
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Maria-Anna Gatou | -- | 9599 | 2024-02-16 08:19:33 | | | |
| 2 | Mona Zou | Meta information modification | 9599 | 2024-02-20 08:29:58 | | |
Video Upload Options
Contemporary technological and industrial advancements have led to increased reliance on chemicals for product innovation, leading to heightened contamination of water sources by traditional pollutants (organic dyes, heavy metals) and disease-causing microorganisms. Wastewater treatment processes now reveal “emerging pollutants”, including pharmaceuticals, endocrine disruptors, and agricultural chemicals. While some are benign, certain emerging pollutants can harm diverse organisms. Researchers seek cost-effective water purification methods that completely degrade pollutants without generating harmful by-products. Semiconductor-based photocatalytic degradation, particularly using titanium dioxide (TiO2), is popular for addressing water pollution.
1. Introduction
2. TiO2-Based Photocatalysts for Effective Elimination of Pharmaceutical Pollutants from Water and Wastewater
3. TiO2-Based Photocatalysts for Effective Elimination of Heavy Metals from Water and Wastewater
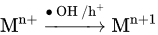
4. TiO2-Based Photocatalysts for Effective Elimination of Organic Dyes from Water and Wastewater
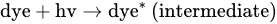
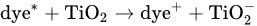
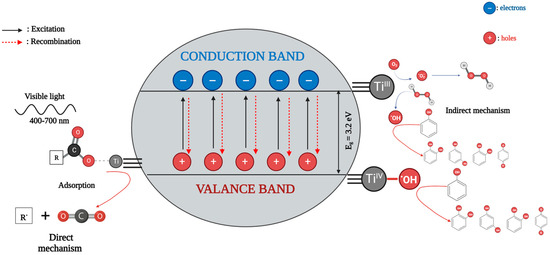
5. TiO2-Based Photocatalysts for Effective Elimination of Pesticides from Water and Wastewater
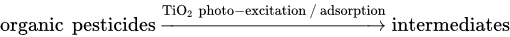
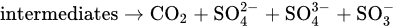
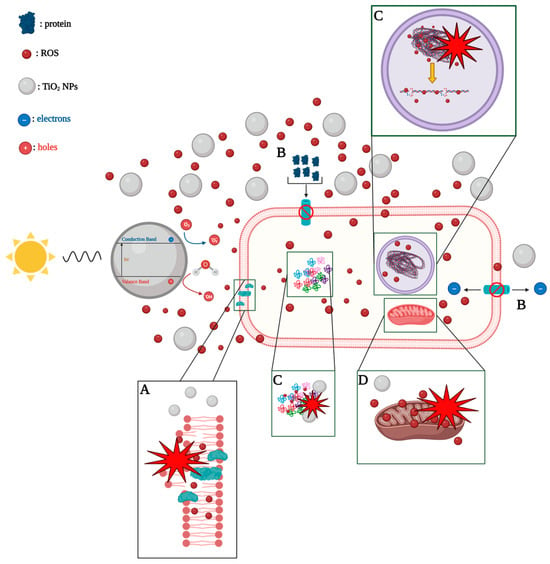
6. TiO2-Based Photocatalysts for Effective Elimination of Microbes from Water and Wastewater
7. TiO2-Based Photocatalysts for Effective Elimination of Hormones and Endocrine Disrupting Compounds (EDCs) from Water and Wastewater
References
- UNESCO. The United Nations World Water Development Report 2020 Water and Climate Change; UNESCO: Paris, France, 2020.
- Chart, H. VTEC enteropathogenicity. Symp. Ser. Soc. Appl. Microbiol. 2000, 29, 12S–23S.
- Fowler, C.C.; Galán, J.E. Decoding a Salmonella typhi regulatory network that controls typhoid toxin expression within human cells. Cell Host Microbe 2018, 23, 65–76.
- Rivera-Chávez, F.; Mekalanos, J.J. Cholera toxin promotes pathogen acquisition of host-derived nutrients. Nature 2019, 572, 244–248.
- Chuang, C.-H.; Wang, Y.-H.; Chang, H.-J.; Chen, H.-L.; Huang, Y.-C.; Lin, T.-Y.; Ozer, E.A.; Allen, J.P.; Hauser, A.R.; Chiu, C.-H. Shanghai fever: A distinct Pseudomonas aeruginosa enteric disease. Gut 2014, 63, 736–743.
- Rajasurya, V.; Surani, S. Legionnaires Disease in Immunocompromised Host; IntechOpen: London, UK, 2020.
- Sansenya, T.; Masri, N.; Chankhanittha, T.; Senasu, T.; Piriyanon, J.; Mukdasai, S.; Nanan, S. Hydrothermal synthesis of ZnO photocatalyst for detoxification of anionic azo dyes and antibiotic. J. Phys. Chem. Solids 2022, 160, 110353.
- Vaez, Z.; Javanbakht, V. Synthesis, characterization and photocatalytic activity of ZSM-5/ZnO nanocomposite modified by Ag nanoparticles for methyl orange degradation. J. Photochem. Photobiol. A 2020, 388, 112064.
- Khataee, A.R.; Pons, M.N.; Zahraa, O. Photocatalytic degradation of three azo dyes using immobilized TiO2 nanoparticles on glass plates activated by UV light irradiation: Influence of dye molecular structure. J. Hazard. Mater. 2009, 168, 451–457.
- Liberatore, L.; Bressan, M.; Belli, C.; Lustrato, G.; Ranalli, G. Chemical and biological combined treatments for the removal of pesticides from wastewaters. Water Air Soil Pollut. 2012, 223, 4751–4759.
- Ballesteros Martín, M.M.; Sánchez Pérez, J.A.; Casas López, J.L.; Oller, I.; Malato Rodríguez, S. Degradation of a four-pesticide mixture by combined photo-Fenton and biological oxidation. Water Res. 2009, 43, 653–660.
- Bressan, M.; Liberatore, L.; d’Alessandro, N.; Tonucci, L.; Belli, C.; Ranalli, G. Improved combined chemical and biological treatments of olive oil mill wastewaters. J. Agric. Food Chem. 2004, 52, 1228–1233.
- Tahir, M.S.; Saleem, M.; Malik, S.R.; Khan, J.R.; Siebenhofer, M. An innovative and advanced oxidation process for effluent treatment through wet tube-type electrostatic precipitation. Chem. Eng. Process. Process Intensif. 2012, 52, 16–20.
- Ben Mansour, L.; Kesentini, I. Treatment of effluents from cardboard industry by coagulation-electroflotation. J. Hazard. Mater. 2008, 153, 1067–1070.
- Hanay, Ö.; Hasar, H. Effect of anions on removing Cu2+, Mn2+ and Zn2+ in electrocoagulation process using aluminum electrodes. J. Hazard. Mater. 2011, 189, 572–576.
- Centi, G.; Perathoner, S.; Torre, T.; Verduna, M.G. Catalytic wet oxidation with H2O2 of carboxylic acids on homogeneous and heterogeneous Fenton-type catalysts. Catal. Today 2000, 55, 61–69.
- Ghosh, P.; Samanta, A.N.; Ray, S. Reduction of COD and removal of Zn2+ from rayon industry wastewater by combined electro-Fenton treatment and chemical precipitation. Desalination 2011, 266, 213–217.
- Lin, S.H.; Chang, C.C. Treatment of landfill leachate by combined electro-Fenton oxidation and sequencing batch reactor method. Water Res. 2000, 34, 4243–4249.
- Ayodele, O.B.; Hameed, B.H. Synthesis of copper pillared bentonite ferrioxalate catalyst for degradation of 4-nitrophenol in visible light assisted Fenton process. J. Ind. Eng. Chem. 2013, 19, 966–974.
- Monteagudo, J.M.; Durán, A.; San Martín, I.; Aguirre, M. Effect of light source on the catalytic degradation of protocatechuic acid in a ferrioxalate-assisted photo-Fenton process. Appl. Catal. B 2010, 96, 486–495.
- John Peter, I.; Praveen, E.; Vignesh, G.; Nithiananthi, P. ZnO nanostructures with different morphology for enhanced photocatalytic activity. Mater. Res. Express 2017, 4, 124003.
- Zhang, D.E.; Ren, L.Z.; Hao, X.Y.; Pan, B.B.; Wang, M.Y.; Ma, J.J.; Li, F.; Li, S.A.; Tong, Z.W. Synthesis and photocatalytic property of multilayered Co3O4. Appl. Surf. Sci. 2015, 355, 547–552.
- Ani, I.J.; Akpan, U.G.; Olutoye, M.A.; Hameed, B.H. Photocatalytic degradation of pollutants in petroleum refinery wastewater by TiO2- and ZnO-based photocatalysts: Recent development. J. Clean. Prod. 2018, 205, 930–954.
- Tanji, K.; Navio, J.A.; Chaqroune, A.; Naja, J.; Puga, F.; Hidalgo, M.C.; Kherbeche, A. Fast photodegradation of rhodamine B and caffeine using ZnO-hydroxyapatite composites under UV-light illumination. Catal. Today 2022, 388–389, 176–186.
