M.; Malfliet, A.; Dolphens, M.; Danneels, L.; Nijs, J.; Cagnie, B. Structural and functional brain abnormalities in chronic low back pain: A systematic review. Semin. Arthritis Rheum. 2015, 45, 229–237.
- Albrecht, D.S.; Forsberg, A.; Sandstrom, A.; Bergan, C.; Kadetoff, D.; Protsenko, E.; Lampa, J.; Lee, Y.C.; Hoglund, C.O.; Catana, C.; et al. Brain glial activation in fibromyalgia—A multi-site positron emission tomography investigation. Brain Behav. Immun. 2019, 75, 72–83.
- Arendt-Nielsen, L.; Morlion, B.; Perrot, S.; Dahan, A.; Dickenson, A.; Kress, H.G.; Wells, C.; Bouhassira, D.; Mohr Drewes, A. Assessment and manifestation of central sensitisation across different chronic pain conditions. Eur. J. Pain 2018, 22, 216–241.
- Kuner, R.; Flor, H. Structural plasticity and reorganisation in chronic pain. Nat. Rev. Neurosci. 2016, 18, 20–30.
- Smith, P.A. BDNF: No gain without pain? Neuroscience 2014, 283, 107–123.
- Kowianski, P.; Lietzau, G.; Czuba, E.; Waskow, M.; Steliga, A.; Morys, J. BDNF: A Key Factor with Multipotent Impact on Brain Signaling and Synaptic Plasticity. Cell Mol. Neurobiol. 2018, 38, 579–593.
- Dussan-Sarria, J.A.; da Silva, N.R.J.; Deitos, A.; Stefani, L.C.; Laste, G.; Souza, A.; Torres, I.L.S.; Fregni, F.; Caumo, W. Higher Cortical Facilitation and Serum BDNF Are Associated with Increased Sensitivity to Heat Pain and Reduced Endogenous Pain Inhibition in Healthy Males. Pain Med. 2018, 19, 1578–1586.
- Wei, X.; Wang, L.; Hua, J.; Jin, X.H.; Ji, F.; Peng, K.; Zhou, B.; Yang, J.; Meng, X.W. Inhibiting BDNF/TrkB.T1 receptor improves resiniferatoxin-induced postherpetic neuralgia through decreasing ASIC3 signaling in dorsal root ganglia. J. Neuroinflamm. 2021, 18, 96.
- Lima Giacobbo, B.; Doorduin, J.; Klein, H.C.; Dierckx, R.; Bromberg, E.; de Vries, E.F.J. Brain-Derived Neurotrophic Factor in Brain Disorders: Focus on Neuroinflammation. Mol. Neurobiol. 2019, 56, 3295–3312.
- Notaras, M.; van den Buuse, M. Neurobiology of BDNF in fear memory, sensitivity to stress, and stress-related disorders. Mol. Psychiatry 2020, 25, 2251–2274.
- Teixeira, A.L.; Barbosa, I.G.; Diniz, B.S.; Kummer, A. Circulating levels of brain-derived neurotrophic factor: Correlation with mood, cognition and motor function. Biomark. Med. 2010, 4, 871–887.
- Park, H.; Poo, M.M. Neurotrophin regulation of neural circuit development and function. Nat. Rev. Neurosci. 2013, 14, 7–23.
- Mattson, M.P.; Maudsley, S.; Martin, B. BDNF and 5-HT: A dynamic duo in age-related neuronal plasticity and neurodegenerative disorders. Trends Neurosci. 2004, 27, 589–594.
- Hempstead, B.L. Brain-Derived Neurotrophic Factor: Three Ligands, Many Actions. Trans. Am. Clin. Climatol. Assoc. 2015, 126, 9–19.
- Zhang, J.C.; Yao, W.; Hashimoto, K. Brain-derived Neurotrophic Factor (BDNF)-TrkB Signaling in Inflammation-related Depression and Potential Therapeutic Targets. Curr. Neuropharmacol. 2016, 14, 721–731.
- Andero, R.; Choi, D.C.; Ressler, K.J. BDNF-TrkB receptor regulation of distributed adult neural plasticity, memory formation, and psychiatric disorders. Prog. Mol. Biol. Transl. Sci. 2014, 122, 169–192.
- Cui, Y.H.; Zhou, S.F.; Liu, Y.; Wang, S.; Li, F.; Dai, R.P.; Hu, Z.L.; Li, C.Q. Injection of Anti-proBDNF Attenuates Hippocampal-Dependent Learning and Memory Dysfunction in Mice With Sepsis-Associated Encephalopathy. Front. Neurosci. 2021, 15, 665757.
