Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Isabella Buttino | -- | 1565 | 2023-12-22 09:42:24 | | | |
| 2 | Wendy Huang | Meta information modification | 1565 | 2023-12-22 13:09:22 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Feng, J.; Mazzei, M.; Di Gregorio, S.; Niccolini, L.; Vitiello, V.; Ye, Y.; Guo, B.; Yan, X.; Buttino, I. Characterization and Biodegradation Role of the Copepod-Associated Microbiome. Encyclopedia. Available online: https://encyclopedia.pub/entry/53059 (accessed on 24 May 2026).
Feng J, Mazzei M, Di Gregorio S, Niccolini L, Vitiello V, Ye Y, et al. Characterization and Biodegradation Role of the Copepod-Associated Microbiome. Encyclopedia. Available at: https://encyclopedia.pub/entry/53059. Accessed May 24, 2026.
Feng, Jiantong, Maurizio Mazzei, Simona Di Gregorio, Luca Niccolini, Valentina Vitiello, Yingying Ye, Baoying Guo, Xiaojun Yan, Isabella Buttino. "Characterization and Biodegradation Role of the Copepod-Associated Microbiome" Encyclopedia, https://encyclopedia.pub/entry/53059 (accessed May 24, 2026).
Feng, J., Mazzei, M., Di Gregorio, S., Niccolini, L., Vitiello, V., Ye, Y., Guo, B., Yan, X., & Buttino, I. (2023, December 22). Characterization and Biodegradation Role of the Copepod-Associated Microbiome. In Encyclopedia. https://encyclopedia.pub/entry/53059
Feng, Jiantong, et al. "Characterization and Biodegradation Role of the Copepod-Associated Microbiome." Encyclopedia. Web. 22 December, 2023.
Copy Citation
Copepods are the most abundant organisms in marine zooplankton and the primary components of the food chain. They are hotspots for highly adaptable microorganisms, which are pivotal in biogeochemical cycles. The microbiome, encompassing microorganisms within and surrounding marine planktonic organisms, holds considerable potential for biotechnological advancements. Despite marine microbiome research interests expanding, the understanding of the ecological interactions between microbiome and copepods remains limited.
zooplankton
microorganisms
bacteria
aquaculture
biodegradation
1. Introduction
Plankton, including both phyto- and zooplanktonic organisms, are at the base of trophic webs in all aquatic ecosystems and contribute significantly to the biodiversity of marine ecosystems [1][2]. These organisms confer ecological benefits to marine environments; other than functioning as a crucial food source for many organisms, they participate in the removal of carbon dioxide from the atmosphere, generating oxygen [3]. Additionally, they contribute to the decomposition of deceased plants and animals, leading to the sequestration of both temporary and permanent carbon in the deep ocean. They participate in this carbon sequestration process alongside the microbial carbon pump, which involves mechanisms like the conversion of organic matter into dissolved inorganic carbon (DIC) and the subsequent temporary storage of this carbon in deeper waters until it is resurfaced through thermohaline circulation (Figure 1).
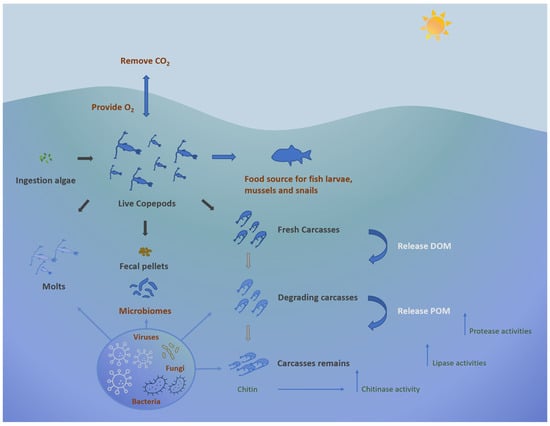
Figure 1. Ecological role of copepods in marine environment.
