
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Felice Valzano | -- | 1984 | 2024-01-29 13:38:21 | | | |
| 2 | Wendy Huang | Meta information modification | 1984 | 2024-01-30 07:44:25 | | |
Video Upload Options
Multidrug-resistant (MDR) bacteria can spread in the healthcare setting by different ways. The most important are direct contact transmission occurring when an individual comes into physical contact with an infected or colonized patient (which can involve healthcare workers, patients, or visitors) and indirect contact transmission occurring when a person touches contaminated objects or surfaces in the hospital environment. Different sites in restrooms, from toilets and hoppers to drains and siphons, can become contaminated with MDR bacteria that can persist there for long time periods. Therefore, shared toilets may play an important role in the transmission of nosocomial infections since they could represent a reservoir for MDR bacteria. Such pathogens can be further disseminated by bioaerosol and/or droplets potentially produced during toilet use or flushing and be transmitted by inhalation and contact with contaminated fomites.
1. Introduction
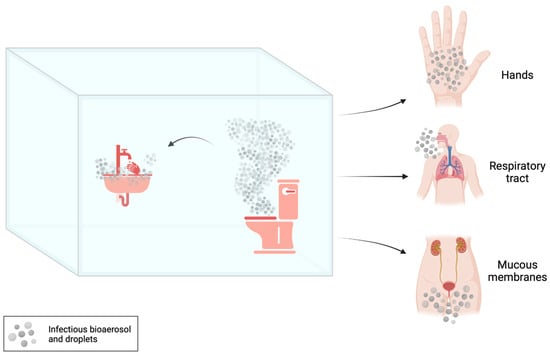
2. Contamination of Toilet Bowl, Seat and Rim
3. Contamination of Toilet Plumbing Systems
4. Contamination of Toilet Flushing-Generated Bioaerosol
References
- World Health Organization (WHO). Antimicrobial Resistance: Global Report on Surveillance, Geneva. 2014. Available online: https://iris.who.int/bitstream/handle/10665/112642/9789241564748_eng.pdf;jsessionid=831AB5113EE962A27DA6BBAD68754A76?sequence=1 (accessed on 10 November 2023).
- Centers for Disease Control and Prevention (CDC). Antibiotic Resistance Threats in the United States. 2019. Available online: https://stacks.cdc.gov/view/cdc/82532 (accessed on 10 November 2023).
- European Centre for Disease Prevention and Control (ECDC). Assessing the Health Burden of Infections with Antibiotic-Resistant Bacteria in the EU/EEA, 2016–2020. 2022. Available online: https://www.ecdc.europa.eu/en/publications-data/health-burden-infections-antibiotic-resistant-bacteria-2016-2020 (accessed on 10 November 2023).
- Balsells, E.; Shi, T.; Leese, C.; Lyell, I.; Burrows, J.; Wiuff, C.; Campbell, H.; Kyaw, M.H.; Nair, H. Global Burden of Clostridium Difficile Infections: A Systematic Review and Meta-Analysis. J. Glob. Health 2019, 9, 010407.
- Leffler, D.A.; Lamont, J.T. Clostridium Difficile Infection. N. Engl. J. Med. 2015, 372, 1539–1548.
- Blanco, N.; O’Hara, L.M.; Harris, A.D. Transmission Pathways of Multidrug-Resistant Organisms in the Hospital Setting: A Scoping Review. Infect. Control Hosp. Epidemiol. 2019, 40, 447–456.
- Lax, S.; Gilbert, J.A. Hospital-Associated Microbiota and Implications for Nosocomial Infections. Trends Mol. Med. 2015, 21, 427–432.
- Centers for Disease Control and Prevention (CDC). Reduce Risk from Water. 2019. Available online: https://www.cdc.gov/hai/prevent/environment/water.html (accessed on 10 November 2023).
- Park, S.C.; Parikh, H.; Vegesana, K.; Stoesser, N.; Barry, K.E.; Kotay, S.M.; Dudley, S.; Peto, T.E.A.; Crook, D.W.; Walker, A.S.; et al. Risk Factors Associated with Carbapenemase-Producing Enterobacterales (CPE) Positivity in the Hospital Wastewater Environment. Appl. Environ. Microbiol. 2020, 86, e01715–e01720.
- Kizny Gordon, A.E.; Mathers, A.J.; Cheong, E.Y.L.; Gottlieb, T.; Kotay, S.; Walker, A.S.; Peto, T.E.A.; Crook, D.W.; Stoesser, N. The Hospital Water Environment as a Reservoir for Carbapenem-Resistant Organisms Causing Hospital-Acquired Infections-A Systematic Review of the Literature. Clin. Infect. Dis. 2017, 64, 1435–1444.
