Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yong Xiao | -- | 1938 | 2023-12-06 18:53:04 | | | |
| 2 | Wendy Huang | Meta information modification | 1938 | 2023-12-07 13:11:07 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Hazzan, O.O.; Zhao, B.; Xiao, Y. Enhancing Extracellular Electron Transfer in Environmental Biotechnology. Encyclopedia. Available online: https://encyclopedia.pub/entry/52460 (accessed on 10 May 2026).
Hazzan OO, Zhao B, Xiao Y. Enhancing Extracellular Electron Transfer in Environmental Biotechnology. Encyclopedia. Available at: https://encyclopedia.pub/entry/52460. Accessed May 10, 2026.
Hazzan, Oluwadamilola Oluwatoyin, Biyi Zhao, Yong Xiao. "Enhancing Extracellular Electron Transfer in Environmental Biotechnology" Encyclopedia, https://encyclopedia.pub/entry/52460 (accessed May 10, 2026).
Hazzan, O.O., Zhao, B., & Xiao, Y. (2023, December 06). Enhancing Extracellular Electron Transfer in Environmental Biotechnology. In Encyclopedia. https://encyclopedia.pub/entry/52460
Hazzan, Oluwadamilola Oluwatoyin, et al. "Enhancing Extracellular Electron Transfer in Environmental Biotechnology." Encyclopedia. Web. 06 December, 2023.
Copy Citation
Extracellular electron transfer (EET) is a biological mechanism that plays a crucial role in various bioelectrochemical systems (BESs) and has substantial implications for renewable energy production. By utilizing the metabolic capacities of exoelectrogens, BESs offer a viable and environmentally friendly approach to electricity generation and chemical production; however, the diminished effectiveness of EET remains a hindrance to their optimal application in practical contexts.
extracellular electron transfer
bioelectrochemical systems (BESs)
direct electron transfer (DET)
indirect electron transfer (IET)
microorganisms
temperature
pH
electron acceptors
1. Introduction
Microorganisms have devised specific processes to transmit electrons to minerals that are located near the surface of the cell, and this process is termed extracellular electron transfer (EET) [1]. The microbial EET process serves as a source of energy for the growth and maintenance of microorganisms. These microorganisms capable of conducting the EET process are referred to as electroactive microorganisms (EAMs) [2]. EAMs are now used for many applications in many environmental biotechnologies such as energy recovery and environmental remediation. For bioelectrochemical systems like microbial electrolysis cells (MECs) and microbial fuel cells (MFCs), the power production, fuel generation rates, and overall energy recovery from organic matter are all improved by increasing EET efficiency [3]. In addition to the generation of bioelectricity, the biotransformation of valuable chemicals, and the bioremediation of environmentally hazardous pollutants, EAMs are now useful for biosensing and even biocomputing [4]. EET enhancement is important in studying and harnessing microbe–metal interactions [5][6][7][8][9], but the complexity of electron transfer mechanisms, the wide range of microbes in question, the influence of environmental factors, and the scaling-up of EET processes are among the key challenges that researchers and engineers face in this field [9][10]. Overcoming these challenges will pave the way for the development of innovative applications for harnessing the potential of EET.
There are now several techniques used in environmental biotechnology to improve EET. To improve the EET capabilities of EAMs like Shewanella or Geobacter species, genetic engineering approaches can be used. This includes adding exogenous electron transfer mediators or overexpressing crucial EET genes or pathways [3]. Biofilm growth can also improve EET between microorganisms and electrodes. Optimizing growth conditions, surface modification, and biofilm engineering approaches are methods to improve the formation of biofilm [11][12]. Another method of improving EET is to optimize the material of the electrode. EET can be enhanced by modifying the composition and surface features of the electrodes [13][14][15]. This includes enhancing the contact between microbes and electrodes by employing conductive materials, such as carbon nanotubes or conductive polymers [2]. Redox mediators like riboflavin, neutral red, or anthraquinone derivatives can improve EET by promoting electron transport between microorganisms and solid surfaces or electron acceptors [16][17].