- Lagopati, N.; Tsilibary, E.P.; Falaras, P.; Papazafiri, P.; Pavlatou, E.A.; Kotsopoulou, E.; Kitsiou, P. Effect of nanostructured TiO2 crystal phase on photoinduced apoptosis of breast cancer epithelial cells. Int. J. Nanomed. 2014, 9, 3219–3230.
- Lagopati, N.; Kitsiou, P.; Kontos, A.; Venieratos, P.; Kotsopoulou, E.; Kontos, A.; Dionysiou, D.; Pispas, S.; Tsilibary, E.; Falaras, P. Photo-induced treatment of breast epithelial cancer cells using nanostructured titanium dioxide solution. J. Photochem. Photobiol. A Chem. 2010, 214, 215–223.
- Anitha, B.; Khadar, M.A. Anatase-rutile phase transformation and photocatalysis in peroxide gel route prepared TiO2 nanocrystals: Role of defect states. Solid State Sci. 2020, 108, 106392.
- Lagopati, N.; Kotsinas, A.; Veroutis, D.; Evangelou, K.; Papaspyropoulos, A.; Arfanis, M.; Falaras, P.; Kitsiou, P.V.; Pateras, I.; Bergonzini, A.; et al. Biological effect of silver-modified nanostructured titanium dioxide in cancer. Cancer Genom. Proteom. 2021, 18 (Suppl. S3), 425–439.
- Nur, A.S.M.; Sultana, M.; Mondal, A.; Islam, S.; Robel, F.N.; Islam, A.; Sumi, M.S.A. A review on the development of elemental and codoped TiO2 photocatalysts for enhanced dye degradation under UV–vis irradiation. J. Water Process. Eng. 2022, 47, 102728.
- Kowalska, E.; Mahaney, O.O.P.; Abe, R.; Ohtani, B. Visible-light-induced photocatalysis through surface plasmon excitation of gold on titania surfaces. Phys. Chem. Chem. Phys. 2010, 12, 2344–2355.
- Kümmerer, K. Antibiotics in the aquatic environment—A review–part I. Chemosphere 2009, 75, 417–434.
- Rasouli, S.; Rezaei, N.; Hamedi, H.; Zendehboudi, S.; Duan, X. Superhydrophobic and superoleophilic membranes for oil-water separation application: A comprehensive review. Mater. Des. 2021, 204, 109599.
- Zareei Pour, F.; Sabzehmeidani, M.M.; Karimi, H.; Madadi Avargani, V.; Ghaedi, M. Superhydrophobic–superoleophilic electrospun nanofibrous membrane modified by the chemical vapor deposition of dimethyl dichlorosilane for efficient oil-water separation. J. Appl. Polym. Sci. 2019, 136, 47621.
- Ahmadpour, N.; Sayadi, M.H.; Sobhani, S.; Hajiani, M. A potential natural solar light active photocatalyst using magnetic ZnFe2O4@TiO2/Cu nanocomposite as a high performance and recyclable platform for degradation of naproxen from aqueous solution. J. Clean. Prod. 2020, 268, 122023.
- Kumar, A.; Kumar, A.; Sharma, G.; Al-Muhtaseb, A.H.; Naushad, M.; Ghfar, A.A.; Stadler, F.J. Quaternary magnetic BiOCl/g-C3N4/Cu2O/Fe3O4 nano-junction for visible light and solar powered degradation of sulfamethoxazole from aqueous environment. Chem. Eng. J. 2018, 334, 462–478.
- Ahmadpour, N.; Sayadi, M.H.; Sobhani, S.; Hajiani, M. Photocatalytic degradation of model pharmaceutical pollutant by novel magnetic TiO2@ZnFe2O4/Pd nanocomposite with enhanced photocatalytic activity and stability under solar light irradiation. J. Environ. Manag. 2020, 271, 110964.
- Naushad, M. Surfactant assisted nano-composite cation exchanger: Development, characterization and applications for the removal of toxic Pb2+ from aqueous medium. Chem. Eng. J. 2014, 235, 100–108.
- Sayadi, M.H.; Ahmadpour, N.; Homaeigohar, S. Photocatalytic and antibacterial properties of Ag-CuFe2O4@WO3 magnetic nanocomposite. Nanomaterials 2021, 11, 298.
- Liu, F.; Liang, J.; Chen, L.; Tong, M.; Liu, W. Photocatalytic removal of diclofenac by Ti doped BiOI microspheres under visible light irradiation: Kinetics, mechanism, and pathways. J. Mol. Liq. 2019, 275, 807–814.
- Comber, S.; Gardner, M.; Sörme, P.; Ellor, B. The removal of pharmaceuticals during wastewater treatment: Can it be predicted accurately? Sci. Total Environ. 2019, 676, 222–230.
- Awfa, D.; Ateia, M.; Fujii, M.; Johnson, M.S.; Yoshimura, C. Photodegradation of pharmaceuticals and personal care products in water treatment using carbonaceous-TiO2 composites: A critical review of recent literature. Water Res. 2018, 142, 26–45.
- Mudgal, S.; De Toni, A.; Lockwood, S.; Sales, K.; Backhaus, T.; Sorensen, B.H. Study on the environmental risks of medicinal products. In BIO Intelligence Service; European Commission: Brussels, Belgium, 2013; Available online: https://hal.science/hal-03859880 (accessed on 12 January 2024).
- Sheng, C.; Agwu Nnanna, A.G.; Liu, Y.; Vargo, J.D. Removal of trace pharmaceuticals from water using coagulation and powdered activated carbon as pretreatment to ultrafiltration membrane system. Sci. Total Environ. 2016, 550, 1075–1083.
- Bai, X.; Acharya, K. Removal of trimethoprim, sulfamethoxazole, and triclosan by the green alga Nannochloris sp. J. Hazard. Mater. 2016, 315, 70–75.
- Williams, R.T.; Cook, J.C. Exposure to pharmaceuticals present in the environment. Ther. Innov. Regul. Sci. 2007, 41, 133–141.
- Oluwole, A.O.; Omotola, E.O.; Olatunji, O.S. Pharmaceuticals and personal care products in water and wastewater: A review of treatment processes and use of photocatalyst immobilized on functionalized carbon in AOP degradation. BMC Chem. 2020, 14, 62.
- Kümmerer, K.; Steger-Hartmann, T.; Meyer, M. Biodegradability of the anti-tumour agent ifosfamide and its occurrence in hospital effluents and communal sewage. Water Res. 1997, 31, 2705–2710.
- Kumar, A.; Sharma, G.; Naushad, M.; Ahamad, T.; Cataluña Veses, R.; Stadler, F.J. Highly visible active Ag2CrO4/Ag/BiFeO3@RGO nano-junction for photoreduction of CO2 and photocatalytic removal of ciprofloxacin and bromate ions: The triggering effect of Ag and RGO. Chem. Eng. J. 2019, 370, 148–165.
- Layton, A.; Gregory, B.; Seward, J.; Schultz, T.; Sayler, G. Mineralization of steroidal hormones by biosolids in wastewater treatment systems in Tennessee USA. Environ. Sci. Technol. 2000, 34, 3925–3931.
- Kokkinos, P.; Venieri, D.; Mantzavinos, D. Advanced oxidation processes for water and wastewater viral disinfection. a systematic review. Food Environ. Virol. 2021, 13, 283–302.
- Kanakaraju, D.; Glass, B.D.; Oelgemöller, M. Titanium dioxide photocatalysis for pharmaceutical wastewater treatment. Environ. Chem. Lett. 2014, 12, 27–47.
- Zhang, Y.; Zhou, Z.; Chen, T.; Wang, H.; Lu, W. Graphene TiO2 nanocomposites with high photocatalytic activity for the degradation of sodium pentachlorophenol. J. Environ. Sci. 2014, 26, 2114–2122.
- Sharma, G.; Alothman, Z.A.; Kumar, A.; Sharma, S.; Kumar Ponnusamy, S.; Naushad, M. Fabrication and characterization of a nanocomposite hydrogel for combined photocatalytic degradation of a mixture of malachite green and fast green dye. Nanotechnol. Environ. Eng. 2017, 2, 4.
- Akter, S.; Islam, S.; Kabir, H.; Shaikh, A.A.; Gafur, A. UV/TiO2 photodegradation of metronidazole, ciprofloxacin and sulfamethoxazole in aqueous solution: An optimization and kinetic study. Arab. J. Chem. 2022, 15, 103900.
- Sharma, M.; Yadav, A.; Mandal, M.K.; Dubey, K.K. TiO2 based photocatalysis: A valuable approach for the removal of pharmaceuticals from aquatic environment. Int. J. Environ. Sci. Technol. 2023, 20, 4569–4584.
- Manasa, M.; Chandewar, P.R.; Mahalingam, H. Photocatalytic degradation of ciprofloxacin & norfloxacin and disinfection studies under solar light using boron & cerium doped TiO2 catalysts synthesized by green EDTA-citrate method. Catal. Today 2020, 375, 522–536.
