
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Mohammad Zarei | -- | 4232 | 2023-11-27 15:06:56 | | | |
| 2 | Lindsay Dong | Meta information modification | 4232 | 2023-11-28 09:08:20 | | |
Video Upload Options
Edible insects are abundant in protein content, encompassing all essential amino acids required for a balanced diet. This is a viable alternative protein source for feeding options to the expanding world population. It has significant potential for enhancing food security, offering a sustainable and environmentally conscious alternative to conventional protein sources.
1. Edible Insects
| Insect | Chicken | Pig | Cow | |
|---|---|---|---|---|
| Greenhouse gases released per kg of live weight, g | 2 | NA | 1130 | 2850 |
| Feed required per kg of live weight, kg | 1.7 | 2.5 | 5 | 10 |
| Land required per g of protein | 18 | 51 | 63 | 254 |
| Water required per g of protein, liter | 23 | 34 | 57 | 112 |
2. Insects Nutritional Value of Edible Insects
2.1. Protein Content of Edible Insects
| Insect’s Name | Protein Content Range (%) | Reference |
|---|---|---|
| Larvae | 26–45 | [16][17][18][19][20] |
| Cricket, Grasshoppers, Locusts | 6–77 | [16][17] |
| Grasshopper | 20–56 | [17][18] |
| Beetles, Grubes | 8–69 | [17][21][22] |
| Termites | 20–43 | [17][18][22] |
| Bees, Ants | 5–66 | [17][22] |
| Dragonfly | 26–54 | [23] |
| Cockroaches | 43–66 | [22] |
| Flies | 35–64 | [22] |
| True Bugs | 27–71 | [22] |
| Butterflies, Moths | 18–60 | [22] |
| Dragonflies, Damselflies | 54–56 | [22] |
2.2. Amino Acid Composition
| Source | Lys * | His ** | Arg ** | Asp | Thr * | Ser | Glu | Pro | Gly | Ala | Met * | Cys | Val * | Ile * | Leu * | Phe * | Tyr ** | Trp * | AAS *** (%) | Reference |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Zonocerus variegatus | 48.4 | 39.2 | 60.6 | 81.9 | 30.7 | 46.7 | 133.7 | 43.0 | 44.9 | 36.6 | 18.9 | 6.5 | 35.4 | 36.7 | 50.6 | 30.5 | 25.3 | - | 66.4 | [25] |
| Periplaneta americana L.a | 40.0 | 20.0 | 51.0 | - | 36.0 | 45.0 | 130 | 65.0 | 71.0 | 61.0 | 36.0 | 20.0 | 65.0 | 31.0 | 56.0 | 31.0 | 69.0 | 6.0 | 76.3 | [26] |
| Rhynchophoris phoenicis (larvae) | 45.0 | 38.9 | 79.2 | 30.6 | 39.0 | 156.0 | 50.1 | 47.2 | 52.5 | 19.7 | 20.2 | 35.0 | 39 | 54.2 | 47.5 | 29.0 | - | 71.9 | [27] | |
| Sciphophorus acupunctatus (larvae) | 53.5 | 14.7 | 44.0 | - | 40.4 | - | - | - | - | - | 20.2 | 26.7 | 62.0 | 48.2 | 78.2 | 46.1 | 63.5 | 8.1 | 83.4 | [28][29] |
| Ephydra hians (larvae) | 55.0 | 10.0 | - | 49.0 | - | - | - | - | - | 19.0 | - | 61.0 | 40.0 | 74.0 | 54.0 | 51.0 | 7.1 | 74.3 | [30] | |
| Hoplophorion monograma | 55.0 | 15.0 | - | 45.0 | - | - | - | - | - | 19.0 | - | 74.0 | 41.0 | 77.0 | 47.0 | 90.0 | 9.6 | 84.1 | [30] | |
| Atta mexicanah | 49.0 | 25.0 | - | 43.0 | - | - | - | - | - | 34.0 | - | 64.0 | 53.0 | 80.0 | 88.0 | 47.0 | 6.0 | 89.3 | [30] | |
| Liometopum apiculatumd | 58.0 | 29 | 50.0 | 42.0 | - | - | - | - | - | 18.0 | 14.0 | 60.0 | 49.0 | 76.0 | 39.0 | 68.0 | 8.0 | 88.0 | [30] | |
| Macrotermes bellicosus | 54.2 | 51.4 | 69.4 | 27.5 | - | - | - | - | - | 7.5 | 18.7 | 73.3 | 51.1 | 78.3 | 43.8 | 30.2 | 14.3 | 97.3 | [28] | |
| Bombyx mori (larvae) | 47.3 | 25.8 | 41.9 | - | 31.2 | 36.6 | 100.0 | 34.4 | 60.2 | 45.2 | 14.0 | 8.6 | 40.9 | 32.3 | 52.7 | 29.0 | 31.2 | 7.5 | 64.8 | [31] |