Electrochemical techniques are essential in studying the behavior of microorganisms for various purposes. One popular electrochemical method for examining the redox behavior of electroactive species in a system is cyclic voltammetry (CV). CV is used in bioelectrochemical systems to describe the electrochemical activity of redox-active mediators and microorganisms involved in EET. CV offers insights into the kinetics and thermodynamics of EET processes by assisting in determining the redox potentials of electron-donor and electron-acceptor compounds. Chronocoulometry (CC) is an additional electrochemical method utilized to quantify the overall electric charge that traversed an electrochemical cell within a designated time interval. This method can be performed by imparting a constant potential to the working electrode and measuring the resulting current as a function of time. The term “chronocoulometry” is derived from the words “coulometry”, which quantifies the amount of electricity (charge) exchanged within a system, and “chrono”, which denotes time. Pyocyanin (PYO) is an identified redox mediator molecule produced by Pseudomonas aeruginosa. The electrochemical technique utilizing CV and CC was employed in a recent study [18], to facilitate the analysis of the adsorption process of PYO onto carbon-based electrodes using glassy carbon. By utilizing these electrochemical methods in conjunction with FT-IR analysis, the authors deduced that P. aeruginosa NEJ07R establishes electronic communication with the electrode via PYO adsorption on the carbon surface. The research endeavors to enhance the engagement between redox mediators, microorganisms, and electrodes. This, in turn, may contribute to the advancement of microbial electrochemical technologies that address ecological challenges including, but not limited to, waste valorization, wastewater treatment, and renewable energy generation.
Gaining a comprehensive understanding of the electrochemical characteristics exhibited by bio-anodes is crucial to enhancing the overall efficiency and effectiveness of bioelectrochemical systems. Electrochemical impedance spectroscopy (EIS) is frequently employed for the comprehensive investigation of these characteristics. In a study conducted by Heijne et al. [19], the researchers aimed to measure the properties of bio-anodes, specifically biofilm capacitance, charge transfer, biofilm resistance, and diffusion resistance. To ensure accurate measurements without the influence of electrode capacitance, the researchers utilized Fluorinated Tin Oxide (FTO) as the electrode material for cultivating the electroactive biofilm. FTO has favorable characteristics as an electrode material in the electrochemical investigation of bio-anodes. This is primarily due to its exceptional stability and significantly lower capacitance, which typically ranges in the tens of microfarads per square centimeter (μF cm−2), in contrast to conventional carbon electrodes, such as graphite plates, that possess a capacitance of approximately 1 millifarad per square centimeter (~1 mF cm−2). The researchers conducted a study to observe the formation of an electroactive biofilm on FTO (by the utilization of EIS and polarization experiments). The findings of the study demonstrate that the capacitance of the biofilm exhibited a significant rise from an initial value of 2 μF cm−2 to a final value of 450 μF cm−2 as the biofilm developed. This observation suggests a correlation between the capacitance and both the current and the total charge generated. The findings of the study additionally indicate that biofilm capacitance can serve as an indicator of the quantity of active biomass present in bioelectrochemical systems.
Scanning electrochemical microscopy (SECM) is an additional method that enables the visualization of electrochemical phenomena at the microscale. In the field of bioelectrochemical systems, this technique can be employed to see and analyze the spatial distribution of electroactive species as well as the level of microbial activity on electrode surfaces. SECM offers valuable insights into the electrochemical activity of biofilms, enabling a deeper understanding of the variability in electron transport mechanisms across microbial communities. The technique has also demonstrated its utility in various applications, including the evaluation of membrane permeability, neurotransmitter levels, and intracellular parameters [20].
Furthermore, the utilization of potentiostatic and galvanostatic operation is an electrochemical technique wherein the potential or current, accordingly, is regulated within an electrochemical system. Within the field of bioelectrochemical systems, potentiostatic and galvanostatic techniques are employed to manipulate the electrochemical conditions, modulate the rate of electron transfer, and examine the reaction of microorganisms to varying applied potentials or currents [21][22]. These electrochemical techniques play an important role in the characterization, optimization, and understanding of the intricate mechanisms associated with extracellular electron transfer in bioelectrochemical systems. Their contributions are crucial in the advancement of microbial electrochemistry and the pragmatic implementation of these systems in the fields of energy generation, wastewater treatment, and environmental monitoring.
2. Direct Electron Transfer (DET)
Microorganisms can carry out EET in different ways as depicted in Figure 1. For DET, electrons can move from the bacteria to electrodes independent of external mediators. This method necessitates the proximity of microorganisms to the outer electron recipient and entails the transfer of electrons via outer membrane proteins known as c-type cytochromes [23][24]. The limited distance of electron transfer in DET is known to be a contributing factor to the improved EET process. The transfer of electrons through the cytochrome pathway has been extensively studied in Geobacter sulfurreducens. This has been achieved through the use of mutant strains, wherein the gene responsible for encoding cytochrome c proteins has been either overexpressed or deleted [25].
