
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Pablo Baldi | -- | 3552 | 2024-01-19 13:51:33 | | | |
| 2 | Lindsay Dong | Meta information modification | 3552 | 2024-01-22 02:05:34 | | |
Video Upload Options
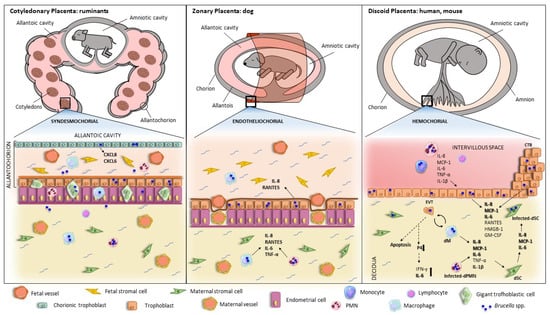
Infection by Brucella species in pregnant animals and humans is associated with an increased risk of abortion, preterm birth, and transmission of the infection to the offspring. The pathogen has a marked tropism for the placenta and the pregnant uterus and has the ability to invade and replicate within cells of the maternal–fetal unit, including trophoblasts and decidual cells. Placentitis is a common finding in infected pregnant animals. Several proinflammatory factors have been found to be increased in both the placenta of Brucella-infected animals and in trophoblasts or decidual cells infected in vitro. As normal pregnancies require an anti-inflammatory placental environment during most of the gestational period, Brucella-induced placentitis is thought to be associated with the obstetric complications of brucellosis.
1. Introduction
2. Epidemiology of Brucella-Induced Pregnancy Complications
| Brucella Species | Hosts | Gestational Manifestations | Vertical Transmission | Contagion Source |
|---|---|---|---|---|
| B. melitensis | Small ruminants | Abortion, weak offspring, reduced milk yields | + | Contaminated placenta or aborted fetus. Milk |
| B. abortus | Bovines | Abortion, weak offspring, reduced milk yields | + | Contaminated placenta or aborted fetus. Milk |
| B. suis (biovars 1, 2, 3) | Swine | Abortion, weak offspring | + | Contaminated placenta or aborted fetus. Milk. Contaminated semen |
| B. canis | Canines | Abortion, weak offspring | + | Contaminated placenta or aborted fetus. Milk. Contaminated semen |
| B. ovis | Sheep | Abortion, weak offspring (rare) | Not reported | Close contact or mating with infected rams. |
| B. melitensis, B. abortus, B. suis | Humans | Abortion, preterm birth, intrauterine fetal death, neonatal or maternal death | + | Contaminated milk and dairy products. Tissues or secretions from infected animals. Contaminated aerosols. |
Although abortion in animals caused by Brucella infection is well known, for several years, there has been controversy about the relationship between human brucellosis and pregnancy outcomes. In pregnant women, the seroprevalence of brucellosis varies from 1.3 to 12.2% [14][15][16][17][18]. Such variability depends on livestock contact, as pregnant women without animal-related occupations show a lower seroprevalence of brucellosis [19] than women from agro-pastoral communities, who do most of the work associated with the care and harvest of livestock products [20]. In the last decades, there have been more reports of adverse outcomes in Brucella-infected pregnant women [15][21]. Spontaneous miscarriage rates range from 18.6 to 73.3% [14][22]. Potential factors affecting the rate of miscarriage include the infecting species (B. melitensis is usually regarded as more virulent), the infection route (food versus other sources), and the median age of the mothers. Interestingly, most of the cases are documented to occur during the first and second trimesters of gestation and differ from the time of abortion occurrence in animals, commonly manifested at later gestational stages [23].
3. Brucella Vaccines and Gestational Complications in Animals
4. Pathological Findings in the Infected Placenta
5. Brucella Infection and Replication in Placental Cells
6. Brucella-Induced Inflammatory Responses in Trophoblasts and Other Cells from the Maternal—Fetal Unit
7. Conclusions
References
- Pappas, G.; Papadimitriou, P.; Akritidis, N.; Christou, L.; Tsianos, E.V. The New Global Map of Human Brucellosis. Lancet Infect. Dis. 2006, 6, 91–99.