| Acheta domesticus (adults) | 53.7 | 23.4 | 61.0 | - | 36.1 | 49.8 | 104.9 | 56.1 | 50.7 | 87.8 | 14.6 | 8.3 | 52.2 | 45.9 | 100.0 | 31.7 | 48.8 | 6.3 | 78.2 | [31] |
| Boopedon flaviventrish | 55.0 | 24.0 | - | 44.0 | - | - | - | - | - | 18.0 | - | 57.0 | 47.0 | 88.0 | 41.0 | 74.0 | 6.0 | 83.1 | ||
| Fish (Clarias anguillaris) | 50.2 | 11.8 | 47.8 | 70.4 | 20.8 | 19.2 | 118 | 24.5 | 31.1 | 24.8 | 23.4 | 7.3 | 28.0 | 25.8 | 64.7 | 38.7 | 24.6 | - | - | [32] |
| Beef | 45 | 20 | 33 | 52 | 25 | 27 | 90 | 28 | 24 | 30 | 16 | 5.9 | 20 | 16 | 42 | 24 | 22 | - | - | [33] |
2.3. Fatty Acids Composition
| Common Name | Fat Content |
Saturate d Fatty Acids (SFA) | Monounsaturated Fatty Acids (MUFA) | Polyunsaturated Fatty Acids (PUFA) | Linoleic Acid (18:2) | Alfa Linolenic Acid (18:3) | Arachidonic Acid (18:4) |
Reference |
|---|---|---|---|---|---|---|---|---|
| Winged termite | 44.82 | 35.05 | 52.77 | 12.18 | 10.75 | 1.43 | [34][35] | |
| White spotted flower beetle | 26.70 | 23.61 | 95.20 | 10.40 | 9.10 | 0.40 | 0.70 | [16][36] |
| Desert locust | 13.00 | 25.30 | 39.35 | 26.28 | 14.04 | 11.35 | [37] | |
| Dung beetle * | 13.50 | 733.46 | 85.65 | 1514.32 | - | 39.82 | 934.95 | [38][39] |
| Black soldier fly larvae | 35.00 | 36.20 | 28.70 | 35.00 | 13.00 | 1.70 | - | [40][41][42] |
| Sugarcane termite | 46.00 | 32.17 | 56.10 | 11.73 | 11.54 | 0.20 | - | [35] |
| Tropical house cricket | 20.00 | 33.74 | 34.33 | 31.91 | 29.78 | 2.13 | - | [37] |
| Guizhou black ant |
15.20 | 23.90 | 72.40 | 3.70 | 2.10 | 1.00 | 0.20 | [43] |
2.4. Micronutrient and Vitamin Composition Variability
3. Production of Protein, Protein Hydrolysate, and Peptides
4. Hydrolysis of Insect Proteins
5. Bioactive Peptides
| Insect Source | Enzyme Used | Identified Peptides | Bioactivity | Reference |
|---|---|---|---|---|
| Silkworm/domestic silk moth (Bombyx mori) | Pepsin, trypsin, R-chymotrypsin | simulated the human gastrointestinal hydrolysate | ACE inhibitory effects, with 100% activity in hydrolysates | [58] |
| Cotton leafworm (Spodoptera littoralis) | Pepsin, trypsin and α-chymotrypsin | Ala-Val-Phe | In vitro ACE inhibitory activity (IC50: 2123 μM) | [59] |
| Mealworm larva (Tenebrio molitor) | Alcalase | Tyr–Ala–Asn | ACE inhibitory activity (IC50: 0.017 mg/mL) | [60] |
| Silkworm pupae (Bombyx mori) | Alcalase, Prolyve, Flavourzyme, Brewers Clarex | SWFVTPF, NDVLFF | Antioxidant activity (ROS reduction, superoxide dismutase (SOD) expression and glutathione (GSH) production activity) | [61] |
| Tropical house crickets (Gryllodes sigillatus) | Alcalase-generated protein hydrolysates | YKPRP, PHGAP, VGPPQ | Anti-hypertensive, anti-glycemic, anti-inflammatory activities | [62] |
| Black soldier fly (Hermetia Illucens) | -------------------------- | Hill-Cec1, Hill-Cec10 | Antimicrobial activity against various pathogens | [63] |
| Silkworm pupae (Bombyx mori) | Acidic protease, Neutral protease | FKGPACA, SVLGTGC | Antioxidant activity, ABTS radical scavenging | [64] |
| Crickets (Gryllus bimaculatus) | Alcalase | TEAPLNPK, EVGA, KLL, TGNLPGAAHPLLL, AHLLT, LSPLYE, AGVL, VAAV, VAGL, QLL | Antioxidant activity | [65] |
| White-spotted flower chafer larva (Protaetia brevitarsis) | Flavourzyme | Ser-Tyr, Pro-Phe, Tyr-Pro-Tyr, Trp-Ile | ACE inhibitory activity, NO production in cells | [66] |
