Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Rahul Mishra | -- | 2677 | 2024-02-17 01:02:38 | | | |
| 2 | Peter Tang | Meta information modification | 2677 | 2024-02-17 07:01:36 | | | | |
| 3 | Lindsay Dong | -11 word(s) | 2666 | 2024-02-18 02:24:44 | | | | |
| 4 | Lindsay Dong | -1 word(s) | 2665 | 2024-02-26 09:51:13 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Mishra, R.; Zokaei Nikoo, M.; Veeraballi, S.; Singh, A. Venetoclax and Hypomethylating Agent Combination in Myeloid Malignancies. Encyclopedia. Available online: https://encyclopedia.pub/entry/55102 (accessed on 31 May 2026).
Mishra R, Zokaei Nikoo M, Veeraballi S, Singh A. Venetoclax and Hypomethylating Agent Combination in Myeloid Malignancies. Encyclopedia. Available at: https://encyclopedia.pub/entry/55102. Accessed May 31, 2026.
Mishra, Rahul, Maedeh Zokaei Nikoo, Sindhusha Veeraballi, Abhay Singh. "Venetoclax and Hypomethylating Agent Combination in Myeloid Malignancies" Encyclopedia, https://encyclopedia.pub/entry/55102 (accessed May 31, 2026).
Mishra, R., Zokaei Nikoo, M., Veeraballi, S., & Singh, A. (2024, February 17). Venetoclax and Hypomethylating Agent Combination in Myeloid Malignancies. In Encyclopedia. https://encyclopedia.pub/entry/55102
Mishra, Rahul, et al. "Venetoclax and Hypomethylating Agent Combination in Myeloid Malignancies." Encyclopedia. Web. 17 February, 2024.
Copy Citation
There has been a widespread adoption of hypomethylating agents (HMA: 5-Azacytidine (5-Aza)/decitabine) and venetoclax (Ven) for the treatment of acute myeloid leukemia (AML); however, the mechanisms behind the combination’s synergy are poorly understood. Monotherapy often encounters resistance, leading to suboptimal outcomes; however, the combination of HMA and Ven has demonstrated substantial improvements in treatment responses.
Venetoclax-5-Azacytidine
hypomethylating agents
acute myeloid leukemia
myeloid malignancies
1. Introduction
Acute myeloid leukemia (AML) is a heterogenous condition, generally affecting older individuals [1]. The ineligibility of such patients to intesive chemotherapy has led to the search for newer therapies in recent years. One such example of a newer combination of hypomethylating agents (HMAs) and a selective oral BCL-2 inhibitor (Venetoclax, Ven) has made significant strides and gained FDA approval in recent years [2].
HMAs, such as 5-azacytidine (Aza) and decitabine (Dec), function by promoting the blocking of DNA methyltransferase 1 (DNMT1). This process results in the demethylation of CpG islands [3] and an increase in the expression of genes necessary for normal myeloid hematopoiesis. HMAs, thus, play an integral role in the treatment of myelodysplastic neoplasms and acute myeloid leukemia (AML), when cellular differentiation blocks arrest maturation [4][5][6].
Normal cellular senescence via apoptosis, regulated by a balance between pro-apoptotic (Bax, Bak, Bad, Bid, Puma, Bim, Noxa) and anti-apoptotic proteins (Bcl-2, Bcl-xL, Bcl-w, Mcl-1), is lost among cancer cells, characterized by increased expression of anti-apoptotic protiens [7]. An anti-apoptotic protein prevents apoptosis by binding to and clearing pro-apoptotic proteins.
In 2014, Pan et al. [8] showed the efficacy of Ven (ABT-199), a potent, selective oral inhibitor of BCL-2, on multiple AML cell lines and patient-derived xenografts at nanomolar concentrations. Notably, the cytotoxic effects of Ven (Venetoclax) were predominantly unaffected by mutational status and were sustained in primary AML blast cells that were resistant to chemotherapy. In a phase II clinical trial, Konopleva et al. [9] showed evidence for the efficacy of Ven monotherapy (19% overall response rate), particularly among patients with IDH1/2 mutations.
2. Mechanisms of Resistance against Monotherapies with Ven or HMA in AML Cell Lines
2.1. Resistance against Ven
The details of Ven resistance have been described elsewhere [10]. Briefly, the primary mechanism involves the overexpression of anti-apoptotic proteins other than BCL-2, i.e., BCL2-A1, MCL-1, and BCL-xL, that sequester pro-apoptotic proteins (e.g., BIM, BAX) [11][12][13]. Broadly, these mechanisms include the inability to trigger apoptosis due to inactivation of BAX/BIM, TP53, and PMA1P1 (NOXA); decreased BCL2 expression, hence lesser activity of Ven (a selective BCL-2 inhibitor); and alterations in mitochondrial function and cellular metabolism [14].
