
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Raphaël Duval | -- | 2752 | 2023-10-23 14:07:20 | | | |
| 2 | Alfred Zheng | Meta information modification | 2752 | 2023-10-24 03:25:44 | | |
Video Upload Options
Amphotericin B is the oldest antifungal molecule which is still currently widely used in clinical practice. Recently, its redox properties (i.e., both prooxidant and antioxidant) have been highlighted in the literature as mechanisms involved in both its activity and its toxicity. Interestingly, similar properties can be described for inorganic nanoparticles. Moreover, in a majority of the published studies, inorganic nanoparticles carrying Amphotericin B are described as having a synergistic activity directly related to the rupture of the redox homeostasis of the pathogen (i.e., fungi, parasites, and yeasts). Consequently, due to the unique properties of inorganic nanoparticles (e.g., magnetism, intrinsic anti-infectious properties, stimuli-triggered responses, etc.), these nanomaterials (i.e., inorganic nanoparticles carrying Amphotericin B) may represent a new generation of medicine that can synergistically enhance the antimicrobial properties of Amphotericin B.
1. Introduction
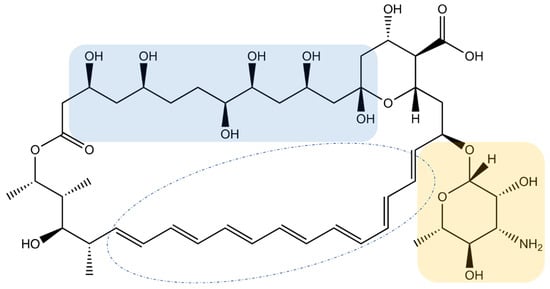
2. Inorganic Nanoparticles Carrying Amphotericin B
2.1. The State-of-the-Art of Lipidic Formulations of Amphotericin B on the Market or under Clinical Trials
2.2. Inorganic Nanoparticles as Modulator of AmB Redox Properties
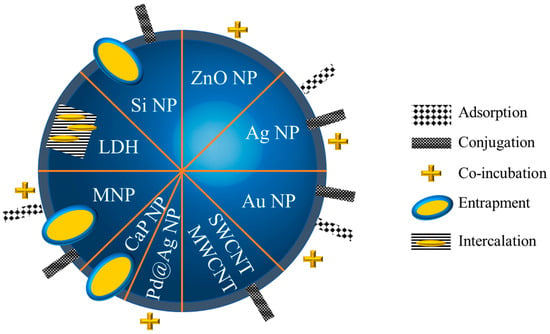
2.2.1. Strategies to Functionalize Inorganic Nanoparticles with Amphotericin B
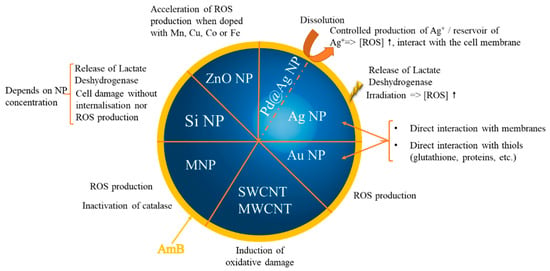
2.2.2. Inorganic Nanoparticles as Synergic Prooxidants
2.2.3. Inorganic Nanoparticles as Synergic Antioxidants
References
- Nicolaou, K.C.; Chen, J.S.; Dalby, S.M. From Nature to the Laboratory and into the Clinic. Bioorg. Med. Chem. 2009, 17, 2290–2303.
- Volmer, A.A.; Szpilman, A.M.; Carreira, E.M. Synthesis and Biological Evaluation of Amphotericin B Derivatives. Nat. Prod. Rep. 2010, 27, 1329–1349.
- Pound, M.W.; Townsend, M.L.; Dimondi, V.; Wilson, D.; Drew, R.H. Overview of Treatment Options for Invasive Fungal Infections. Med. Mycol. 2011, 49, 561–580.
- Cavassin, F.B.; Baú-Carneiro, J.L.; Vilas-Boas, R.R.; Queiroz-Telles, F. Sixty Years of Amphotericin B: An Overview of the Main Antifungal Agent Used to Treat Invasive Fungal Infections. Infect. Dis. Ther. 2021, 10, 115–147.
