| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Marcin Dębowski | -- | 5386 | 2024-01-17 08:57:23 | | | |
| 2 | Mona Zou | Meta information modification | 5386 | 2024-01-18 07:55:29 | | |
Video Upload Options
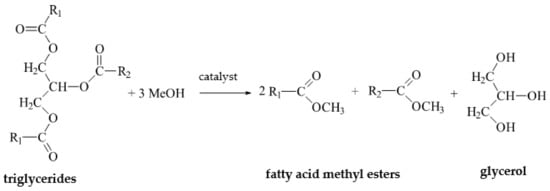
The supply of waste glycerol is rising steadily, partially due to the increased global production of biodiesel. Global biodiesel production totals about 47.1 billion liters and is a process that involves the co-production of waste glycerol, which accounts for over 12% of total esters produced. Waste glycerol is also generated during bioethanol production and is estimated to account for 10% of the total sugar consumed on average. Therefore, there is a real need to seek new technologies for reusing and neutralizing glycerol waste, as well as refining the existing ones. Biotechnological means of valorizing waste glycerol include converting it into gas biofuels via anaerobic fermentation processes. Glycerol-to-bioenergy conversion can be improved through the implementation of new technologies, the use of carefully selected or genetically modified microbial strains, the improvement of their metabolic efficiency, and the synthesis of new enzymes.
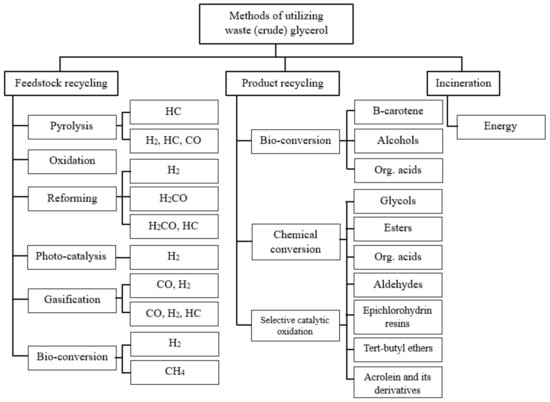
1. Glycerol Production and Applications
2. Biomethane Production
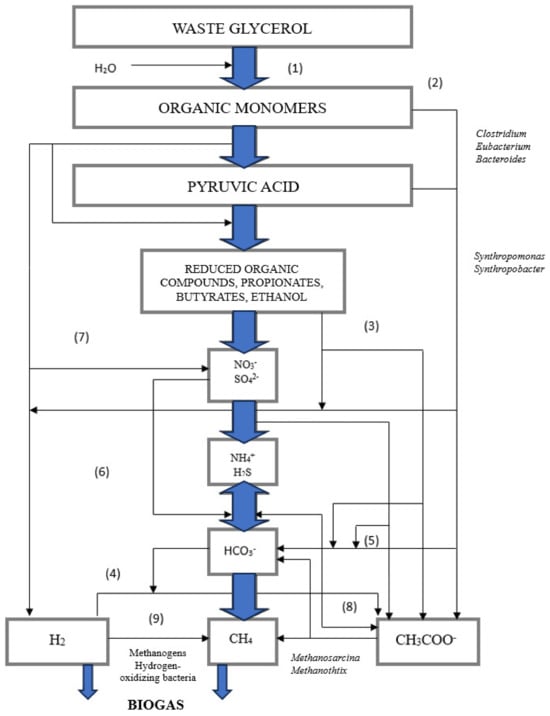
3. Biohydrogen Production
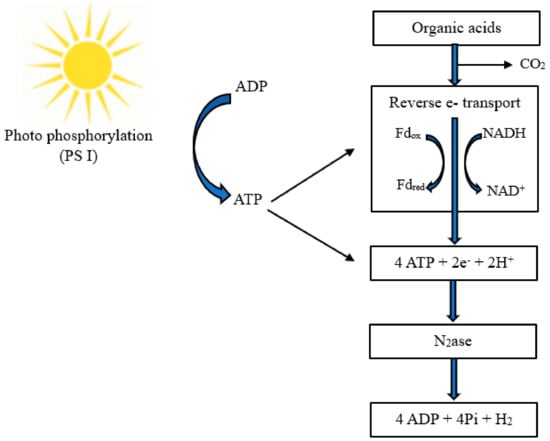
3.1. Photofermentation
3.2. Dark Fermentation
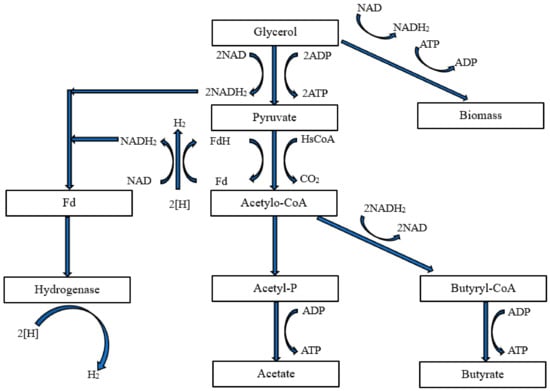
3.3. Mixed Fermentation
4. Biohythane Production
References
- García Martín, J.F.; Torres García, M.; Álvarez Mateos, P. Special Issue on “Biodiesel Production Processes and Technology”. Processes 2022, 11, 25.