Another resistance mechanism includes stimulation of signaling pathways by mutant KRAS/PTPN11 or FMS-like tyrosine kinase 3 (FLT3) proteins. These mutations could either be intrinsic or emerge secondary to Ven-based therapy, and they carry an adverse prognosis [15]. The anti-apoptotic factors MCL-1 and BCL2-A1 are upregulated due to a mutation in the KRAS gene, whereas a mutation in PTPN11 results in the upregulation of the anti-apoptotic factors MCL-1 and BCL-xL. KRAS mutations also downregulate BCL-2 and BAX [13]. Venetoclax resistance is seen in FLT3-ITD mutations due to upregulation of the anti-apoptotic factors BCL-xL and MCL-1 via complex downstream pathways [16][17][18].
2.2. Resistance against HMA
The resistance mechanisms against HMA (Aza or Dec) are related factors intrinsic to tumor cells and extrinsic factors related to immune cells or other cells in the bone marrow, as discussed elsewhere [4]. Tumor cell-intrinsic factors are related to HMA transport (via human concentrative or equilibrative nucleoside transporter 1; hCNT1 or hENT1) into cells, activation (via kinases; UCK or DCK), incorporation into nucleic acids, and DNMT inhibition and metabolism via cytidine deaminases (CDAs). Low levels of transporters and decreased expression of kinases, leading to decreased incorporation of Aza or Dec into nucleic acids, contribute to HMA resistance [19][20][21][22].
HMAs can induce major antioxidant pathways mediated by the NF-E2-related factor (Nrf2), thus contributing to self-resistance [23]. Cheng JX et al. also reported that leukemia cells resistant to 5-Aza exhibit increased active chromatin organization associated with one of the methylcytosine (RNA: m5C) methytransferases, namely, NSUN1. NSUN1 establishes interactions with BRD4 and RNA polymerase II, resulting in the formation of an active chromatin structure resistant to 5-Aza but responsive to the inhibition of BRD4 [24]. Extrinsic factors are commonly related to, but not limited to, the bone marrow microenvironment.
3. Ven–HMA Synergy Mechanisms
3.1. Preclinical Data
3.1.1. HMA-Mediated Downregulation of MCL-1 Levels
Tsao et al. [25] initially reported that co-administration of 5-Aza and ABT-737 (inhibitor of BCL-2 and Bcl-xL) could increase mitochondrial outer membrane permeability among AML cells. This was reflected via the activation of BAX protein and loss of the mitochondrial membrane potential. They noted that the 5-Aza and ABT-737 combination synergistically induced apoptosis independent of p53 expression in these cells, which was not the case when these agents were used as single agents. Elevated levels of anti-apoptotic MCL-1 are known for conferring resistance to ABT-737. The application of 5-AZA reduced the expression of MCL-1 in these cells, independent of p53, leading to enhanced AML cell apoptosis.
The role of MCL-1 inhibition (by S63845, a specific MCL-1 inhibitor) in synergizing venetoclax activity was further assessed by Hormi et al. [26]. They observed that despite relatively weaker expression of MCL-1 compared to BCL-2, S63845 prompted apoptosis in AML cells and exhibited a robust synergistic effect with Venetoclax.
3.1.2. BCL2 Family Protein as 5-Aza-Sensitizing Targets
Bogenberger et al. [27] noted greater sensitization to 5-Aza (three- to fourfold) in AML cells with RNAi-mediated knockdown of BCL-xL. Furthermore, they focused on the involvement of three prominent anti-apoptotic proteins—BCL-2, MCL-1, and BCL-XL—in influencing cell proliferation and responsiveness to 5-Aza. Notably, the siRNA-mediated silencing of BCL-XL or MCL-1 substantially diminished viability across the majority of the examined AML cell lines. In contrast, the impact of BCL-2 siRNA was comparatively less pronounced. The silencing of BCL-xL strongly sensitized particular types of erythroid cells, while MCL-1 sensitized more broadly across AML cells.
Navitoclax (ABT-263, its tool compound was ABT-737), a combined inhibitor of BCL-xL, BCL-w, and BCL2 and Venetoclax (ABT-199, selective BCL2 inhibitor) were utilized to assess the relative potency of these agents in sensitizing the AML cells for 5-Aza. Higher concentrations of ABT-199 compared to ABT-737 were required for the enhancement of Aza activity. The association was dose-dependent with ABT-737 but not with ABT-199, except for MDS-L cells. A greater synergy was noted with ABT-737 than ABT-199 in most cell lines. The sensitization was mediated by enhancing apoptosis; hence, they concluded that ABT-737 had greater potency when compared to ABT-199 and in combination with 5-Aza.
3.1.3. Combination Disrupts Energy Metabolism and Targets Leukemia Stem Cells (LSCs)
Extended exposure to azacitidine (oral; CC-486) harnesses sustained DNMT1 loss and sustained hypomethylation. This can lead to preferential targeting of immature LSCs, potentially addressing myeloid maturation arrests by facilitating differentiation [28]. Pollyea et al. [29] reported that the combination of Ven + 5-Aza altered the tricarboxylic acid cycle, as indicated by a decrease in α-ketoglutarate levels and an increase in succinate levels in LSCs. This suggested a targeted inhibition of electron transport chain complex II, specifically in LSCs. In vitro modeling further validated the inhibition of complex II through the diminished glutathionylation of succinate dehydrogenase.