Among zooplankton, copepods, a class within the Crustacean subphylum, predominate as the most abundant multicellular organisms on Earth, potentially comprising 80% of the biomass of medium-sized zooplankton [4][5][6]. Current knowledge identifies more than 11,300 copepod species, showcasing their remarkable diversity and wide distribution across aquatic environments while also occurring in moist terrestrial habitats [7][8][9]. Copepods can be benthic-pelagic organisms, inhabiting the water column and sediment, and even include parasites [10][11]. Moreover, marine copepods play a critical role in the microbial loop, significantly contributing to the cycling of dissolved organic material (DOM) and microelements through their feeding activities [6][12][13]. Copepods also contribute to the release of dissolved organic carbon (DOC) and particulate organic carbon (POC) during feeding and excretion, thus providing nutrients for associated microorganisms [14][15]. Collectively, copepods regulate microbial populations, recycle nutrients, and facilitate the transfer of energy within the food web. Due to their feeding habits, nutrient excretion, and vertical migration, copepods contribute to the overall productivity and function of aquatic ecosystems and are keystone organisms in marine food webs.
Due to the position of copepods in marine food chains, they are an important alternative to traditional live feed in aquaculture. In fact, in comparison to more conventional feeds such as Artemia and Rotifers, copepods demonstrate superior capacity in ensuring nutritional quality and enhancing the digestibility of food for animals [16]. Utilizing copepods in aquaculture offers several advantages, including improved growth, survival, tolerance, and a reduced incidence of deformities in fish larvae [17][18]. Extensive research has been dedicated to exploring the benefits of copepods in aquaculture [19][20][21][22], and the observed enhancements in fish larvae performance are also due to the microbiome associated with copepod cultures [23].
In recent years, symbiotic interactions in crustacean arthropods, such as copepods, have gained significant attention [24]. The microbiome associated with copepods is a key factor in regulating the overall energy balance of zooplankton, ensuring homeostasis, and influencing the cycling of organic matter in aquatic ecosystems [25][26]. Moreover, the gut microbiota of copepods acts as a driving force for their adaptation and acclimation to harmful cyanobacterial blooms by facilitating the degradation of toxic substances released by cyanobacteria [27]. Furthermore, microbial communities associated with copepod carcasses participate in the metabolic processing of nutrient uptake, including denitrification, particularly in anoxic environments [28][29].
2. Characterization of the Copepod-Associated Microbiome
Identifying microbiomes associated with aquatic organisms involves a combination of techniques to characterize the microbial communities living in and on these organisms. These techniques help to understand the diversity, composition, and functionality of the microbiota. Prior to the advent of molecular techniques, the identification and characterization of microbiota in environmental samples involved a culture-dependent approach. The method relies on growing microorganisms in a laboratory setting, usually on culture media, to isolate and identify them. Microorganisms were grown in isolated conditions under controlled conditions of temperature, pH, and oxygen [30]. This method allows the isolation and pure culture of specific microorganisms, providing the opportunity to study the physiology, metabolism, and characteristics of individual microorganisms. Microorganisms were identified through techniques such as microscopy, biochemical testing, and genetic sequencing. However, the culturomics approach has limitations due to the specific growth requirements of different organisms, and conventional microbiological techniques only capture a small fraction of the microbiota present in environmental samples. The process is time-consuming, as some microorganisms can take days or even weeks to grow. Additionally, laboratory culture conditions may not accurately represent the natural environment; they may overlook the most abundant microorganisms in the environment, including those that are not easily cultivable, leading to bias in the types of microorganisms that can be isolated [31].
Culture-independent molecular techniques, such as next-generation sequencing (NGS) and meta-barcoding, have advanced our understanding of microbial diversity in marine and freshwater environments [32]. It is currently the most used method to analyze the microbiome, designed to study microorganisms without requiring their culture.
Analysis of genetic material directly from environmental samples can allow the identification and characterization of microorganisms [33]. It provides a more comprehensive view of the microbial community, as it can detect unculturable and rare microorganisms, allowing the study of the genetic content and biodiversity of entire microbiomes [34].
The advancement of genome sequencing technologies has revolutionized the field of microbiome research, with many studies employing Illumina short-read and nanopore long-read technologies for sequencing [35][36]. Illumina next-generation sequencing is a DNA sequencing technology used to determine the order of base pairs in DNA [37]. Currently, the most commonly used method for analyzing microbial communities associated with aquatic organisms is high-throughput sequencing of the 16S rRNA gene, predominantly performed on the Illumina platform [38][39][40]. The 16S rRNA gene contains nine hypervariable regions with distinct characteristics, with the V3 and V4 regions being the most frequently sequenced [41]. This technology can be utilized for various applications, such as whole genome and fragment sequencing, metagenomics, transcriptome analysis, and methylation analysis [42]. The process involves three fundamental steps: amplification, sequencing, and analysis [43].