- Fasanya, O.O.; Osigbesan, A.A.; Avbenake, O.P. Biodiesel Production from Non-Edible and Waste Lipid Sources. In Biodiesel Technology and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2021; pp. 389–427.
- Nayab, R.; Imran, M.; Ramzan, M.; Tariq, M.; Taj, M.B.; Akhtar, M.N.; Iqbal, H.M.N. Sustainable Biodiesel Production via Catalytic and Non-Catalytic Transesterification of Feedstock Materials—A Review. Fuel 2022, 328, 125254.
- Alagha, S.M.; Salih, R. Review the Studies of Mass Transfer and Kinetic Modeling for Production the Biodiesel by the Transesterification Method and the Impact of Some Selected Factors. IOP Conf. Ser. Earth Environ. Sci. 2023, 1232, 012014.
- Shunaia, A.A.; Jazie, A.A. Applications of Super Critical Technology in Biodiesel Production. IOP Conf. Ser. Earth Environ. Sci. 2023, 1232, 012011.
- Ma, Q.; Fu, K.; Zhang, J.; Li, M.; Han, X.; Chen, Z.; Ma, L.; Chang, C. New Bio-Based Polyurethane (PU) Foams Synthesized Using Crude Glycerol-Based Biopolyol and Humin-Based Byproducts from Biomass Hydrolysis. Ind. Crops Prod. 2023, 205, 117548.
- Tardiolo, G.; Nicolò, M.S.; Drago, C.; Genovese, C.; Fava, G.; Gugliandolo, C.; D’antona, N. Orange Peel Waste as Feedstock for the Production of Glycerol-Free Biodiesel by the Microalgae Nannochloropsis Oculata. Molecules 2023, 28, 6846.
- Sawasdee, V.; Vikromvarasiri, N.; Pisutpaisal, N. Optimization of Ethanol Production from Co-Substrate of Waste Glycerol and Acetic Acid by Enterobacter Aerogenes. Biomass Convers. Biorefinery 2023, 13, 10505–10512.
- Vivek, N.; Sindhu, R.; Madhavan, A.; Anju, A.J.; Castro, E.; Faraco, V.; Pandey, A.; Binod, P. Recent Advances in the Production of Value Added Chemicals and Lipids Utilizing Biodiesel Industry Generated Crude Glycerol as a Substrate—Metabolic Aspects, Challenges and Possibilities: An Overview. Bioresour. Technol. 2017, 239, 507–517.
- Murashchenko, L.; Abbas, C.; Dmytruk, K.; Sibirny, A. Overexpression of the Truncated Version of ILV2 Enhances Glycerol Production in Saccharomyces Cerevisiae. Yeast 2016, 33, 463–469.
- Stoumpou, V.; Novakovic, J.; Kontogianni, N.; Barampouti, E.M.; Mai, S.; Moustakas, K.; Malamis, D.; Loizidou, M. Assessing Straw Digestate as Feedstock for Bioethanol Production. Renew. Energy 2020, 153, 261–269.
- Osman, A.I.; Qasim, U.; Jamil, F.; Al-Muhtaseb, A.H.; Jrai, A.A.; Al-Riyami, M.; Al-Maawali, S.; Al-Haj, L.; Al-Hinai, A.; Al-Abri, M.; et al. Bioethanol and Biodiesel: Bibliometric Mapping, Policies and Future Needs. Renew. Sustain. Energy Rev. 2021, 152, 111677.
- Blomberg, A.; Morrissey, J. Yeast Osmoregulation—Glycerol Still in Pole Position. FEMS Yeast Res. 2022, 22, foac035.
- Tse, T.J.; Wiens, D.J.; Reaney, M.J.T. Production of Bioethanol—A Review of Factors Affecting Ethanol Yield. Fermentation 2021, 7, 268.
- Moshtagh, B.; Hawboldt, K.; Zhang, B. Biosurfactant Production by Native Marine Bacteria (Acinetobacter Calcoaceticus P1-1A) Using Waste Carbon Sources: Impact of Process Conditions. Can. J. Chem. Eng. 2021, 99, 2386–2397.
- Zahid, I.; Ayoub, M.; Abdullah, B.B.; Nazir, M.H.; Ameen, M.; Zulqarnain; Mohd Yusoff, M.H.; Inayat, A.; Danish, M. Production of Fuel Additive Solketal via Catalytic Conversion of Biodiesel-Derived Glycerol. Ind. Eng. Chem. Res. 2020, 59, 20961–20978.
- Asopa, R.P.; Bhoi, R.; Saharan, V.K. Valorization of Glycerol into Value-Added Products: A Comprehensive Review on Biochemical Route. Bioresour. Technol. Rep. 2022, 20, 101290.
- Pirzadi, Z.; Meshkani, F. From Glycerol Production to Its Value-Added Uses: A Critical Review. Fuel 2022, 329, 125044.
- Dimian, A.C.; Kiss, A.A. Eco-Efficient Processes for Biodiesel Production from Waste Lipids. J. Clean. Prod. 2019, 239, 118073.