Together, these data suggest that the azacitidine-mediated differentiation of LSCs coupled with Ven-mediated programmed cell death further synergizes each agent’s activity. While the Ven–HMA combination targets oxidative phosphorylation in LSCs, there may be an additional role of HMA-mediated LSC differentiation that augments durable responses in AML patients.
3.1.4. Reactive Oxygen Species (ROS)-Dependent Antileukemic Activity
Ven augments HMA by inhibiting Nrf2 antioxidant pathway activation, induced by HMA. This leads to oxidative killing of AML cells, via an indirect underlying mechanism. The NF-E2-related factor 2 (Nrf2) antioxidant pathway is the key cellular mechanism that prevents oxidative cell damage. In the presence of oxidative stress and the generation of ROS, the transcription factor Nrf2 dissociates from its cytoplasmic assembly with Kelch-like ECH-associated protein 1 (Keap-1). Under normal circumstances, Keap-1 facilitates the ubiquitination and degradation of Nrf2 [30][31]. Upon release, Nrf2 translocates to the nucleus, where it binds to the antioxidant response elements in the promoters of several genes, including heme oxygenase-1 (HO-1) and NADP-quinone oxidoreductase-1 (NQO-1). These genes play a role in neutralizing ROS [30][31].
3.1.5. HMA (5-Aza) Induced “Priming” of the AML Cells for Ven-Induced Apoptosis
Jin et al. [32] demonstrated a novel non-epigenetic mechanism of action for 5-Aza and its synergistic activity with Ven through the integrated stress response (ISR)-mediated induction of PMAIP1 transcripts. They reported that 5-Aza pretreated AML cells in vitro were more sensitive to Ven than DMSO pretreated cells. The number of cells with active caspase-3/-7 was higher immediately following treatment with combined 5-Aza and Ven exposure than with either agent alone. Molecular analysis revealed 5-Aza dose- and time- dependent increases in the pro-apoptotic protein factors NOXA (PMAIP1) and PUMA (BBC3) in cells obtained from AML patients. Yet, chronic 5-Aza therapy did not exert a substantial impact on gene expression or methylation. The promoter regions and transcripts of PMAIP1 and BBC3 genes remained unmethylated and unchanged. They postulated a role for the ISR pathway in Aza-induced priming. ATF4, the ISR pathway’s main effector, can promote apoptosis through transcriptional activation of proapoptotic factors (including PMAIP1 (NOXA) and BBC3 (PUMA)). When AML cells were exposed to the Ven–Aza combination, the levels of activating transcription factor 4 (ATF4) were significantly elevated within 24 h, indicating the activation of the ISR pathway by 5-Aza. These preclinical findings underscore the clinical importance of initiating both therapies concomitantly, preferably within 24 h of HMA initiation [32][33]. The relative impact of NOXA and PUMA was delineated by deleting their respective genes (PMAIP1 and BBC3). A reduction in the magnitude of cell death from the 5-Aza/Ven combination was observed in PMAIP1-deleted cells but not in BBC3-deleted cells. Therefore, they came to the conclusion that, whereas 5-Aza therapy induces both NOXA and PUMA, only NOXA is essential for sensitizing AML cells to 5-Aza/Ven-induced apoptosis [32].
3.1.6. Overexpression or HMA-Mediated Restoration of Caspase-3/GSDME Significantly Increases Ven-Induced Pyroptosis
Ven induces pyroptosis through the cleavage of the gasdermin (GSDM) protein, a process mediated by caspase-3. Cleaved GSDM forms membrane pores, leading to cytokine release and/or programed lytic cell death, called pyroptosis [34]. Recently, Ye et al., via in vivo and in vitro experiments, showed that GSDME, but not GSDMA, GSDMB, GSDMC, or GSDMD, was cleaved upon Ven treatment. GSDME is suppressed in AML cell lines through promoter methylation, and reduced GSDME expression is strongly correlated with an unfavorable prognosis. They showed that the overexpression of GSDME, achieved through the demethylation of the GSDME gene or the restoration of GSDME expression mediated by HMA, markedly enhanced Ven-induced pyroptosis in AML [35].
3.1.7. Ven Augments HMA via Inhibiting De Novo Pyrimidine Synthesis
Upregulated de novo pyrimidine synthesis is the major adaptive resistance mechanism against HMA (Aza and decitabine) in MDS/AML cells [36]. The mitochondrial membrane potential is required for the functioning of the mitochondrial enzyme DHODH, a key enzyme of the de novo pyrimidine synthesis in cells. Ven, by inhibiting BCL-2, depolarizes the mitochondrial membrane and, hence, inhibits the DHODH enzyme. At concentrations that do not induce apoptosis, both Ven and the DHODH inhibitor teriflunomide substantially reduce the levels of cytidine- and deoxycytidine triphosphate (CTP, dCTP) in AML cells. This contributes to the downregulation of de novo pyrimidine synthesis, hence overcoming resistance against hypomethylating agents (HMAs) [37][38].