| Brown seaweed (Laminaria digitate) | Hydrolysates, fermentation generated peptides or generated from high-pressure processing | YIGNNPAKGGLF, IGNNPAKGGLF, and others (130 in total) | ACE-1 inhibitory activity, Potential DPP-IV inhibition | [67] |
6. Bioactive Peptides from Insect Proteins: Functional Potential and Applications
6.1. Protein Solubility
Zielińska, Karaś, and Baraniak [48] studied the functional properties of proteins isolated from three species of edible insects including the tropical house cricket, desert locust, and mealworm, and evaluated the solubility of the isolated proteins. Their research found that the proteins exhibited their lowest solubility at a pH level of approximately 5 across all three protein samples, while the highest solubility was achieved at around pH 11. A high protein solubility was also shown within the pH range of 2 to 4. Similar results have been reported for larvae of an edible insect, the black soldier fly, in which the lowest solubility was shown at pH values of 4 to 5. However, the solubility increased beyond this range [68]. Mexican fruit fly protein also showed the minimum protein solubility at pH 5 whereas its maximum solubility was at pH 10 [45].
6.2. Edible Insect Proteins as Emulsifiers
Zielińska, Karaś, and Baraniak [48] compared the emulsifying properties of three insect species in which the highest value was noted in the tropical house cricket protein preparation (72.62%) with an emulsion stability of 38.3%. Similarly, Omotoso [69] reported an emulsion activity of 75% for silkworms but with a lower emulsion stability (23%). The emulsion activities for the whole insects in the Zielińska, Baraniak, Karaś, Rybczyńska, and Jakubczyk [37] study were found to have consistent values ranging from 62% to 69.17%. Moreover, pretreatment with pulsed-field electricity (PFE) could improve the emulsifying capacity (EC) of proteins extracted from the house cricket flour. The highest intense PFE increased the emulsifying capacity to 74.7%, while the lowest increase in the EC was 22.1%.
6.3. Foaming Properties of Insect Proteins
Egg-white proteins are the most widely utilized proteins in spray applications. Furthermore, milk proteins such as whey proteins and caseins or soy proteins are also utilized for spray utilization. The characteristics of foam inherent to these proteins have been investigated, especially within the most popular edible insect proteins. These protein sources exhibit potential for integration into food formulations for foaming purposes under specific pH and ionic strength conditions [70]. The foaming activity and stability are different among various insects, even when subjected to a similar processing approach. One of the main factors affecting the foaming properties is the amino acid composition of insect proteins, as shown in Kim, et al. [71]. In that study, the salt-soluble protein fraction of mealworms revealed significantly higher foamability than water-soluble fractions. Having a polar water-soluble group (amphiphilicity) is a critical parameter in foaming properties. No foam activity was observed in the African migratory locust flours at a pH of less than 3, while a significant foam formation of around 200% was shown at pH 5 [72].