- Kongjan, P.; Reungsang, A.; Sittijunda, S. Conversion of Glycerol Derived from Biodiesel Production to Butanol and 1,3-Propanediol. In Chemical Substitutes from Agricultural and Industrial By-Products; John Wiley & Sons: Hoboken, NJ, USA, 2023; pp. 337–353.
- Ayude, M.A.; Doumic, L.I.; Cassanello, M.C.; Nigam, K.D.P. Clean Catalytic Oxidation for Derivatization of Key Biobased Platform Chemicals: Ethanol, Glycerol, and Hydroxymethyl Furfural. Ind. Eng. Chem. Res. 2019, 58, 16077–16095.
- Sedghi, R.; Shahbeik, H.; Rastegari, H.; Rafiee, S.; Peng, W.; Nizami, A.S.; Gupta, V.K.; Chen, W.H.; Lam, S.S.; Pan, J.; et al. Turning Biodiesel Glycerol into Oxygenated Fuel Additives and Their Effects on the Behavior of Internal Combustion Engines: A Comprehensive Systematic Review. Renew. Sustain. Energy Rev. 2022, 167, 112805.
- Salgaonkar, N.; Kadamkode, V.; Kumaran, S.; Mallemala, P.; Christy, E.; Appavoo, S.; Majumdar, A.; Mitra, R.; Dasgupta, A. Glycerol Fermentation by Skin Bacteria Generates Lactic Acid and Upregulates the Expression Levels of Genes Associated with the Skin Barrier Function. Exp. Dermatol. 2022, 31, 1364–1372.
- Guerfali, M.; Ayadi, I.; Sassi, H.E.; Belhassen, A.; Gargouri, A.; Belghith, H. Biodiesel-Derived Crude Glycerol as Alternative Feedstock for Single Cell Oil Production by the Oleaginous Yeast Candida Viswanathii Y-E4. Ind. Crops Prod. 2020, 145, 112103.
- Marciniak, P.; Możejko-Ciesielska, J. What Is New in the Field of Industrial Wastes Conversion into Polyhydroxyalkanoates by Bacteria? Polymers 2021, 13, 1731.
- Banaszuk, P.; Kamocki, A.K.; Wysocka-Czubaszek, A.; Czubaszek, R.; Roj-Rojewski, S. Closing the Loop—Recovery of Nutrients and Energy from Wetland Biomass. Ecol. Eng. 2020, 143, 105643.
- Kisielewska, M.; Dębowski, M.; Zieliński, M.; Kazimierowicz, J.; Quattrocelli, P.; Bordiean, A. Effects of Liquid Digestate Treatment on Sustainable Microalgae Biomass Production. Bioenergy Res. 2022, 15, 357–370.
- Teleszewski, T.J.; Z’ukowski, M. Analysis of Heat Loss of a Biogas Anaerobic Digester in Weather Conditions in Poland. J. Ecol. Eng. 2018, 19, 242–250.
- Czubaszek, R.; Wysocka-Czubaszek, A.; Wichtmann, W.; Banaszuk, P. Specific Methane Yield of Wetland Biomass in Dry and Wet Fermentation Technologies. Energies 2021, 14, 8373.
- Czubaszek, R.; Wysocka-Czubaszek, A.; Banaszuk, P. GHG Emissions and Efficiency of Energy Generation through Anaerobic Fermentation of Wetland Biomass. Energies 2020, 13, 6497.
- Teleszewski, T.; Żukowski, M. The Influence of Sludge on Thermal Performance of Heat Exchanger Tubes Inside in an Anaerobic Digester. Annu. Set Environ. Prot. 2018, 20, 763–779.
- Kazimierowicz, J.; Dębowski, M. Aerobic Granular Sludge as a Substrate in Anaerobic Digestion—Current Status and Perspectives. Sustainability 2022, 14, 10904.
- Khalid, S.A.; Elsherif, W.M. Types of Microorganisms for Biodegradation. In Handbook of Biodegradable Materials; Springer: Cham, Switzerland, 2022; pp. 1–27.
- Gnanasekaran, L.; Priya, A.K.; Thanigaivel, S.; Hoang, T.K.A.; Soto-Moscoso, M. The Conversion of Biomass to Fuels via Cutting-Edge Technologies: Explorations from Natural Utilization Systems. Fuel 2023, 331, 125668.
- Detman, A.; Bucha, M.; Treu, L.; Chojnacka, A.; Pleśniak, Ł.; Salamon, A.; Łupikasza, E.; Gromadka, R.; Gawor, J.; Gromadka, A.; et al. Evaluation of Acidogenesis Products’ Effect on Biogas Production Performed with Metagenomics and Isotopic Approaches. Biotechnol. Biofuels 2021, 14, 125.
- Cardoso, W.; di Felice, R.; Baptista, R.C. A Critical Overview of Development and Innovations in Biogas Upgrading. Smart Innov. Syst. Technol. 2022, 295 SIST, 42–50.
- Vítězová, M.; Kohoutová, A.; Vítěz, T.; Hanišáková, N.; Kushkevych, I. Methanogenic Microorganisms in Industrial Wastewater Anaerobic Treatment. Processes 2020, 8, 1546.