3.2. Clinical Data: Molecular Predictors for Response to Ven–HMA Combination
The FDA approved Ven–HMA as a frontline treatment for older adults aged 75 years or above, or those with comorbidities that make them unsuitable candidates for intensive induction chemotherapy [2][39]. In a pivotal randomized clinical trial, there was a significant improvement in the median OS (14.7 months for Ven + HMA vs. 9.6 months for placebo + HMA) and complete response rates (37% vs. 18%, respectively) [40].
The analysis of data from a phase 1b/2 study involving 209 patients treated with Ven 400 mg or 600 mg in combination with either HMA or LDAC revealed a CR rate of 83.7% for pts with IDH1/IDH2 mutations, 84.6% for patients with NPM1 mutations, 59.5% for patients with TP53 mutations, and 53.3% for patients with FLT3 mutations [41]. In the pivotal VIALE-A trial [40] (n = 431, median age of 76-years), patients with IDH1 or IDH2 mutations at baseline had better OS at 12 months, with 5-Aza + Ven (66.8%), as compared to 35.7% with 5-Aza + placebo group (with the hazard ratio for death, 0.35; 95% CI, 0.20 to 0.60; p < 0.001). Superior responses were noted with Ven + HMA in patients with IDH1/2, FLT3, NPM1, and TP53 genes. In another study, DiNardo et al. reported high response rates and sustained remissions among patients with NPM1 or IDH2 mutations (2-year OS of 71.8% and 79.5%, respectively). The study identified that primary and adaptive resistance to Ven-based combinations was predominantly associated with mutations in FLT3, RAS, or TP53 genes [42].
Morisa et al. retrospectively performed analysis of 86 patients (44 newly diagnosed with AML and 42 with relapsed/refractory AML) who received an HMA–Ven combination. In the upfront setting, logistic regression analyses revealed that the presence of the CEPBA mutation conferred improved CR/CRi rates. Each of the four patients with CEPBA mutation attained CR, in contrast to 51% (18 of 35) of CEPBA wild-type patients. The median OS for patients receiving upfront HMA–Ven was 11 months (95% CI; 8–23 months). It was higher among those recieving CR compared to those not achieving complete response (17 months vs. 3 months). For relapsed/refractory disease, the presence of JAK2 (p = 0.03), DNMT3A (p < 0.01), and BCOR (p = 0.04) mutations was identified as predictors of CR/CRi. Median overall survival for relapsed/refractory AML patients was 5 months (95% CI, 3–9 months), which was higher among those patients achieving complete response (15 months vs. 3 months) vs. those not achieving CR/CRi [43].
3.3. Superior Response with Ven–HMA Compared to Ven-LDAC
Clinical studies have reported superior response with Ven–HMA compared with Ven low-dose cytarabine (LDAC) therapy in newly diagnosed (CCR rate: 66.4% (Ven–HMA in VIAL-A trial) vs. 48% (LDAC-Ven in VIAL-C trial)) and r/r AML patients (CCR rate of 37% vs. 11%, respectively, in retrospective study) [40][44][45].
The precise mechanism of this difference in response rates is yet to be explored and likely extends beyond the different study populations in the two landmark studies that led to the FDA approval of both regimens. It relates to the mechanism of action or potential drug–drug interactions, patient characteristics, and specific genetic mutations, all of which can significantly influence response rates. HMA and Ven synergize together via multiple mechanisms (Figure 1). However, LDAC, also known as, arabinosylcytosine (ARA-C), undergoes conversion into its triphosphate form within the cell. This form competes with cytidine to integrate into the DNA. The sugar moiety of cytarabine impedes the rotation of the molecule within the DNA, leading to the cessation of DNA replication, particularly during the S phase of the cell cycle. Additionally, DNA replication and repair are halted due to the inhibition of DNA polymerase by cytarabine [46]. Variation in single nucleotide polymorphisms (SNPs) within the genes responsible for the transport, activation, and inactivation of cytarabine can impact the intracellular ara-CTP levels. These SNPs can influence the expression and activity of these genes and, consequently, can affect the clinical outcome of patients treated with ara-C [47].

