
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Khaled M. Aboshanab | -- | 3380 | 2023-11-02 09:24:28 | | | |
| 2 | Lindsay Dong | Meta information modification | 3380 | 2023-11-06 01:40:09 | | |
Video Upload Options
Marine actinomycetes produce a multitude of active metabolites, some of which acquire antifouling properties. These antifouling compounds have chemical structures that fall under the terpenoids, polyketides, furanones, and alkaloids chemical groups. These compounds demonstrate eminent antimicrobial vigor associated with antiquorum sensing and antibiofilm potentialities against both Gram-positive and -negative bacteria. They have also constrained larval settlements and the acetylcholinesterase enzyme, suggesting a strong anti-macrofouling activity. Despite their promising in vitro and in vivo biological activities, scaled-up production of natural antifouling agents retrieved from marine actinomycetes remains inapplicable and challenging. This might be attributed to their relatively low yield, the unreliability of in vitro tests, and the need for optimization before scaled-up manufacturing.
1. Introduction
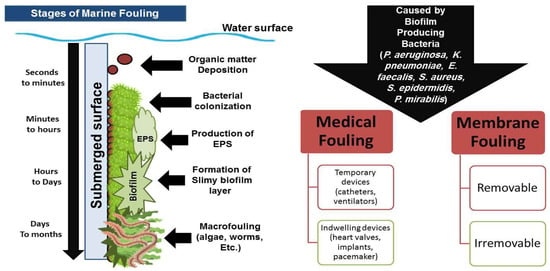
2. Fouling
3. Synthetic Antifouling Strategies and Toxicities Associated with Conventional Antifouling Coatings
4. Natural Antifouling Agents (AFs) as an Alternative to Synthetic Antifouling Coatings
5. Marine Actinomycetes as Sources of Natural Antifouling Agents
Actinomycetes, in particular, marine actinomyctes, are highly important industrial sources of secondary metabolites that include a wide range of antimicrobial, antibacterial, and antifouling agents. They belong to the Gram-positive bacterial order Actinomycetales and display a wide range of distinctive features, such as habitat, ideal pH, thermophilicity, and moisture tolerance. They interact with a wide range of aquatic animals, including invertebrates, like sponges, corals, and echinoderms, as well as vertebrates, like pufferfish corals, and a variety of invertebrates [76][77]. The evolution of secondary metabolic pathways may be influenced by these interactions, which may promote particular chemical ecologies. Although most strains have been identified from sediments, marine actinomycetes can coexist with other species and live in both planktonic and biofilm habitats [13].
5.1. Antifouling Agents from the Streptomycetaceae Family
5.2. Antifouling Agents from Micromonosporaceae, Nocardiaceae, and Pseudonocardiaceae Families
6. Obstacles Facing Commercial Use of Natural Antifouling Agents
7. Conclusions
References
- Al-Juboori, R.A.; Yusaf, T. Biofouling in RO system: Mechanisms, monitoring and controlling. Desalination 2012, 302, 1–23.
- Luoma, E.; Laurila-Pant, M.; Altarriba, E.; Nevalainen, L.; Helle, I.; Granhag, L.; Lehtiniemi, M.; Srėbalienė, G.; Olenin, S.; Lehikoinen, A. A multi-criteria decision analysis model for ship biofouling management in the Baltic Sea. Sci. Total Environ. 2022, 852, 158316.
- Qiu, H.; Feng, K.; Gapeeva, A.; Meurisch, K.; Kaps, S.; Li, X.; Yu, L.; Mishra, Y.K.; Adelung, R.; Baum, M. Functional polymer materials for modern marine biofouling control. Prog. Polym. Sci. 2022, 127, 101516.
- Giraud, T. Biological globalisation: Bioinvasions and their impacts on nature, the economy and public health. Environ. Sci. 2008, 5, 214–216.
- Callow, J.A.; Callow, M.E. Trends in the development of environmentally friendly fouling-resistant marine coatings. Nat. Commun. 2011, 2, 244.
- Srinivasan, M.; Swain, G.W. Managing the use of copper-based antifouling paints. Environ. Manag. 2007, 39, 423–441.
- Yebra, D.M.; Kiil, S.; Dam-Johansen, K. Antifouling technology—Past, present and future steps towards efficient and environmentally friendly antifouling coatings. Prog. Org. Coat. 2004, 50, 75–104.