- Carolus, H.; Pierson, S.; Lagrou, K.; Van Dijck, P. Amphotericin B and Other Polyenes-Discovery, Clinical Use, Mode of Action and Drug Resistance. J. Fungi 2020, 6, 321.
- Cowen, L.E.; Sanglard, D.; Howard, S.J.; Rogers, P.D.; Perlin, D.S. Mechanisms of Antifungal Drug Resistance. Cold Spring Harb. Perspect. Med. 2015, 5, a019752.
- Brunet, K.; Diop, C.A.B.; Chauzy, A.; Prébonnaud, N.; Marchand, S.; Rammaert, B.; Tewes, F. Improved In Vitro Anti-Mucorales Activity and Cytotoxicity of Amphotericin B with a Pegylated Surfactant. J. Fungi 2022, 8, 121.
- Pappas, P.G.; Kauffman, C.A.; Andes, D.R.; Clancy, C.J.; Marr, K.A.; Ostrosky-Zeichner, L.; Reboli, A.C.; Schuster, M.G.; Vazquez, J.A.; Walsh, T.J.; et al. Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2016, 62, e1–e50.
- Pathak, A.; Pien, F.D.; Carvalho, L. Amphotericin B Use in a Community Hospital, with Special Emphasis on Side Effects. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 1998, 26, 334–338.
- Gursoy, V.; Ozkalemkas, F.; Ozkocaman, V.; Serenli Yegen, Z.; Ethem Pinar, I.; Ener, B.; Akalın, H.; Kazak, E.; Ali, R.; Ersoy, A. Conventional Amphotericin B Associated Nephrotoxicity in Patients With Hematologic Malignancies. Cureus 2021, 13, e16445.
- Jafari, M.; Abolmaali, S.S.; Tamaddon, A.M.; Zomorodian, K.; Sarkari, B.S. Nanotechnology Approaches for Delivery and Targeting of Amphotericin B in Fungal and Parasitic Diseases. Nanomedicine 2021, 16, 857–877.
- Alakkad, A.; Stapleton, P.; Schlosser, C.; Murdan, S.; Odunze, U.; Schatzlein, A.; Uchegbu, I.F. Amphotericin B Polymer Nanoparticles Show Efficacy against Candida Species Biofilms. Pathogens 2022, 11, 73.
- Faustino, C.; Pinheiro, L. Lipid Systems for the Delivery of Amphotericin B in Antifungal Therapy. Pharmaceutics 2020, 12, 29.
- Chen, Y.-C.; Su, C.-Y.; Jhan, H.-J.; Ho, H.-O.; Sheu, M.-T. Physical Characterization and in Vivo Pharmacokinetic Study of Self-Assembling Amphotericin B-Loaded Lecithin-Based Mixed Polymeric Micelles. Int. J. Nanomed. 2015, 10, 7265–7274.
- Serrano, D.R.; Lalatsa, A.; Dea-Ayuela, M.A.; Bilbao-Ramos, P.E.; Garrett, N.L.; Moger, J.; Guarro, J.; Capilla, J.; Ballesteros, M.P.; Schätzlein, A.G.; et al. Oral Particle Uptake and Organ Targeting Drives the Activity of Amphotericin B Nanoparticles. Mol. Pharm. 2015, 12, 420–431.
- Wang, C.; Makvandi, P.; Zare, E.N.; Tay, F.R.; Niu, L. Advances in Antimicrobial Organic and Inorganic Nanocompounds in Biomedicine. Adv. Ther. 2020, 3, 2000024.
- Jiang, S.; Win, K.Y.; Liu, S.; Teng, C.P.; Zheng, Y.; Han, M.-Y. Surface-Functionalized Nanoparticles for Biosensing and Imaging-Guided Therapeutics. Nanoscale 2013, 5, 3127–3148.
- Huang, H.; Feng, W.; Chen, Y.; Shi, J. Inorganic Nanoparticles in Clinical Trials and Translations. Nano Today 2020, 35, 100972.
- Liu, M.; Chen, M.; Yang, Z. Design of Amphotericin B Oral Formulation for Antifungal Therapy. Drug Deliv. 2017, 24, 1–9.