- Laine, C.G.; Johnson, V.E.; Scott, H.M.; Arenas-Gamboa, A.M. Global Estimate of Human Brucellosis Incidence. Emerg. Infect. Dis. 2023, 29, 1789–1797.
- Bosilkovski, M.; Arapović, J.; Keramat, F. Human Brucellosis in Pregnancy—An Overview. Bosn. J. Basic Med. Sci. 2020, 20, 415–422.
- Mor, G.; Aldo, P.; Alvero, A.B. The Unique Immunological and Microbial Aspects of Pregnancy. Nat. Rev. Immunol. 2017, 17, 469–482.
- Krishnan, L.; Nguyen, T.; Mccomb, S. From Mice to Women: The Conundrum of Immunity to Infection during Pregnancy. J. Reprod. Immunol. 2013, 97, 62–73.
- Tsai, A.Y.; Byndloss, M.X.; Seyffert, N.; Winter, M.G.; Young, B.M.; Tsolis, R.M. Tumor Necrosis Factor Alpha Contributes to Inflammatory Pathology in the Placenta during Brucella abortus Infection. Infect. Immun. 2022, 90, e00013-22.
- Fernández, A.G.; Ferrero, M.C.; Hielpos, M.S.; Fossati, C.A.; Baldi, P.C. Proinflammatory Response of Human Trophoblastic Cells to Brucella abortus Infection and upon Interactions with Infected Phagocytes. Biol. Reprod. 2016, 94, 131706.
- Ren, J.; Hou, H.; Zhao, W.; Wang, J.; Peng, Q. Administration of Exogenous Progesterone Protects Against Brucella abortus Infection-Induced Inflammation in Pregnant Mice. J. Infect. Dis. 2021, 224, 532–543.
- Rossetti, C.A.; Maurizio, E.; Rossi, U.A. Comparative Review of Brucellosis in Small Domestic Ruminants. Front. Vet. Sci. 2022, 9, 7671.
- Wanke, M.M. Canine Brucellosis. Anim. Reprod. Sci. 2004, 82–83, 195–207.
- Olsen, S.; Tatum, F. Swine Brucellosis: Current Perspectives. Vet. Med. 2016, 8, 1–12.
- Grilló, M.J.; Marín, C.M.; Barberán, M.; Blasco, J.M. Experimental Brucella ovis Infection in Pregnant Ewes. Vet. Rec. 1999, 144, 555–558.
- Ris, D.R. The Bacteriology and Serology of Ewes Inoculated with Viable Brucella ovis Organisms. N. Z. Vet. J. 1970, 18, 2–7.
- Khan, M.Y.; Mah, M.W.; Memish, Z.A. Brucellosis in Pregnant Women. Clin. Infect. Dis. 2001, 32, 1172–1177.
- Elshamy, M.; Ahmed, A.I. The Effects of Maternal Brucellosis on Pregnancy Outcome. J. Infect. Dev. Ctries. 2008, 2, 230–234.
- Kurdoglu, M.; Adali, E.; Kurdoglu, Z.; Karahocagil, M.K.; Kolusari, A.; Yildizhan, R.; Kucukaydin, Z.; Sahin, H.G.; Kamaci, M.; Akdeniz, H. Brucellosis in Pregnancy: A 6-Year Clinical Analysis. Arch. Gynecol. Obstet. 2010, 281, 201–206.
- Makala, R.; Majigo, M.V.; Bwire, G.M.; Kibwana, U.; Mirambo, M.M.; Joachim, A. Seroprevalence of Brucella Infection and Associated Factors among Pregnant Women Receiving Antenatal Care around Human, Wildlife and Livestock Interface in Ngorongoro Ecosystem, Northern Tanzania. A Cross-Sectional Study. BMC Infect. Dis. 2020, 20, 152.
- Ali, S.; Akhter, S.; Neubauer, H.; Scherag, A.; Kesselmeier, M.; Melzer, F.; Khan, I.; El-Adawy, H.; Azam, A.; Qadeer, S.; et al. Brucellosis in Pregnant Women from Pakistan: An Observational Study. BMC Infect. Dis. 2016, 16, 468.