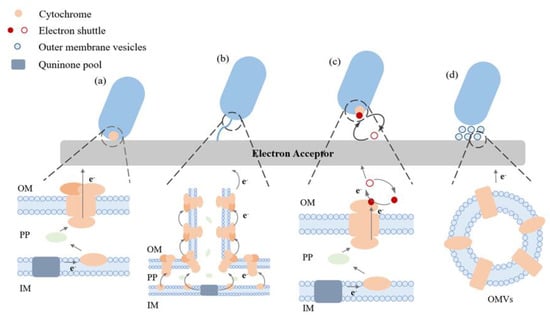
Figure 1. Elucidating the extracellular electron transport pathways that are commonly observed in electroactive microorganisms. (a) DET facilitated by cytochrome proteins, (b) the process of DET facilitated through nanowires, (c) the mode of IET facilitated by electron shuttles, (d) the method of indirect electron transfer facilitated through exterior vesicles of the membrane (OMVs). Outer membrane (OM), interior membrane (IM), and periplasm (PP).
The DET process is not exhibited by all microorganisms; hence, there has been a focus on investigating an alternative mechanism that is comparable but less comprehended, referred to as microbial nanowires. This mechanism has been occasionally referred to as a variant of DET. Nanowires are cellular membrane protrusions that bear a resemblance to pili. Due to their relatively elevated conductivity, it is postulated that they facilitate the conduction of electrons. Consequently, this confers an advantage to microorganisms by enabling them to inhabit a location that is distanced from the metal oxide or electrode. Additionally, this could facilitate inter-species electron transfer within a biofilm [23].
3. Indirect Electron Transfer (IET)
In IET, microorganisms can generate and secrete diminutive molecules that can undergo redox cycling, thereby acting as an ES (Figure 1). They function as the final electron acceptor and, upon reduction, are capable of transferring electrons to iron oxides, subsequently undergoing reoxidation. This type of shuttle offers a means for an indirect reduction process. The repetitive cycling of a solitary shuttle molecule can significantly influence the turnover rate of the terminal oxidant, such as iron, within a specific situation. Compounds such as humic compounds, quinones, phenazines, and molecules containing thiols have the potential to fulfil this function [26][27][28][29]. Electron shuttles can be classified into two distinct categories, specifically exogenous and endogenous, depending on their presence in the environment or the inherent ability of the cell to produce them. Thionine is the predominant exogenous mediator utilized, while other compounds such as quinine, phenoxazine, phenazine, and phenothiazine have also been employed in this capacity. On the other hand, endogenous mediators are released from cells in a reduced state and undergo oxidation to enable electron transfer at the electrode. The mediators under consideration in this category consist of flavin compounds produced by S. oneidensis MR-1 and 2-amino-3-carboxy-1,4-naphthoquinone, which is excreted by S. putrefaciens [24].
4. Factors Affecting EET Efficiency
EET is subject to the influence of multiple factors, encompassing temperature, pH levels, the existence of electron acceptors and donors, redox potential, as well as the properties of the electrode. Temperature affects the rate of chemical reactions and the availability of electron transfer [30]. High temperatures can enhance EET efficiency by increasing the rate of enzymatic reactions involved in electron transfer. The pH level is an essential determinant that affects both the activity of enzymes involved in EET and the proton gradient across cellular membranes. Enzymes involved in extracellular electron transfer often exhibit pH-dependent activity, and an increase in pH can disrupt the proton gradient and impede EET efficiency [31]. Electron acceptors and donors are also critical factors in EET efficiency. Oxygen is a highly favorable electron recipient because of its strong reduction potential and abundance in many natural environments [32]. In environments with a limited availability of electron acceptors, microorganisms may employ alternative strategies for EET. Organic substances like sugars, fatty acids, alcohols, and hydrogen gas are typical electron donors [33]. Redox potential is another crucial factor in EET processes. Microorganisms can utilize various electron acceptors with different redox potentials, such as solid metal oxides and metals like gold and platinum. The redox potential of these metal oxides influences their ability to accept electrons from microbial donors [34]. Electrode properties also play a significant role in EET efficiency. Conductive materials like graphite, carbon nanotubes, and metals like gold and platinum are often employed as electrodes because they possess strong electrical conductivity [35][36]. Biochar, a representative carbon-based material, has also been shown to serve as an ES to mediate the DIET process of Methanosarcina and Geobacter [37]. Thus, temperature, pH level, the presence of electron acceptors and donors, redox potential, and the characteristics of the electrodes are a few factors that generally affect the efficiency of EET.
References
- Spietz, R.L.; Payne, D.; Szilagyi, R.; Boyd, E.S. Reductive biomining of pyrite by methanogens. Trends Microbiol. 2022, 30, 1072–1083.