- Ahmad, I. Implementation and Optimizing Methane Content in Biogas for the Production of Electricity. Int. J. Eng. Res. Technol. 2015, 4, 1326–1329.
- Santibáñez, C.; Varnero, M.T.; Bustamante, M. Glicerol Residual de La Producción de Biodiesel, Residuo o Potencial Fuente de Energía: Una Revisión. Chil. J. Agric. Res. 2011, 71, 469–475.
- Battista, F.; Strazzera, G.; Valentino, F.; Gottardo, M.; Villano, M.; Matos, M.; Silva, F.M.; Reis, M.A.; Mata-Alvarez, J.; Astals, S.; et al. New Insights in Food Waste, Sewage Sludge and Green Waste Anaerobic Fermentation for Short-Chain Volatile Fatty Acids Production: A Review. J. Environ. Chem. Eng. 2022, 10, 108319.
- Adames, L.V.; Jacobus, A.P.; Sakamoto, I.K.; Lazaro, C.Z.; Pires, L.O.; Maintinguer, S.I. Bioenergy Recovery from Anaerobic Co-Digestion of Crude Glycerol and Domestic Sewage In-Series Reactor: Microbial Characterization and System Performance. Bioenergy Res. 2022, 15, 2145–2158.
- Ferreira, C.M.; Akisue, R.A.; de Sousa Júnior, R. Mathematical Modeling and Computational Simulation Applied to the Study of Glycerol and/or Molasses Anaerobic Co-Digestion Processes. Processes 2023, 11, 2121.
- Adames, L.V.; Pires, L.O.; Maintinguer, S.I. Continuous Long-Term Anaerobic Co-Digestion of Crude Glycerol and Domestic Sewage: Plug-Flow In-Series Reactor Performance and Microbiota Acclimatization. Bioenergy Res. 2023, 16, 1876–1888.
- Bułkowska, K.; Mikucka, W.; Pokój, T. Enhancement of Biogas Production from Cattle Manure Using Glycerine Phase as a Co-Substrate in Anaerobic Digestion. Fuel 2022, 317, 123456.
- Dębowski, M.; Zieliński, M.; Kazimierowicz, J.; Kujawska, N.; Talbierz, S. Microalgae Cultivation Technologies as an Opportunity for Bioenergetic System Development—Advantages and Limitations. Sustainability 2020, 12, 9980.
- Catenacci, A.; Boniardi, G.; Mainardis, M.; Gievers, F.; Farru, G.; Asunis, F.; Malpei, F.; Goi, D.; Cappai, G.; Canziani, R. Processes, Applications and Legislative Framework for Carbonized Anaerobic Digestate: Opportunities and Bottlenecks. A Critical Review. Energy Convers. Manag. 2022, 263, 115691.
- Fan, Q. Methane Potential of Municipal Sludge in Anaerobic Co- Digestion Process Boosted with Glycerol. Ph.D. Thesis, Savonia-ammattikorkeakoulu, Kuopio, Finland, 2018.
- Sittijunda, S.; Reungsang, A. Methane Production from the Co-Digestion of Algal Biomass with Crude Glycerol by Anaerobic Mixed Cultures. Waste Biomass Valorization 2020, 11, 1873–1881.
- Dos Santos Ferreira, J.; Volschan, I.; Cammarota, M.C. Co-Digestion of Sewage Sludge with Crude or Pretreated Glycerol to Increase Biogas Production. Environ. Sci. Pollut. Res. 2018, 25, 21811–21821.
- Zahedi, S.; Rivero, M.; Solera, R.; Perez, M. Mesophilic Anaerobic Co-Digestion of Sewage Sludge with Glycerine: Effect of Solids Retention Time. Fuel 2018, 215, 285–289.
- He, Q.; McNutt, J.; Yang, J. Utilization of the Residual Glycerol from Biodiesel Production for Renewable Energy Generation. Renew. Sustain. Energy Rev. 2017, 71, 63–76.
- Nartker, S.; Ammerman, M.; Aurandt, J.; Stogsdil, M.; Hayden, O.; Antle, C. Increasing Biogas Production from Sewage Sludge Anaerobic Co-Digestion Process by Adding Crude Glycerol from Biodiesel Industry. Waste Manag. 2014, 34, 2567–2571.
- Baba, Y.; Tada, C.; Watanabe, R.; Fukuda, Y.; Chida, N.; Nakai, Y. Anaerobic Digestion of Crude Glycerol from Biodiesel Manufacturing Using a Large-Scale Pilot Plant: Methane Production and Application of Digested Sludge as Fertilizer. Bioresour. Technol. 2013, 140, 342–348.
- Athanasoulia, E.; Melidis, P.; Aivasidis, A. Co-Digestion of Sewage Sludge and Crude Glycerol from Biodiesel Production. Renew. Energy 2014, 62, 73–78.
- Nghiem, L.D.; Nguyen, T.T.; Manassa, P.; Fitzgerald, S.K.; Dawson, M.; Vierboom, S. Co-Digestion of Sewage Sludge and Crude Glycerol for on-Demand Biogas Production. Int. Biodeterior. Biodegrad. 2014, 95, 160–166.