- Voltan, A.R.; Quindós, G.; Alarcón, K.P.M.; Fusco-Almeida, A.M.; Mendes-Giannini, M.J.S.; Chorilli, M. Fungal Diseases: Could Nanostructured Drug Delivery Systems Be a Novel Paradigm for Therapy? Int. J. Nanomed. 2016, 11, 3715–3730.
- Bekersky, I.; Fielding, R.M.; Buell, D.; Lawrence, I. Lipid-Based Amphotericin B Formulations: From Animals to Man. Pharm. Sci. Technol. Today 1999, 2, 230–236.
- Fernández-García, R.; de Pablo, E.; Ballesteros, M.P.; Serrano, D.R. Unmet Clinical Needs in the Treatment of Systemic Fungal Infections: The Role of Amphotericin B and Drug Targeting. Int. J. Pharm. 2017, 525, 139–148.
- Zager, R.A. Polyene Antibiotics: Relative Degrees of in Vitro Cytotoxicity and Potential Effects on Tubule Phospholipid and Ceramide Content. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2000, 36, 238–249.
- Hnik, P.; Wasan, E.K.; Wasan, K.M. Safety, Tolerability, and Pharmacokinetics of a Novel Oral Amphotericin B Formulation (iCo-019) Following Single-Dose Administration to Healthy Human Subjects: An Alternative Approach to Parenteral Amphotericin B Administration. Antimicrob. Agents Chemother. 2020, 64, e01450-20.
- Ahmad, A.; Wei, Y.; Syed, F.; Tahir, K.; Taj, R.; Khan, A.U.; Hameed, M.U.; Yuan, Q. Amphotericin B-Conjugated Biogenic Silver Nanoparticles as an Innovative Strategy for Fungal Infections. Microb. Pathog. 2016, 99, 271–281.
- Anwar, A.; Siddiqui, R.; Raza Shah, M.; Ahmed Khan, N. Gold Nanoparticles Conjugation Enhances Antiacanthamoebic Properties of Nystatin, Fluconazole and Amphotericin B. J. Microbiol. Biotechnol. 2019, 29, 171–177.
- Ahmadpour Kermani, S.; Salari, S.; Ghasemi Nejad Almani, P. Comparison of Antifungal and Cytotoxicity Activities of Titanium Dioxide and Zinc Oxide Nanoparticles with Amphotericin B against Different Candida Species: In Vitro Evaluation. J. Clin. Lab. Anal. 2021, 35, e23577.
- Chintalacharuvu, K.R.; Matolek, Z.A.; Pacheco, B.; Carriera, E.M.; Beenhouwer, D.O. Complexing Amphotericin B with Gold Nanoparticles Improves Fungal Clearance from the Brains of Mice Infected with Cryptococcal Neoformans. Med. Mycol. 2021, 59, 1085–1091.
- Patra, J.K.; Baek, K.-H. Green Biosynthesis of Magnetic Iron Oxide (Fe3O4) Nanoparticles Using the Aqueous Extracts of Food Processing Wastes under Photo-Catalyzed Condition and Investigation of Their Antimicrobial and Antioxidant Activity. J. Photochem. Photobiol. B 2017, 173, 291–300.
- Patra, J.K.; Baek, K.-H. Comparative Study of Proteasome Inhibitory, Synergistic Antibacterial, Synergistic Anticandidal, and Antioxidant Activities of Gold Nanoparticles Biosynthesized Using Fruit Waste Materials. Int. J. Nanomed. 2016, 11, 4691–4705.
- Souza, J.A.S.; Alves, M.M.; Barbosa, D.B.; Lopes, M.M.; Pinto, E.; Figueiral, M.H.; Delbem, A.C.B.; Mira, N.P. Study of the Activity of Punica Granatum-Mediated Silver Nanoparticles against Candida Albicans and Candida Glabrata, Alone or in Combination with Azoles or Polyenes. Med. Mycol. 2020, 58, 564–567.
- Wypij, M.; Czarnecka, J.; Dahm, H.; Rai, M.; Golinska, P. Silver Nanoparticles from Pilimelia Columellifera Subsp. Pallida SL19 Strain Demonstrated Antifungal Activity against Fungi Causing Superficial Mycoses. J. Basic Microbiol. 2017, 57, 793–800.