6.4. Gelling Properties of Insect Proteins
6.5. Water-Holding Capacity of Insect Proteins
All conditions associated with the protein matrix’s capacity to retain the maximum amount of water per gram of the sample material, even when subjected to the force of gravity, are called the water-holding capacity (WHC). Water-holding capacity (WHC), water-binding capacity (WBC), and water absorption capacity (WAC) are all the same terms used to elucidate how a protein is able to absorb water. Despite potential variations in measurement methodologies, these three terminologies are commonly employed interchangeably. This functional characteristic is indeed related to the gelation and gelling properties of proteins. Furthermore, the water-binding capacity is enhanced through thermal denaturation. This feature is also linked to a better texture and wetness that is of considerable relevance in food formulation. Certain insects like mealworm and African palm weevil (Rhynchophorus phoenicis) have greater WHC values than pulse protein flours and are similar to concentrated soy and milk protein levels. These interesting results show that specific concentrated insect protein or flour may be included as functional agents in food formulation [70].
6.6. Oil Absorption Capacity of Insect Proteins
7. Edible Insects and Their Proteins in Meat Analogs and Cereal Products
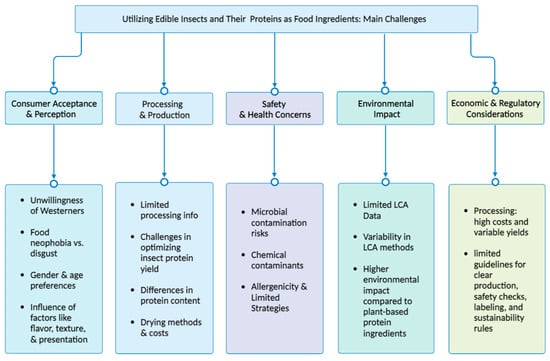
8. Challenges, Food Safety, and Considerations in Utilizing Edible Insects and Their Proteins as Food Ingredients
9. Halal and Kosher Considerations for Insect-Based Food Products
References
- Kröger, T.; Dupont, J.; Büsing, L.; Fiebelkorn, F. Acceptance of Insect-Based Food Products in Western Societies: A Systematic Review. Front. Nutr. 2021, 8, 759885.
- Liceaga, A.M.; Aguilar-Toalá, J.E.; Vallejo-Cordoba, B.; González-Córdova, A.F.; Hernández-Mendoza, A. Insects as an alternative protein source. Annu. Rev. Food Sci. Technol. 2022, 13, 19–34.
- Aidoo, O.F.; Osei-Owusu, J.; Asante, K.; Dofuor, A.K.; Boateng, B.O.; Debrah, S.K.; Ninsin, K.D.; Siddiqui, S.A.; Chia, S.Y. Insects as food and medicine: A sustainable solution for global health and environmental challenges. Front. Nutr. 2023, 10, 1113219.
- Hazarika, A.K.; Kalita, U. Human consumption of insects. Science 2023, 379, 140–141.
- Cadinu, L.A.; Barra, P.; Torre, F.; Delogu, F.; Madau, F.A. Insect rearing: Potential, challenges, and circularity. Sustainability 2020, 12, 4567.
- Aiello, D.; Barbera, M.; Bongiorno, D.; Cammarata, M.; Censi, V.; Indelicato, S.; Mazzotti, F.; Napoli, A.; Piazzese, D.; Saiano, F. Edible Insects an Alternative Nutritional Source of Bioactive Compounds: A Review. Molecules 2023, 28, 699.
- Alexander, P.; Brown, C.; Arneth, A.; Dias, C.; Finnigan, J.; Moran, D.; Rounsevell, M.D.A. Could consumption of insects, cultured meat or imitation meat reduce global agricultural land use? Glob. Food Secur. 2017, 15, 22–32.
- de Sousa, A.; Warren, H.; Rekomaa, R. Bugs Are Coming Soon to Your Dinner Table. Available online: https://www.bloomberg.com/graphics/2018-insects-as-food/ (accessed on 5 July 2018).
- de Castro, R.J.S.; Ohara, A.; dos Santos Aguilar, J.G.; Domingues, M.A.F. Nutritional, functional and biological properties of insect proteins: Processes for obtaining, consumption and future challenges. Trends Food Sci. Technol. 2018, 76, 82–89.
- Johnson, D.V. The contribution of edible forest insects to human nutrition and to forest management. In Forest Insects as Food: Humans Bite Back; FAO of the United Nations Regional Office for Asia and the Pacific: Bangkok, Thailand, 2010; pp. 5–22.