- Suwannaruang, T.; Kidkhunthod, P.; Chanlek, N.; Soontaranon, S.; Wantala, K. High anatase purity of nitrogen-doped TiO2 nanorice particles for the photocatalytic treatment activity of pharmaceutical wastewater. Appl. Surf. Sci. 2019, 478, 1–14.
- Martins, P.; Kappert, S.; Nga Le, H.; Sebastian, V.; Kühn, K.; Alves, M.; Pereira, L.; Cuniberti, G.; Melle-Franco, M.; Lanceros-Méndez, S. Enhanced photocatalytic activity of Au/TiO2 nanoparticles against ciprofloxacin. Catalysts 2020, 10, 234.
- Cabrera-Reina, A.; Martínez-Piernas, A.B.; Bertakis, Y.; Xekoukoulotakis, N.P.; Agüera, A.; Sánchez-Pérez, J.A. Ti photocatalysis under natural solar radiation for the degradation of the carbapenem antibiotics imipenem and meropenem in aqueous solutions at pilot plant scale. Water Res. 2019, 166, 115037.
- Truppi, A.; Petronella, F.; Placido, T.; Margiotta, V.; Lasorella, G.; Giotta, L.; Giannini, C.; Sibillano, T.; Murgolo, S.; Mascolo, G.; et al. Gram-scale synthesis of UV-vis light active plasmonic photocatalytic nanocomposite based on TiO2/Au nanorods for degradation of pollutants in water. Appl. Catal. B Environ. 2019, 243, 604–613.
- Gómez-Avilés, A.; Peñas-Garzón, M.; Bedia, J.; Rodriguez, J.J.; Belver, C. C-modified TiO2 using lignin as carbon precursor for the solar photocatalytic degradation of acetaminophen. Chem. Eng. J. 2019, 358, 1574–1582.
- Peñas-Garzón, M.; Gómez-Avilés, A.; Belver, C.; Rodriguez, J.J.; Bedia, J. Degradation pathways of emerging contaminants using TiO2-activated carbon heterostructures in aqueous solution under simulated solar light. Chem. Eng. J. 2020, 392, 124867.
- Murgolo, S.; Moreira, I.S.; Piccirillo, C.; Castro, P.M.L.; Ventrella, G.; Cocozza, C.; Mascolo, G. Photocatalytic degradation of diclofenac by hydroxyapatite–TiO2 composite material: Identification of transformation products and assessment of toxicity. Materials 2018, 11, 1779.
- Czech, B.; Tyszczuk-Rotko, K. Visible-light-driven photocatalytic removal of acetaminophen from water using a novel MWCNT-TiO2-SiO2 photocatalysts. Separ. Purif. Technol. 2018, 206, 343–355.
- Payan, A.; Akbar Isari, A.; Gholizade, N. Catalytic decomposition of sulfamethazine antibiotic and pharmaceutical wastewater using Cu-TiO2@functionalized SWCNT ternary porous nanocomposite: Influential factors, mechanism, and pathway studies. Chem. Eng. J. 2019, 361, 1121–1141.
- Abdelraheem, W.H.M.; Nadagouda, M.N.; Dionysiou, D.D. Solar light-assisted remediation of domestic wastewater by N,B-TiO2 nanoparticles for potable reuse. Appl. Catal. B Environ. 2020, 269, 118807.
- Carbuloni, C.F.; Savoia, J.E.; Santos, J.S.P.; Pereira, C.A.A.; Marques, R.G.; Ribeiro, V.A.S.; Ferrari, A.M. Degradation of metformin in water by TiO2–ZrO2 photocatalysis. J. Environ. Manag. 2020, 262, 110347.
- Escudeiro de Oliveira, M.; Barroso, B.L.; de Almeida, J.; Moraes, M.L.L.; de Arruda Rodrigues, C. Photoelectrocatalytic degradation of 17a-ethinylestradiol and estrone under UV and visible light using nanotubular oxide arrays grown on Ti-0.5wt%W. Environ. Res. 2020, 191, 110044.
- Gurung, K.; Ncibi, M.C.; Thangaraj, S.K.; Jänis, J.; Seyedsalehi, M.; Sillanpää, M. Removal of pharmaceutically active compounds (PhACs) from real membrane bioreactor (MBR) effluents by photocatalytic degradation using composite Ag2O/P-25 photocatalyst. Separ. Purif. Technol. 2019, 215, 317–328.
- Gomathi Devi, L.; Kavitha, R. A review on plasmonic metal TiO2 composite for generation, trapping, storing and dynamic vectorial transfer of photogenerated electrons across the Schottky junction in a photocatalytic system. Appl. Surf. Sci. 2016, 360, 601–622.
- Yin, X.; Que, W.; Liao, Y.; Xie, H.; Fei, D. Ag-TiO2 nanocomposites with improved photocatalytic properties prepared by a low temperature process in polyethylene glycol. Colloids Surf. A 2012, 410, 153–158.
- Fei, J.; Li, J. Controlled preparation of porous TiO2-Ag nanostructures through supramolecular assembly for plasmon-enhanced photocatalysis. Adv. Mater. 2015, 27, 314–319.
- Hou, W.; Cronin, S.B. A review of surface plasmon resonance-enhanced photocatalysis. Adv. Funct. Mater. 2013, 23, 1612–1619.
- Wang, T.; Wang, H.-J.; Lin, J.-S.; Yang, J.-L.; Zhang, F.-L.; Lin, X.-M.; Zhang, Y.-J.; Jin, S.; Li, J.-F. Plasmonic photocatalysis: Mechanism, applications and perspectives. Chin. J. Struct. Chem. 2023, 42, 100066.
- Kaur, R.; Pal, B. Co-catalysis effect of different morphological facets of as prepared Ag nanostructures for the photocatalytic oxidation reaction by Ag-TiO2 aqueous slurry. Mater. Chem. Phys. 2013, 143, 393–399.
- Gang, R.; Xia, Y.; Xu, L.; Zhang, L.; Ju, S.; Wang, Z.; Koppala, S. Size controlled Ag decorated TiO2 plasmonic photocatalysts for tetracycline degradation under visible light. Surf. Interfaces 2022, 31, 102018.
- Litter, M.I. Mechanisms of removal of heavy metals and arsenic from water by TiO2-heterogeneous photocatalysis. Pure Appl. Chem. 2015, 87, 557–567.
- Zhang, D.; Zhang, C.L.; Zhou, P. Preparation of porous nano-calcium titanate microspheres and its adsorption behavior for heavy metal ion in water. J. Hazard. Mater. 2011, 186, 971–977.
- Shaheen, N.; Irfan, N.M.; Khan, I.N.; Islam, S.; Islam, M.S.; Ahmed, M.K. Presence of heavy metals in fruits and vegetables: Health risk implications in Bangladesh. Chemosphere 2016, 152, 431–438.
- Khan, A.; Khan, S.; Khan, M.A.; Qamar, Z.; Waqas, M. The uptake and bioaccumulation of heavy metals by food plants, their effects on plants nutrients, and associated health risk: A review. Environ. Sci. Pollut. Res. 2015, 22, 13772–13799.
- Kyung, H.; Lee, J.; Choi, W. Simultaneous and synergistic conversion of dyes and heavy metal ions in aqueous TiO2 suspensions under visible-light illumination. Environ. Sci. Technol. 2005, 39, 2376–2382.
- Mahdavi, S.; Jalali, M.; Afkhami, A. Heavy metals removal from aqueous solutions using TiO2, MgO, and Al2O3 nanoparticles. Chem. Eng. Commun. 2013, 200, 448–470.
- Poursani, A.S.; Nilchi, A.; Hassani, A.; Shariat, S.M.; Nouri, J. The synthesis of nano TiO2 and its use for removal of lead ions from aqueous solution. J. Water Resour. Protect. 2016, 8, 438–448.
- Luo, T.; Cui, J.; Hu, S.; Huang, Y.; Jing, C. Arsenic removal and recovery from copper smelting wastewater using TiO2. Environ. Sci. Technol. 2010, 44, 9094–9098.
- Yan, M.; Zeng, G.; Li, X.; Zhao, C.; Yang, G.; Gong, J.; Chen, G.; Tang, L.; Huang, D. Titanium dioxide nanotube arrays with silane coupling agent modification for heavy metal reduction and persistent organic pollutant degradation. New J. Chem. 2017, 41, 4377–4389.
- Skubal, L.R.; Meshkov, N.K. Reduction and removal of mercury from water using arginine-modified TiO2. J. Photochem. Photobiol. A Chem. 2002, 148, 211–214.
- Zhang, F.-S.; Nriagu, J.O.; Itoh, H. Photocatalytic removal and recovery of mercury from water using TiO2-modified sewage sludge carbon. J. Photochem. Photobiol. A Chem. 2004, 167, 223–228.