- Brady, R.F. Fouling-release coatings for warships. Def. Sci. J. 2005, 55, 75.
- Amara, I.; Miled, W.; Slama, R.B.; Ladhari, N. Antifouling processes and toxicity effects of antifouling paints on marine environment. A review. Environ. Toxicol. Pharmacol. 2018, 57, 115–130.
- Arrhenius, A.; Backhaus, T.; Grönvall, F.; Junghans, M.; Scholze, M.; Blanck, H. Effects of three antifouling agents on algal communities and algal reproduction: Mixture toxicity studies with TBT, Irgarol, and Sea-Nine. Arch. Environ. Contam. Toxicol. 2006, 50, 335–345.
- Qian, P.Y.; Chen, L.; Xu, Y. Mini-review: Molecular mechanisms of antifouling compounds. Biofouling 2013, 29, 381–400.
- Fusetani, N. Biofouling and antifouling. Nat. Prod. Rep. 2004, 21, 94–104.
- Qian, P.Y.; Li, Z.; Xu, Y.; Li, Y.; Fusetani, N. Mini-review: Marine natural products and their synthetic analogs as antifouling compounds: 2009–2014. Biofouling 2015, 31, 101–122.
- Jagannathan, S.V.; Manemann, E.M.; Rowe, S.E.; Callender, M.C.; Soto, W. Marine actinomycetes, new sources of biotechnological products. Mar. Drugs 2021, 19, 365.
- Ghanem, N.B.; Sabry, S.A.; El-Sherif, Z.M.; El-Ela, G.A. Isolation and enumeration of marine actinomycetes from seawater and sediments in Alexandria. J. Gen. Appl. Microbiol. 2000, 46, 105–111.
- Sarkar, G.; Suthindhiran, K. Diversity and biotechnological potential of marine actinomycetes from india. Indian. J. Microbiol. 2022, 62, 475–493.
- Almeida, J.R.; Vasconcelos, V. Natural antifouling compounds: Effectiveness in preventing invertebrate settlement and adhesion. Biotechnol. Adv. 2015, 33, 343–357.
- Wang, K.L.; Wu, Z.H.; Wang, Y.; Wang, C.Y.; Xu, Y. Mini-Review: Antifouling natural products from marine microorganisms and their synthetic analogs. Mar. Drugs 2017, 15, 266.
- Levy, J.L.; Angel, B.M.; Stauber, J.L.; Poon, W.L.; Simpson, S.L.; Cheng, S.H.; Jolley, D.F. Uptake and internalisation of copper by three marine microalgae: Comparison of copper-sensitive and copper-tolerant species. Aquat. Toxicol. 2008, 89, 82–93.
- Qi, S.H.; Ma, X. Antifouling compounds from marine invertebrates. Mar. Drugs 2017, 15, 263.
- Banerjee, I.; Pangule, R.C.; Kane, R.S. Antifouling coatings: Recent developments in the design of surfaces that prevent fouling by proteins, bacteria, and marine organisms. Adv. Mater. 2011, 23, 690–718.
- Bixler, G.D.; Bhushan, B. Biofouling: Lessons from nature. Philos. Trans. R. Soc. A 2012, 370, 2381–2417.
- Kazemzadeh-Narbat, M.; Cheng, H.; Chabok, R.; Alvarez, M.M.; De La Fuente-Nunez, C.; Phillips, K.S.; Khademhosseini, A. Strategies for antimicrobial peptide coatings on medical devices: A review and regulatory science perspective. Crit. Rev. Biotechnol. 2021, 41, 94–120.
- Wi, Y.M.; Patel, R. Understanding biofilms and novel approaches to the diagnosis, prevention, and treatment of medical device-associated infections. Infect. Dis. Clin. N. Am. 2018, 32, 915–929.
- Mirghani, R.; Saba, T.; Khaliq, H.; Mitchell, J.; Do, L.; Chambi, L.; Diaz, k.; Kennedy, T.; Alkassab, K.; Huynh, T. Biofilms: Formation, drug resistance and alternatives to conventional approaches. AIMS Microbiol. 2022, 8, 239.