- Kledmanee, K.; Liabsuetrakul, T.; Sretrirutchai, S. Seropositivities against Brucellosis, Coxiellosis, and Toxoplasmosis and Associated Factors in Pregnant Women with Adverse Pregnancy Outcomes: A Cross-Sectional Study. PLoS ONE 2019, 14, e0216652.
- Te-Chaniyom, T.; Geater, A.F.; Kongkaew, W.; Chethanond, U.; Chongsuvivatwong, V. Goat Farm Management and Brucella Serological Test among Goat Keepers and Livestock Officers, 2011–2012, Nakhon Si Thammarat Province, Southern Thailand. One Health 2016, 2, 126–130.
- Vilchez, G.; Espinoza, M.; D’Onadio, G.; Saona, P.; Gotuzzo, E. Brucellosis in Pregnancy: Clinical Aspects and Obstetric Outcomes. Int. J. Infect. Dis. 2015, 38, 95–100.
- Al-Tawfiq, A.J.; Memish, A.Z. Pregnancy Associated Brucellosis. Recent Pat. Anti-Infect. Drug Discov. 2013, 8, 47–50.
- Poester, F.P.; Samartino, L.E.; Santos, R.I. Pathogenesis and Pathobiology of Brucellosis in Livestock. Rev. Sci. Tech. 2013, 32, 105–115.
- Alton, G.G. Control of Brucella melitensis Infection in Sheep and Goats-a Review. Trop. Anim. Health Prod. 1987, 19, 65–74.
- Zundel, E.; Verger, J.M.; Grayon, M.; Michel, R. Conjunctival Vaccination of Pregnant Ewes and Goats with Brucella melitensis Rev 1 Vaccine: Safety and Serological Responses. Ann. Rech. Vet. 1992, 23, 177–188.
- Jiménez de Bagués, M.P.; Marin, C.M.; Barberán, M.; Blasco, J.M. Responses of Ewes to B. Melitensis Rev1 Vaccine Administered by Subcutaneous or Conjunctival Routes at Different Stages of Pregnancy. Ann. Rech. Vet. 1989, 20, 205–213.
- Hensel, M.E.; Garcia-Gonzalez, D.G.; Chaki, S.P.; Hartwig, A.; Gordy, P.W.; Bowen, R.; Ficht, T.A.; Arenas-Gamboa, A.M. Vaccine Candidate Brucella melitensis 16M ΔvjbR Is Safe in a Pregnant Sheep Model and Confers Protection. mSphere 2020, 5, e00120-20.
- Sangari, F.J.; García-Lobo, J.M.; Agüero, J. The Brucella abortus Vaccine Strain B19 Carries a Deletion in the Erythritol Catabolic Genes. FEMS Microbiol. Lett. 1994, 121, 337–342.
- Schurig, G.G.; Roop, R.M.; Bagchi, T.; Boyle, S.; Buhrman, D.; Sriranganathan, N. Biological Properties of RB51; A Stable Rough Strain of Brucella abortus. Vet. Microbiol. 1991, 28, 171–188.
- de Oliveira, M.M.; Pereira, C.R.; de Oliveira, I.R.C.; Godfroid, J.; Lage, A.P.; Dorneles, E.M.S. Efficacy of Brucella abortus S19 and RB51 Vaccine Strains: A Systematic Review and Meta-Analysis. Transbound. Emerg. Dis. 2022, 69, e32–e51.
- Schurig, G.G.; Sriranganathan, N.; Corbel, M.J. Brucellosis Vaccines: Past, Present and Future. Vet. Microbiol. 2002, 90, 479–496.
- Kudi, A.C.; Kalla, D.J.U.; Kudi, M.C.; Kapio, G.I. Brucellosis in Camels. J. Arid Environ. 1997, 37, 413–417.
- Miller, W.G.; Adams, L.G.; Ficht, T.A.; Cheville, N.F.; Payeur, J.P.; Harley, D.R.; House, C.; Ridgway, S.H. Brucella-Induced Abortions and Infection in Bottlenose Dolphins (Tursiops truncatus). J. Zoo Wildl. Med. 1999, 30, 100–110.