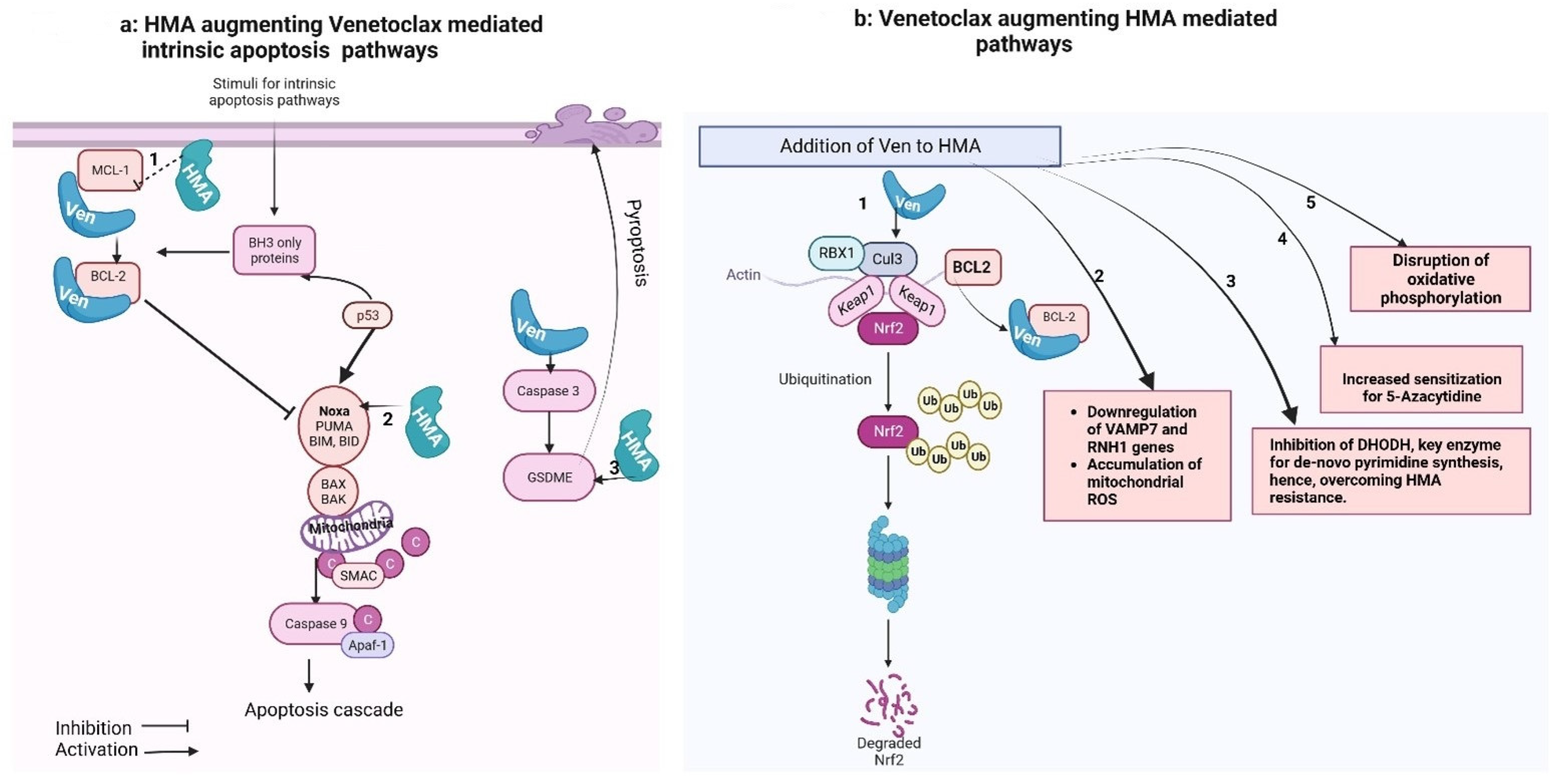
Figure 1. Venetoclax–hypomethylating synergy mechanisms. These figures were made using Biorender website (Biorender.com).
Figure 1a shows that the intrinsic apoptotic pathway is regulated by proapoptotic (BAX, BAK, BID, BAD, BIM, PUMA) and anti-apoptotic (BCL-2, MCL-1, BCL-xL) factors. Pro-apoptotic activator proteins, once triggered by a stimulus, activate the central effectors of apoptosis (BAX and BAK) that undergo confirmational change and form pores in the mitochondrial membrane, causing miMOMP (minority-mitochondrial outer membrane permeabilization) and release of cytochrome-C. Free cytochrome-C then binds with Apaf-1 (apoptosome), which activates caspase-9 and triggers the apoptotic cascade. Anti-apoptotic proteins, when released, bind with and neutralize anti-apoptotic proteins.
Figure 1b shows that Venetoclax synergizes with HMA-mediated cancer cell killing: (1) The primary cellular mechanism for averting oxidative cell damage is the NF-E2-related factor 2 (Nrf2) antioxidant pathway. Exposure to oxidative stress and reactive oxygen species (ROS) leads to dissociation of the transcription factor Nrf2 from its cytoplasmic adaptor Kelch-like ECH-associated protein 1 (Keap-1). In the absence of oxidative stress, Keap-1 mediates the ubiquitination and degradation of Nrf2. Once released, Nrf2 translocate to the nucleus and activates genes, which mediate neutralization of ROS (Baird and Dinkova-Kostova, 2011 [31]; Nguyen et al., 2009 [30]). This pathway is also induced by HMA.
References
- Key Statistics for Acute Myeloid Leukemia (AML). Available online: https://www.cancer.org/cancer/types/acute-myeloid-leukemia/about/key-statistics.html (accessed on 29 October 2023).
- FDA. FDA Grants Regular Approval to Venetoclax in Combination for Untreated Acute Myeloid Leukemia. 11 June 2021. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-regular-approval-venetoclax-combination-untreated-acute-myeloid-leukemia (accessed on 24 September 2023).