- Liu, D.; Shu, H.; Zhou, J.; Bai, X.; Cao, P. Research Progress on New Environmentally Friendly Antifouling Coatings in Marine Settings: A Review. Biomimetics 2023, 8, 200.
- Ping Chu, H.; Li, X.Y. Membrane fouling in a membrane bioreactor (MBR): Sludge cake formation and fouling characteristics. Biotechnol. Bioeng. 2005, 90, 323–331.
- Chen, L.; Qian, P.Y. Review on molecular mechanisms of antifouling compounds: An update since 2012. Mar. Drugs 2017, 15, 264.
- Beg, M.A.; Beg, M.A.; Zargar, U.R.; Sheikh, I.A.; Bajouh, O.S.; Abuzenadah, A.M.; Rehan, M. Organotin antifouling compounds and sex-steroid nuclear receptor perturbation: Some structural insights. Toxic 2022, 11, 25.
- Romeu, M.J.; Mergulhão, F. Development of antifouling strategies for marine applications. Microorganisms 2023, 11, 1568.
- Alzieu, C. Environmental problems caused by TBT in France: Assessment, regulations, prospects. Mar. Environ. Res. 1991, 32, 7–17.
- Waite, M.E.; Waldock, M.J.; Thain, J.E.; Smith, D.J.; Milton, S.M. Reductions in TBT concentrations in UK estuaries following legislation in 1986 and 1987. Mar. Environ. Res. 1991, 32, 89–111.
- Chesworth, J.C.; Donkin, M.E.; Brown, M.T. The interactive effects of the antifouling herbicides Irgarol 1051 and Diuron on the seagrass Zostera marina (L.). Aquat. Toxicol. 2004, 66, 293–305.
- Soon, Z.Y.; Jung, J.H.; Jang, M.; Kang, J.H.; Jang, M.C.; Lee, J.S.; Lee, J.S.; Kim, M. Zinc Pyrithione (ZnPT) as an antifouling biocide in the marine environment—A Literature Review of Its Toxicity, Environmental Fates, and Analytical Methods. Wat. Air Soil. Poll. 2019, 230, 310.
- Jiang, C.; Wang, G.; Hein, R.; Liu, N.; Luo, X.; Davis, J.J. Antifouling strategies for selective in vitro and in vivo sensing. Chem. Rev. 2020, 120, 3852–3889.
- Veerachamy, S.; Yarlagadda, T.; Manivasagam, G.; Yarlagadda, P.K. Bacterial adherence and biofilm formation on medical implants: A review. Proc. Inst. Mech. Eng. 2014, 228, 1083–1099.
- Li, F.; Huang, T.; Pasic, P.; Easton, C.D.; Voelcker, N.H.; Heath, D.E.; O’Brien-Simpson, N.M.; O’Connor, A.J.; Thissen, H. One step antimicrobial coatings for medical device applications based on low fouling polymers containing selenium nanoparticles. J. Chem. Eng. 2023, 467, 143546.
- Ozkan, E.; Mondal, A.; Douglass, M.; Hopkins, S.P.; Garren, M.; Devine, R.; Pandey, R.; Manuel, J.; Singha, P.; Warnock, J.; et al. Bioinspired ultra-low fouling coatings on medical devices to prevent device-associated infections and thrombosis. J. Colloid. Interface Sci. 2022, 608, 1015–1024.
- Haugen, H.J.; Makhtari, S.; Ahmadi, S.; Hussain, B. The antibacterial and cytotoxic effects of silver nanoparticles coated titanium implants: A narrative review. Materials 2022, 15, 5025.
- Yazdani, J.; Ahmadian, E.; Sharifi, S.; Shahi, S.; Dizaj, S.M. A short view on nanohydroxyapatite as coating of dental implants. Biomed. Pharmacother. 2018, 105, 553–557.
- Khatoon, Z.; McTiernan, C.D.; Suuronen, E.J.; Mah, T.F.; Alarcon, E.I. Bacterial biofilm formation on implantable devices and approaches to its treatment and prevention. Heliyon 2018, 4, e01067.
- Gupta, P.; Sarkar, S.; Das, B.; Bhattacharjee, S.; Tribedi, P. Biofilm, pathogenesis and prevention—A journey to break the wall: A review. Arch. Microbiol. 2016, 198, 1–15.