- Woo, N.H.; Teng, H.K.; Siao, C.J.; Chiaruttini, C.; Pang, P.T.; Milner, T.A.; Hempstead, B.L.; Lu, B. Activation of p75NTR by proBDNF facilitates hippocampal long-term depression. Nat. Neurosci. 2005, 8, 1069–1077.
- Lu, B.; Pang, P.T.; Woo, N.H. The yin and yang of neurotrophin action. Nat. Rev. Neurosci. 2005, 6, 603–614.
- Hofer, M.; Pagliusi, S.R.; Hohn, A.; Leibrock, J.; Barde, Y.A. Regional distribution of brain-derived neurotrophic factor mRNA in the adult mouse brain. EMBO J. 1990, 9, 2459–2464.
- Brady, R.; Zaidi, S.I.; Mayer, C.; Katz, D.M. BDNF is a target-derived survival factor for arterial baroreceptor and chemoafferent primary sensory neurons. J. Neurosci. 1999, 19, 2131–2142.
- Biane, J.; Conner, J.M.; Tuszynski, M.H. Nerve growth factor is primarily produced by GABAergic neurons of the adult rat cortex. Front. Cell Neurosci. 2014, 8, 220.
- Bejot, Y.; Prigent-Tessier, A.; Cachia, C.; Giroud, M.; Mossiat, C.; Bertrand, N.; Garnier, P.; Marie, C. Time-dependent contribution of non neuronal cells to BDNF production after ischemic stroke in rats. Neurochem. Int. 2011, 58, 102–111.
- Taves, S.; Berta, T.; Chen, G.; Ji, R.R. Microglia and spinal cord synaptic plasticity in persistent pain. Neural Plast. 2013, 2013, 753656.
- Yan, Q.; Rosenfeld, R.D.; Matheson, C.R.; Hawkins, N.; Lopez, O.T.; Bennett, L.; Welcher, A.A. Expression of brain-derived neurotrophic factor protein in the adult rat central nervous system. Neuroscience 1997, 78, 431–448.
- Nakahashi, T.; Fujimura, H.; Altar, C.A.; Li, J.; Kambayashi, J.; Tandon, N.N.; Sun, B. Vascular endothelial cells synthesize and secrete brain-derived neurotrophic factor. FEBS Lett. 2000, 470, 113–117.
- Tamura, S.; Suzuki, H.; Hirowatari, Y.; Hatase, M.; Nagasawa, A.; Matsuno, K.; Kobayashi, S.; Moriyama, T. Release reaction of brain-derived neurotrophic factor (BDNF) through PAR1 activation and its two distinct pools in human platelets. Thromb. Res. 2011, 128, e55–e61.
- Le Blanc, J.; Fleury, S.; Boukhatem, I.; Belanger, J.C.; Welman, M.; Lordkipanidze, M. Platelets Selectively Regulate the Release of BDNF, But Not That of Its Precursor Protein, proBDNF. Front. Immunol. 2020, 11, 575607.
- Meacham, K.; Shepherd, A.; Mohapatra, D.P.; Haroutounian, S. Neuropathic Pain: Central vs. Peripheral Mechanisms. Curr. Pain. Headache Rep. 2017, 21, 28.
- Latremoliere, A.; Woolf, C.J. Central sensitization: A generator of pain hypersensitivity by central neural plasticity. J. Pain 2009, 10, 895–926.
- Retamal, J.; Reyes, A.; Ramirez, P.; Bravo, D.; Hernandez, A.; Pelissier, T.; Villanueva, L.; Constandil, L. Burst-Like Subcutaneous Electrical Stimulation Induces BDNF-Mediated, Cyclotraxin B-Sensitive Central Sensitization in Rat Spinal Cord. Front. Pharmacol. 2018, 9, 1143.
- Wang, H.; Wei, Y.; Pu, Y.; Jiang, D.; Jiang, X.; Zhang, Y.; Tao, J. Brain-derived neurotrophic factor stimulation of T-type Ca(2+) channels in sensory neurons contributes to increased peripheral pain sensitivity. Sci. Signal 2019, 12, eaaw2300.
- Echeverry, S.; Shi, X.Q.; Yang, M.; Huang, H.; Wu, Y.; Lorenzo, L.E.; Perez-Sanchez, J.; Bonin, R.P.; De Koninck, Y.; Zhang, J. Spinal microglia are required for long-term maintenance of neuropathic pain. Pain 2017, 158, 1792–1801.
- Ramer, L.M.; McPhail, L.T.; Borisoff, J.F.; Soril, L.J.; Kaan, T.K.; Lee, J.H.; Saunders, J.W.; Hwi, L.P.; Ramer, M.S. Endogenous TrkB ligands suppress functional mechanosensory plasticity in the deafferented spinal cord. J. Neurosci. 2007, 27, 5812–5822.