This technique is faster and more efficient than a culture-dependent method. Its disadvantage is that it does not provide information on the physiological and metabolic characteristics of individual microorganisms and requires specialized equipment and expertise for genetic analysis. The functional roles of the copepod-associated microbiome are also being revealed [9]. For instance, Moisander et al. [39] employed 16S rRNA amplicon sequencing to identify a high abundance of the Proteobacteria phylum in copepods, along with Planctomycetes. Shoemaker and Moisander [44] highlighted the importance of the copepod gut microbiome on the function of bacterioplankton in seawater, suggesting that despite low microbial abundance, copepods can contribute to bacterioplankton function across diverse oceanic regions. Yeh et al. [45] utilized 16S rDNA metabarcoding to analyze the eukaryotic and prokaryotic diversity in the gut contents of the copepod Calanus finmarchicus in the North Atlantic Ocean. This approach provided insights into the diet, microbiome, parasites, and pathogens of copepods. Notably, while Vibrio spp. Was commonly observed in culture-dependent studies, it was relatively rare in high-throughput sequencing analyses [9]. Overall, the combination of these techniques provides a more comprehensive understanding of the microbiome and its ecological role associated with aquatic life and is tailored to the specific research goals and the nature of the microbial community being studied.
3. Biodegradation Role of the Copepod-Associated Microbiome
Zooplankton and microorganisms can release and consume large amounts of particulate matter and dissolve organic compounds, and bacteria can thrive in the microhabitat of zooplankton by obtaining organic and inorganic nutrients. De Corte et al. [46] showed that the zooplankton-associated bacterial assemblage was able to metabolize chitin, taurine, and other organic macromolecules. This association mediates biogeochemical processes in environmental waters through the proliferation of specific bacterial populations.
Wäge et al. [47] found the presence of gut-specific prokaryotic taxa and indicator species of methanogenic pathways in both copepods. The relative abundance of archaea and methanogenic bacteria was examined and showed a high degree of variability among individual copepods, highlighting intra- and interspecific variation in copepod-associated prokaryotic communities. The results reveal that the guts of Temora sp. And Acartia sp. Have the potential to produce methane in trace amounts.
Gorokhova et al. [48] speculated that it is possible that the copepod gut, fecal pellets, and carcasses contained mercury-methylated bacteria [49]. In a clade-specific quantitative PCR assay of copepods and cladocerans, the authors found that the hgcA gene of the methylation-associated bacterial cluster was carried in both copepods, while it was not found in cladocerans. In contrast, the Hg methylation efficiency of fecal pellets was higher in copepods, and it is hypothesized that endogenous Hg methylation in zooplankton contributes to the regulation of methylmercury in marine fish. Its methylation capacity varied synchronously in the microbiome, and this observation contributes to the dynamics of methylmercury in marine food webs.
Sadaiappan et al. [50] studied five copepod genera, Acartia spp., Calanus spp., Centropages sp., Pleuromamma spp., and Temora spp., and their associated bacteriobiomes. Their meta-analysis revealed five copepod genera with bacteriobiomes capable of mediating methanogenesis and methane oxidation. Among them, the bacteriobiomes of Pleuromamma spp. Had potential genes for methanogenesis and nitrogen fixation, and the bacteriobiomes of Temora spp. Were involved in assimilatory sulfate reduction and cyanocobalamin synthesis. The bacteriobiomes of Pleuromamma spp. And Temora spp. Had potential genes responsible for iron transport. There are also flavobacteria clade members that degrade high-molecular-weight organic matter such as cellulose and chitin, which have symbiotic or parasitic interactions with zooplankton and can use the intestines of copepods as a host environment [51].
References
- Mauchline, J.; Blaxter, J.H.S.; Douglas, B.; Tyler, P.A. The Biology of Calanoid Copepods, Introduction; Academic Press: Cambridge, MA, USA, 1998; pp. 1–13.