- Dou, B.; Chen, H. Removal of toxic mercury(II) from aquatic solutions by synthesized TiO2 nanoparticles. Desalination 2011, 269, 260–265.
- Xu, S.; Du, A.J.; Liu, J.; Ng, J.; Sun, D.D. Highly efficient CuO incorporated TiO2 nanotube photocatalyst for hydrogen production from water. Int. J. Hydrogen Energy 2011, 36, 6538–6545.
- Xu, S.C.; Pan, S.S.; Xu, Y.; Luo, Y.Y.; Zhang, Y.X.; Li, G.H. Efficient removal of Cr (VI) from wastewater under sunlight by Fe(II)-doped TiO2 spherical shell. J. Hazard. Mater. 2014, 283, 7–13.
- Chen, Z.; Li, Y.; Guo, M.; Xu, F.; Wang, P.; Du, Y.; Na, P. One-pot synthesis of Mn-doped TiO2 grown on graphene and the mechanism for removal of Cr(VI) and Cr (III). J. Hazard. Mater. 2016, 310, 188–198.
- Gullipilli, S.; Rupakula, R.B. Adsorption studies of Cr(VI) and Cu(II) metal ions from aqueous solutions by synthesized Ag and Mg co-doped TiO2 nanoparticles. Separ. Sci. Technol. 2018, 54, 2983–2992.
- Eddy, D.R.; Rahayu, I.; Wyantuti, S.; Hartati, Y.W.; Firdaus, M.L.; Bahti, H.H. Photocatalytic activity of gadolinium doped TiO2 particles for decreasing heavy metal chromium (VI) concentration. J. Phys. Conf. Ser. 2018, 1080, 012013.
- Luo, Z.; Qu, L.; Jia, J.; Wang, J.; Jiang, S.; Wu, Z.; Wu, X. TiO2/EDTA-rich carbon composites: Synthesis, characterization and visible-light-driven photocatalytic reduction of Cr(VI). Chin. Chem. Lett. 2018, 29, 547–550.
- Kobayashi, Y.; Kanasaki, R.; Nozaki, T.; Shoji, R.; Sato, K. Improving effect of MnO2 addition on TiO2-photocatalytic removal of lead ion from water. J. Water Environ. Technol. 2017, 15, 35–42.
- Zhang, Y.; Hu, H.; Chang, M.; Chen, D.; Zhang, M.; Wu, L.; Li, X. Nonuniform doping outperforms uniform doping for enhancing the photocatalytic efficiency of Au-doped TiO2 nanotubes in organic dye degradation. Ceram. Int. 2017, 43, 9053–9059.
- Zhang, Y.; Hu, Z.; Cui, X.; Yao, W.; Duan, T.; Zhu, W. Capture of Cs+ and Sr2+ from aqueous solutions by using Cr doped TiO2 nanotubes. J. Nanosci. Nanotechnol. 2017, 17, 3943–3950.
- Garza-Arévalo, J.I.; García-Montes, I.; Reyes, M.H.; Guzmán-Mar, J.L.; Rodríguez-González, V.; Reyes, L.H. Fe doped TiO2 photocatalyst for the removal of As (III) under visible radiation and its potential application on the treatment of As-contaminated groundwater. Mater. Res. Bull. 2016, 73, 145–152.
- Seema, K.M.; Mamba, B.B.; Njuguna, J.; Bakhtizin, R.Z.; Mishra, A.K. Removal of lead (II) from aqeouos waste using (CD-PCL-TiO2) bio-nanocomposites. Int. J. Biol. Macromol. 2018, 109, 136–142.
- Zhao, Y.; Chang, W.; Huang, Z.; Feng, X.; Ma, L.; Qi, X.; Li, Z. Enhanced removal of toxic Cr(VI) in tannery wastewater by photoelectrocatalysis with synthetic TiO2 hollow spheres. Appl. Surf. Sci. 2017, 405, 102–110.
- Wang, Q.; Zheng, Q.; Jin, R.; Gao, S.; Yuan, Q.; Rong, W.; Wang, R. Photoelectrocatalytic removal of organic dyes and Cr(VI) ions using Ag3PO4 nanoparticles sensitized TiO2 nanotube arrays. Mater. Chem. Phys. 2017, 199, 209–215.
- Hang, Y.; Yin, H.; Ji, Y.; Liu, Y.; Lu, Z.; Wang, A.; Shen, L.; Yin, H. Adsorption performances of naked and 3-aminopropyl triethoxysilane-modified mesoporous TiO2 hollow nanospheres for Cu2+, Cd2+, Pb2+, Cr(VI) ions. J. Nanosci. Nanotechnol. 2017, 17, 5539–5549.
- Abbas, K.K.; Al-Ghaban, A.M.H.A. Enhanced solar light photoreduction of innovative TiO2 nanospherical shell by reduced graphene oxide for removal silver ions from aqueous media. J. Environ. Chem. Eng. 2019, 7, 103168.
- Gan, W.; Shang, X.; Li, X.H.; Zhang, J.; Fu, X. Achieving high adsorption capacity and ultrafast removal of methylene blue and Pb2+ by graphene-like TiO2@C. Colloids Surf. A 2019, 561, 218–225.
- Ajmal, A.; Majeed, I.; Malik, R.N.; Idriss, H.; Nadeem, M.A. Principles and mechanisms of photocatalytic dye degradation on TiO2-based photocatalysts: A comparative overview. RSC Adv. 2014, 4, 37003–37026.
- Konstantinou, I.K.; Albanis, T.A. TiO2-assisted photocatalytic degradation of azo dyes in aqueous solution: Kinetic and mechanistic investigations: A review. Appl. Catal. B Environ. 2004, 49, 1–14.
- Zhang, F.; Zhao, J.; Shen, T.; Hidaka, H.; Pelizzetti, E.; Serpone, N. TiO2-assisted photodegradation of dye pollutants II. Adsorption and degradation kinetics of eosin in TiO2 dispersions under visible light irradiation. Appl. Catal. B Environ. 1998, 15, 147–156.
- Sakthivel, S.; Neppolian, B.; Shankar, M.V.; Arabindoo, B.; Palanichamy, M.; Murugesan, V. Solar photocatalytic degradation of azo dye: Comparison of photocatalytic efficiency of ZnO and TiO2. Sol. Energy Mater. Sol. Cells 2003, 77, 65–82.
- Chen, C.; Li, X.; Ma, W.; Zhao, J.; Hidaka, H.; Serpone, N. Effect of transition metal ions on the TiO2-assisted photodegradation of dyes under visible irradiation: A probe for the interfacial electron transfer process and reaction mechanism. J. Phys. Chem. B 2002, 106, 318–324.
- Gola, D.; Malik, A.; Namburath, M.; Ahammad, S.Z. Removal of industrial dyes and heavy metals by Beauveria bassiana: FTIR, SEM, TEM and AFM investigations with Pb(II). Environ. Sci. Pollut. Res. 2018, 25, 20486–20496.
- Chung, K.T. Azo dyes and human health: A review. J. Environ. Sci. Health Part C Environ. Carcinog. Ecotoxicol. Rev. 2016, 34, 233–261.
- Raman, C.D.; Kanmani, S. Textile dye degradation using nano zero valent iron: A review. J. Environ. Manag. 2016, 177, 341–355.
- Chandra, R.; Mukhopadhyay, S.; Nath, M. TiO2@ZIF-8: A novel approach of modifying micro-environment for enhanced photo-catalytic dye degradation and high usability of TiO2 nanoparticles. Mater. Lett. 2016, 164, 571–574.
- Dariani, R.S.; Esmaeili, A.; Mortezaali, A.; Dehghanpour, S. Photocatalytic reaction and degradation of methylene blue on TiO2 nano-sized particles. Optik 2016, 127, 7143–7154.
- Singh, R.; Kumari, P.; Chavan, P.D.; Datta, S.; Dutta, S. Synthesis of solvothermal derived TiO2 nanocrystals supported on ground nano egg shell waste and its utilization for the photocatalytic dye degradation. Opt. Mater. 2017, 73, 377–383.
- Ljubas, D.; Smoljanić, G.; Juretić, H. Degradation of Methyl Orange and Congo Red dyes by using TiO2 nanoparticles activated by the solar and the solar-like radiation. J. Environ. Manag. 2015, 161, 83–91.
- Zulfiqar, M.; Chowdhury, S.; Sufian, S.; Omar, A.A. Enhanced photocatalytic activity of Orange II in aqueous solution using solvent-based TiO2 nanotubes: Kinetic, equilibrium and thermodynamic studies. J. Clean. Prod. 2018, 203, 848–859.
- Pol, R.; Guerrero, M.; García-Lecina, E.; Altube, A.; Rossinyol, E.; Garroni, S.; Baró, M.D.; Pons, J.; Sort, J.; Pellicer, E. Ni-, Pt- and (Ni/Pt)-doped TiO2 nanophotocatalysts: A smart approach for sustainable degradation of Rhodamine B dye. Appl. Catal. B Environ. 2016, 181, 270–278.