- Uy, N.; Singh, A.; Gore, S.D.; Prebet, T. Hypomethylating agents (HMA) treatment for myelodysplastic syndromes: Alternatives in the frontline and relapse settings. Expert Opin. Pharmacother. 2017, 18, 1213–1224.
- Stomper, J.; Rotondo, J.C.; Greve, G.; Lübbert, M. Hypomethylating agents (HMA) for the treatment of acute myeloid leukemia and myelodysplastic syndromes: Mechanisms of resistance and novel HMA-based therapies. Leukemia 2021, 35, 1873–1889.
- Šorm, F.; Pískala, A.; Čihák, A.; Veselý, J. 5-Azacytidine, a new, highly effective cancerostatic. Cell. Mol. Life Sci. 1964, 20, 202–203.
- Singh, A.; Carraway, H.E.M. Overview of the Management of Higher-Risk Myelodysplastic Syndromes. Cancer J. 2023, 29, 160–167.
- Wong, R.S.Y. Apoptosis in cancer: From pathogenesis to treatment. J. Exp. Clin. Cancer Res. 2011, 30, 87.
- Pan, R.; Hogdal, L.J.; Benito, J.M.; Bucci, D.; Han, L.; Borthakur, G.; Cortes, J.; DeAngelo, D.J.; Debose, L.; Mu, H.; et al. Selective BCL-2 inhibition by ABT-199 causes on-target cell death in acute myeloid leukemia. Cancer Discov. 2014, 4, 362–375.
- Konopleva, M.; Pollyea, D.A.; Potluri, J.; Chyla, B.; Hogdal, L.; Busman, T.; McKeegan, E.; Salem, A.H.; Zhu, M.; Ricker, J.L.; et al. Efficacy and Biological Correlates of Response in a Phase II Study of Venetoclax Monotherapy in Patients with Acute Myelogenous Leukemia. Cancer Discov. 2016, 6, 1106–1117.
- Ong, F.; Kim, K.; Konopleva, M.Y. Venetoclax resistance: Mechanistic insights and future strategies. Cancer Drug Resist. 2022, 5, 380–400.
- Bisaillon, R.; Moison, C.; Thiollier, C.; Krosl, J.; Bordeleau, M.E.; Lehnertz, B.; Lavallée, V.P.; MacRae, T.; Mayotte, N.; Labelle, C.; et al. Genetic characterization of ABT-199 sensitivity in human AML. Leukemia 2020, 34, 63–74.
- Lin, K.H.; Winter, P.S.; Xie, A.; Roth, C.; Martz, C.A.; Stein, E.M.; Anderson, G.R.; Tingley, J.P.; Wood, K.C. Targeting MCL-1/BCL-XL Forestalls the Acquisition of Resistance to ABT-199 in Acute Myeloid Leukemia. Sci. Rep. 2016, 6, 27696.
- Zhang, H.; Nakauchi, Y.; Köhnke, T.; Stafford, M.; Bottomly, D.; Thomas, R.; Wilmot, B.; McWeeney, S.K.; Majeti, R.; Tyner, J.W. Integrated analysis of patient samples identifies biomarkers for venetoclax efficacy and combination strategies in acute myeloid leukemia. Nat. Cancer 2020, 1, 826–839.
- Chyla, B.; Daver, N.; Doyle, K.; McKeegan, E.; Huang, X.; Ruvolo, V.; Wang, Z.; Chen, K.; Souers, A.; Leverson, J.; et al. Genetic Biomarkers Of Sensitivity and Resistance to Venetoclax Monotherapy in Patients With Relapsed Acute Myeloid Leukemia. Am. J. Hematol. 2018, 93, E202–E205.
- Yoshimoto, G.; Miyamoto, T.; Jabbarzadeh-Tabrizi, S.; Iino, T.; Rocnik, J.L.; Kikushige, Y.; Mori, Y.; Shima, T.; Iwasaki, H.; Takenaka, K.; et al. FLT3-ITD up-regulates MCL-1 to promote survival of stem cells in acute myeloid leukemia via FLT3-ITD-specific STAT5 activation. Blood 2009, 114, 5034–5043.
- Dumon, S.; Santos, S.; Debierre-Grockiego, F.; Gouilleux-Gruart, V.; Cocault, L.; Boucheron, C.; Mollat, P.; Gisselbrecht, S.; Gouilleux, F. IL-3 dependent regulation of Bcl-xL gene expression by STAT5 in a bone marrow derived cell line. Oncogene 1999, 18, 4191–4199.
- Kasper, S.; Breitenbuecher, F.; Heidel, F.; Hoffarth, S.; Markova, B.; Schuler, M.; Fischer, T. Targeting MCL-1 sensitizes FLT3-ITD-positive leukemias to cytotoxic therapies. Blood Cancer J. 2012, 2, e60.
- Kuusanmäki, H.; Leppä, A.-M.; Pölönen, P.; Kontro, M.; Dufva, O.; Deb, D.; Yadav, B.; Brück, O.; Kumar, A.; Everaus, H.; et al. Phenotype-based drug screening reveals association between venetoclax response and differentiation stage in acute myeloid leukemia. Haematologica 2019, 105, 708–720.