- Sikandar, S.; Minett, M.S.; Millet, Q.; Santana-Varela, S.; Lau, J.; Wood, J.N.; Zhao, J. Brain-derived neurotrophic factor derived from sensory neurons plays a critical role in chronic pain. Brain 2018, 141, 1028–1039.
- Zhang, Z.; Wang, X.; Wang, W.; Lu, Y.G.; Pan, Z.Z. Brain-derived neurotrophic factor-mediated downregulation of brainstem K+-Cl- cotransporter and cell-type-specific GABA impairment for activation of descending pain facilitation. Mol. Pharmacol. 2013, 84, 511–520.
- Lu, V.B.; Ballanyi, K.; Colmers, W.F.; Smith, P.A. Neuron type-specific effects of brain-derived neurotrophic factor in rat superficial dorsal horn and their relevance to ‘central sensitization’. J. Physiol. 2007, 584, 543–563.
- Woolf, C.J. Central sensitization: Implications for the diagnosis and treatment of pain. Pain 2011, 152, S2–S15.
- Nijs, J.; Meeus, M.; Versijpt, J.; Moens, M.; Bos, I.; Knaepen, K.; Meeusen, R. Brain-derived neurotrophic factor as a driving force behind neuroplasticity in neuropathic and central sensitization pain: A new therapeutic target? Expert. Opin. Ther. Targets 2015, 19, 565–576.
- Wang, C.S.; Kavalali, E.T.; Monteggia, L.M. BDNF signaling in context: From synaptic regulation to psychiatric disorders. Cell 2022, 185, 62–76.
- Cappoli, N.; Tabolacci, E.; Aceto, P.; Dello Russo, C. The emerging role of the BDNF-TrkB signaling pathway in the modulation of pain perception. J. Neuroimmunol. 2020, 349, 577406.
- Zhuo, M. A synaptic model for pain: Long-term potentiation in the anterior cingulate cortex. Mol. Cells 2007, 23, 259–271.
- Cheng, Q.; Song, S.H.; Augustine, G.J. Calcium-Dependent and Synapsin-Dependent Pathways for the Presynaptic Actions of BDNF. Front. Cell Neurosci. 2017, 11, 75.
- Metsis, M.; Timmusk, T.; Arenas, E.; Persson, H. Differential usage of multiple brain-derived neurotrophic factor promoters in the rat brain following neuronal activation. Proc. Natl. Acad. Sci. USA 1993, 90, 8802–8806.
- Zheng, Y.; Zhou, Y.; Wu, Q.; Yue, J.; Ying, X.; Li, S.; Lou, X.; Yang, G.; Tu, W.; Zhou, K.; et al. Effect of electroacupuncture on the expression of P2 x 4, GABAA gamma 2 and long-term potentiation in spinal cord of rats with neuropathic pain. Brain Res. Bull. 2020, 162, 1–10.
- Hollins, M.; Bryen, C.P.; Taylor, D. Effects of chronic pain history on perceptual and cognitive inhibition. Exp. Brain Res. 2020, 238, 321–332.
- Caumo, W.; Deitos, A.; Carvalho, S.; Leite, J.; Carvalho, F.; Dussan-Sarria, J.A.; Lopes Tarrago Mda, G.; Souza, A.; Torres, I.L.; Fregni, F. Motor Cortex Excitability and BDNF Levels in Chronic Musculoskeletal Pain According to Structural Pathology. Front. Hum. Neurosci. 2016, 10, 357.
- Simis, M.; Imamura, M.; de Melo, P.S.; Marduy, A.; Pacheco-Barrios, K.; Teixeira, P.E.P.; Battistella, L.; Fregni, F. Increased motor cortex inhibition as a marker of compensation to chronic pain in knee osteoarthritis. Sci. Rep. 2021, 11, 24011.
- Soldatelli, M.D.; Siepmann, T.; Illigens, B.M.; Souza Dos Santos, V.; Lucena da, S.T.I.; Fregni, F.; Caumo, W. Mapping of predictors of the disengagement of the descending inhibitory pain modulation system in fibromyalgia: An exploratory study. Br. J. Pain 2021, 15, 221–233.
- Zhao, S.; Wang, F.; Wang, L.; Xu, Y.; Lv, L.; Duan, W.; Bai, R.; Meng, Z.; Shao, X. Involvement of the BDNF-TrkB-KCC2 pathway in neuropathic pain after brachial plexus avulsion. Brain Behav. 2022, 12, e2464.
- Kitayama, T. The Role of K(+)-Cl(-)-Cotransporter-2 in Neuropathic Pain. Neurochem. Res. 2018, 43, 110–115.