- Meyer-Rochow, V.B.; Gahukar, R.T.; Ghosh, S.; Jung, C. Chemical Composition, Nutrient Quality and Acceptability of Edible Insects Are Affected by Species, Developmental Stage, Gender, Diet, and Processing Method. Foods 2021, 10, 1036.
- Kim, T.-K.; Yong, H.I.; Kim, Y.-B.; Kim, H.-W.; Choi, Y.-S. Edible insects as a protein source: A review of public perception, processing technology, and research trends. Food Sci. Anim. Resour. 2019, 39, 521.
- Melo, V.; Garcia, M.; Sandoval, H.; Jiménez, H.D.; Calvo, C. Quality proteins from edible indigenous insect food of Latin America and Asia. Emir. J. Food Agric. 2011, 23, 283.
- Schlüter, O.; Rumpold, B.; Holzhauser, T.; Roth, A.; Vogel, R.F.; Quasigroch, W.; Vogel, S.; Heinz, V.; Jäger, H.; Bandick, N. Safety aspects of the production of foods and food ingredients from insects. Mol. Nutr. Food Res. 2017, 61, 1600520.
- Durst, P.B.; Johnson, D.V.; Leslie, R.N.; Shono, K. Forest insects as food: Humans bite back. RAP Publ. 2010, 1, 1–241.
- Ghosh, S.; Lee, S.-M.; Jung, C.; Meyer-Rochow, V. Nutritional composition of five commercial edible insects in South Korea. J. Asia-Pac. Entomol. 2017, 20, 686–694.
- Banjo, A.; Lawal, O.; Songonuga, E. The nutritional value of fourteen species of edible insects in southwestern Nigeria. Afr. J. Biotechnol. 2006, 5, 298–301.
- Siulapwa, N.; Mwambungu, A.; Lungu, E.; Sichilima, W. Nutritional value of four common edible insects in Zambia. Int. J. Sci. Res 2014, 3, 876–884.
- Ekpo, K.; Onigbinde, A. Nutritional potentials of the larva of Rhynchophorus phoenicis (F). Pak. J. Nutr. 2005, 4, 287–290.
- Ravzanaadii, N.; Kim, S.-H.; Choi, W.-H.; Hong, S.-J.; Kim, N.-J. Nutritional value of mealworm, Tenebrio molitor as food source. Int. J. Ind. Entomol. 2012, 25, 93–98.
- Adesina, A. Proximate and anti-nutritional composition of two common edible insects: Yam beetle (Heteroligus meles) and palm weevil (Rhynchophorus phoenicis). Elixir Food Sci. 2012, 49, 9782–9786.
- Rumpold, B.A.; Schlüter, O.K. Nutritional composition and safety aspects of edible insects. Mol. Nutr. Food Res. 2013, 57, 802–823.
- Ramos-Elorduy, J. Insects: A hopeful food source. In Ecological Implications of Minilivestock; CRC Press: Boca Raton, FL, USA, 2005; pp. 263–291.
- Hawkey, K.J.; Lopez-Viso, C.; Brameld, J.M.; Parr, T.; Salter, A.M. Insects: A potential source of protein and other nutrients for feed and food. Annu. Rev. Anim. Biosci. 2021, 9, 333–354.
- Adeyeye, E. Amino acid composition of variegated grasshopper, Zonocerus variegatus. Trop. Sci. 2005, 45, 141–143.
- Blásquez, J.R.-E.; Moreno, J.M.P.; Camacho, V.H.M. Could grasshoppers be a nutritive meal. Food Nutr. Sci. 2012, 3, 164.
- Onyeike, E.N.; Ayalogu, E.O.; Okaraonye, C.C. Nutritive value of the larvae of raphia palm beetle (Oryctes rhinoceros) and weevil (Rhyncophorus pheonicis). J. Sci. Food Agric. 2005, 85, 1822–1828.
- Bukkens, S.G. The nutritional value of edible insects. Ecol. Food Nutr. 1997, 36, 287–319.
- Williams, J.; Williams, J.; Kirabo, A.; Chester, D.; Peterson, M. Nutrient content and health benefits of insects. In Insects as Sustainable Food Ingredients; Elsevier: Amsterdam, The Netherlands, 2016; pp. 61–84.
- Ladrón de Guevara, O.; Padilla, P.; García, L.; Pino, J.; Ramos-Elorduy, J. Amino acid determination in some edible Mexican insects. Amino Acids 1995, 9, 161–173.