- Uppal, G.; Thakur, A.; Chauhan, A.; Bala, S. Magnesium based implants for functional bone tissue regeneration–A review. J. Magnes. Alloy 2022, 10, 356–386.
- Rahim, S.A.; Joseph, M.; Sampath Kumar, T. Recent progress in surface modification of Mg alloys for biodegradable orthopedic applications. Front. Mater. 2022, 9, 848980.
- Riedel, T.; Riedelová-Reicheltová, Z.; Májek, P.; Rodriguez-Emmenegger, C.; Houska, M.; Dyr, J.E.; Houska, M.; Dyr, J.E.; Brynda, E. Complete identification of proteins responsible for human blood plasma fouling on poly (ethylene glycol)-based surfaces. Langmuir 2013, 29, 3388–3397.
- Asha, A.B.; Chen, Y.; Narain, R. Bioinspired dopamine and zwitterionic polymers for non-fouling surface engineering. Chem. Soc. Rev. 2021, 50, 11668–11683.
- Yi, E.; Kang, H.S.; Lim, S.M.; Heo, H.J.; Han, D.; Kim, J.F.; Park, A.; Choi, D.H.; Park, Y.I.; Park, H.; et al. Superamphiphobic blood-repellent surface modification of porous fluoropolymer membranes for blood oxygenation applications. J. Membr. Sci. 2022, 648, 120363.
- Zhang, Z.Q.; Ren, K.F.; Ji, J. Silane coupling agent in biomedical materials. Biointerphases 2023, 18, 030801.
- Zhang, W.; Yang, Z.; Kaufman, Y.; Bernstein, R. Surface and anti-fouling properties of a polyampholyte hydrogel grafted onto a polyethersulfone membrane. J. Colloid. Interface Sci. 2018, 517, 155–165.
- Klemm, S.; Baum, M.; Qiu, H.; Nan, Z.; Cavalheiro, M.; Teixeira, M.C.; Tendero, C.; Gapeeva, A.; Adelung, R.; Dague, E. Development of polythiourethane/ZnO-based anti-fouling materials and evaluation of the adhesion of Staphylococcus aureus and Candida glabrata using single-cell force spectroscopy. Nanomaterials 2021, 11, 271.
- Gu, J.; Li, L.; Huang, D.; Jiang, L.; Liu, L.; Li, F.; Pang, A.; Guo, X.; Tao, B. In situ synthesis of grapheme cuprous oxide nanocomposite incorporated marine antifouling coating with elevated antifouling performance. Open J. Org. Polym. Mater. 2019, 9, 47–62.
- Castro, J.D.; Lima, M.J.; Carvalho, S. Wetting and corrosion properties of CuxOy films deposited by magnetron sputtering for maritime applications. Appl. Surf. Sci. 2022, 584, 152582.
- Zeng, H.; Xie, Q.; Ma, C.; Zhang, G. Silicone elastomer with surface-enriched, nonleaching amphiphilic side chains for inhibiting marine biofouling. ACS Appl. Polym. Mater. 2019, 1, 1689–1696.
- Kolle, S.; Ahanotu, O.; Meeks, A.; Stafslien, S.; Kreder, M.; Vanderwal, L.; Cohen, L.; Waltz, G.; Lim, C.S.; Slocum, D. On the mechanism of marine fouling-prevention performance of oil-containing silicone elastomers. Sci. Rep. 2022, 12, 11799.
- Seo, E.; Lee, J.W.; Lee, D.; Seong, M.R.; Kim, G.H.; Hwang, D.S.; Lee, S.J. Eco-friendly erucamide–polydimethylsiloxane coatings for marine anti-biofouling. Colloids Surf. B 2021, 207, 112003.
- Long, Y.; Yin, X.; Mu, P.; Wang, Q.; Hu, J.; Li, J. Slippery liquid-infused porous surface (SLIPS) with superior liquid repellency, anti-corrosion, anti-icing and intensified durability for protecting substrates. Chem. Eng. J. 2020, 401, 126137.
- Yandi, W.; Mieszkin, S.; di Fino, A.; Martin-Tanchereau, P.; Callow, M.E.; Callow, J.A.; Tyson, L.; Clare, A.S.; Ederth, T. Charged hydrophilic polymer brushes and their relevance for understanding marine biofouling. Biofouling 2016, 32, 609–625.