- Ho, J.S. Phylogenetic analysis of copepod orders. J. Crustac. Biol. 1990, 10, 528–536.
- Barbier, E.B. Marine ecosystem services. Curr. Biol. 2017, 27, R507–R510.
- Boxshall, G.A.; Halsey, S.H. An Introduction to Copepod Diversity; Ray Society: Andover, UK, 2004.
- Hammervold, S.H.; Glud, R.N.; Evjemo, J.O.; Hagemann, A.; Hansen, B.W. A new large egg type from the marine live feed calanoid copepod Acartia tonsa (Dana) perspectives for selective breeding of designer feed for hatcheries. Aquaculture 2015, 436, 114–120.
- Verity, P.; Smetacek, V. Organism life cycles, predation, and the structure of marine pelagic ecosystems. Mar. Ecol. Prog. Ser. 1996, 130, 277–293.
- Walter, T.C.; Boxshall, G. World of Copepods Database. 2021. Available online: http://www.marinespecies.org/copepoda (accessed on 2 January 2023).
- Reid, J.W.; Williamson, C.E. Copepoda. In Ecology and Classification of North American Freshwater Invertebrates; Academic Press: Cambridge, MA, USA, 2010; pp. 829–899.
- Bass, D.; Rueckert, S.; Stern, R.; Cleary, A.C.; Taylor, J.D.; Ward, G.M.; Huys, R. Parasites, pathogens, and other symbionts of copepods. Trends Parasitol. 2021, 37, 875–889.
- Huys, R.; Boxshall, G.A. (Eds.) Copepod Evolution; Ray Society: London, UK, 1991.
- González, M.T.; Castro, R.; Muñoz, G.; López, Z. Sea lice (Siphonostomatoida: Caligidae) diversity on littoral fishes from the southeastern Pacific coast determined from morphology and molecular analysis, with description of a new species (Lepeophtheirus Confusum). Parasitol. Int. 2016, 65, 685–695.
- Azam, F.; Fenchel, T.; Field, J.G.; Gray, J.S.; Meyer-Reil, L.; Thingstad, F. The ecological role of water-column microbes in the sea. Mar. Ecol. Prog. Ser. 1983, 10, 257–263.
- Turner, J.T. The importance of small planktonic copepods and their roles in pelagic marine food webs. Zool. Stud. 2004, 43, 255–266.
- Thor, P.; Dam, H.G.; Rogers, D.R. Fate of organic carbon released from decomposing copepod fecal pellets in relation to bacterial production and ectoenzymatic activity. Aquat. Microb. Ecol. 2003, 33, 279–288.
- Møller, E.F.; Thor, P.; Nielsen, T.G. Production of DOC by Calanus finmarchicus, C. glacialis, and C. hyperboreus through sloppy feeding and leakage from fecal pellets. Mar. Ecol. Prog. Ser. 2003, 262, 185–191.
- Melianawati, R.; Rarastoeti, P.; Nyoman, P.; Pudji, A. The effect of various kinds of live feeds on the digestive enzyme activity of coral trout Plectropomus leopardus (Lacepède, 1802) larvae. Int. J. Fish. Aquat. Stud. 2015, 3, 83–88.
- Liu, G.; Xu, D. Effects of calanoid copepod Schmackeria poplesia as a live food on the growth, survival and fatty acid composition of larvae and juveniles of Japanese flounder, Paralichthys olivaceus. J. Ocean Univ. China 2009, 8, 359–365.
- Karlsen, Ø.; van der Meeren, T.; Rønnestad, I.; Mangor-Jensen, A.; Galloway, T.F.; Kjørsvik, E.; Hamre, K. Copepods enhance nutritional status, growth and development in Atlantic cod (Gadus morhua L.) larvae—Can we identify the underlying factors? PeerJ 2015, 3, e902.
- Buttino, I.; Ianora, A.; Buono, S.; Vitiello, V.; Malzone, M.G.; Rico, C.; Langellotti, A.L.; Sansone, G.; Gennari, L.; Miralto, A. Experimental cultivation of the Mediterranean calanoid copepods Temora stylifera and Centropages typicus in a pilot re-circulating system. Aquacult. Res. 2012, 43, 247–259.