- Gnanasekaran, L.; Hemamalini, R.; Saravanan, R.; Ravichandran, K.; Gracia, F.; Gupta, V.K. Intermediate state created by dopant ions (Mn, Co and Zr) into TiO2 nanoparticles for degradation of dyes under visible light. J. Mol. Liq. 2016, 223, 652–659.
- Kerkez-Kuyumcu, Ö.; Kibar, E.; Dayioʇlu, K.; Gedik, F.; Akin, A.N.; Özkara Aydinoǧlu, S. A comparative study for removal of different dyes over M/TiO2(M ¼ Cu, Ni, Co, Fe, Mn and Cr) photocatalysts under visible light irradiation. J. Photochem. Photobiol. A Chem. 2015, 311, 176–185.
- Kaur, N.; Kaur, S.; Singh, V. Preparation, characterization and photocatalytic degradation kinetics of Reactive Red dye 198 using N, Fe codoped TiO2 nanoparticles under visible light. Desalin. Water Treat. 2016, 57, 9237–9246.
- Sood, S.; Umar, A.; Kumar Mehta, S.; Sinha, A.S.K.; Kansal, S.K. Efficient photocatalytic degradation of brilliant green using Sr-doped TiO2 nanoparticles. Ceram. Int. 2015, 41, 3533–3540.
- Brindha, A.; Sivakumar, T. Visible active N, S co-doped TiO2/graphene photocatalysts for the degradation of hazardous dyes. J. Photochem. Photobiol. A Chem. 2017, 340, 146–156.
- McManamon, C.; O’Connell, J.; Delaney, P.; Rasappa, S.; Holmes, J.D.; Morris, M.A. A facile route to synthesis of S-doped TiO2 nanoparticles for photocatalytic activity. J. Mol. Catal. A Chem. 2015, 406, 51–57.
- Shao, J.; Sheng, W.; Wang, M.; Li, S.; Chen, J.; Zhang, Y.; Cao, S. In situ synthesis of carbon-doped TiO2 single-crystal nanorods with a remarkably photocatalytic efficiency. Appl. Catal. B Environ. 2017, 209, 311–319.
- Zhang, Q.; Fu, Y.; Wu, Y.; Zhang, Y.N.; Zuo, T. Low-cost Y-doped TiO2 nanosheets film with highly reactive facets from CRT waste and enhanced photocatalytic removal of Cr(VI) and methyl orange. ACS Sustain. Chem. Eng. 2016, 4, 1794–1803.
- Li, Q.; Zong, L.; Li, C.; Yang, J. Reprint of photocatalytic reduction of CO2 on MgO/TiO2 nanotube films. Appl. Surf. Sci. 2014, 319, 16–20.
- Dahl, M.; Liu, Y.; Yin, Y. Composite titanium dioxide nanomaterials. Chem. Rev. 2014, 114, 9853–9889.
- Anwar, D.I.; Mulyadi, D. Synthesis of Fe-TiO2 composite as a photocatalyst for degradation of methylene blue. Procedia Chem 2015, 17, 49–54.
- Lei, P.; Wang, F.; Gao, X.; Ding, Y.; Zhang, S.; Zhao, J.; Liu, S.; Yang, M. Immobilization of TiO2 nanoparticles in polymeric substrates by chemical bonding for multi-cycle photodegradation of organic pollutants. J. Hazard. Mater. 2012, 227–228, 185–194.
- Talebi, S.; Chaibakhsh, N.; Moradi-Shoeili, Z. Application of nanoscale ZnS/TiO2 composite for optimized photocatalytic decolorization of a textile dye. J. Appl. Res. Technol. 2017, 15, 378–385.
- Hui, C.; Lei, Z.; Xitang, W.; Shujing, L.; Zhongxing, L. Preparation of nanoporous TiO2/SiO2 composite with rice husk as template and its photocatalytic property. Rare Met. Mater. Eng. 2015, 44, 1607–1611.
- Li, J.; Zhen, D.; Sui, G.; Zhang, C.; Deng, Q.; Jia, L. Nanocomposite of Cu-TiO2-SiO2 with high photoactive performance for degradation of rhodamine B dye in aqueous wastewater. J. Nanosci. Nanotechnol. 2012, 12, 6265–6270.
- Liu, J.; Liu, J.; Shi, F.; Hu, S.; Jiang, S.; Liu, S.; Liu, D.; Tian, X. F/W codoped TiO2-SiO composite aerogels with improved visible light-driven photocatalytic activity. J. Solid State Chem. 2019, 275, 8–15.
- Shao, L.; Liu, H.; Zeng, W.; Zhou, C.; Li, D.; Wang, L.; Lan, Y.; Xu, F.; Liu, G. Immobilized and photocatalytic performances of PDMS-SiO2-chitosan@TiO2 composites on pumice under simulated sunlight irradiation. Appl. Surf. Sci. 2019, 478, 1017–1026.
- Tang, Y.; Di, W.; Zhai, X.; Yang, R.; Qin, W. NIR-responsive photocatalytic activity and mechanism of NaYF4:Yb,Tm@TiO2 core-shell nanoparticles. ACS Catal. 2013, 3, 405–412.
- Djellabi, R.; Ghorab, M.F.; Cerrato, G.; Morandi, S.; Gatto, S.; Oldani, V.; Di Michele, A.; Bianchi, C.L. Photoactive TiO2-montmorillonite composite for degradation of organic dyes in water. J. Photochem. Photobiol. A Chem. 2015, 295, 57–63.
- Alagarasi, A.; Rajalakshmi, P.U.; Shanthi, K.; Selvam, P. Solar light photocatalytic activity of mesoporous nanocrystalline TiO2, SnO2, and TiO2-SnO2 composites. Mater. Today Sustain. 2019, 5, 100016.
- Magdalane, C.M.; Kanimozhi, K.; Arularasu, M.V.; Ramalingam, G.; Kaviyarasu, K. Self-cleaning mechanism of synthesized SnO2/TiO2 nanostructure for photocatalytic activity application for waste water treatment. Surf. Interfaces 2019, 17, 100346.
- Li, Y.; Zhang, W.; Li, L.; Yi, C.; Lv, H.; Song, Q. Litchi-like CdS/CdTiO3-TiO2 composite: Synthesis and enhanced photocatalytic performance for crystal violet degradation and hydrogen production. RSC Adv. 2016, 6, 51374–51386.
- Hamdy, M.S.; Saputera, W.H.; Groenen, E.J.; Mul, G. A novel TiO2 composite for photocatalytic wastewater treatment. J. Catal. 2014, 310, 75–83.
- Rahimi, B.; Jafari, N.; Abdolahnejad, A.; Farrokhzadeh, H.; Ebrahimi, A. Application of efficient photocatalytic process using a novel BiVO/TiO2-NaY zeolite composite for removal of acid orange 10 dye in aqueous solutions: Modeling by response surface methodology (RSM). J. Environ. Chem. Eng. 2019, 7, 103253.
- Saleh, R.; Taufik, A.; Prakoso, S.P. Fabrication of Ag2O/TiO2 composites on nanographene platelets for the removal of organic pollutants: Influence of oxidants and inorganic anions. Appl. Surf. Sci. 2019, 480, 697–708.
- Visa, M.; Isac, L.; Duta, A. New fly ash TiO2 composite for the sustainable treatment of wastewater with complex pollutants load. Appl. Surf. Sci. 2015, 339, 62–68.
- Jaseela, P.K.; Garvasis, J.; Joseph, A. Selective adsorption of methylene blue (MB) dye from aqueous mixture of MB and methyl orange (MO) using mesoporous titania (TiO2)-poly vinyl alcohol (PVA) nanocomposite. J. Mol. Liq. 2019, 286, 110908.
- Yun, J.; Jin, D.; Lee, Y.S.; Kim, H. Photocatalytic treatment of acidic waste water by electrospun composite nanofibers of pH-sensitive hydrogel and TiO2. Mater. Lett. 2010, 64, 2431–2434.
- Cheng, H.; Hu, M.; Zhai, Q.; Li, S.; Jiang, Y. Polydopamine tethered CPO/HRPTiO2 nano-composites with high bio-catalytic activity, stability and reusability: Enzyme-photo bifunctional synergistic catalysis in water treatment. Chem. Eng. J. 2018, 347, 703–710.
- Saleh, T.A.; Gupta, V.K. Photo-catalyzed degradation of hazardous dye methyl orange by use of a composite catalyst consisting of multi-walled carbon nanotubes and titanium dioxide. J. Colloid Interface Sci. 2012, 371, 101–106.
- Rajamanickam, D.; Shanthi, M. Photocatalytic degradation of an azo dye Sunset Yellow under UV-A light using TiO2/CAC composite catalysts. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 128, 100–108.
- Hassan, M.E.; Liu, G.; Omer, E.O.M.; Goja, A.M.; Acharya, S. Silver embedded C-TiO2 exhibits improved photocatalytic properties with potential application in waste water treatment. Arab. J. Chem. 2018, 12, 1134–1140.