- Li, J.; Xie, Z.; Wang, G.; Ding, C.; Jiang, H.; Wang, P. Preparation and evaluation of amphiphilic polymer as fouling-release coating in marine environment. J. Coat. Technol. Res. 2017, 14, 1237–1245.
- Villardi de Oliveira, C.; Petitbois, J.; Fay, F.; Sanchette, F.; Schuster, F.; Alhussein, A.; Schuster, F.; Alhussein, A.; Chaix-Pluchery, O.; Deschanvres, J.L.; et al. Marine antibiofouling properties of TiO2 and Ti-Cu-O films deposited by aerosol-assisted chemical vapor deposition. Coatings 2020, 10, 779.
- Statz, A.; Finlay, J.; Dalsin, J.; Callow, M.; Callow, J.A.; Messersmith, P.B. Algal antifouling and fouling-release properties of metal surfaces coated with a polymer inspired by marine mussels. Biofouling 2006, 22, 391–399.
- Fyrner, T.; Lee, H.H.; Mangone, A.; Ekblad, T.; Pettitt, M.E.; Callow, M.E.; Callow, J.A.; Conlan, S.L.; Mutton, R.; Clare, A.S. Saccharide-functionalized alkanethiols for fouling-resistant self-assembled monolayers: Synthesis, monolayer properties, and antifouling behavior. Langmuir 2011, 27, 15034–15047.
- Lambropoulou, D.A.; Sakkas, V.A.; Albanis, T.A. Headspace solid phase microextraction for the analysis of the new antifouling agents Irgarol 1051 and Sea Nine 211 in natural waters. Anal. Chim. Acta 2002, 468, 171–180.
- Pittol, M.; Tomacheski, D.; Simões, D.N.; Ribeiro, V.F.; Santana, R.M.C. Antimicrobial performance of thermoplastic elastomers containing zinc pyrithione and silver nanoparticles. Mater. Res. 2017, 20, 1266–1273.
- Fusetani, N. Antifouling marine natural products. Nat. Prod. Rep. 2011, 28, 400–410.
- De Nys, R.; Leya, T.; Maximilien, R.; Afsar, A.; Nair, P.; Steinberg, P. The need for standardised broad scale bioassay testing: A case study using the red alga Laurencia rigida. Biofouling 1996, 10, 213–224.
- König, G.M.; Wright, A.D. Laurencia rigida: Chemical investigations of its antifouling dichloromethane extract. J. Nat. Prod. 1997, 60, 967–970.
- Lau, S.C.; Qian, P.Y. Phlorotannins and related compounds as larval settlement inhibitors of the tube-building polychaete Hydroides elegans. Mar. Ecol. Prog. Ser. 1997, 159, 219–227.
- Qi, S.; Zhang, S.; Qian, P.; Xu, H. Antifeedant and antifouling briaranes from the South China Sea gorgonian Junceella juncea. Chem. Nat. Compd. 2009, 45, 49–54.
- Bianco, É.M.; Rogers, R.; Teixeira, V.L.; Pereira, R.C. Antifoulant diterpenes produced by the brown seaweed Canistrocarpus cervicornis. J. Appl. Phycol. 2009, 21, 341–346.
- Gozari, M.; Alborz, M.; El-Seedi, H.R.; Jassbi, A.R. Chemistry, biosynthesis and biological activity of terpenoids and meroterpenoids in bacteria and fungi isolated from different marine habitats. Eur. J. Med. Chem. 2021, 210, 112957.
- Núñez-Pons, L.; Shilling, A.; Verde, C.; Baker, B.J.; Giordano, D. Marine terpenoids from polar latitudes and their potential applications in biotechnology. Mar. Drugs 2020, 18, 401.
- Kwong, T.F.N.; Miao, L.; Li, X.; Qian, P.Y. Novel antifouling and antimicrobial compound from a marine-derived fungus Ampelomyces sp. Mar. Biotechnol. 2006, 8, 634–640.
- Xing, Q.; Gan, L.S.; Mou, X.F.; Wang, W.; Wang, C.Y.; Wei, M.Y.; Wang, C.Y.; Wei, M.Y.; Shao, C.L. Isolation, resolution and biological evaluation of pestalachlorides E and F containing both point and axial chirality. RSC Adv. 2016, 6, 22653–22658.