- Naaz, T.; Kumar, A.; Vempaty, A.; Singhal, N.; Pandit, S.; Gautam, P.; Jung, S.P. Recent advances in biological approaches towards anode biofilm engineering for improvement of extracellular electron transfer in microbial fuel cells. Environ. Eng. Res. 2023, 28, 220666.
- Wang, Y.X.; Hou, N.; Liu, X.L.; Mu, Y. Advances in interfacial engineering for enhanced microbial extracellular electron transfer. Bioresour. Technol. 2022, 345, 126562.
- Zou, L.; Huang, Y.-H.; Long, Z.-E.; Qiao, Y. On-going applications of Shewanella species in microbial electrochemical system for bioenergy, bioremediation and biosensing. World J. Microbiol. Biotechnol. 2019, 35, 1–9.
- Chiranjeevi, P.; Patil, S.A. Strategies for improving the electroactivity and specific metabolic functionality of microorganisms for various microbial electrochemical technologies. Biotechnol. Adv. 2020, 39, 107468.
- Sibi, G. Environmental Biotechnology: Fundamentals to Modern Techniques; CRC Press: Boca Raton, FL, USA, 2022.
- Tucci, M.; Viggi, C.C.; Núnez, A.E.; Schievano, A.; Rabaey, K.; Aulenta, F. Empowering electroactive microorganisms for soil remediation: Challenges in the bioelectrochemical removal of petroleum hydrocarbons. Chem. Eng. J. 2021, 419, 130008.
- Wang, R.; Li, H.; Sun, J.; Zhang, L.; Jiao, J.; Wang, Q.; Liu, S. Nanomaterials Facilitating Microbial Extracellular Electron Transfer at Interfaces. Adv. Mater. 2021, 33, 2004051.
- Zhao, J.; Li, F.; Cao, Y.; Zhang, X.; Chen, T.; Song, H.; Wang, Z. Microbial extracellular electron transfer and strategies for engineering electroactive microorganisms. Biotechnol. Adv. 2020, 53, 107682.
- Jiang, Y.; Zeng, R.J. Bidirectional extracellular electron transfers of electrode-biofilm: Mechanism and application. Bioresour. Technol. 2019, 271, 439–448.
- Wen, L.; Huang, L.; Wang, Y.; Yuan, Y.; Zhou, L. Facet-engineered hematite boosts microbial electrogenesis by synergy of promoting electroactive biofilm formation and extracellular electron transfer. Sci. Total Environ. 2022, 819, 153154.
- You, Z.; Li, J.; Wang, Y.; Wu, D.; Li, F.; Song, H. Advances in mechanisms and engineering of electroactive biofilms. Biotechnol. Adv. 2023, 66, 108170.
- Chen, L.; Yu, H.; Zhang, J.; Qin, H. A short review of graphene in the microbial electrosynthesis of biochemicals from carbon dioxide. RSC Adv. 2022, 12, 22770–22782.
- Ma, J.; Zhang, J.; Zhang, Y.; Guo, Q.; Hu, T.; Xiao, H.; Lu, W.; Jia, J. Progress on anodic modification materials and future development directions in microbial fuel cells. J. Power Sources 2023, 556, 232486.
- Tawalbeh, M.; Javed, R.M.N.; Al-Othman, A.; Almomani, F. The novel advancements of nanomaterials in biofuel cells with a focus on electrodes’ applications. Fuel 2022, 322, 124237.
- Gemünde, A.; Lai, B.; Pause, L.; Krömer, J.; Holtmann, D. Redox mediators in microbial electrochemical systems. ChemElectroChem 2022, 9, e202200216.
- Wang, X.; Chen, T.; Gao, C.; Xie, Y.; Zhang, A. Use of extracellular polymeric substances as natural redox mediators to enhance denitrification performance by accelerating electron transfer and carbon source metabolism. Bioresour. Technol. 2022, 345, 126522.
- Pérez-García, J.A.; Bacame-Valenzuela, F.J.; Manríquez, J.; Castañeda-Zaldívar, F.; Reyes-Vidal, Y. Electrochemical analysis of extracellular electron transfer process of Pseudomonas aeruginosa NEJ07R using pyocyanin on a carbon electrode. J. Environ. Chem. Eng. 2023, 11, 110708.
- Heijne, A.t.; Liu, D.; Sulonen, M.; Sleutels, T.; Fabregat-Santiago, F. Quantification of bio-anode capacitance in bioelectrochemical systems using Electrochemical Impedance Spectroscopy. J. Power Sources 2018, 400, 533–538.