- Wu, P.; Geng, S.; Weng, J.; Deng, C.; Lu, Z.; Luo, C.; Du, X. The hENT1 and DCK genes underlie the decitabine response in patients with myelodysplastic syndrome. Leuk. Res. 2015, 39, 216–220.
- Valencia, A.; Masala, E.; Rossi, A.; Martino, A.; Sanna, A.; Buchi, F.; Canzian, F.; Cilloni, D.; Gaidano, V.; Voso, M.T.; et al. Expression of nucleoside-metabolizing enzymes in myelodysplastic syndromes and modulation of response to azacitidine. Leukemia 2014, 28, 621–628.
- Gruber, E.; Franich, R.L.; Shortt, J.; Johnstone, R.W.; Kats, L.M. Distinct and overlapping mechanisms of resistance to azacytidine and guadecitabine in acute myeloid leukemia. Leukemia 2020, 34, 3388–3392.
- Qin, T.; Jelinek, J.; Si, J.; Shu, J.; Issa, J.-P.J. Mechanisms of resistance to 5-aza-2′-deoxycytidine in human cancer cell lines. Blood 2009, 113, 659–667.
- Nguyen, L.X.T.; Troadec, E.; Kalvala, A.; Kumar, B.; Hoang, D.H.; Viola, D.; Zhang, B.; Nguyen, D.Q.; Aldoss, I.; Ghoda, L.; et al. The Bcl-2 inhibitor venetoclax inhibits Nrf2 antioxidant pathway activation induced by hypomethylating agents in AML. J. Cell. Physiol. 2019, 234, 14040–14049.
- Cheng, J.X.; Chen, L.; Li, Y.; Cloe, A.; Yue, M.; Wei, J.; Watanabe, K.A.; Shammo, J.M.; Anastasi, J.; Shen, Q.J.; et al. RNA cytosine methylation and methyltransferases mediate chromatin organization and 5-azacytidine response and resistance in leukaemia. Nat. Commun. 2018, 9, 1163, Erratum in Nat. Commun. 2018, 9, 2286.
- Tsao, T.; Shi, Y.; Kornblau, S.; Lu, H.; Konoplev, S.; Antony, A.; Ruvolo, V.; Qiu, Y.H.; Zhang, N.; Coombes, K.R.; et al. Concomitant inhibition of DNA methyltransferase and BCL-2 protein function synergistically induce mitochondrial apoptosis in acute myelogenous leukemia cells. Ann. Hematol. 2012, 91, 1861–1870.
- Hormi, M.; Birsen, R.; Belhadj, M.; Huynh, T.; Aguilar, L.C.; Grignano, E.; Haddaoui, L.; Guillonneau, F.; Mayeux, P.; Hunault, M.; et al. Pairing MCL-1 inhibition with venetoclax improves therapeutic efficiency of BH3-mimetics in AML. Eur. J. Haematol. 2020, 105, 588–596.
- Bogenberger, J.M.; Kornblau, S.M.; E Pierceall, W.; Lena, R.; Chow, D.; Shi, C.-X.; Mantei, J.; Ahmann, G.; Gonzales, I.M.; Choudhary, A.; et al. BCL-2 family proteins as 5-Azacytidine-sensitizing targets and determinants of response in myeloid malignancies. Leukemia 2014, 28, 1657–1665.
- Jeyaraju, D.V.; Alapa, M.; Polonskaia, A.; Risueño, A.; Subramanyam, P.; Anand, A.; Ghosh, K.; Kyriakopoulos, C.; Hemerich, D.; Hurren, R.; et al. Extended exposure to low doses of azacitidine induces differentiation of leukemic stem cells through activation of myeloperoxidase. Haematologica, 2023; Epub ahead of printing.
- Pollyea, D.A.; Stevens, B.M.; Jones, C.L.; Winters, A.; Pei, S.; Minhajuddin, M.; D’Alessandro, A.; Culp-Hill, R.; Riemondy, K.A.; Gillen, A.E.; et al. Venetoclax with azacitidine disrupts energy metabolism and targets leukemia stem cells in patients with acute myeloid leukemia. Nat. Med. 2018, 24, 1859–1866.
- Nguyen, T.; Nioi, P.; Pickett, C.B. The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J. Biol. Chem. 2009, 284, 13291–13295.
- Baird, L.; Dinkova-Kostova, A.T. The cytoprotective role of the Keap1-Nrf2 pathway. Arch. Toxicol. 2011, 85, 241–272.
- Jin, S.; Cojocari, D.; Purkal, J.J.; Popovic, R.; Talaty, N.N.; Xiao, Y.; Solomon, L.R.; Boghaert, E.R.; Leverson, J.D.; Phillips, D.C. 5-Azacitidine Induces NOXA to Prime AML Cells for Venetoclax-Mediated Apoptosis. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2020, 26, 3371–3383.