- Mackie, J.T.; Blyde, D.; Harris, L.; Roe, W.D.; Keyburn, A.L. Brucellosis Associated with Stillbirth in a Bottlenose Dolphin in Australia. Aust. Vet. J. 2020, 98, 92–95.
- Rebollada-Merino, A.; García-Seco, T.; Pérez-Sancho, M.; Domínguez, L.; Rodríguez-Bertos, A. Histopathologic and Immunohistochemical Findings in the Placentas and Fetuses of Domestic Swine Naturally Infected with Brucella suis Biovar 2. J. Vet. Diagn. Investig. 2023, 35, 258–265.
- Carmichael, L.E.; Kenney, R.M. Canine Abortion Caused by Brucella canis. J. Am. Vet. Med. Assoc. 1968, 152, 605–616.
- Gyuranecz, M.; Szeredi, L.; Rónai, Z.; Dénes, B.; Dencso, L.; Dán, Á.; Pálmai, N.; Hauser, Z.; Lami, E.; Makrai, L.; et al. Detection of Brucella canis-Induced Reproductive Diseases in a Kennel. J. Vet. Diagn. Investig. 2011, 23, 143–147.
- Payne, J.M. Changes in the Rat Placenta and Foetus Following Experimental Infection with Various Species of Bacteria. J. Pathol. Bacteriol. 1958, 75, 367–385.
- Payne, J.M. The Pathogenesis of Experimental Brucellosis in the Pregnant Cow. J. Pathol. Bacteriol. 1959, 78, 447–463.
- Anderson, T.D.; Meador, V.P.; Cheville, N.F. Pathogenesis of Placentitis in the Goat Inoculated with Brucella abortus. I. Gross and Histologic Lesions. Vet. Pathol. 1986, 23, 219–226.
- Siddiqur, R.M.; Kirl, B.B. Clinical and Pathological Findings in Experimental Brucellosis in Pregnant Rats. J. Infect. Dev. Ctries. 2008, 2, 226–229.
- Anderson, T.D.; Cheville, N.F. Ultrastructural Morphometric Analysis of Brucella abortus-Infected Trophoblasts in Experimental Placentitis. Bacterial Replication Occurs in Rough Endoplasmic Reticulum. Am. J. Pathol. 1986, 124, 226.
- Meador, V.P.; Deyoe, B.L. Intracellular Localization of Brucella abortus in Bovine Placenta. Vet. Pathol. 1989, 26, 513–515.
- Carvalho Neta, A.V.; Stynen, A.P.R.; Paixão, T.A.; Miranda, K.L.; Silva, F.L.; Roux, C.M.; Tsolis, R.M.; Everts, R.E.; Lewin, H.A.; Adams, L.G.; et al. Modulation of the Bovine Trophoblastic Innate Immune Response by Brucella abortus. Infect. Immun. 2008, 76, 1897–1907.
- Carvalho Neta, A.V.; Mol, J.P.S.; Xavier, M.N.; Paixão, T.A.; Lage, A.P.; Santos, R.L. Pathogenesis of Bovine Brucellosis. Vet. J. 2010, 184, 146–155.
- Bosseray, N. Brucella Infection and Immunity in Placenta. Ann. l’Institut Pasteur. Microbiol. 1987, 138, 110–113.
- Bosseray, N. Colonization of Mouse Placentas by Brucella abortus Inoculated during Pregnancy. Br. J. Exp. Pathol. 1980, 61, 361–368.
- Poveda-Urkixo, I.; Ramírez, G.A.; Grilló, M.J. Kinetics of Placental Infection by Different Smooth Brucella Strains in Mice. Pathogens 2022, 11, 279.
- Keestra-Gounder, A.M.; Byndloss, M.X.; Seyffert, N.; Young, B.M.; Chávez-Arroyo, A.; Tsai, A.Y.; Cevallos, S.A.; Winter, M.G.; Pham, O.H.; Tiffany, C.R.; et al. NOD1 and NOD2 Signalling Links ER Stress with Inflammation. Nature 2016, 532, 394–397.