- Ji, R.R.; Nackley, A.; Huh, Y.; Terrando, N.; Maixner, W. Neuroinflammation and Central Sensitization in Chronic and Widespread Pain. Anesthesiology 2018, 129, 343–366.
- Xiong, H.Y.; Zhang, Z.J.; Wang, X.Q. Bibliometric Analysis of Research on the Comorbidity of Pain and Inflammation. Pain Res. Manag. 2021, 2021, 6655211.
- Ji, R.R.; Chamessian, A.; Zhang, Y.Q. Pain regulation by non-neuronal cells and inflammation. Science 2016, 354, 572–577.
- Bathina, S.; Das, U.N. Brain-derived neurotrophic factor and its clinical implications. Arch. Med. Sci. 2015, 11, 1164–1178.
- Zhou, T.T.; Wu, J.R.; Chen, Z.Y.; Liu, Z.X.; Miao, B. Effects of dexmedetomidine on P2X4Rs, p38-MAPK and BDNF in spinal microglia in rats with spared nerve injury. Brain Res. 2014, 1568, 21–30.
- Coull, J.A.; Beggs, S.; Boudreau, D.; Boivin, D.; Tsuda, M.; Inoue, K.; Gravel, C.; Salter, M.W.; De Koninck, Y. BDNF from microglia causes the shift in neuronal anion gradient underlying neuropathic pain. Nature 2005, 438, 1017–1021.
- Ferrini, F.; Trang, T.; Mattioli, T.A.; Laffray, S.; Del’Guidice, T.; Lorenzo, L.E.; Castonguay, A.; Doyon, N.; Zhang, W.; Godin, A.G.; et al. Morphine hyperalgesia gated through microglia-mediated disruption of neuronal Cl(-) homeostasis. Nat. Neurosci. 2013, 16, 183–192.
- Marini, A.M.; Jiang, X.; Wu, X.; Tian, F.; Zhu, D.; Okagaki, P.; Lipsky, R.H. Role of brain-derived neurotrophic factor and NF-kappaB in neuronal plasticity and survival: From genes to phenotype. Restor. Neurol. Neurosci. 2004, 22, 121–130.
- Mannion, R.J.; Costigan, M.; Decosterd, I.; Amaya, F.; Ma, Q.P.; Holstege, J.C.; Ji, R.R.; Acheson, A.; Lindsay, R.M.; Wilkinson, G.A.; et al. Neurotrophins: Peripherally and centrally acting modulators of tactile stimulus-induced inflammatory pain hypersensitivity. Proc. Natl. Acad. Sci. USA 1999, 96, 9385–9390.
- Li, H.; Liu, T.; Sun, J.; Zhao, S.; Wang, X.; Luo, W.; Luo, R.; Shen, W.; Luo, C.; Fu, D. Up-Regulation of ProBDNF/p75(NTR) Signaling in Spinal Cord Drives Inflammatory Pain in Male Rats. J. Inflamm. Res. 2023, 16, 95–107.
- Lang, B.C.; Zhang, Z.; Lv, L.Y.; Liu, J.; Wang, T.Y.; Yang, L.H.; Liao, D.Q.; Zhang, W.S.; Wang, T.H. OECs transplantation results in neuropathic pain associated with BDNF regulating ERK activity in rats following cord hemisection. BMC Neurosci. 2013, 14, 80.
- Hempstead, B.L. The many faces of p75NTR. Curr. Opin. Neurobiol. 2002, 12, 260–267.
- Chao, M.V. Neurotrophins and their receptors: A convergence point for many signalling pathways. Nat. Rev. Neurosci. 2003, 4, 299–309.
- Slack, S.E.; Pezet, S.; McMahon, S.B.; Thompson, S.W.; Malcangio, M. Brain-derived neurotrophic factor induces NMDA receptor subunit one phosphorylation via ERK and PKC in the rat spinal cord. Eur. J. Neurosci. 2004, 20, 1769–1778.
- Sarchielli, P.; Mancini, M.L.; Floridi, A.; Coppola, F.; Rossi, C.; Nardi, K.; Acciarresi, M.; Pini, L.A.; Calabresi, P. Increased levels of neurotrophins are not specific for chronic migraine: Evidence from primary fibromyalgia syndrome. J. Pain 2007, 8, 737–745.
- Jablochkova, A.; Backryd, E.; Kosek, E.; Mannerkorpi, K.; Ernberg, M.; Gerdle, B.; Ghafouri, B. Unaltered low nerve growth factor and high brain-derived neurotrophic factor levels in plasma from patients with fibromyalgia after a 15-week progressive resistance exercise. J. Rehabil. Med. 2019, 51, 779–787.