- Silvestre, G.; Fernández, B.; Bonmatí, A. Addition of Crude Glycerine as Strategy to Balance the C/N Ratio on Sewage Sludge Thermophilic and Mesophilic Anaerobic Co-Digestion. Bioresour. Technol. 2015, 193, 377–385.
- Jensen, P.D.; Astals, S.; Lu, Y.; Devadas, M.; Batstone, D.J. Anaerobic Codigestion of Sewage Sludge and Glycerol, Focusing on Process Kinetics, Microbial Dynamics and Sludge Dewaterability. Water Res. 2014, 67, 355–366.
- Alves, I.R.F.S.; Mahler, C.F.; Oliveira, L.B.; Reis, M.M.; Bassin, J.P. Assessing the Use of Crude Glycerol from Biodiesel Production as an Alternative to Boost Methane Generation by Anaerobic Co-Digestion of Sewage Sludge. Biomass Bioenergy 2020, 143, 105831.
- Alves, I.R.F.S.; Mahler, C.F.; Oliveira, L.B.; Reis, M.M.; Bassin, J.P. Investigating the Effect of Crude Glycerol from Biodiesel Industry on the Anaerobic Co-Digestion of Sewage Sludge and Food Waste in Ternary Mixtures. Energy 2022, 241, 122818.
- Siles, J.A.; Martín, M.A.; Chica, A.F.; Martín, A. Anaerobic Co-Digestion of Glycerol and Wastewater Derived from Biodiesel Manufacturing. Bioresour. Technol. 2010, 101, 6315–6321.
- Fountoulakis, M.S.; Manios, T. Enhanced Methane and Hydrogen Production from Municipal Solid Waste and Agro-Industrial by-Products Co-Digested with Crude Glycerol. Bioresour. Technol. 2009, 100, 3043–3047.
- Farias, C.B.B.; Barreiros, R.C.S.; da Silva, M.F.; Casazza, A.A.; Converti, A.; Sarubbo, L.A. Use of Hydrogen as Fuel: A Trend of the 21st Century. Energies 2022, 15, 311.
- Kazimierowicz, J.; Dębowski, M.; Zieliński, M. Effectiveness of Hydrogen Production by Bacteroides Vulgatus in Psychrophilic Fermentation of Cattle Slurry. Clean Technol. 2022, 4, 806–814.
- Dębowski, M.; Dudek, M.; Zieliński, M.; Nowicka, A.; Kazimierowicz, J. Microalgal Hydrogen Production in Relation to Other Biomass-Based Technologies—A Review. Energies 2021, 14, 6025.
- Meier, K.; Kurtz, C.; Weckerle, C.; Hubner, M.; Bürger, I. Air-Conditioning System for Vehicles with on-Board Hydrogen. Appl. Therm. Eng. 2018, 129, 1150–1159.
- Capurso, T.; Stefanizzi, M.; Torresi, M.; Camporeale, S.M. Perspective of the Role of Hydrogen in the 21st Century Energy Transition. Energy Convers. Manag. 2022, 251, 114898.
- Tashie-Lewis, B.C.; Nnabuife, S.G. Hydrogen Production, Distribution, Storage and Power Conversion in a Hydrogen Economy—A Technology Review. Chem. Eng. J. Adv. 2021, 8, 100172.
- Younas, M.; Shafique, S.; Hafeez, A.; Javed, F.; Rehman, F. An Overview of Hydrogen Production: Current Status, Potential, and Challenges. Fuel 2022, 316, 123317.
- Ahmed, S.F.; Mofijur, M.; Nuzhat, S.; Rafa, N.; Musharrat, A.; Lam, S.S.; Boretti, A. Sustainable Hydrogen Production: Technological Advancements and Economic Analysis. Int. J. Hydrogen Energy 2022, 47, 37227–37255.
- Pareek, A.; Dom, R.; Gupta, J.; Chandran, J.; Adepu, V.; Borse, P.H. Insights into Renewable Hydrogen Energy: Recent Advances and Prospects. Mater. Sci. Energy Technol. 2020, 3, 319–327.
- Bhatia, S.K.; Jagtap, S.S.; Bedekar, A.A.; Bhatia, R.K.; Rajendran, K.; Pugazhendhi, A.; Rao, C.V.; Atabani, A.E.; Kumar, G.; Yang, Y.H. Renewable Biohydrogen Production from Lignocellulosic Biomass Using Fermentation and Integration of Systems with Other Energy Generation Technologies. Sci. Total Environ. 2021, 765, 144429.
- Martinez-Burgos, W.J.; de Souza Candeo, E.; Pedroni Medeiros, A.B.; Cesar de Carvalho, J.; Oliveira de Andrade Tanobe, V.; Soccol, C.R.; Sydney, E.B. Hydrogen: Current Advances and Patented Technologies of Its Renewable Production. J. Clean. Prod. 2021, 286, 124970.
- Akroum-Amrouche, D.; Akroum, H.; Lounici, H. Green Hydrogen Production by Rhodobacter Sphaeroides. Energy Sources Part A Recover. Util. Environ. Eff. 2023, 45, 2862–2880.