- Nadhe, S.B.; Singh, R.; Wadhwani, S.A.; Chopade, B.A. Acinetobacter Sp. Mediated Synthesis of AgNPs, Its Optimization, Characterization and Synergistic Antifungal Activity against C. Albicans. J. Appl. Microbiol. 2019, 127, 445–458.
- Tutaj, K.; Szlazak, R.; Szalapata, K.; Starzyk, J.; Luchowski, R.; Grudzinski, W.; Osinska-Jaroszuk, M.; Jarosz-Wilkolazka, A.; Szuster-Ciesielska, A.; Gruszecki, W.I. Amphotericin B-Silver Hybrid Nanoparticles: Synthesis, Properties and Antifungal Activity. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 1095–1103.
- Sadat Akhavi, S.; Moradi Dehaghi, S. Drug Delivery of Amphotericin B through Core-Shell Composite Based on PLGA/Ag/Fe3O4: In Vitro Test. Appl. Biochem. Biotechnol. 2020, 191, 496–510.
- Kumar, P.; Shivam, P.; Mandal, S.; Prasanna, P.; Kumar, S.; Prasad, S.R.; Kumar, A.; Das, P.; Ali, V.; Singh, S.K.; et al. Synthesis, Characterization, and Mechanistic Studies of a Gold Nanoparticle-Amphotericin B Covalent Conjugate with Enhanced Antileishmanial Efficacy and Reduced Cytotoxicity. Int. J. Nanomed. 2019, 14, 6073–6101.
- Wu, W.; Wieckowski, S.; Pastorin, G.; Benincasa, M.; Klumpp, C.; Briand, J.-P.; Gennaro, R.; Prato, M.; Bianco, A. Targeted Delivery of Amphotericin B to Cells by Using Functionalized Carbon Nanotubes. Angew. Chem. Int. Ed Engl. 2005, 44, 6358–6362.
- Trikeriotis, M.; Ghanotakis, D.F. Intercalation of Hydrophilic and Hydrophobic Antibiotics in Layered Double Hydroxides. Int. J. Pharm. 2007, 332, 176–184.
- Ahmad, A.; Wei, Y.; Syed, F.; Khan, S.; Khan, G.M.; Tahir, K.; Khan, A.U.; Raza, M.; Khan, F.U.; Yuan, Q. Isatis Tinctoria Mediated Synthesis of Amphotericin B-Bound Silver Nanoparticles with Enhanced Photoinduced Antileishmanial Activity: A Novel Green Approach. J. Photochem. Photobiol. B 2016, 161, 17–24.
- Yu, D.; Wang, L.; Zhou, H.; Zhang, X.; Wang, L.; Qiao, N. Fluorimetric Detection of Candida Albicans Using Cornstalk N-Carbon Quantum Dots Modified with Amphotericin B. Bioconjug. Chem. 2019, 30, 966–973.
- Balabathula, P.; Whaley, S.G.; Janagam, D.R.; Mittal, N.K.; Mandal, B.; Thoma, L.A.; Rogers, P.D.; Wood, G.C. Lyophilized Iron Oxide Nanoparticles Encapsulated in Amphotericin B: A Novel Targeted Nano Drug Delivery System for the Treatment of Systemic Fungal Infections. Pharmaceutics 2020, 12, E247.
- Gudz, K.Y.; Permyakova, E.S.; Matveev, A.T.; Bondarev, A.V.; Manakhov, A.M.; Sidorenko, D.A.; Filippovich, S.Y.; Brouchkov, A.V.; Golberg, D.V.; Ignatov, S.G.; et al. Pristine and Antibiotic-Loaded Nanosheets/Nanoneedles-Based Boron Nitride Films as a Promising Platform to Suppress Bacterial and Fungal Infections. ACS Appl. Mater. Interfaces 2020, 12, 42485–42498.
- Hwang, I.; Hwang, J.H.; Choi, H.; Kim, K.-J.; Lee, D.G. Synergistic Effects between Silver Nanoparticles and Antibiotics and the Mechanisms Involved. J. Med. Microbiol. 2012, 61, 1719–1726.
- Jamaran, S.; Zarif, B.R. Synergistic Effect of Silver Nanoparticles with Neomycin or Gentamicin Antibiotics on Mastitis-Causing Staphylococcus Aureus. Open J. Ecol. 2016, 6, 452–459.