- Haas, L.; Portela, L.V.; Bohmer, A.E.; Oses, J.P.; Lara, D.R. Increased plasma levels of brain derived neurotrophic factor (BDNF) in patients with fibromyalgia. Neurochem. Res. 2010, 35, 830–834.
- Simao, A.P.; Mendonca, V.A.; de Oliveira Almeida, T.M.; Santos, S.A.; Gomes, W.F.; Coimbra, C.C.; Lacerda, A.C. Involvement of BDNF in knee osteoarthritis: The relationship with inflammation and clinical parameters. Rheumatol. Int. 2014, 34, 1153–1157.
- Laske, C.; Stransky, E.; Eschweiler, G.W.; Klein, R.; Wittorf, A.; Leyhe, T.; Richartz, E.; Kohler, N.; Bartels, M.; Buchkremer, G.; et al. Increased BDNF serum concentration in fibromyalgia with or without depression or antidepressants. J. Psychiatr. Res. 2007, 41, 600–605.
- Stefani, L.C.; Leite, F.M.; da Graca, L.T.M.; Zanette, S.A.; de Souza, A.; Castro, S.M.; Caumo, W. BDNF and serum S100B levels according the spectrum of structural pathology in chronic pain patients. Neurosci. Lett. 2019, 706, 105–109.
- Nugraha, B.; Korallus, C.; Gutenbrunner, C. Serum level of brain-derived neurotrophic factor in fibromyalgia syndrome correlates with depression but not anxiety. Neurochem. Int. 2013, 62, 281–286.
- Zanette, S.A.; Dussan-Sarria, J.A.; Souza, A.; Deitos, A.; Torres, I.L.; Caumo, W. Higher serum S100B and BDNF levels are correlated with a lower pressure-pain threshold in fibromyalgia. Mol. Pain. 2014, 10, 46.
- Bidari, A.; Ghavidel-Parsa, B.; Gharibpoor, F. Comparison of the serum brain-derived neurotrophic factor (BDNF) between fibromyalgia and nociceptive pain groups; and effect of duloxetine on the BDNF level. BMC Musculoskelet. Disord. 2022, 23, 411.
- Klein, K.; Aeschlimann, A.; Jordan, S.; Gay, R.; Gay, S.; Sprott, H. ATP induced brain-derived neurotrophic factor expression and release from osteoarthritis synovial fibroblasts is mediated by purinergic receptor P2X4. PLoS ONE 2012, 7, e36693.
- Grimsholm, O.; Guo, Y.; Ny, T.; Forsgren, S. Expression patterns of neurotrophins and neurotrophin receptors in articular chondrocytes and inflammatory infiltrates in knee joint arthritis. Cells Tissues Organs 2008, 188, 299–309.
- Gowler, P.R.W.; Li, L.; Woodhams, S.G.; Bennett, A.J.; Suzuki, R.; Walsh, D.A.; Chapman, V. Peripheral brain-derived neurotrophic factor contributes to chronic osteoarthritis joint pain. Pain 2020, 161, 61–73.
- Ferrini, F.; Salio, C.; Boggio, E.M.; Merighi, A. Interplay of BDNF and GDNF in the Mature Spinal Somatosensory System and Its Potential Therapeutic Relevance. Curr. Neuropharmacol. 2021, 19, 1225–1245.
- Charlton, T.; Prowse, N.; McFee, A.; Heiratifar, N.; Fortin, T.; Paquette, C.; Hayley, S. Brain-derived neurotrophic factor (BDNF) has direct anti-inflammatory effects on microglia. Front. Cell Neurosci. 2023, 17, 1188672.
- Hu, X.M.; Cao, S.B.; Zhang, H.L.; Lyu, D.M.; Chen, L.P.; Xu, H.; Pan, Z.Q.; Shen, W. Downregulation of miR-219 enhances brain-derived neurotrophic factor production in mouse dorsal root ganglia to mediate morphine analgesic tolerance by upregulating CaMKIIgamma. Mol. Pain 2016, 12, 1744806916666283.
- Cejas, P.J.; Martinez, M.; Karmally, S.; McKillop, M.; McKillop, J.; Plunkett, J.A.; Oudega, M.; Eaton, M.J. Lumbar transplant of neurons genetically modified to secrete brain-derived neurotrophic factor attenuates allodynia and hyperalgesia after sciatic nerve constriction. Pain 2000, 86, 195–210.
- Jiang, Y.; Wei, N.; Zhu, J.; Lu, T.; Chen, Z.; Xu, G.; Liu, X. Effects of brain-derived neurotrophic factor on local inflammation in experimental stroke of rat. Mediat. Inflamm. 2010, 2010, 372423.