- Sağır, E.; Hallenbeck, P.C. Photofermentative Hydrogen Production. In Biomass, Biofuels and Biochemical: Biohydrogen, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 141–157.
- Ghiasian, M. Biophotolysis-Based Hydrogen Production by Cyanobacteria. Prospect. Renew. Bioprocess. Future Energy Syst. 2019, 10, 161–184.
- Budiman, P.M.; Wu, T.Y. Role of Chemicals Addition in Affecting Biohydrogen Production through Photofermentation. Energy Convers. Manag. 2018, 165, 509–527.
- Akhlaghi, N.; Najafpour-Darzi, G. A Comprehensive Review on Biological Hydrogen Production. Int. J. Hydrogen Energy 2020, 45, 22492–22512.
- Du Toit, J.P.; Pott, R.W.M. Heat-Acclimatised Strains of Rhodopseudomonas palustris Reveal Higher Temperature Optima with Concomitantly Enhanced Biohydrogen Production Rates. Int. J. Hydrogen Energy 2021, 46, 11564–11572.
- Pott, R.W.M.; Howe, C.J.; Dennis, J.S. Photofermentation of Crude Glycerol from Biodiesel Using Rhodopseudomonas palustris: Comparison with Organic Acids and the Identification of Inhibitory Compounds. Bioresour. Technol. 2013, 130, 725–730.
- Sabourin-Provost, G.; Hallenbeck, P.C. High Yield Conversion of a Crude Glycerol Fraction from Biodiesel Production to Hydrogen by Photofermentation. Bioresour. Technol. 2009, 100, 3513–3517.
- Ghosh, D.; Sobro, I.F.; Hallenbeck, P.C. Stoichiometric Conversion of Biodiesel Derived Crude Glycerol to Hydrogen: Response Surface Methodology Study of the Effects of Light Intensity and Crude Glycerol and Glutamate Concentration. Bioresour. Technol. 2012, 106, 154–160.
- Zhang, D.; Xiao, N.; Mahbubani, K.T.; del Rio-Chanona, E.A.; Slater, N.K.H.; Vassiliadis, V.S. Bioprocess Modelling of Biohydrogen Production by Rhodopseudomonas palustris: Model Development and Effects of Operating Conditions on Hydrogen Yield and Glycerol Conversion Efficiency. Chem. Eng. Sci. 2015, 130, 68–78.
- Aziz, M.; Darmawan, A.; Juangsa, F.B. Hydrogen Production from Biomasses and Wastes: A Technological Review. Int. J. Hydrogen Energy 2021, 46, 33756–33781.
- Rodrigues, C.V.; Nespeca, M.G.; Sakamoto, I.K.; de Oliveira, J.E.; Amâncio Varesche, M.B.; Maintinguer, S.I. Bioconversion of Crude Glycerol from Waste Cooking Oils into Hydrogen by Sub-Tropical Mixed and Pure Cultures. Int. J. Hydrogen Energy 2019, 44, 144–154.
- Baeyens, J.; Zhang, H.; Nie, J.; Appels, L.; Dewil, R.; Ansart, R.; Deng, Y. Reviewing the Potential of Bio-Hydrogen Production by Fermentation. Renew. Sustain. Energy Rev. 2020, 131, 110023.
- Bernal, M.; Tinoco, L.K.; Torres, L.; Malagón-Romero, D.; Montoya, D. Evaluating Colombian Clostridium Spp. Strains’ Hydrogen Production Using Glycerol as Substrate. Electron. J. Biotechnol. 2013, 16, 6.
- Zahedi, S.; Solera, R.; García-Morales, J.L.; Sales, D. Effect of the Addition of Glycerol on Hydrogen Production from Industrial Municipal Solid Waste. Fuel 2016, 180, 343–347.
- Trchounian, K.; Trchounian, A. Hydrogen Production from Glycerol by Escherichia Coli and Other Bacteria: An Overview and Perspectives. Appl. Energy 2015, 156, 174–184.
- Hu, H.; Wood, T.K. An Evolved Escherichia Coli Strain for Producing Hydrogen and Ethanol from Glycerol. Biochem. Biophys. Res. Commun. 2010, 391, 1033–1038.
- Sanchez-Torres, V.; Mohd Yusoff, M.Z.; Nakano, C.; Maeda, T.; Ogawa, H.I.; Wood, T.K. Influence of Escherichia Coli Hydrogenases on Hydrogen Fermentation from Glycerol. Int. J. Hydrogen Energy 2013, 38, 3905–3912.
- Tran, K.T.; Maeda, T.; Sanchez-Torres, V.; Wood, T.K. Beneficial Knockouts in Escherichia Coli for Producing Hydrogen from Glycerol. Appl. Microbiol. Biotechnol. 2015, 99, 2573–2581.
- Trchounian, A. Mechanisms for Hydrogen Production by Different Bacteria during Mixed-Acid and Photo-Fermentation and Perspectives of Hydrogen Production Biotechnology. Crit. Rev. Biotechnol. 2015, 35, 103–113.
- Lo, Y.C.; Chen, X.J.; Huang, C.Y.; Yuan, Y.J.; Chang, J.S. Dark Fermentative Hydrogen Production with Crude Glycerol from Biodiesel Industry Using Indigenous Hydrogen-Producing Bacteria. Int. J. Hydrogen Energy 2013, 38, 15815–15822.