- Finke, M.D. Complete nutrient composition of commercially raised invertebrates used as food for insectivores. Zoo Biol. Publ. Affil. Am. Zoo Aquar. Assoc. 2002, 21, 269–285.
- Adeyeye, E. Amino acid composition of three species of Nigerian fish: Clarias anguillaris, Oreochromis niloticus and Cynoglossus senegalensis. Food Chem. 2009, 113, 43–46.
- Van Huis, A.; Van Itterbeeck, J.; Klunder, H.; Mertens, E.; Halloran, A.; Muir, G.; Vantomme, P. Edible Insects: Future Prospects for Food and Feed Security; Food and Agriculture Organization of the United Nations: Rome, Italy, 2013.
- Weru, J.; Chege, P.; Kinyuru, J. Nutritional potential of edible insects: A systematic review of published data. Int. J. Trop. Insect Sci. 2021, 41, 2015–2037.
- Kinyuru, J.N.; Konyole, S.O.; Roos, N.; Onyango, C.A.; Owino, V.O.; Owuor, B.O.; Estambale, B.B.; Friis, H.; Aagaard-Hansen, J.; Kenji, G.M. Nutrient composition of four species of winged termites consumed in western Kenya. J. Food Compos. Anal. 2013, 30, 120–124.
- Yeo, H.; Youn, K.; Kim, M.; Yun, E.Y.; Hwang, J.S.; Jeong, W.S.; Jun, M. Fatty Acid Composition and Volatile Constituents of Protaetia brevitarsis Larvae. Prev. Nutr. Food Sci. 2013, 18, 150–156.
- Zielińska, E.; Baraniak, B.; Karaś, M.; Rybczyńska, K.; Jakubczyk, A. Selected species of edible insects as a source of nutrient composition. Food Res. Int. 2015, 77, 460–466.
- Raksakantong, P.; Meeso, N.; Kubola, J.; Siriamornpun, S. Fatty acids and proximate composition of eight Thai edible terricolous insects. Food Res. Int. 2010, 43, 350–355.
- Bophimai, P.; Siri, S. Fatty acid composition of some edible dung beetles in Thailand. Int. Food Res. J. 2010, 17, 1025–1030.
- Ewald, N.; Vidakovic, A.; Langeland, M.; Kiessling, A.; Sampels, S.; Lalander, C. Fatty acid composition of black soldier fly larvae (Hermetia illucens)—Possibilities and limitations for modification through diet. Waste Manag. 2020, 102, 40–47.
- Mai, H.C.; Dao, N.D.; Lam, T.D.; Nguyen, B.V.; Nguyen, D.C.; Bach, L.G. Purification process, physicochemical properties, and fatty acid composition of black soldier fly (Hermetia illucens Linnaeus) larvae oil. J. Am. Oil Chem. Soc. 2019, 96, 1303–1311.
- Müller, A.; Wolf, D.; Gutzeit, H.O. The black soldier fly, Hermetia illucens—A promising source for sustainable production of proteins, lipids and bioactive substances. Z. Naturforschung C 2017, 72, 351–363.
- Oranut, S.; Subhachai, B.; Shen, L.-R.; Duo, L. Lipids and fatty acid composition of dried edible red and black ants. Agric. Sci. China 2010, 9, 1072–1077.
- Churchward-Venne, T.A.; Pinckaers, P.J.; van Loon, J.J.; van Loon, L.J. Consideration of insects as a source of dietary protein for human consumption. Nutr. Rev. 2017, 75, 1035–1045.
- Del Valle, F.; Mena, M.; Bourges, H. An Investigation into insect protein. J. Food Process. Preserv. 1982, 6, 99–110.
- Bußler, S.; Rumpold, B.A.; Jander, E.; Rawel, H.M.; Schlüter, O.K. Recovery and techno-functionality of flours and proteins from two edible insect species: Meal worm (Tenebrio molitor) and black soldier fly (Hermetia illucens) larvae. Heliyon 2016, 2, e00218.
- Choi, B.D.; Wong, N.A.; Auh, J.-H. Defatting and sonication enhances protein extraction from edible insects. Korean J. Food Sci. Anim. Resour. 2017, 37, 955.
- Zielińska, E.; Karaś, M.; Baraniak, B. Comparison of functional properties of edible insects and protein preparations thereof. LWT 2018, 91, 168–174.