- Olivotto, I.; Avella, A.M.; Buttino, I.; Borroni, M.; Cutignano, A.; Carnevali, O. Calanoid copepod administration improves yellowtail clownfish (Amphiprion clarkii) larviculture: Biochemical and molecular implications. Aquac. Aquar. Conserv. Legis. Int. J. Bioflux Soc. 2009, 2, 355–367.
- Drillet, G.; Frouël, S.; Sichlau, M.H.; Jepsen, P.M.; Højgaard, J.K.; Joarder, A.K.; Hansen, B.W. Status and recommendations on marine copepod cultivation for use as live feed. Aquaculture 2011, 315, 155–166.
- Hansen, B.W. Advances using copepods in aquaculture. J. Plankton Res. 2017, 39, 972–974.
- Rong, R.L.; Gagnat, M.R.; Attramadal, Y.; Vadstein, O. Microbial water quality of the copepod Acartia tonsa in cultures for use as live feed. Aquaculture 2022, 560, 378439.
- Krueger, D.M.; Gustafson, R.G.; Cavanaugh, C.M. Vertical transmission of chemoautotrophic symbionts in the bivalve Solemya velum (Bivalvia: Protobranchia). Biol. Bull. 1996, 190, 195–202.
- Nuester, J.; Shema, S.; Vermont, A.; Fields, D.M.; Twining, B.S. The regeneration of highly bioavailable iron by meso- and microzooplankton. Limnol. Oceanogr. 2014, 59, 1399–1409.
- Schmidt, K.; Schlosser, C.; Atkinson, A.; Fielding, S.; Venables, H.J.; Waluda, C.M.; Achterberg, E.P. Zooplankton gut passage mobilizes lithogenic iron for ocean productivity. Curr. Biol. 2016, 26, 2667–2673.
- Gorokhova, E.; El-Shehawy, R.; Lehtiniemi, M.; Garbaras, A. How Copepods Can Eat Toxins Without Getting Sick: Gut Bacteria Help Zooplankton to Feed in Cyanobacteria Blooms. Front. Microbiol. 2021, 11, 589816.
- Glud, R.N.; Grossart, H.P.; Larsen, M.; Tang, K.W.; Arendt, K.E.; Rysgaard, S. Copepod carcasses as microbial hot spots for pelagic denitrification. Limnol. Oceanogr. 2015, 60, 2026–2036.
- Stief, P.; Lundgaard, A.B.; Morales-Ramirez, A.; Thamdrup, B.; Glud, R.N. Fixed-nitrogen loss associated with sinking zooplankton carcasses in a coastal oxygen minimum zone (Golfo Dulce, Costa Rica). Front. Mar. Sci. 2017, 4, 152.
- Orphan, V.J.; Taylor, L.T.; Hafenbradl, D.; Delong, E. Culture-dependent and culture-independent characterization of microbial assemblages associated with high-temperature petroleum reservoirs. Appl. Environ. Microbiol. 2000, 66, 700–711.
- Joint, I.; Mühling, M.; Querellou, J. Culturing marine bacteria—An essential prerequisite for biodiscovery. Microb. Biotechnol. 2010, 3, 564–575.
- Doytchinov, V.V.; Dimov, S.G. Microbial community composition of the Antarctic ecosystems: Review of the bacteria, fungi, and archaea identified through an NGS-based metagenomics approach. Life 2022, 12, 916.
- Su, C.; Lei, L.; Duan, Y.; Zhang, K.Q.; Yang, J. Culture-independent methods for studying environmental microorganisms: Methods, application, and perspective. Appl. Microbiol. Biotechnol. 2012, 93, 993–1003.
- Liu, S.; Moon, C.D.; Zheng, N.; Huws, S.; Zhao, S.; Wang, J. Opportunities and challenges of using metagenomic data to bring uncultured microbes into cultivation. Microbiome 2022, 10, 76.
- Akaçin, İ.; Ersoy, Ş.; Doluca, O.; Güngörmüşler, M. Comparing the significance of the utilization of next generation and third generation sequencing technologies in microbial metagenomics. Microbiol. Res. 2022, 264, 127154.
