
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ralf Weiskirchen | -- | 1684 | 2024-01-03 06:56:41 | | | |
| 2 | Catherine Yang | Meta information modification | 1684 | 2024-01-03 07:24:22 | | | | |
| 3 | Catherine Yang | Meta information modification | 1684 | 2024-01-05 07:16:14 | | |
Video Upload Options
The liver is the largest gland in the human body, mediating essential functions in homeostasis, metabolism, serum protein production, storage of glycogen, drug detoxification, immune system, and production and secretion of bile acids. The ultimate goal of liver tissue engineering (LTE) is to restore partial or total function of the liver during liver failure. A fully functional liver is the ultimate aim of LTE, and functional liver tissue can be used for drug testing. Moreover, LTE has the potential to develop an extracorporeal liver support (ECLS) system performing the essential functions of the liver to reduce mortality or to bridge a patient to a liver transplant. With recent advances in the field of regenerative medicine, tissue engineering holds high potential to progress as an alternative or supplement to LT. A major advance in the LTE area was published by Chhabra and colleagues, reporting the development of a vascularized liver model to understand liver regeneration.
1. Focus on Extracellular Matrix: Use of Biodegradable Biomaterials
2. Knowledge from Studies on Post-Hepatectomy Liver Failures
| Regeneration Stage | Growth Factors/ Cytokines |
Major Roles | Enhances Regeneration |
|---|---|---|---|
| Priming stage | TNF-α, IL-6 | Essential for liver regeneration | Yes |
| Proliferation stage | PDGF-Rα | Replaceable with EGFR | Yes |
| HGF | Essential for liver regeneration | Yes | |
| IGF | Liver growth, development, and regeneration | Yes | |
| Termination stage | TGF-β | DNA synthesis inhibition in hepatocytes, ECM remodeling | No |
3. Whole-Organ Bioengineering Approach for Liver Tissue Engineering
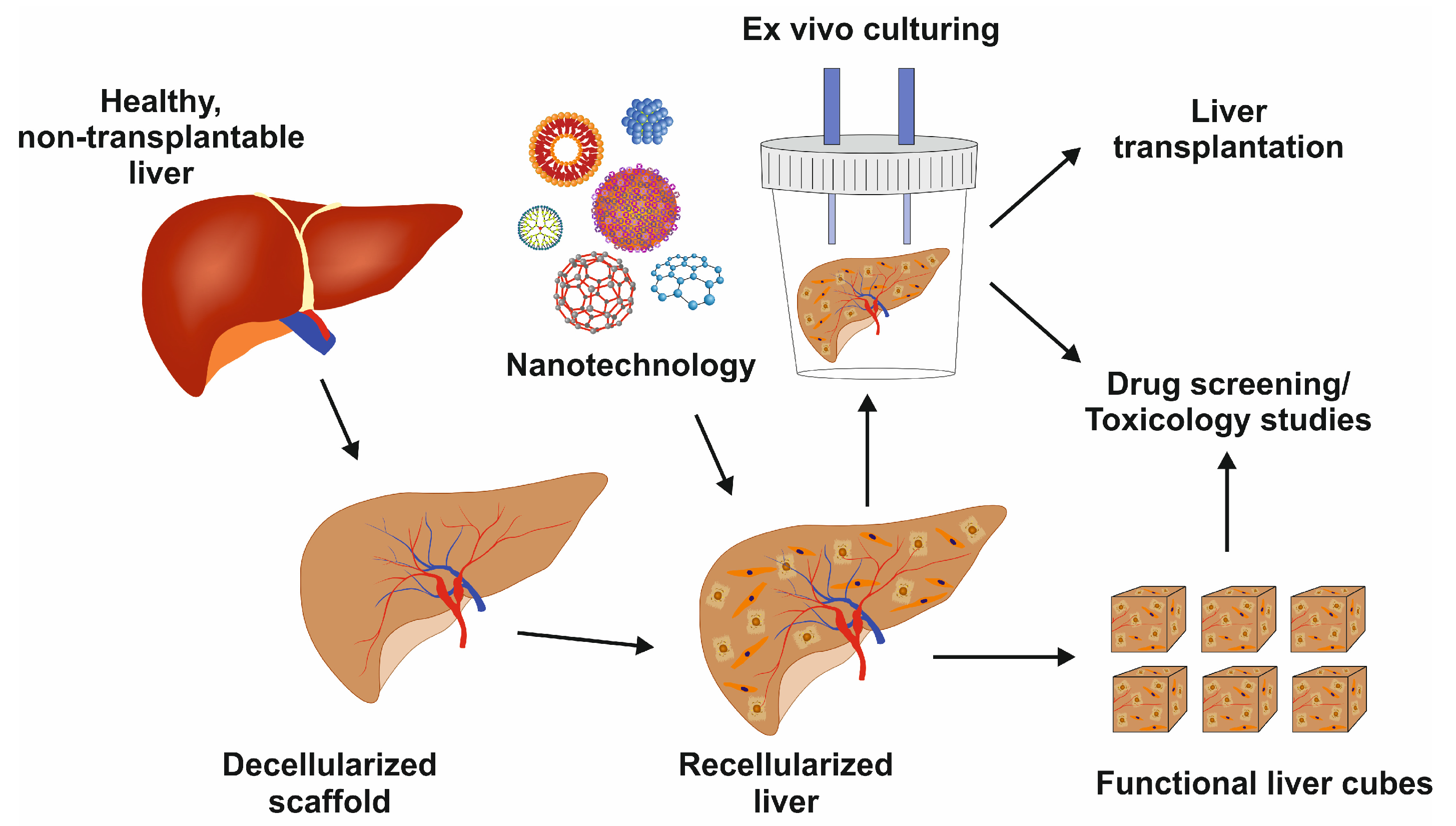
4. Recellularization in Liver Tissue Engineering
5. Liver Cell Culture Methods
References
- Lv, W.; Zhou, H.; Aazmi, A.; Yu, M.; Xu, X.; Yang, H.; Huang, Y.Y.S.; Ma, L. Constructing biomimetic liver models through biomaterials and vasculature engineering. Regen. Biomater. 2022, 9, rbac079.