- Jonas, B.A.; Pollyea, D.A. How we use venetoclax with hypomethylating agents for the treatment of newly diagnosed patients with acute myeloid leukemia. Leukemia 2019, 33, 2795–2804.
- Xia, S.; Hollingsworth, L.R.; Wu, H. Mechanism and Regulation of Gasdermin-Mediated Cell Death. Cold Spring Harb. Perspect. Biol. 2020, 12, a036400.
- Ye, F.; Zhang, W.; Fan, C.; Dong, J.; Peng, M.; Deng, W.; Zhang, H.; Yang, L. Antileukemic effect of venetoclax and hypomethylating agents via caspase-3/GSDME-mediated pyroptosis. J. Transl. Med. 2023, 21, 606.
- Gu, X.; Tohme, R.; Tomlinson, B.; Sakre, N.; Hasipek, M.; Durkin, L.; Schuerger, C.; Grabowski, D.; Zidan, A.M.; Radivoyevitch, T.; et al. Decitabine- and 5-azacytidine resistance emerges from adaptive responses of the pyrimidine metabolism network. Leukemia 2020, 35, 1023–1036.
- Gu, X.; Tohme, R.; Goldfinger, M.; Tomlinson, B.K.; Sakre, N.; Hanmer, S.; Jha, B.K.; Maciejewski, J.P.; Verma, A.; Saunthararajah, Y. Venetoclax Inhibition of Pyrimidine Synthesis Guides Methods for Integration with Decitabine or 5-Azacytidine That Are Non-Myelosuppressive. Blood 2020, 136 (Suppl. 1), 26–27.
- Saunthararajah, Y. Mysteries of partial dihydroorotate dehydrogenase inhibition and leukemia terminal differentiation. Haematologica 2020, 105, 2191–2193.
- FDA. FDA Approves Venetoclax in Combination for AML in Adults. 20 December 2019. Available online: https://www.fda.gov/drugs/fda-approves-venetoclax-combination-aml-adults (accessed on 24 September 2023).
- DiNardo, C.D.; Jonas, B.A.; Pullarkat, V.; Thirman, M.J.; Garcia, J.S.; Wei, A.H.; Konopleva, M.; Döhner, H.; Letai, A.; Fenaux, P.; et al. Azacitidine and Venetoclax in Previously Untreated Acute Myeloid Leukemia. N. Engl. J. Med. 2020, 383, 617–629.
- Chyla, B.J.; Harb, J.; Mantis, C.; Riehm, J.J.; Ross, J.A.; Sun, Y.; Huang, X.; Jiang, Q.; Dail, M.; Peale, F.V., Jr.; et al. Response to Venetoclax in Combination with Low Intensity Therapy (LDAC or HMA) in Untreated Patients with Acute Myeloid Leukemia Patients with IDH, FLT3 and Other Mutations and Correlations with BCL2 Family Expression. Blood 2019, 134, 546.
- DiNardo, C.D.; Tiong, I.S.; Quaglieri, A.; MacRaild, S.; Loghavi, S.; Brown, F.C.; Thijssen, R.; Pomilio, G.; Ivey, A.; Salmon, J.M.; et al. Molecular patterns of response and treatment failure after frontline venetoclax combinations in older patients with AML. Blood 2020, 135, 791–803.
- Morsia, E.; McCullough, K.; Joshi, M.; Cook, J.; Alkhateeb, H.B.; Al-Kali, A.; Begna, K.; Elliott, M.; Hogan, W.; Litzow, M.; et al. Venetoclax and hypomethylating agents in acute myeloid leukemia: Mayo Clinic series on 86 patients. Am. J. Hematol. 2020, 95, 1511–1521.
- Stahl, M.; Menghrajani, K.; Derkach, A.; Chan, A.; Xiao, W.; Glass, J.; King, A.C.; Daniyan, A.F.; Famulare, C.; Cuello, B.M.; et al. Clinical and molecular predictors of response and survival following venetoclax therapy in relapsed/refractory AML. Blood Adv. 2021, 5, 1552–1564.
- Wei, A.H.; Montesinos, P.; Ivanov, V.; DiNardo, C.D.; Novak, J.; Laribi, K.; Kim, I.; Stevens, D.A.; Fiedler, W.; Pagoni, M.; et al. Venetoclax plus LDAC for newly diagnosed AML ineligible for intensive chemotherapy: A phase 3 randomized placebo-controlled trial. Blood 2020, 135, 2137–2145.
- Faruqi, A.; Tadi, P. Cytarabine. In StatPearls ; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: http://www.ncbi.nlm.nih.gov/books/NBK557680/ (accessed on 28 October 2023).
- Megías-Vericat, J.E.; Montesinos, P.; Herrero, M.J.; Bosó, V.; Martínez-Cuadrón, D.; Poveda, J.L.; Sanz, M.; Aliño, S.F. Pharmacogenomics and the treatment of acute myeloid leukemia. Pharmacogenomics 2016, 17, 1245–1272.
More
Information
Subjects:
Oncology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
604
Revisions:
4 times
(View History)
Update Date:
26 Feb 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