- Hocquet, D.; Muller, A.; Bertrand, X. What Happens in Hospitals Does Not Stay in Hospitals: Antibiotic-Resistant Bacteria in Hospital Wastewater Systems. J. Hosp. Infect. 2016, 93, 395–402.
- Gerba, C.P.; Wallis, C.; Melnick, J.L. Microbiological Hazards of Household Toilets: Droplet Production and the Fate of Residual Organisms. Appl. Microbiol. 1975, 30, 229–237.
- Leone, C.M.; Tang, C.; Sharp, J.; Jiang, X.; Fraser, A. Presence of Human Noroviruses on Bathroom Surfaces: A Review of the Literature. Int. J. Environ. Health Res. 2016, 26, 420–432.
- Grabowski, M.E.; Kang, H.; Wells, K.M.; Sifri, C.D.; Mathers, A.J.; Lobo, J.M. Provider Role in Transmission of Carbapenem-Resistant Enterobacteriaceae. Infect. Control Hosp. Epidemiol. 2017, 38, 1329–1334.
- Gerhardts, A.; Hammer, T.R.; Balluff, C.; Mucha, H.; Hoefer, D. A Model of the Transmission of Micro-Organisms in a Public Setting and Its Correlation to Pathogen Infection Risks. J. Appl. Microbiol. 2012, 112, 614–621.
- Jessen, C.U. Airborne Microorganisms: Occurrance and Control; G.E.C. Gads Forlag: Copenhagen, Denmark, 1955.
- Knowlton, S.D.; Boles, C.L.; Perencevich, E.N.; Diekema, D.J.; Nonnenmann, M.W. CDC Epicenters Program Bioaerosol Concentrations Generated from Toilet Flushing in a Hospital-Based Patient Care Setting. Antimicrob. Resist. Infect. Control 2018, 7, 16.
- Li, Y.-Y.; Wang, J.-X.; Chen, X. Can a Toilet Promote Virus Transmission? From a Fluid Dynamics Perspective. Phys. Fluids 2020, 32, 065107.
- Smismans, A.; Ho, E.; Daniels, D.; Ombelet, S.; Mellaerts, B.; Obbels, D.; Valgaeren, H.; Goovaerts, A.; Huybrechts, E.; Montag, I.; et al. New Environmental Reservoir of CPE in Hospitals. Lancet Infect. Dis. 2019, 19, 580–581.
- Jolivet, S.; Couturier, J.; Vuillemin, X.; Gouot, C.; Nesa, D.; Adam, M.; Brissot, E.; Mohty, M.; Bonnin, R.A.; Dortet, L.; et al. Outbreak of OXA-48-Producing Enterobacterales in a Haematological Ward Associated with an Uncommon Environmental Reservoir, France, 2016 to 2019. Eurosurveillance 2021, 26, 2000118.
- European Centre for Disease Prevention and Control (ECDC). Antimicrobial Resistance Surveillance in Europe 2023: 2021 Data. Available online: https://op.europa.eu/en/publication-detail/-/publication/97ee4ab5-0994-11ee-b12e-01aa75ed71a1/language-en (accessed on 10 November 2023).
- Saliba, R.; Ghelfenstein-Ferreira, T.; Lomont, A.; Pilmis, B.; Carbonnelle, E.; Seytre, D.; Nasser-Ayoub, E.; Zahar, J.-R.; Karam-Sarkis, D. Risk Factors for the Environmental Spread of Different Multidrug-Resistant Organisms: A Prospective Cohort Study. J. Hosp. Infect. 2021, 111, 155–161.
- Noble, M.A.; Isaac-Renton, J.L.; Bryce, E.A.; Roscoe, D.L.; Roberts, F.J.; Walker, M.; Scharf, S.; Walsh, A.; Altamirano-Dimas, M.; Gribble, M. The Toilet as a Transmission Vector of Vancomycin-Resistant Enterococci. J. Hosp. Infect. 1998, 40, 237–241.
- Ellis Mulligan, M.; Rolfe, R.D.; Finegold, S.M.; George, W.L. Contamination of a Hospital Environment by Clostridium Difficile. Curr. Microbiol. 1979, 3, 173–175.
- Reigadas, E.; Vázquez-Cuesta, S.; Villar-Gómara, L.; Onori, R.; Alcalá, L.; Marín, M.; Muñoz, P.; Bouza, E. Role of Clostridioides Difficile in Hospital Environment and Healthcare Workers. Anaerobe 2020, 63, 102204.
- Alcalá, L.; Reigadas, E.; Marín, M.; Martín, A.; Catalán, P.; Bouza, E. Spanish Clostridium difficile Study Group Impact of Clinical Awareness and Diagnostic Tests on the Underdiagnosis of Clostridium Difficile Infection. Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 1515–1525.