- Mishyna, M.; Keppler, J.K.; Chen, J. Techno-functional properties of edible insect proteins and effects of processing. Curr. Opin. Colloid Interface Sci. 2021, 56, 101508.
- Villaseñor, V.M.; Enriquez-Vara, J.N.; Urías-Silva, J.E.; Mojica, L. Edible insects: Techno-functional properties food and feed applications and biological potential. Food Rev. Int. 2021, 38, 866–892.
- EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA); Turck, D.; Bohn, T.; Castenmiller, J.; De Henauw, S.; Hirsch-Ernst, K.I.; Maciuk, A.; Mangelsdorf, I.; McArdle, H.J.; Naska, A.; et al. Safety of frozen and dried formulations from whole yellow mealworm (Tenebrio molitor larva) as a novel food pursuant to Regulation (EU) 2015/2283. EFSA J. 2021, 19, e06778.
- Akhtar, Y.; Isman, M.B. Insects as an alternative protein source. In Proteins in Food Processing; Elsevier: Amsterdam, The Netherlands, 2018; pp. 263–288.
- Batish, I.; Brits, D.; Valencia, P.; Miyai, C.; Rafeeq, S.; Xu, Y.; Galanopoulos, M.; Sismour, E.; Ovissipour, R. Effects of Enzymatic Hydrolysis on the Functional Properties, Antioxidant Activity and Protein Structure of Black Soldier Fly (Hermetia illucens) Protein. Insects 2020, 11, 876.
- Zhao, X.; Vázquez-Gutiérrez, J.L.; Johansson, D.P.; Landberg, R.; Langton, M. Yellow mealworm protein for food purposes-extraction and functional properties. PLoS ONE 2016, 11, e0147791.
- Gould, J.; Wolf, B. Interfacial and emulsifying properties of mealworm protein at the oil/water interface. Food Hydrocoll. 2018, 77, 57–65.
- Zarei, M.; Muhialdin, B.J.; Hassanzadeh, K.; Yea, C.S.; Ahmadi, R. Enzymatic Hydrolysis of Proteins. In Bioactive Peptides from Food: Sources, Analysis, and Functions; CRC Press: Boca Raton, FL, USA, 2022; pp. 189–208.
- Leni, G.; Soetemans, L.; Caligiani, A.; Sforza, S.; Bastiaens, L. Degree of hydrolysis affects the techno-functional properties of lesser mealworm protein hydrolysates. Foods 2020, 9, 381.
- Vercruysse, L.; Smagghe, G.; Herregods, G.; Van Camp, J. ACE inhibitory activity in enzymatic hydrolysates of insect protein. J. Agric. Food Chem. 2005, 53, 5207–5211.
- Vercruysse, L.; Smagghe, G.; Matsui, T.; Van Camp, J. Purification and identification of an angiotensin I converting enzyme (ACE) inhibitory peptide from the gastrointestinal hydrolysate of the cotton leafworm, Spodoptera littoralis. Process Biochem. 2008, 43, 900–904.
- Dai, C.; Ma, H.; Luo, L.; Yin, X. Angiotensin I-converting enzyme (ACE) inhibitory peptide derived from Tenebrio molitor (L.) larva protein hydrolysate. Eur. Food Res. Technol. 2013, 236, 681–689.
- Cermeño, M.; Bascón, C.; Amigo-Benavent, M.; Felix, M.; FitzGerald, R.J. Identification of peptides from edible silkworm pupae (Bombyx mori) protein hydrolysates with antioxidant activity. J. Funct. Foods 2022, 92, 105052.
- Hall, F.; Reddivari, L.; Liceaga, A.M. Identification and characterization of edible cricket peptides on hypertensive and glycemic in vitro inhibition and their anti-inflammatory activity on RAW 264.7 macrophage cells. Nutrients 2020, 12, 3588.
- Van Moll, L.; De Smet, J.; Paas, A.; Tegtmeier, D.; Vilcinskas, A.; Cos, P.; Van Campenhout, L. In vitro evaluation of antimicrobial peptides from the black soldier fly (Hermetia illucens) against a selection of human pathogens. Microbiol. Spectr. 2022, 10, e01664-21.
- Zhang, Y.; Wang, J.; Zhu, Z.; Li, X.; Sun, S.; Wang, W.; Sadiq, F.A. Identification and characterization of two novel antioxidant peptides from silkworm pupae protein hydrolysates. Eur. Food Res. Technol. 2021, 247, 343–352.