- Ciuffreda, L.; Rodríguez-Pérez, H.; Flores, C. Nanopore sequencing and its application to the study of microbial communities. Comput. Struct. Biotechnol. J. 2021, 19, 1497–1511.
- Ansorge, W.J. Next-generation DNA sequencing techniques. New Biotechnol. 2009, 25, 195–203.
- De Corte, D.; Lekunberri, I.; Sintes, E.; Garcia, J.A.L.; Gonzales, S.; Herndl, G.J. Linkage between copepods and bacteria in the North Atlantic Ocean. Aquat. Microb. Ecol. 2014, 72, 215–225.
- Moisander, P.H.; Sexton, A.D.; Daley, M.C. Stable associations masked by temporal variability in the marine copepod microbiome. PLoS ONE 2015, 10, e0138967.
- Dorosz, J.N.A.; Castro-Mejia, J.L.; Hansen, L.H.; Nielsen, D.S.; Skovgaard, A. Different microbiomes associated with the copepods Acartia tonsa and Temora longicornis from the same marine environment. Aquat. Microb. Ecol. 2020, 78, 1–9.
- Ortiz-Estrada, Á.M.; Gollas-Galván, T.; Martínez-Córdova, L.R.; Martínez-Porchas, M. Predictive functional profiles using metagenomic 16S rRNA data: A novel approach to understanding the microbial ecology of aquaculture systems. Rev. Aquac. 2019, 11, 234–245.
- Meyer, M.; Kircher, M. Illumina sequencing library preparation for highly multiplexed target capture and sequencing. Cold Spring Harb. Protoc. 2010, 2010, t5448.
- Mardis, E.R. The impact of next-generation sequencing technology on genetics. Trends Genet. 2008, 24, 133–141.
- Shoemaker, K.M.; Moisander, P.H. Seasonal variation in the copepod gut microbiome in the subtropical North Atlantic Ocean. Environ. Microbiol. 2017, 19, 3087–3097.
- Yeh, H.D.; Questel, J.M.; Maas, K.R.; Bucklin, A. Metabarcoding analysis of regional variation in gut contents of the copepod Calanus finmarchicus in the North Atlantic Ocean. Deep Sea Res. Part II Top. Stud. Oceanogr. 2020, 180, 104738.
- De Corte, D.; Srivastava, A.; Koski, M.; Garcia, J.A.L.; Takaki, Y.; Yokokawa, T.; Nunoura, T.; Elisabeth, N.H.; Sintes, E.; Herndl, G.J. Metagenomic insights into zooplankton-associated bacterial communities. Environ. Microbiol. 2018, 20, 492–505.
- Wäge, J.; Strassert, J.F.; Landsberger, A.; Loick-Wilde, N.; Schmale, O.; Stawiarski, B.; Labrenz, M. Microcapillary sampling of Baltic Sea copepod gut microbiomes indicates high variability among individuals and the potential for methane production. FEMS Microbiol. Ecol. 2019, 95, fiz024.
- Gorokhova, E.; Soerensen, A.L.; Motwani, N.H. Mercury-methylating bacteria are associated with copepods: A proof-of-principle survey in the Baltic Sea. PLoS ONE 2020, 15, e0230310.
- Sunderland, E.M.; Krabbenhoft, D.P.; Moreau, J.W.; Strode, S.A.; Landing, W.M. Mercury sources, distribution, and bioavailability in the North Pacific Ocean: Insights from data and models. Glob. Biogeochem. Cycles 2009, 23, GB2010.
- Sadaiappan, B.; PrasannaKumar, C.; Nambiar, V.U.; Subramanian, M.; Gauns, M.U. Meta-analysis cum machine learning approaches address the structure and biogeochemical potential of marine copepod associated bacteriobiomes. Sci. Rep. 2021, 11, 3312.
- Glöckner, F.O.; Fuchs, B.M.; Amann, R. Bacterioplankton compositions of lakes and oceans: A first comparison based on fluorescence in situ hybridization. Appl. Environ. Microbiol. 1999, 65, 3721–3726.
More
Information
Subjects:
Marine & Freshwater Biology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
480
Revisions:
2 times
(View History)
Update Date:
26 Dec 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