- Shao, C.L.; Xu, R.F.; Wang, C.Y.; Qian, P.Y.; Wang, K.L.; Wei, M.Y. Potent antifouling marine dihydroquinolin-2(1h)-one-containing alkaloids from the gorgonian coral-derived fungus Scopulariopsis sp. Mar. Biotechnol. 2015, 17, 408–415.
- Pérez, M.; Pis Diez, C.M.; Belén Valdez, M.; García, M.; Paola, A.; Avigliano, E.; Palermo, J.A.; Blustein, G. Isolation and antimacrofouling activity of indole and furoquinoline alkaloids from ‘Guatambú’ trees (Aspidosperma australe and Balfourodendron riedelianum). Chem. Biodivers. 2019, 16, e1900349.
- ELnahas, M.; Elkhateeb, W.; Daba, G. Marine actinomycetes the past, the present and the future. Pharm. Res. 2021, 5, 000241.
- Wu, Z.; Xie, L.; Xia, G.; Zhang, J.; Nie, Y.; Hu, J.; Wang, S.; Zhang, R. A new tetrodotoxin-producing actinomycete, Nocardiopsis dassonvillei, isolated from the ovaries of puffer fish Fugu rubripes. Toxicon 2005, 45, 851–859.
- Taddei, A.; Rodríguez, M.J.; Márquez-Vilchez, E.; Castelli, C. Isolation and identification of Streptomyces spp. from Venezuelan soils: Morphological and biochemical studies. Microbiol. Res. 2006, 161, 222–231.
- Barka, E.A.; Vatsa, P.; Sanchez, L.; Gaveau-Vaillant, N.; Jacquard, C.; Klenk, H.P.; Clément, C.; Ouhdouch, Y.; van Wezel, G.P. Taxonomy, physiology, and natural products of Actinobacteria. Microbiol. Mol. Biol. Rev. 2016, 80, 1–43.
- Bafghi, M.F.; Heidarieh, P.; Soori, T.; Saber, S.; Meysamie, A.; Gheitoli, K.; Habibnia, S.; Nasab, M.R.; Eshraghi, S.S. Nocardia isolation from clinical samples with the paraffin baiting technique. Germs 2015, 5, 12.
- Teo, W.F.A.; Tan, G.Y.A.; Li, W.J. Taxonomic note on the family Pseudonocardiaceae based on phylogenomic analysis and descriptions of Allosaccharopolyspora gen. nov. and Halosaccharopolyspora gen. nov. Int. J. Syst. Evol. Microbiol. 2021, 71, 005075.
- Takamura, H.; Kinoshita, Y.; Yorisue, T.; Kadota, I. Chemical synthesis and antifouling activity of monoterpene–furan hybrid molecules. Org. Biomol. Chem. 2023, 21, 632–638.
- Takamura, H.; Ohashi, T.; Kikuchi, T.; Endo, N.; Fukuda, Y.; Kadota, I. Late-stage divergent synthesis and antifouling activity of geraniol–butenolide hybrid molecules. Org. Biomol. Chem. 2017, 15, 5549–5555.
- Moodie, L.W.; Cervin, G.; Trepos, R.; Labriere, C.; Hellio, C.; Pavia, H.; Svenson, J. Design and biological evaluation of antifouling dihydrostilbene oxime hybrids. Mar. Biotechnol. 2018, 20, 257–267.
- Kato, T.; Sato, T.; Kashiwagi, Y.; Hosokawa, S. Synthetic studies on aculeximycin: Synthesis of C24-C40 segment by Kobayashi aldolization and epoxide rearrangements. Org. Lett. 2015, 17, 2274–2277.
- Kim, T.; Kim, Y.J.; Jeong, K.H.; Park, Y.T.; Kwon, H.; Choi, P.; Ju, H.N.; Yoon, C.H.; Kim, J.Y.; Ham, J. The efficient synthesis and biological evaluation of justicidin B. Nat. Prod. Res. 2023, 37, 56–62.
- Qian, P.Y.; Xu, Y.; Fusetani, N. Natural products as antifouling compounds: Recent progress and future perspectives. Biofouling 2009, 26, 223–234.