- Ali, M.; Payne, S.L. Biomaterial-based cell delivery strategies to promote liver regeneration. Biomater. Res. 2021, 25, 5.
- Sanyal, A.J.; Yoon, S.K.; Lencioni, R. The etiology of hepatocellular carcinoma and consequences for treatment. Oncologist 2010, 15, 14–22.
- Nair, D.G.; Weiskirchen, R.; Al-Musharafi, S.K. The use of marine-derived bioactive compounds as potential hepatoprotective agents. Acta Pharmacol. Sin. 2015, 36, 158–170.
- Turner, W.S.; Schmelzer, E.; McClelland, R.; Wauthier, E.; Chen, W.; Reid, L.M. Human hepatoblast phenotype maintained by hyaluronan hydrogels. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 82B, 156–168.
- Richert, L.; Binda, D.; Hamilton, G.; Viollon-Abadie, C.; Alexandre, E.; Bigot-Lasserre, D.; Bars, R.; Coassolo, P.; LeCluyse, E. Evaluation of the effect of culture configuration on morphology, survival time, antioxidant status and metabolic capacities of cultured rat hepatocytes. Toxicol. In Vitro 2002, 16, 89–99.
- Bizzaro, D.; Russo, F.P.; Burra, P. New perspectives in liver transplantation: From regeneration to bioengineering. Bioengineering 2019, 6, 81.
- Jain, E.; Damania, A.; Kumar, A. Biomaterials for liver tissue engineering. Hepatol. Int. 2014, 8, 185–197.
- Bhandari, R.N.B.; Riccalton, L.A.; Lewis, A.L.; Fry, J.R.; Hammond, A.H.; Tendler, S.J.B.; Shakesheff, K.M. Liver tissue engineering: A role for co-culture systems in modifying hepatocyte function and viability. Tissue Eng. 2001, 7, 345–357.
- Hoffmann, K.; Nagel, A.J.; Tanabe, K.; Fuchs, J.; Dehlke, K.; Ghamarnejad, O.; Lemekhova, A.; Mehrabi, A. Markers of liver regeneration-the role of growth factors and cytokines: A systematic review. BMC Surg. 2020, 20, 31.
- Shi, J.-H.; Line, P.-D. Effect of liver regeneration on malignant hepatic tumors. World J. Gastroenterol. 2014, 20, 16167–16177.
- Peloso, A.; Dhal, A.; Zambon, J.P.; Li, P.; Orlando, G.; Atala, A.; Soker, S. Current achievements and future perspectives in whole-organ bioengineering. Stem Cell Res. Ther. 2015, 6, 107.
- The New York Times. Discarded Kidney. Opinion (Editorial). Available online: http://www.nytimes.com/2012/09/25/opinion/discarded-kidneys.html (accessed on 19 December 2023).
- Uygun, B.E.; Yarmush, M.L.; Uygun, K. Application of whole-organ tissue engineering in hepatology. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 738–744.