- Kumar, P.; Sharma, R.; Ray, S.; Mehariya, S.; Patel, S.K.S.; Lee, J.K.; Kalia, V.C. Dark Fermentative Bioconversion of Glycerol to Hydrogen by Bacillus Thuringiensis. Bioresour. Technol. 2015, 182, 383–388.
- Ito, T.; Nakashimada, Y.; Senba, K.; Matsui, T.; Nishio, N. Hydrogen and Ethanol Production from Glycerol-Containing Wastes Discharged after Biodiesel Manufacturing Process. J. Biosci. Bioeng. 2005, 100, 260–265.
- Trchounian, K.; Abrahamyan, V.; Poladyan, A.; Trchounian, A. Escherichia Coli Growth and Hydrogen Production in Batch Culture upon Formate Alone and with Glycerol Co-Fermentation at Different PHs. Int. J. Hydrogen Energy 2015, 40, 9935–9941.
- Trchounian, K.; Poladyan, A.; Trchounian, A. Enhancement of Escherichia Coli Bacterial Biomass and Hydrogen Production by Some Heavy Metal Ions and Their Mixtures during Glycerol vs. Glucose Fermentation at a Relatively Wide Range of PH. Int. J. Hydrogen Energy 2017, 42, 6590–6597.
- Pachapur, V.L.; Sarma, S.J.; Brar, S.K.; Le Bihan, Y.; Buelna, G.; Verma, M. Surfactant Mediated Enhanced Glycerol Uptake and Hydrogen Production from Biodiesel Waste Using Co-Culture of Enterobacter Aerogenes and Clostridium Butyricum. Renew. Energy 2016, 95, 542–551.
- De Faber, M.O.; Ferreira-Leitão, V.S. Optimization of Biohydrogen Yield Produced by Bacterial Consortia Using Residual Glycerin from Biodiesel Production. Bioresour. Technol. 2016, 219, 365–370.
- Sharma, Y.; Parnas, R.; Li, B. Bioenergy Production from Glycerol in Hydrogen Producing Bioreactors (HPBs) and Microbial Fuel Cells (MFCs). Int. J. Hydrogen Energy 2011, 36, 3853–3861.
- Chookaew, T.; Prasertsan, P.; Ren, Z.J. Two-Stage Conversion of Crude Glycerol to Energy Using Dark Fermentation Linked with Microbial Fuel Cell or Microbial Electrolysis Cell. New Biotechnol. 2014, 31, 179–184.
- Khalil, M.E.; Jain, A.; Das, E.; Yang, K.L.; Rajagopalan, G. A Robust and Efficient Bioprocess of Hydrogen Production from Crude Glycerol by Clostridium Beijerinckii G117. Int. J. Hydrogen Energy 2023, 48, 7604–7620.
- Sinharoy, A.; Kumar, M.; Pakshirajan, K. An Overview of Bioreactor Configurations and Operational Strategies for Dark Fermentative Biohydrogen Production. In Bioreactors: Sustainable Design and Industrial Applications in Mitigation of GHG Emissions; Academic Press: Cambridge, MA, USA, 2020; pp. 249–288.
- Toledo-Alarcón, J.; Cabrol, L.; Jeison, D.; Trably, E.; Steyer, J.P.; Tapia-Venegas, E. Impact of the Microbial Inoculum Source on Pre-Treatment Efficiency for Fermentative H2 Production from Glycerol. Int. J. Hydrogen Energy 2020, 45, 1597–1607.
- Sivaramakrishnan, R.; Shanmugam, S.; Sekar, M.; Mathimani, T.; Incharoensakdi, A.; Kim, S.H.; Parthiban, A.; Edwin Geo, V.; Brindhadevi, K.; Pugazhendhi, A. Insights on Biological Hydrogen Production Routes and Potential Microorganisms for High Hydrogen Yield. Fuel 2021, 291, 120136.
- Niño-Navarro, C.; Chairez, I.; Christen, P.; Canul-Chan, M.; García-Peña, E.I. Enhanced Hydrogen Production by a Sequential Dark and Photo Fermentation Process: Effects of Initial Feedstock Composition, Dilution and Microbial Population. Renew. Energy 2020, 147, 924–936.
- Mishra, P.; Krishnan, S.; Rana, S.; Singh, L.; Sakinah, M.; Ab Wahid, Z. Outlook of Fermentative Hydrogen Production Techniques: An Overview of Dark, Photo and Integrated Dark-Photo Fermentative Approach to Biomass. Energy Strateg. Rev. 2019, 24, 27–37.
- Manish, S.; Banerjee, R. Comparison of Biohydrogen Production Processes. Int. J. Hydrogen Energy 2008, 33, 279–286.
- Szewczyk, K.W. Biological Production of Hydrogen. Prog. Microbiol. 2008, 47, 241–247.
- Chookaew, T.; O-Thong, S.; Prasertsan, P. Biohydrogen Production from Crude Glycerol by Immobilized Klebsiella Sp. TR17 in a UASB Reactor and Bacterial Quantification under Non-Sterile Conditions. Int. J. Hydrogen Energy 2014, 39, 9580–9587.