- Barrientos, R.M.; Sprunger, D.B.; Campeau, S.; Watkins, L.R.; Rudy, J.W.; Maier, S.F. BDNF mRNA expression in rat hippocampus following contextual learning is blocked by intrahippocampal IL-1beta administration. J. Neuroimmunol. 2004, 155, 119–126.
- Wang, M.; Pan, W.; Xu, Y.; Zhang, J.; Wan, J.; Jiang, H. Microglia-Mediated Neuroinflammation: A Potential Target for the Treatment of Cardiovascular Diseases. J. Inflamm. Res. 2022, 15, 3083–3094.
- Egan, M.F.; Kojima, M.; Callicott, J.H.; Goldberg, T.E.; Kolachana, B.S.; Bertolino, A.; Zaitsev, E.; Gold, B.; Goldman, D.; Dean, M.; et al. The BDNF val66met polymorphism affects activity-dependent secretion of BDNF and human memory and hippocampal function. Cell 2003, 112, 257–269.
- Yu, Y.B.; Zuo, X.L.; Zhao, Q.J.; Chen, F.X.; Yang, J.; Dong, Y.Y.; Wang, P.; Li, Y.Q. Brain-derived neurotrophic factor contributes to abdominal pain in irritable bowel syndrome. Gut 2012, 61, 685–694.
- Vossen, H.; Kenis, G.; Rutten, B.; van Os, J.; Hermens, H.; Lousberg, R. The genetic influence on the cortical processing of experimental pain and the moderating effect of pain status. PLoS ONE 2010, 5, e13641.
- Zhao, M.; Chen, L.; Yang, J.; Han, D.; Fang, D.; Qiu, X.; Yang, X.; Qiao, Z.; Ma, J.; Wang, L.; et al. BDNF Val66Met polymorphism, life stress and depression: A meta-analysis of gene-environment interaction. J. Affect. Disord. 2018, 227, 226–235.
- de Oliveira Franco, A.; de Oliveira Venturini, G.; da Silveira Alves, C.F.; Alves, R.L.; Vicuna, P.; Ramalho, L.; Tomedi, R.; Bruck, S.M.; Torres, I.L.S.; Fregni, F.; et al. Functional connectivity response to acute pain assessed by fNIRS is associated with BDNF genotype in fibromyalgia: An exploratory study. Sci. Rep. 2022, 12, 18831.
- da Silveira Alves, C.F.; Caumo, W.; Silvestri, J.M.; Zortea, M.; Dos Santos, V.S.; Cardoso, D.F.; Regner, A.; de Souza, A.H.; Simon, D. Pain catastrophizing is associated with the Val66Met polymorphism of the brain-derived neurotrophic factor in fibromyalgia. Adv. Rheumatol. 2020, 60, 39.
- Tian, Y.; Liu, X.; Jia, M.; Yu, H.; Lichtner, P.; Shi, Y.; Meng, Z.; Kou, S.; Ho, I.H.T.; Jia, B.; et al. Targeted Genotyping Identifies Susceptibility Locus in Brain-derived Neurotrophic Factor Gene for Chronic Postsurgical Pain. Anesthesiology 2018, 128, 587–597.
- Lamy, J.C.; Boakye, M. BDNF Val66Met polymorphism alters spinal DC stimulation-induced plasticity in humans. J. Neurophysiol. 2013, 110, 109–116.
- Cheeran, B.; Talelli, P.; Mori, F.; Koch, G.; Suppa, A.; Edwards, M.; Houlden, H.; Bhatia, K.; Greenwood, R.; Rothwell, J.C. A common polymorphism in the brain-derived neurotrophic factor gene (BDNF) modulates human cortical plasticity and the response to rTMS. J. Physiol. 2008, 586, 5717–5725.
- Kerr, J.I.; Burri, A. Genetic and epigenetic epidemiology of chronic widespread pain. J. Pain Res. 2017, 10, 2021–2029.
- D’Agnelli, S.; Arendt-Nielsen, L.; Gerra, M.C.; Zatorri, K.; Boggiani, L.; Baciarello, M.; Bignami, E. Fibromyalgia: Genetics and epigenetics insights may provide the basis for the development of diagnostic biomarkers. Mol. Pain 2019, 15, 1744806918819944.
- Crow, M.; Denk, F.; McMahon, S.B. Genes and epigenetic processes as prospective pain targets. Genome Med. 2013, 5, 12.
- Mauceri, D. Role of Epigenetic Mechanisms in Chronic Pain. Cells 2022, 11, 2613.
- Tan, M.; Shen, L.; Hou, Y. Epigenetic modification of BDNF mediates neuropathic pain via miR-30a-3p/EP300 axis in CCI rats. Biosci. Rep. 2020, 40, BSR20194442.