- Sabetkish, S.; Kajbafzadeh, A.-M.; Sabetkish, N.; Khorramirouz, R.; Akbarzadeh, A.; Seyedian, S.L.; Pasalar, P.; Orangian, S.; Beigi, R.S.H.; Aryan, Z.; et al. Whole-organ tissue engineering: Decellularization and recellularization of three-dimensional matrix liver scaffolds. J. Biomed. Mater. Res. A 2015, 103, 1498–1508.
- Baptista, P.M.; Siddiqui, M.M.; Lozier, G.; Rodriguez, S.R.; Atala, A.; Soker, S. The use of whole organ decellularization for the generation of a vascularized liver organoid. Hepatology 2011, 53, 604–617.
- Takebe, T.; Sekine, K.; Kimura, M.; Yoshizawa, E.; Ayano, S.; Koido, M.; Funayama, S.; Nakanishi, N.; Hisai, T.; Kobayashi, T.; et al. Massive and reproducible production of liver buds entirely from human pluripotent stem cells. Cell Rep. 2017, 21, 2661–2670.
- Toprakhisar, B.; Verfaillie, C.M.; Kumar, M. Advances in recellularization of decellularized liver grafts with different liver (stem) cells: Towards clinical applications. Cells 2023, 12, 301.
- Pan, J.; Yan, S.; Gao, J.-J.; Wang, Y.-Y.; Lu, Z.-J.; Cui, C.-W.; Zhang, Y.-H.; Wang, Y.; Meng, X.-Q.; Zhou, L.; et al. In-vivo organ engineering: Perfusion of hepatocytes in a single liver lobe scaffold of living rats. Int. J. Biochem. Cell Biol. 2016, 80, 124–131.
- Tricot, T.; De Boeck, J.; Verfaillie, C. Alternative cell sources for liver parenchyma repopulation: Where do we stand? Cells 2020, 9, 566.
- Debnath, T.; Mallarpu, C.S.; Chelluri, L.K. Development of bioengineered organ using biological acellular rat liver scaffold and hepatocytes. Organogenesis 2020, 16, 61–72.
- Elchaninov, A.; Fatkhudinov, T.; Makarov, A.; Vorobieva, I.; Lokhonina, A.; Usman, N.; Kananykhina, E.; Vishnyakova, P.; Nikitina, M.; Goldshtein, D.; et al. Inherent control of hepatocyte proliferation after subtotal liver resection. Cell Biol. Int. 2020, 44, 80–88.
- Elchaninov, A.; Fatkhudinov, T.; Usman, N.; Kananykhina, E.; Arutyunyan, I.; Makarov, A.; Bolshakova, G.; Goldshtein, D.; Sukhikh, G. Molecular survey of cell source usage during subtotal hepatectomy-induced liver regeneration in rats. PLoS ONE 2016, 11, e0162613.
- Yang, W.; Wang, X.; Wang, Z. Engineered liver tissue in vitro to mimic liver functions and its biomedical applications. Mater. Adv. 2022, 3, 4132–4154.
- Kamishibahara, Y.; Okamoto, S.; Ohkuma, T.; Taniguchi, H. Stabilized generation of human iPSC-derived liver organoids using a modified coating approach. Biol. Methods Protoc. 2023, 8, bpac034.
- Kajiwara, M.; Aoi, T.; Okita, K.; Takahashi, R.; Inoue, H.; Takayama, N.; Endo, H.; Eto, K.; Toguchida, J.; Uemoto, S.; et al. Donor-dependent variations in hepatic differentiation from human-induced pluripotent stem cells. Proc. Natl. Acad. Sci. USA 2012, 109, 12538–12543.
- Acun, A.; Oganesyan, R.; Jaramillo, M.; Yarmush, M.L.; Uygun, B.E. Human-origin iPSC-based recellularization of decellularized whole rat livers. Bioengineering 2022, 9, 219.
- Park, K.-M.; Hussein, K.H.; Hong, S.-H.; Ahn, C.; Yang, S.-R.; Park, S.-M.; Kweon, O.-K.; Kim, B.-M.; Woo, H.-M. Decellularized liver extracellular matrix as promising tools for transplantable bioengineered liver promotes hepatic lineage commitments of induced pluripotent stem cells. Tissue Eng. Part A 2016, 22, 449–460.
- Mazza, G.; Al-Akkad, W.; Telese, A.; Longato, L.; Urbani, L.; Robinson, B.; Hall, A.; Kong, K.; Frenguelli, L.; Marrone, G.; et al. Rapid production of human liver scaffolds for functional tissue engineering by high shear stress oscillation-decellularization. Sci. Rep. 2017, 7, 5534.