- Chookaew, T.; O-Thong, S.; Prasertsan, P. Biohydrogen Production from Crude Glycerol by Two Stage of Dark and Photo Fermentation. Int. J. Hydrogen Energy 2015, 40, 7433–7438.
- Rodrigues, C.V.; Rios Alcaraz, F.A.; Nespeca, M.G.; Rodrigues, A.V.; Motteran, F.; Tallarico Adorno, M.A.; Varesche, M.B.A.; Maintinguer, S.I. Biohydrogen Production in an Integrated Biosystem Using Crude Glycerol from Waste Cooking Oils. Renew. Energy 2020, 162, 701–711.
- Kazimierowicz, J.; Dębowski, M.; Zieliński, M. Biohythane Production in Hydrogen-Oriented Dark Fermentation of Aerobic Granular Sludge (AGS) Pretreated with Solidified Carbon Dioxide (SCO2). Int. J. Mol. Sci. 2023, 24, 4442.
- Roy, S.; Das, D. Biohythane Production from Organic Wastes: Present State of Art. Environ. Sci. Pollut. Res. 2016, 23, 9391–9410.
- Liu, X.; Li, R.; Ji, M.; Han, L. Hydrogen and Methane Production by Co-Digestion of Waste Activated Sludge and Food Waste in the Two-Stage Fermentation Process: Substrate Conversion and Energy Yield. Bioresour. Technol. 2013, 146, 317–323.
- Fu, S.F.; Xu, X.H.; Dai, M.; Yuan, X.Z.; Guo, R.B. Hydrogen and Methane Production from Vinasse Using Two-Stage Anaerobic Digestion. Process Saf. Environ. Prot. 2017, 107, 81–86.
- Nkemka, V.N.; Gilroyed, B.; Yanke, J.; Gruninger, R.; Vedres, D.; McAllister, T.; Hao, X. Bioaugmentation with an Anaerobic Fungus in a Two-Stage Process for Biohydrogen and Biogas Production Using Corn Silage and Cattail. Bioresour. Technol. 2015, 185, 79–88.
- Cheng, J.; Liu, Y.; Lin, R.; Xia, A.; Zhou, J.; Cen, K. Cogeneration of Hydrogen and Methane from the Pretreated Biomass of Algae Bloom in Taihu Lake. Int. J. Hydrogen Energy 2014, 39, 18793–18802.
- Wang, X.; Zhao, Y.-c. A Bench Scale Study of Fermentative Hydrogen and Methane Production from Food Waste in Integrated Two-Stage Process. Int. J. Hydrogen Energy 2009, 34, 245–254.
- Lateef, S.A.; Beneragama, N.; Yamashiro, T.; Iwasaki, M.; Umetsu, K. Batch Anaerobic Co-Digestion of Cow Manure and Waste Milk in Two-Stage Process for Hydrogen and Methane Productions. Bioprocess Biosyst. Eng. 2014, 37, 355–363.
- Chu, C.F.; Li, Y.Y.; Xu, K.Q.; Ebie, Y.; Inamori, Y.; Kong, H.N. A PH- and Temperature-Phased Two-Stage Process for Hydrogen and Methane Production from Food Waste. Int. J. Hydrogen Energy 2008, 33, 4739–4746.
- Luo, G.; Xie, L.; Zou, Z.; Wang, W.; Zhou, Q.; Shim, H. Anaerobic Treatment of Cassava Stillage for Hydrogen and Methane Production in Continuously Stirred Tank Reactor (CSTR) under High Organic Loading Rate (OLR). Int. J. Hydrogen Energy 2010, 35, 11733–11737.
- Buitrón, G.; Kumar, G.; Martinez-Arce, A.; Moreno, G. Hydrogen and Methane Production via a Two-Stage Processes (H2-SBR + CH4-UASB) Using Tequila Vinasses. Int. J. Hydrogen Energy 2014, 39, 19249–19255.
- Kongjan, P.; Jariyaboon, R.; O-Thong, S. Anaerobic Digestion of Skim Latex Serum (SLS) for Hydrogen and Methane Production Using a Two-Stage Process in a Series of up-Flow Anaerobic Sludge Blanket (UASB) Reactor. Int. J. Hydrogen Energy 2014, 39, 19343–19348.
- Rivero, M.; Solera, R.; Perez, M. Anaerobic Mesophilic Co-Digestion of Sewage Sludge with Glycerol: Enhanced Biohydrogen Production. Int. J. Hydrogen Energy 2014, 39, 2481–2488.
- Jehlee, A.; Rodjaroen, S.; Waewsak, J.; Reungsang, A.; O-Thong, S. Improvement of Biohythane Production from Chlorella Sp. TISTR 8411 Biomass by Co-Digestion with Organic Wastes in a Two-Stage Fermentation. Int. J. Hydrogen Energy 2019, 44, 17238–17247.
- Dounavis, A.; Νtaikou, Ι.; Lyberatos, G. Production of Advanced Biobased Hydrogen Enriched Methane from Waste Glycerol in a Two-Stage Continuous System. Waste Biomass Valorization 2016, 7, 677–689.