- Fashakin, O.O.; Tangjaidee, P.; Unban, K.; Klangpetch, W.; Khumsap, T.; Sringarm, K.; Rawdkuen, S.; Phongthai, S. Isolation and Identification of Antioxidant Peptides Derived from Cricket (Gryllus bimaculatus) Protein Fractions. Insects 2023, 14, 674.
- Lee, J.H.; Kim, T.-K.; Yong, H.I.; Cha, J.Y.; Song, K.-M.; Lee, H.G.; Je, J.-G.; Kang, M.-C.; Choi, Y.-S. Peptides inhibiting angiotensin-I-converting enzyme: Isolation from flavourzyme hydrolysate of Protaetia brevitarsis larva protein and identification. Food Chem. 2023, 399, 133897.
- Purcell, D.; Packer, M.A.; Hayes, M. Identification of Bioactive Peptides from a Laminaria digitata Protein Hydrolysate Using In Silico and In Vitro Methods to Identify Angiotensin-1-Converting Enzyme (ACE-1) Inhibitory Peptides. Mar. Drugs 2023, 21, 90.
- Mintah, B.K.; He, R.; Agyekum, A.A.; Dabbour, M.; Golly, M.K.; Ma, H. Edible insect protein for food applications: Extraction, composition, and functional properties. J. Food Process. Eng. 2020, 43, e13362.
- Omotoso, O.T. An Evaluation of the Nutrients and Some Anti-nutrients in Silkworm, Bombyxmori L. (Bombycidae: Lepidoptera). Jordan J. Biol. Sci. 2015, 8, 45–50.
- Gravel, A.; Doyen, A. The use of edible insect proteins in food: Challenges and issues related to their functional properties. Innov. Food Sci. Emerg. Technol. 2020, 59, 102272.
- Kim, T.-K.; Yong, H.I.; Jeong, C.H.; Han, S.G.; Kim, Y.-B.; Paik, H.-D.; Choi, Y.-S. Technical functional properties of water-and salt-soluble proteins extracted from edible insects. Food Sci. Anim. Resour. 2019, 39, 643.
- Purschke, B.; Meinlschmidt, P.; Horn, C.; Rieder, O.; Jäger, H. Improvement of techno-functional properties of edible insect protein from migratory locust by enzymatic hydrolysis. Eur. Food Res. Technol. 2018, 244, 999–1013.
- Yi, L.; Lakemond, C.M.; Sagis, L.M.; Eisner-Schadler, V.; van Huis, A.; van Boekel, M.A. Extraction and characterisation of protein fractions from five insect species. Food Chem. 2013, 141, 3341–3348.
- Dion-Poulin, A.; Laroche, M.; Doyen, A.; Turgeon, S.L. Functionality of cricket and mealworm hydrolysates generated after pretreatment of meals with high hydrostatic pressures. Molecules 2020, 25, 5366.
- Brishti, F.; Zarei, M.; Muhammad, S.; Ismail-Fitry, M.; Shukri, R.; Saari, N. Evaluation of the functional properties of mung bean protein isolate for development of textured vegetable protein. Int. Food Res. J. 2017, 24, 1595–1605.
- Malik, M.A.; Saini, C.S. Gamma irradiation of alkali extracted protein isolate from dephenolized sunflower meal. LWT 2017, 84, 204–211.
- Asgar, M.; Fazilah, A.; Huda, N.; Bhat, R.; Karim, A. Nonmeat protein alternatives as meat extenders and meat analogs. Compr. Rev. Food Sci. Food Saf. 2010, 9, 513–529.
- Azzollini, D.; Derossi, A.; Fogliano, V.; Lakemond, C.M.M.; Severini, C. Effects of formulation and process conditions on microstructure, texture and digestibility of extruded insect-riched snacks. Innov. Food Sci. Emerg. Technol. 2018, 45, 344–353.
- Duda, A.; Adamczak, J.; Chełmińska, P.; Juszkiewicz, J.; Kowalczewski, P. Quality and nutritional/textural properties of durum wheat pasta enriched with cricket powder. Foods 2019, 8, 46.
- Tajudeen, A.L. Halal Certification of Insect-Based Food: A critique. Int. J. Islam. Bus. Ethics 2020, 5, 100–112.