- Xu, C.; Cui, A.; Xu, Y.; Fu, X. Graphene oxide-TiO2 composite filtration membranes and their potential application for water purification. Carbon 2013, 62, 465–471.
- Ahmed, A.S.; Ahamad, T.; Ahmad, N.; Khan, M.Z. Removal enhancement of acid navy blue dye by GO-TiO2 nanocomposites synthesized using sonication method. Mater. Chem. Phys. 2019, 238, 121906.
- Mohamed, H.H.; Alsanea, A.A. TiO2/carbon dots decorated reduced graphene oxide composites from waste car bumper and TiO2 nanoparticles for photocatalytic applications. Arab. J. Chem. 2018, 13, 3082–3091.
- Ranjith, R.; Renganathan, V.; Chen, S.M.; Selvan, N.S.; Rajam, P.S. Green synthesis of reduced graphene oxide supported TiO2/Co3O4 nanocomposite for photocatalytic degradation of methylene blue and crystal violet. Ceram. Int. 2019, 45, 12926–12933.
- Monga, D.; Basu, S. Enhanced photocatalytic degradation of industrial dye by g-C3N4/TiO2 nanocomposite: Role of shape of TiO2. Adv. Powder Technol. 2019, 30, 1089–1098.
- Zhang, J.J.; Fang, S.S.; Mei, J.Y.; Zheng, G.P.; Zheng, X.C.; Guan, X.X. High-efficiency removal of rhodamine B dye in water using g-C3N4 and TiO2 co-hybridized 3D graphene aerogel composites. Separ. Purif. Technol. 2018, 194, 96–103.
- Zhang, Y.; Cui, W.; An, W.; Liu, L.; Liang, Y.; Zhu, Y. Combination of photoelectrocatalysis and adsorption for removal of bisphenol A over TiO2-graphene hydrogel with 3D network structure. Appl. Catal. B Environ. 2018, 221, 36–46.
- Chi, L.; Qian, Y.; Guo, J.; Wang, X.; Arandiyan, H.; Jiang, Z. Novel g-C3N4/TiO2/PAA/PTFE ultrafiltration membrane enabling enhanced antifouling and exceptional visible-light photocatalytic self-cleaning. Catal. Today 2019, 335, 527–537.
- Daneshvar, N.; Salari, D.; Niaei, A.; Rasoulifard, M.H.; Khataee, A.R. Immobilization of TiO2 nanopowder on glass beads for the photocatalytic decolorization of an azo dye C.I. Direct Red 23. J. Environ. Sci. Health 2005, 40, 1605–1617.
- Cunha, D.L.; Kuznetsov, A.; Achete, C.A.; Machado, A.E.d.H.; Marques, M. Immobilized TiO2 on glass spheres applied to heterogeneous photocatalysis: Photoactivity, leaching and regeneration process. PeerJ 2018, 6, e4464.
- Abbasi, Z.; Farrokhnia, A.; García-López, E.I.; Zargar Shoushtari, M. Codeposition of Fe3O4 nanoparticles sandwiched between g-C3N4 and TiO2 nanosheets: Structure, characterization and high photocatalytic activity for efficiently degradation of dye pollutants. Phys. Chem. Res. 2019, 7, 65–80.
- Gu, W.; Lu, F.; Wang, C.; Kuga, S.; Wu, L.; Huang, Y.; Wu, M. Face-to-Face interfacial assembly of ultrathin g-C3N4 and anatase TiO2 nanosheets for enhanced solar photocatalytic activity. ACS Appl. Mater. Interfaces 2017, 9, 28674–28684.
- Qiu, J.; Liu, F.; Yue, C.; Ling, C.; Li, A. A recyclable nanosheet of Mo/N-doped TiO2 nanorods decorated on carbon nanofibers for organic pollutants degradation under simulated sunlight irradiation. Chemosphere 2019, 215, 280–293.
- Khan, M.A.; Mutahir, S.; Wang, F.; Lei, W.; Xia, M. Sensitization of TiO2 nanosheets with Cu-biphenylamine framework to enhance photocatalytic degradation performance of toxic organic contaminants: Synthesis, mechanism and kinetic studies. Nanotechnology 2018, 29, 375605.
- Nair, A.K.; Jagadeesh Babu, P.E. Ag-TiO2 nanosheet embedded photocatalytic membrane for solar water treatment. J. Environ. Chem. Eng. 2017, 5, 4128–4133.
- Du, F.; Zuo, X.; Yang, Q.; Yang, B.; Li, G.; Ding, Z.; Wu, M.; Ma, Y.; Jin, S.; Zhu, K. Facile assembly of TiO2 nanospheres/SnO2 quantum dots composites with excellent photocatalyst activity for the degradation of methyl orange. Ceram. Int. 2016, 42, 12778–12782.
- Wang, H.; Xiao, L.; Wang, C.; Lin, B.; Lyu, S.; Chu, X.; Chi, Y.; Yang, X.; Wang, X. Pd/TiO2 nanospheres with three-dimensional hyperstructure for enhanced photodegradation of organic dye. Chem. Res. 2019, 35, 667–673.
- Ghaforyan, H.; Ghaffary, T.; Pincak, R.; Ebrahimzadeh, M. Effects of ultrasound waves intensity on the removal of Congo Red color from the textile industry wastewater by Fe3O4@TiO2 core-shell nanospheres. arXiv 2017, arXiv:1708.04683.
- Tetteh, E.K.; Rathilal, S.; Asante-Sackey, D.; Chollom, M.N. Prospects of synthesized magnetic TiO2-based membranes for wastewater treatment: A review. Materials 2021, 14, 3524.
- Cao, J.; Song, X.Z.; Kang, X.; Dai, Z.; Tan, Z. One-pot synthesis of oleic acid modified monodispersed mesoporous TiO2 nanospheres with enhanced visible light photocatalytic performance. Adv. Powder Technol. 2018, 29, 1925–1932.
- Liu, D.; Tian, R.; Wang, J.; Nie, E.; Piao, X.; Li, X.; Sun, Z. Photoelectrocatalytic degradation of methylene blue using F doped TiO2 photoelectrode under visible light irradiation. Chemosphere 2017, 185, 574–581.
- Nurdin, M.; Azis, T.; Maulidiyah, M.; Aladin, A.; Hafid, N.A.; Salim, L.O.A.; Wibowo, D. Photocurrent responses of metanil yellow and remazol red B organic dyes by using TiO2/Ti electrode. IOP Conf. Ser. Mater. Sci. Eng. 2018, 367, 12048.
- Yilmaz, P.; Lacerda, A.M.; Larrosa, I.; Dunn, S. Photoelectrocatalysis of rhodamine B and solar hydrogen production by TiO2 and Pd/TiO2 catalyst systems. Electrochim. Acta 2017, 231, 641–649.
- Likodimos, V. Photonic crystal-assisted visible light activated TiO2 photocatalysis. Appl. Catal. B Environ. 2018, 230, 269–303.
- Lu, Y.; Yu, H.; Chen, S.; Quan, X.; Zhao, H. Integrating plasmonic nanoparticles with TiO2 photonic crystal for enhancement of visible-light-driven photocatalysis. Environ. Sci. Technol. 2012, 46, 1724–1730.
- Liao, G.; Chen, S.; Quan, X.; Chen, H.; Zhang, Y. Photonic crystal coupled TiO2/polymer hybrid for efficient photocatalysis under visible light irradiation. Environ. Sci. Technol. 2010, 44, 3481–3485.
- Wang, Y.; Li, P.; Chen, S.L.; Wang, A.J. Dual-bandgap effect of photonic crystals on TiO2 photocatalytic activity in ultraviolet and visible light regions. Catal. Surv. 2019, 23, 23–32.
- Geng, Z.; Zhang, Y.; Yuan, X.; Huo, M.; Zhao, Y.; Lu, Y.; Qiu, Y. Incorporation of Cu2O nanocrystals into TiO2 photonic crystal for enhanced UV-visible light driven photocatalysis. J. Alloys Compd. 2015, 644, 734–741.
- Chen, H.; Chen, S.; Quan, X.; Zhang, Y. Structuring a TiO2-based photonic crystal photocatalyst with Schottky junction for efficient photocatalysis. Environ. Sci. Technol. 2010, 44, 451–455.
- Li, Y.; Chang, Y.; Liu, F.T.; Zhao, Y.; Wang, J.; Wang, C.W. Photonic crystal structural-induced Cu3SnS4/Ti3+-TiO2 p-n coaxial heterojunction arrays for light-driven H2 production and pollutant degradation. Mater. Des. 2017, 133, 426–434.
- Vela, N.; Pérez-Lucas, G.; Fenoll, J.; Navarro, S. Recent overview on the abatement of pesticide residues in water by photocatalytic treatment using TiO2. In Application of Titanium Dioxide; IntechOpen: London, UK, 2017.