- Deng, Y.; Liu, X.; Duan, K.; Peng, Q. Research Progress on Brucellosis. Curr. Med. Chem. 2018, 26, 5598–5608.
- Santos, R.L.; Silva, T.M.A.; Costa, E.A.; Paixo, T.A.; Tsolis, R.M. Laboratory Animal Models for Brucellosis Research. J. Biomed. Biotechnol. 2011, 2011, 518323.
- Robbins, J.R.; Bakardjiev, A.I. Pathogens and the Placental Fortress. Curr. Opin. Microbiol. 2012, 15, 36–43.
- Starr, T.; Ng, T.W.; Wehrly, T.D.; Knodler, L.A.; Celli, J. Brucella Intracellular Replication Requires Trafficking through the Late Endosomal/Lysosomal Compartment. Traffic 2008, 9, 678–694.
- Comerci, D.J.; Martínez-Lorenzo, M.J.; Sieira, R.; Gorvel, J.P.; Ugalde, R.A. Essential Role of the VirB Machinery in the Maturation of the Brucella abortus-Containing Vacuole. Cell. Microbiol. 2001, 3, 159–168.
- Delrue, R.M.; Martinez-Lorenzo, M.; Lestrate, P.; Danese, I.; Bielarz, V.; Mertens, P.; De Bolle, X.; Tibor, A.; Gorvel, J.P.; Letesson, J.J. Identification of Brucella Spp. Genes Involved in Intracellular Trafficking. Cell. Microbiol. 2001, 3, 487–497.
- Salcedo, S.P.; Chevrier, N.; Lacerda, T.L.S.; Ben Amara, A.; Gerart, S.; Gorvel, V.A.; De Chastellier, C.; Blasco, J.M.; Mege, J.L.; Gorvel, J.P. Pathogenic Brucellae Replicate in Human Trophoblasts. J. Infect. Dis. 2013, 207, 1075–1083.
- García-Méndez, K.B.; Hielpos, S.M.; Soler-Llorens, P.F.; Arce-Gorvel, V.; Hale, C.; Gorvel, J.P.; O’Callaghan, D.; Keriel, A. Infection by Brucella melitensis or Brucella papionis Modifies Essential Physiological Functions of Human Trophoblasts. Cell. Microbiol. 2019, 21, 13019.
- Fernández, A.G.; Hielpos, M.S.; Ferrero, M.C.; Fossati, C.A.; Baldi, P.C. Proinflammatory Response of Canine Trophoblasts to Brucella canis Infection. PLoS ONE 2017, 12, e0186561.
- Brennan, S.J.; Ngeleka, M.; Philibert, H.M.; Forbes, L.B.; Allen, A.L. Canine Brucellosis in a Saskatchewan Kennel. Can. Vet. J. 2008, 49, 703–708.
- Liu, X.; Zhou, M.; Wu, J.; Wang, J.; Peng, Q. HMGB1 Release from Trophoblasts Contributes to Inflammation during Brucella melitensis Infection. Cell. Microbiol. 2019, 21, 13080.
- Zhang, J.; Zhang, Y.; Li, Z.; Liu, J.; Shao, X.; Wu, C.; Wang, Y.; Wang, K.; Li, T.; Liu, L.; et al. Outer Membrane Protein 25 of Brucella Activates Mitogen-Activated Protein Kinase Signal Pathway in Human Trophoblast Cells. Front. Vet. Sci. 2017, 4, 197.
- Dunn, C.L.; Kelly, R.W.; Critchley, H.O.D. Decidualization of the Human Endometrial Stromal Cell: An Enigmatic Transformation. Reprod. Biomed. Online 2003, 7, 151–161.
- Vigliani, M.B.; Bakardjiev, A.I. Intracellular Organisms as Placental Invaders. Fetal Matern. Med. Rev. 2014, 25, 332–338.
- Zavattieri, L.; Ferrero, M.C.; Alonso Paiva, I.M.; Sotelo, A.D.; Canellada, A.M.; Baldi, P.C. Brucella Abortus Pro-liferates in Decidualized and Non-Decidualized Human Endometrial Cells Inducing a Proinflammatory Response. Pathogens 2020, 9, 369.