- Sib, E.; Voigt, A.M.; Wilbring, G.; Schreiber, C.; Faerber, H.A.; Skutlarek, D.; Parcina, M.; Mahn, R.; Wolf, D.; Brossart, P.; et al. Antibiotic Resistant Bacteria and Resistance Genes in Biofilms in Clinical Wastewater Networks. Int. J. Hyg. Environ. Health 2019, 222, 655–662.
- Hamerlinck, H.; Aerssens, A.; Boelens, J.; Dehaene, A.; McMahon, M.; Messiaen, A.-S.; Vandendriessche, S.; Velghe, A.; Leroux-Roels, I.; Verhasselt, B. Sanitary Installations and Wastewater Plumbing as Reservoir for the Long-Term Circulation and Transmission of Carbapenemase Producing Citrobacter Freundii Clones in a Hospital Setting. Antimicrob. Resist. Infect. Control 2023, 12, 58.
- Herruzo, R.; Ruiz, G.; Vizcaino, M.J.; Rivas, L.; Pérez-Blanco, V.; Sanchez, M. Microbial Competition in Environmental Nosocomial Reservoirs and Diffusion Capacity of OXA48-Klebsiella Pneumoniae: Potential Impact on Patients and Possible Control Methods. J. Prev. Med. Hyg. 2017, 58, E34–E41.
- Heireman, L.; Hamerlinck, H.; Vandendriessche, S.; Boelens, J.; Coorevits, L.; De Brabandere, E.; De Waegemaeker, P.; Verhofstede, S.; Claus, K.; Chlebowicz-Flissikowska, M.A.; et al. Toilet Drain Water as a Potential Source of Hospital Room-to-Room Transmission of Carbapenemase-Producing Klebsiella Pneumoniae. J. Hosp. Infect. 2020, 106, 232–239.
- Buchan, B.W.; Graham, M.B.; Lindmair-Snell, J.; Arvan, J.; Ledeboer, N.A.; Nanchal, R.; Munoz-Price, L.S. The Relevance of Sink Proximity to Toilets on the Detection of Klebsiella Pneumoniae Carbapenemase inside Sink Drains. Am. J. Infect. Control 2019, 47, 98–100.
- Marmor, A.; Daveson, K.; Harley, D.; Coatsworth, N.; Kennedy, K. Two Carbapenemase-Producing Enterobacteriaceae Outbreaks Detected Retrospectively by Whole-Genome Sequencing at an Australian Tertiary Hospital. Infect. Dis. Health 2020, 25, 30–33.
- Breathnach, A.S.; Cubbon, M.D.; Karunaharan, R.N.; Pope, C.F.; Planche, T.D. Multidrug-Resistant Pseudomonas Aeruginosa Outbreaks in Two Hospitals: Association with Contaminated Hospital Waste-Water Systems. J. Hosp. Infect. 2012, 82, 19–24.
- Stjärne Aspelund, A.; Sjöström, K.; Olsson Liljequist, B.; Mörgelin, M.; Melander, E.; Påhlman, L.I. Acetic Acid as a Decontamination Method for Sink Drains in a Nosocomial Outbreak of Metallo-β-Lactamase-Producing Pseudomonas Aeruginosa. J. Hosp. Infect. 2016, 94, 13–20.
- Abney, S.E.; Bright, K.R.; McKinney, J.; Ijaz, M.K.; Gerba, C.P. Toilet Hygiene-Review and Research Needs. J. Appl. Microbiol. 2021, 131, 2705–2714.
- Crimaldi, J.P.; True, A.C.; Linden, K.G.; Hernandez, M.T.; Larson, L.T.; Pauls, A.K. Commercial Toilets Emit Energetic and Rapidly Spreading Aerosol Plumes. Sci. Rep. 2022, 12, 20493.
- Mathers, A.J.; Vegesana, K.; German Mesner, I.; Barry, K.E.; Pannone, A.; Baumann, J.; Crook, D.W.; Stoesser, N.; Kotay, S.; Carroll, J.; et al. Intensive Care Unit Wastewater Interventions to Prevent Transmission of Multispecies Klebsiella Pneumoniae Carbapenemase-Producing Organisms. Clin. Infect. Dis. 2018, 67, 171–178.
- Best, E.L.; Sandoe, J.A.T.; Wilcox, M.H. Potential for Aerosolization of Clostridium Difficile after Flushing Toilets: The Role of Toilet Lids in Reducing Environmental Contamination Risk. J. Hosp. Infect. 2012, 80, 1–5.
- Wilson, G.M.; Jackson, V.B.; Boyken, L.D.; Schweizer, M.L.; Diekema, D.J.; Petersen, C.A.; Breheny, P.J.; Nonnenmann, M.W.; Perencevich, E.N. CDC Prevention Epicenter Program Bioaerosols Generated from Toilet Flushing in Rooms of Patients with Clostridioides Difficile Infection. Infect. Control Hosp. Epidemiol. 2020, 41, 517–521.