- Liu, Y.; Liu, F.; Pan, X.; Li, J. Protecting the environment and public health from pesticides. Environ. Sci. Technol. 2012, 46, 5658–5659.
- Shuman-Goodier, M.E.; Propper, C.R. A meta-analysis synthesizing the effects of pesticides on swim speed and activity of aquatic vertebrates. Sci. Total Environ. 2016, 565, 758–766.
- Blair, A.; Ritz, B.; Wesseling, C.; Beane Freeman, L. Pesticides and human health. Occup. Environ. Med. 2015, 72, 81–82.
- Alavanja, M.C.R.; Bonner, M.R. Occupational pesticide exposures and cancer risk: A review. J. Toxicol. Environ. Health Part B 2012, 15, 238–263.
- Vanraes, P.; Ghodbane, H.; Davister, D.; Wardenier, N.; Nikiforov, A.; Verheust, Y.P.; Van Hulle, S.W.H.; Hamdaoui, O.; Vandamme, J.; Van Durme, J.; et al. Removal of several pesticides in a falling water film DBD reactor with activated carbon textile: Energy efficiency. Water Res. 2017, 116, 1–12.
- Farner Budarz, J.; Cooper, E.M.; Gardner, C.; Hodzic, E.; Ferguson, P.L.; Gunsch, C.K.; Wiesner, M.R. Chlorpyrifos degradation via photoreactive TiO2 nanoparticles: Assessing the impact of a multi-component degradation scenario. J. Hazard. Mater. 2019, 372, 61–68.
- Amalraj, A.; Thirumalaisamy, S.; Rajeswari, A.; Pius, A. Photocatalytic degradation of quinalphos and profenofos pesticides using UV irradiated TiO2 nanoparticles-a kinetic study. Mater. Focus 2016, 5, 377–384.
- Cruz, M.; Gomez, C.; Duran-Valle, C.J.; Pastrana-Martínez, L.M.; Faria, J.L.; Silva, A.M.T.; Faraldos, M.; Bahamonde, A. Bare TiO2 and graphene oxide TiO2 photocatalysts on the degradation of selected pesticides and influence of the water matrix. Appl. Surf. Sci. 2017, 416, 1013–1021.
- Zhang, Y.; Han, C.; Zhang, G.; Dionysiou, D.D.; Nadagouda, M.N. PEG-assisted synthesis of crystal TiO2 nanowires with high specific surface area for enhanced photocatalytic degradation of atrazine. Chem. Eng. J. 2015, 268, 170–179.
- Ahmari, H.; Heris, S.Z.; Khayyat, M.H. The effect of titanium dioxide nanoparticles and UV irradiation on photocatalytic degradation of Imidaclopride. Environ. Technol. 2018, 39, 536–547.
- Vela, N.; Calín, M.; Yáñez-Gascón, M.J.; Garrido, I.; Pérez-Lucas, G.; Fenoll, J.; Navarro, S. Photocatalytic oxidation of six pesticides listed as endocrine disruptor chemicals from wastewater using two different TiO2 samples at pilot plant scale under sunlight irradiation. J. Photochem. Photobiol. A Chem. 2018, 353, 271–278.
- Kalantary, R.R.; Dadban Shahamat, Y.; Farzadkia, M.; Esrafili, A.; Asgharnia, H. Photocatalytic degradation and mineralization of diazinon in aqueous solution using nano-TiO2 (Degussa, P25): Kinetic and statistical analysis. Desalin. Water Treat. 2015, 55, 555–563.
- Suhaimy, S.H.M.; Lai, C.W.; Tajuddin, H.A.; Samsudin, E.M.; Johan, M.R. Impact of TiO2 nanotubes’ morphology on the photocatalytic degradation of simazine pollutant. Materials 2018, 11, 66.
- Tabasideh, S.; Maleki, A.; Shahmoradi, B.; Ghahremani, E.; McKay, G. Sonophotocatalytic degradation of diazinon in aqueous solution using iron-doped TiO2 nanoparticles. Separ. Purif. Technol. 2017, 189, 186–192.
- Mermana, J.; Sutthivaiyakit, P.; Blaise, C.; Gagné, F.; Charnsethikul, S.; Kidkhunthod, P.; Sutthivaiyakit, S. Photocatalysis of S-metolachlor in aqueous suspension of magnetic cerium-doped mTiO2 core-shell under simulated solar light. Environ. Sci. Pollut. Res. 2017, 24, 4077–4092.
- Umar, K.; Aris, A.; Ahmad, H.; Parveen, T.; Jaafar, J.; Majid, Z.A.; Reddy, A.V.B.; Talib, J. Synthesis of visible light active doped TiO2 for the degradation of organic pollutants-methylene blue and glyphosate. J. Anal. Sci. Technol. 2016, 7, 29.
- Maddila, S.; Oseghe, E.O.; Jonnalagadda, S.B. Photocatalyzed ozonation by Ce doped TiO2 catalyst degradation of pesticide Dicamba in water. J. Chem. Technol. Biotechnol. 2016, 91, 385–393.
- Quiñones, D.H.; Rey, A.; Álvarez, P.M.; Beltrán, F.J.; Li Puma, G. Boron doped TiO2 catalysts for photocatalytic ozonation of aqueous mixtures of common pesticides: Diuron, o-phenylphenol, MCPA and terbuthylazine. Appl. Catal. B Environ. 2014, 178, 74–81.
- Joseph, A.I.J.; Thiripuranthagan, S. Non-metal doped titania photocatalysts for the degradation of neonicotinoid insecticides under visible light irradiation. J. Nanosci. Nanotechnol. 2018, 18, 3158–3164.
- Komtchou, S.; Dirany, A.; Drogui, P.; Delegan, N.; El Khakani, M.A.; Robert, D.; Lafrance, P. Degradation of atrazine in aqueous solution with electrophotocatalytic process using TiO2-x photoanode. Chemosphere 2016, 157, 79–88.
- Turan, N.B.; Sari Erkan, H.; Çaglak, A.; Bakırdere, S.; Engin, G.O. Optimization of atrazine removal from synthetic groundwater by electrooxidation process using titanium dioxide and graphite electrodes. Separ. Sci. Technol. 2019, 55, 3036–3045.
- Matsunaga, T.; Tomoda, R.; Nakajima, T.; Wake, H. Photoelectrochemical sterilization of microbial cells by semiconductor powders. FEMS Microbiol. Lett. 1985, 29, 211–214. Available online: https://www.sciencedirect.com/science/article/pii/0378109785903003 (accessed on 12 January 2024).
- Maness, P.-C.; Smolinski, S.; Blake, D.M.; Huang, Z.; Wolfrum, E.J.; Jacoby, W.A. Bactericidal activity of photocatalytic TiO2 reaction toward an understanding of its killing mechanism. Appl. Environ. Microbiol. 1999, 65, 4094–4098.
- Cai, R.; Hashimoto, K.; Itoh, K.; Kubota, Y.; Fujishima, A. Photokilling of malignant cells with ultrafine TiO2 powder. Bull. Chem. Soc. Jpn. 1991, 64, 1268–1273.
- Karunakaran, C. Solar Photocatalytic Disinfection of Bacteria. In New and Future Developments in Catalysis; Suib, S.L., Ed.; Elsevier B.V.: Amsterdam, The Netherlands, 2013.
- Prasad, G.K.; Ramacharyulu, P.V.R.K.; Merwyn, S.; Agarwal, G.S.; Srivastava, A.R.; Beer Singh, G.P.R.; Vijayaraghavan, R. Photocatalytic inactivation of spores of Bacillus anthracis using titania nanomaterials. J. Hazard. Mater. 2011, 185, 977–982.
- Nasser, A.M.; Paulman, H.; Sela, O.; Ktaitzer, T.; Cikurel, H.; Zuckerman, I.; Meir, A.; Aharoni, A.; Adin, A. UV disinfection of wastewater effluents for unrestricted irrigation. Water Sci. Technol. 2006, 54, 83–88.
- Egli, T.; Köster, W.; Meile, L. Pathogenic microbes in water and food: Changes and challenges. FEMS Microbiol. Rev. 2002, 26, 111–112.
- Laxma Reddy, P.V.; Kavitha, B.; Kumar Reddy, P.A.; Kim, K.H. TiO2-based photocatalytic disinfection of microbes in aqueous media: A review. Environ. Res. 2017, 154, 296–303.
- Mahmood, M.A.; Baruah, S.; Anal, A.K.; Dutta, J. Heterogeneous photocatalysis for removal of microbes from water. Environ. Chem. Lett. 2012, 10, 145–151.
- Leung, T.L.F.; Bates, A.E. More rapid and severe disease outbreaks for aquaculture at the tropics: Implications for food security. J. Appl. Ecol. 2013, 50, 215–222.
- Ashbolt, N.J. Microbial contamination of drinking water and disease outcomes in developing regions. Toxicology 2004, 198, 229–238.