- Zhang, L.; Feng, H.; Jin, Y.; Zhan, Y.; Han, Q.; Zhao, X.; Li, P. Long Non-coding RNA LINC01119 Promotes Neuropathic Pain by Stabilizing BDNF Transcript. Front. Mol. Neurosci. 2021, 14, 673669.
- Polli, A.; Ghosh, M.; Bakusic, J.; Ickmans, K.; Monteyne, D.; Velkeniers, B.; Bekaert, B.; Godderis, L.; Nijs, J. DNA Methylation and Brain-Derived Neurotrophic Factor Expression Account for Symptoms and Widespread Hyperalgesia in Patients With Chronic Fatigue Syndrome and Comorbid Fibromyalgia. Arthritis Rheumatol. 2020, 72, 1936–1944.
- Paoloni-Giacobino, A.; Luthi, F.; Stenz, L.; Le Carre, J.; Vuistiner, P.; Leger, B. Altered BDNF Methylation in Patients with Chronic Musculoskeletal Pain and High Biopsychosocial Complexity. J. Pain Res. 2020, 13, 1289–1296.
- Menzies, V.; Lyon, D.E.; Archer, K.J.; Zhou, Q.; Brumelle, J.; Jones, K.H.; Gao, G.; York, T.P.; Jackson-Cook, C. Epigenetic alterations and an increased frequency of micronuclei in women with fibromyalgia. Nurs. Res. Pract. 2013, 2013, 795784.
- Xiong, H.Y.; Zheng, J.J.; Wang, X.Q. Non-invasive Brain Stimulation for Chronic Pain: State of the Art and Future Directions. Front. Mol. Neurosci. 2022, 15, 888716.
- Knotkova, H.; Hamani, C.; Sivanesan, E.; Le Beuffe, M.F.E.; Moon, J.Y.; Cohen, S.P.; Huntoon, M.A. Neuromodulation for chronic pain. Lancet 2021, 397, 2111–2124.
- Xiong, H.Y.; Cao, Y.Q.; Du, S.H.; Yang, Q.H.; He, S.Y.; Wang, X.Q. Effects of High-Definition Transcranial Direct Current Stimulation Targeting the Anterior Cingulate Cortex on the Pain Thresholds: A Randomized Controlled Trial. Pain Med. 2023, 24, 89–98.
- Villamar, M.F.; Wivatvongvana, P.; Patumanond, J.; Bikson, M.; Truong, D.Q.; Datta, A.; Fregni, F. Focal modulation of the primary motor cortex in fibromyalgia using 4x1-ring high-definition transcranial direct current stimulation (HD-tDCS): Immediate and delayed analgesic effects of cathodal and anodal stimulation. J. Pain 2013, 14, 371–383.
- Khedr, E.M.; Omran, E.A.H.; Ismail, N.M.; El-Hammady, D.H.; Goma, S.H.; Kotb, H.; Galal, H.; Osman, A.M.; Farghaly, H.S.M.; Karim, A.A.; et al. Effects of transcranial direct current stimulation on pain, mood and serum endorphin level in the treatment of fibromyalgia: A double blinded, randomized clinical trial. Brain Stimul. 2017, 10, 893–901.
- Kuo, H.I.; Bikson, M.; Datta, A.; Minhas, P.; Paulus, W.; Kuo, M.F.; Nitsche, M.A. Comparing cortical plasticity induced by conventional and high-definition 4 x 1 ring tDCS: A neurophysiological study. Brain Stimul. 2013, 6, 644–648.
- Monte-Silva, K.; Kuo, M.F.; Hessenthaler, S.; Fresnoza, S.; Liebetanz, D.; Paulus, W.; Nitsche, M.A. Induction of late LTP-like plasticity in the human motor cortex by repeated non-invasive brain stimulation. Brain Stimul. 2013, 6, 424–432.
- Scarabelot, V.L.; de Oliveira, C.; Medeiros, L.F.; de Macedo, I.C.; Cioato, S.G.; Adachi, L.N.S.; Paz, A.H.; de Souza, A.; Caumo, W.; Torres, I.L.S. Transcranial direct-current stimulation reduces nociceptive behaviour in an orofacial pain model. J. Oral Rehabil. 2019, 46, 40–50.
- Fritsch, B.; Reis, J.; Martinowich, K.; Schambra, H.M.; Ji, Y.; Cohen, L.G.; Lu, B. Direct current stimulation promotes BDNF-dependent synaptic plasticity: Potential implications for motor learning. Neuron 2010, 66, 198–204.
- Lopes, B.C.; Medeiros, L.F.; Silva de Souza, V.; Cioato, S.G.; Medeiros, H.R.; Regner, G.G.; Lino de Oliveira, C.; Fregni, F.; Caumo, W.; Torres, I.L.S. Transcranial direct current stimulation combined with exercise modulates the inflammatory profile and hyperalgesic response in rats subjected to a neuropathic pain model: Long-term effects. Brain Stimul. 2020, 13, 774–782.