- Zhou, Y.; Takahashi, Y.; Fukuma, T.; Matsue, T. Scanning electrochemical microscopy for biosurface imaging. Curr. Opin. Electrochem. 2021, 29, 100739.
- Fatima, S.; Aarti, T.; Sridhar, S. Overview of wastewater treatment approaches related to the microbial electrochemical system. In Advanced Nanomaterials and Nanocomposites for Bioelectrochemical Systems; Elsevier: Amsterdam, The Netherlands, 2023; pp. 57–80.
- Zeppilli, M.; Cristiani, L.; Dell’Armi, E.; Villano, M. Potentiostatic vs galvanostatic operation of a Microbial Electrolysis Cell for ammonium recovery and biogas upgrading. Biochem. Eng. J. 2021, 167, 107886.
- Doyle, L.; Marsili, E. The dynamics and characterization of electroactive biofilms. In Encyclopedia of Interfacial Chemistry: Surface Science and Electrochemistry; Elsevier BV: Amsterdam, The Netherlands, 2018; pp. 524–528.
- Matsena, M.T.; Nkhalambayausi Chirwa, E.M. Advances in microbial fuel cell technology for zero carbon emission energy generation from waste. In Biofuels and Bioenergy; Elsevier: Amsterdam, The Netherlands, 2022; pp. 321–358.
- Zhou, J. Establishing Bisphenol a Degradation and Enhancing Microbial Fuel Cell Performance by Biofilm Optimization of Shewanella Oneidensis MR1. Ph.D. Thesis, Illinois Institute of Technology, Chicago, IL, USA, 2023.
- Hernandez, M.E.; Newman, D.K. Extracellular electron transfer. Cell. Mol. Life Sci. 2001, 58, 1562–1571.
- Kato, S. Biotechnological Aspects of Microbial Extracellular Electron Transfer. Microbes Environ. 2015, 30, 133–139.
- Tavker, N.; Kumar, N. Bioelectrochemical systems: Understanding the basics and overcoming the challenges. In Development in Wastewater Treatment Research and Processes; Elsevier: Amsterdam, The Netherlands, 2023; pp. 79–98.
- Verma, M.; Singh, V.; Mishra, V. Moving towards the enhancement of extracellular electron transfer in electrogens. World J. Microbiol. Biotechnol. 2023, 39, 130.
- Franza, T.; Gaudu, P. Quinones: More than electron shuttles. Res. Microbiol. 2022, 173, 103953.
- Lusk, B.G.; Peraza, I.; Albal, G.; Marcus, A.K.; Popat, S.C.; Torres, C.I. pH Dependency in Anode Biofilms of Thermincola ferriacetica Suggests a Proton-Dependent Electrochemical Response. J. Am. Chem. Soc. 2018, 140, 5527–5534.
- Kracke, F.; Vassilev, I.; Kromer, J.O. Microbial electron transport and energy conservation—The foundation for optimizing bioelectrochemical systems. Front. Microbiol. 2015, 6, 575.
- Thapa, B.S.; Kim, T.; Pandit, S.; Song, Y.E.; Afsharian, Y.P.; Rahimnejad, M.; Kim, J.R.; Oh, S.E. Overview of electroactive microorganisms and electron transfer mechanisms in microbial electrochemistry. Bioresour. Technol. 2022, 347, 126579.
- Levar, C.E.; Hoffman, C.L.; Dunshee, A.J.; Toner, B.M.; Bond, D.R. Redox potential as a master variable controlling pathways of metal reduction by Geobacter sulfurreducens. ISME J. 2017, 11, 741–752.
- Liu, F.; Rotaru, A.-E.; Shrestha, P.M.; Malvankar, N.S.; Nevin, K.P.; Lovley, D.R. Promoting direct interspecies electron transfer with activated carbon. Energy Environ. Sci. 2012, 5, 8982–8989.
- Rotaru, A.-E.; Shrestha, P.M.; Liu, F.; Markovaite, B.; Chen, S.; Nevin, K.P.; Lovley, D.R. Direct interspecies electron transfer between Geobacter metallireducens and Methanosarcina barkeri. Appl. Environ. Microbiol. 2014, 80, 4599–4605.
- Zhang, K.; Deng, Y.; Liu, Z.; Feng, Y.; Hu, C.; Wang, Z. Biochar Facilitated Direct Interspecies Electron Transfer in Anaerobic Digestion to Alleviate Antibiotics Inhibition and Enhance Methanogenesis: A Review. Int. J. Environ. Res. Public Health 2023, 20, 2296.
More
Information
Subjects:
Engineering, Environmental
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
595
Revisions:
2 times
(View History)
Update Date:
07 Dec 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