- Durán, N.; Marcato, P.D.; Durán, M.; Yadav, A.; Gade, A.; Rai, M. Mechanistic Aspects in the Biogenic Synthesis of Extracellular Metal Nanoparticles by Peptides, Bacteria, Fungi, and Plants. Appl. Microbiol. Biotechnol. 2011, 90, 1609–1624.
- Janik, S.; Grela, E.; Stączek, S.; Zdybicka-Barabas, A.; Luchowski, R.; Gruszecki, W.I.; Grudzinski, W. Amphotericin B-Silver Hybrid Nanoparticles Help to Unveil the Mechanism of Biological Activity of the Antibiotic: Disintegration of Cell Membranes. Molecules 2023, 28, 4687.
- Paulo, C.S.O.; Lino, M.M.; Matos, A.A.; Ferreira, L.S. Differential Internalization of Amphotericin B--Conjugated Nanoparticles in Human Cells and the Expression of Heat Shock Protein 70. Biomaterials 2013, 34, 5281–5293.
- Paulo, C.S.O.; Vidal, M.; Ferreira, L.S. Antifungal Nanoparticles and Surfaces. Biomacromolecules 2010, 11, 2810–2817.
- Lino, M.M.; Paulo, C.S.O.; Vale, A.C.; Vaz, M.F.; Ferreira, L.S. Antifungal Activity of Dental Resins Containing Amphotericin B-Conjugated Nanoparticles. Dent. Mater. Off. Publ. Acad. Dent. Mater. 2013, 29, e252–e262.
- Alshahrani, S.M.; Khafagy, E.-S.; Riadi, Y.; Al Saqr, A.; Alfadhel, M.M.; Hegazy, W.A.H. Amphotericin B-PEG Conjugates of ZnO Nanoparticles: Enhancement Antifungal Activity with Minimal Toxicity. Pharmaceutics 2022, 14, 1646.
- Gedda, M.R.; Madhukar, P.; Vishwakarma, A.K.; Verma, V.; Kushwaha, A.K.; Yadagiri, G.; Mudavath, S.L.; Singh, O.P.; Srivastava, O.N.; Sundar, S. Evaluation of Safety and Antileishmanial Efficacy of Amine Functionalized Carbon-Based Composite Nanoparticle Appended with Amphotericin B: An in Vitro and Preclinical Study. Front. Chem. 2020, 8, 510.
- Chaurasia, M.; Singh, P.K.; Jaiswal, A.K.; Kumar, A.; Pawar, V.K.; Dube, A.; Paliwal, S.K.; Chourasia, M.K. Bioinspired Calcium Phosphate Nanoparticles Featuring as Efficient Carrier and Prompter for Macrophage Intervention in Experimental Leishmaniasis. Pharm. Res. 2016, 33, 2617–2629.
- Saldanha, C.A.; Garcia, M.P.; Iocca, D.C.; Rebelo, L.G.; Souza, A.C.O.; Bocca, A.L.; Santos, M.d.F.M.A.; Morais, P.C.; Azevedo, R.B. Antifungal Activity of Amphotericin B Conjugated to Nanosized Magnetite in the Treatment of Paracoccidioidomycosis. PLoS Negl. Trop. Dis. 2016, 10, e0004754.
- Leonhard, V.; Alasino, R.V.; Munoz, A.; Beltramo, D.M. Silver Nanoparticles with High Loading Capacity of Amphotericin B: Characterization, Bactericidal and Antifungal Effects. Curr. Drug Deliv. 2018, 15, 850–859.
- Chernousova, S.; Epple, M. Silver as Antibacterial Agent: Ion, Nanoparticle, and Metal. Angew. Chem. Int. Ed Engl. 2013, 52, 1636–1653.
- Sreeharsha, N.; Chitrapriya, N.; Jang, Y.J.; Kenchappa, V. Evaluation of Nanoparticle Drug-Delivery Systems Used in Preclinical Studies. Ther. Deliv. 2021, 12, 325–336.
- Luo, M.; Boudier, A.; Clarot, I.; Maincent, P.; Schneider, R.; Leroy, P. Gold Nanoparticles Grafted by Reduced Glutathione with Thiol Function Preservation. Colloid Interface Sci. Commun. 2016, 14, 8–12.