- Filho, P.R.; Vercelino, R.; Cioato, S.G.; Medeiros, L.F.; de Oliveira, C.; Scarabelot, V.L.; Souza, A.; Rozisky, J.R.; Quevedo Ada, S.; Adachi, L.N.; et al. Transcranial direct current stimulation (tDCS) reverts behavioral alterations and brainstem BDNF level increase induced by neuropathic pain model: Long-lasting effect. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 64, 44–51.
- Marlatt, M.W.; Potter, M.C.; Lucassen, P.J.; van Praag, H. Running throughout middle-age improves memory function, hippocampal neurogenesis, and BDNF levels in female C57BL/6J mice. Dev. Neurobiol. 2012, 72, 943–952.
- Lima, L.V.; Abner, T.S.S.; Sluka, K.A. Does exercise increase or decrease pain? Central mechanisms underlying these two phenomena. J. Physiol. 2017, 595, 4141–4150.
- Lee, M.; Moon, W.; Kim, J. Effect of yoga on pain, brain-derived neurotrophic factor, and serotonin in premenopausal women with chronic low back pain. Evid. Based Complement. Altern. Med. 2014, 2014, 203173.
- Wang, Y.H.; Zhou, H.H.; Luo, Q.; Cui, S. The effect of physical exercise on circulating brain-derived neurotrophic factor in healthy subjects: A meta-analysis of randomized controlled trials. Brain Behav. 2022, 12, e2544.
- Pedersen, B.K. Physical activity and muscle-brain crosstalk. Nat. Rev. Endocrinol. 2019, 15, 383–392.
- Di-Bonaventura, S.; Fernandez-Carnero, J.; Matesanz-Garcia, L.; Arribas-Romano, A.; Polli, A.; Ferrer-Pena, R. Effect of Different Physical Therapy Interventions on Brain-Derived Neurotrophic Factor Levels in Chronic Musculoskeletal Pain Patients: A Systematic Review. Life 2023, 13, 163.
- Ribeiro, V.G.C.; Mendonca, V.A.; Souza, A.L.C.; Fonseca, S.F.; Camargos, A.C.R.; Lage, V.K.S.; Neves, C.D.C.; Santos, J.M.; Teixeira, L.A.C.; Vieira, E.L.M.; et al. Inflammatory biomarkers responses after acute whole body vibration in fibromyalgia. Braz. J. Med. Biol. Res. 2018, 51, e6775.
- Mojtabavi, H.; Shaka, Z.; Momtazmanesh, S.; Ajdari, A.; Rezaei, N. Circulating brain-derived neurotrophic factor as a potential biomarker in stroke: A systematic review and meta-analysis. J. Transl. Med. 2022, 20, 126.
- Gutierrez, A.; Corey-Bloom, J.; Thomas, E.A.; Desplats, P. Evaluation of Biochemical and Epigenetic Measures of Peripheral Brain-Derived Neurotrophic Factor (BDNF) as a Biomarker in Huntington’s Disease Patients. Front. Mol. Neurosci. 2019, 12, 335.
- Azoulay, D.; Horowitz, N.A. Brain-derived neurotrophic factor as a potential biomarker of chemotherapy-induced peripheral neuropathy and prognosis in haematological malignancies; what we have learned, the challenges and a need for global standardization. Br. J. Haematol. 2020, 191, 17–18.
- Gejl, A.K.; Enevold, C.; Bugge, A.; Andersen, M.S.; Nielsen, C.H.; Andersen, L.B. Associations between serum and plasma brain-derived neurotrophic factor and influence of storage time and centrifugation strategy. Sci. Rep. 2019, 9, 9655.
- Polacchini, A.; Metelli, G.; Francavilla, R.; Baj, G.; Florean, M.; Mascaretti, L.G.; Tongiorgi, E. A method for reproducible measurements of serum BDNF: Comparison of the performance of six commercial assays. Sci. Rep. 2015, 5, 17989.
- Park, D.J.; Kim, S.H.; Nah, S.S.; Lee, J.H.; Kim, S.K.; Lee, Y.A.; Hong, S.J.; Kim, H.S.; Lee, H.S.; Kim, H.A.; et al. Association between brain-derived neurotrophic factor gene polymorphisms and fibromyalgia in a Korean population: A multicenter study. Arthritis Res. Ther. 2018, 20, 220.
- Zorina-Lichtenwalter, K.; Meloto, C.B.; Khoury, S.; Diatchenko, L. Genetic predictors of human chronic pain conditions. Neuroscience 2016, 338, 36–62.