- Joost, U.; Juganson, K.; Visnapuu, M.; Mortimer, M.; Kahru, A.; Nõmmiste, E.; Joost, U.; Kisand, V.; Ivask, A. Photocatalytic antibacterial activity of nano-TiO2 (anatase)-based thin films: Effects on Escherichia coli cells and fatty acids. J. Photochem. Photobiol. B Biol. 2015, 142, 178–185.
- Podporska-Carroll, J.; Panaitescu, E.; Quilty, B.; Wang, L.; Menon, L.; Pillai, S.C. Antimicrobial properties of highly efficient photocatalytic TiO2 nanotubes. Appl. Catal. B Environ. 2015, 176–177, 70–75.
- Long, M.; Wang, J.; Zhuang, H.; Zhang, Y.; Wu, H.; Zhang, J. Performance and mechanism of standard nano-TiO2 (P-25) in photocatalytic disinfection of foodborne microorganisms-Salmonella typhimurium and Listeria monocytogenes. Food Control 2014, 39, 68–74.
- Makropoulou, T.; Panagiotopoulou, P.; Venieri, D. N-doped TiO2 photocatalysts for bacterial inactivation in water. J. Chem. Technol. Biotechnol. 2018, 93, 2518–2526.
- Milosevic, I.; Jayaprakash, A.; Greenwood, B.; van Driel, B.; Rtimi, S.; Bowen, P. Synergistic effect of fluorinated and N doped TiO2 nanoparticles leading to different microstructure and enhanced photocatalytic bacterial inactivation. Nanomaterials 2017, 7, 391.
- Choi, S.Y.; Cho, B. Extermination of influenza virus H1N1 by a new visible-light induced photocatalyst under fluorescent light. Virus Res. 2018, 248, 71–73.
- Zheng, X.; Shen, Z.; Cheng, C.; Shi, L.; Cheng, R.; Yuan, D. Photocatalytic disinfection performance in virus and virus/bacteria system by Cu-TiO2 nanofibers under visible light. Environ. Pollut. 2018, 237, 452–459.
- Rao, G.; Brastad, K.S.; Zhang, Q.; Robinson, R.; He, Z.; Li, Y. Enhanced disinfection of Escherichia coli and bacteriophage MS2 in water using a copper and silver loaded titanium dioxide nanowire membrane. Front. Environ. Sci. Eng. 2016, 10, 11.
- Venieri, D.; Gounaki, I.; Binas, V.; Zachopoulos, A.; Kiriakidis, G.; Mantzavinos, D. Inactivation of MS2 coliphage in sewage by solar photocatalysis using metal-doped TiO2. Appl. Catal. B Environ. 2015, 178, 54–64.
- De Pasquale, I.; Lo Porto, C.; Dell’Edera, M.; Petronella, F.; Agostiano, A.; Curri, M.L.; Comparelli, R. Photocatalytic TiO2-based nanostructured materials for microbial inactivation. Catalysts 2020, 10, 1382.
- Tijani, J.O.; Fatoba, O.O.; Petrik, L.F. A review of pharmaceuticals and endocrinedisrupting compounds: Sources, effects, removal, and detections. Water. Air. Soil Pollut. 2013, 224, 1770.
- Burkhardt-Holm, P. Endocrine disruptors and water quality: A state-of-the-art review. Int. J. Water Resour. Dev. 2010, 26, 477–493.
- Monneret, C. What is an endocrine disruptor? Comptes Rendus Biol. 2017, 340, 403–405.
- Tetreault, G.R.; Bennett, C.J.; Shires, K.; Knight, B.; Servos, M.R.; McMaster, M.E. Intersex and reproductive impairment of wild fish exposed to multiple municipal wastewater discharges. Aquat. Toxicol. 2011, 104, 278–290.
- Tabata, A.; Kashiwada, S.; Ohnishi, Y.; Ishikawa, H.; Miyamoto, N.; Itoh, M.; Magara, Y. Estrogenic influences of estradiol-17 beta, p-nonylphenol and bis-phenol-A on Japanese medaka (Oryzias latipes) at detected environmental concentrations. Water Sci. Technol. 2001, 43, 109–116.
- Villeneuve, S.; Cyr, D.; Lynge, E.; Orsi, L.; Sabroe, S.; Merletti, F.; Gorini, G.; Morales Suarez Varela, M.; Ahrens, W.; Baumgardt-Elms, C.; et al. Occupation and occupational exposure to endocrine disrupting chemicals in male breast cancer: A case-control study in Europe. Occup. Environ. Med. 2010, 67, 837–844.
- Li, D.; Zhou, Z.; Qing, D.; He, Y.; Wu, T.; Miao, M.; Wang, J.; Weng, X.; Ferber, J.R.; Herrinton, L.J.; et al. Occupational exposure to bisphenol-A (BPA) and the risk of self-reported male sexual dysfunction. Hum. Reprod. 2009, 25, 519–527.
- Tong, A.Y.C.; Braund, R.; Warren, D.S.; Peake, B.M. TiO2-assisted photodegradation of pharmaceuticals—A review. Cent. Eur. J. Chem. 2012, 10, 989–1027.
- AlAani, H.; Hashem, S.; Karabet, F. Photocatalytic (UV-A/TiO2) and photolytic (UV-A) degradation of steroid hormones: Ethinyl estradiol, levonorgestrel, and progesterone. Int. J. Chem. Res. 2017, 10, 1061–1070.
- Arlos, M.J.; Liang, R.; Hatat-Fraile, M.M.; Bragg, L.M.; Zhou, N.Y.; Servos, M.R.; Andrews, S.A. Photocatalytic decomposition of selected estrogens and their estrogenic activity by UV-LED irradiated TiO2 immobilized on porous titanium sheets via thermal-chemical oxidation. J. Hazard. Mater. 2016, 318, 541–550.
- Sornalingam, K.; McDonagh, A.; Zhou, J.L.; Johir, M.A.H.; Ahmed, M.B. Photocatalysis of estrone in water and wastewater: Comparison between Au-TiO2 nanocomposite and TiO2, and degradation by-products. Sci. Total Environ. 2018, 610–611, 521–530.
- Zatloukalová, K.; Obalová, L.; Koči, K.; Čapek, L.; Matěj, Z.; Šnajdhaufová, H.; Ryczkowski, J.; Słowik, G. Photocatalytic degradation of endocrine disruptor compounds in water over immobilized TiO2 photocatalysts. Iran. J. Chem. Chem. Eng. IJCCE 2017, 36, 29–38.
- Ramírez-Sánchez, I.M.; Tuberty, S.; Hambourger, M.; Bandala, E.R. Resource efficiency analysis for photocatalytic degradation and mineralization of estriol using TiO2 nanoparticles. Chemosphere 2017, 184, 1270–1285.
- Solcova, O.; Spacilova, L.; Maleterova, Y.; Morozova, M.; Ezechias, M.; Kresinova, Z. Photocatalytic water treatment on TiO2 thin layers. Desalin. Water Treat. 2016, 57, 11631–11638.
- Ruokolainen, M.; Gul, T.; Permentier, H.; Sikanen, T.; Kostiainen, R.; Kotiaho, T. Comparison of TiO2 photocatalysis, electrochemically assisted Fenton reaction and direct electrochemistry for simulation of phase i metabolism reactions of drugs. Eur. J. Pharmaceut. Sci. 2016, 83, 36–44.
- Pazoki, M.; Parsa, M.; Farhadpour, R. Removal of the hormones dexamethasone (DXM) by Ag doped on TiO2 photocatalysis. J. Environ. Chem. Eng. 2016, 4, 4426–4434.
- Chow, K.L.; Man, Y.B.; Tam, N.F.Y.; Liang, Y.; Wong, M.H. Removal of decabromodiphenyl ether (BDE-209) using a combined system involving TiO2 photocatalysis and wetland plants. J. Hazard. Mater. 2017, 322, 263–269.
- Vasapollo, G.; Mele, G.; Del Sole, R.; Pio, I.; Li, J.; Mazzetto, S.E. Use of novel cardanol-porphyrin hybrids and their TiO2-based composites for the photodegradation of 4-Nitrophenol in water. Molecules 2011, 16, 5769–5784.
- Sraw, A.; Kaur, T.; Pandey, Y.; Sobti, A.; Wanchoo, R.K.; Toor, A.P. Fixed bed recirculation type photocatalytic reactor with TiO2 immobilized clay beads for the degradation of pesticide polluted water. J. Environ. Chem. Eng. 2018, 6, 7035–7043.
- Wang, H.; Liang, Y.; Liu, L.; Hu, J.; Cui, W. Highly ordered TiO2 nanotube arrays wrapped with g-C3N4 nanoparticles for efficient charge separation and increased photoelectrocatalytic degradation of phenol. J. Hazard. Mater. 2018, 344, 369–380.
- Martins, A.S.; Nuñez, L.; Lanza, M.R.d.V. Enhanced photoelectrocatalytic performance of TiO2 nanotube array modified with WO3 applied to the degradation of the endocrine disruptor propyl paraben. J. Electroanal. Chem. 2017, 802, 33–39.





