
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ng Shong Li | -- | 4130 | 2023-10-12 22:07:10 | | | |
| 2 | Lindsay Dong | Meta information modification | 4130 | 2023-10-15 14:13:06 | | | | |
| 3 | Lindsay Dong | Meta information modification | 4130 | 2023-10-23 05:17:43 | | |
Video Upload Options
Cancer, also known as malignant tumour or neoplasm, is a leading cause of death worldwide. One distinct feature from normal cells is that cancerous cells often overexpress protein on the cell membrane—for instance, the overexpression of human epidermal growth factor receptor 2. The expression of a specific protein on the cancerous cell surface acts as a marker that differentiates the normal cell and facilitates the recognition of cancerous cells. An emerging anticancer treatment, Antibody–Drug Conjugates (ADCs), utilises this unique feature to kill cancerous cells. ADCs consist of an antibody linked with a cytotoxic payload, mainly targeting the antigen found on cancerous cells. This design can increase the specificity in delivering the cytotoxin to the drug target, thus increasing the drug efficacy and reducing the side effect of cancer treatment due to off-target toxicities.
1. Introduction
2. Principle of Antibody–Drug Conjugate
| Components of ADC | ||
|---|---|---|
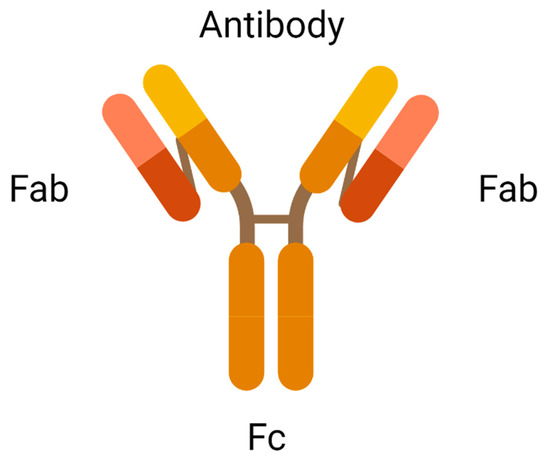
| Antibody | Subunit | Function |
| Antigen-binding Fragment (Fab) | Mediate antigen recognition expressed on tumour cells. | |
| Constant Fragment (Fc) | Facilitate binding of Fab to immune cells. | |
| Linker | Type | Mechanism of Drug Release |
| Cleavable | Cleavage depends on the physiological environment.
|
|
| Non-cleavable | It depends on lysosomal proteolytic degradation and requires optimal trafficking to lysosome. Thioether linker is cleaved by proteases in the cytosolic milieu, e.g., Trastuzumab Emtansine (T–DM1). | |
| Cytotoxic Payload | Target Site | Examples of Payload with Mechanism of Action |
| DNA |
|
|
| Tubulin |
|
|
2.1. Antibody
2.2. Cytotoxic Payload
2.3. Linker
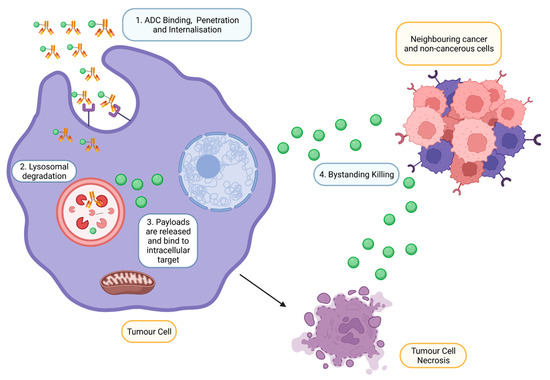
2.4. Mechanism of Action of ADC
3. Antibody–Drug Conjugate Approved by FDA
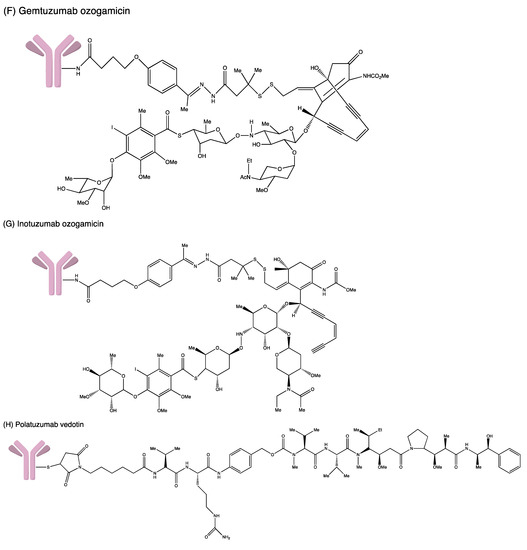
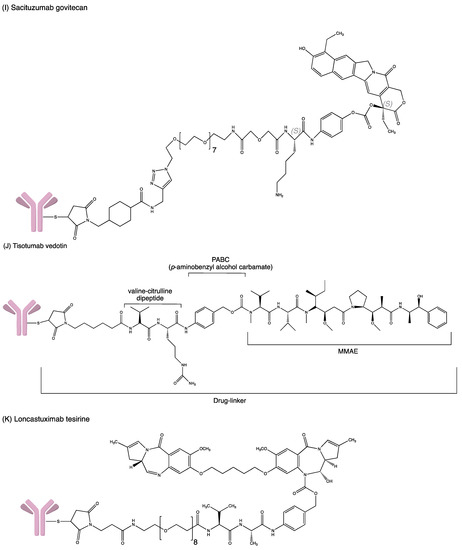
| ADC | Antibody | Target Antigen | Linker | Cytotoxic Payload | Indication | Year of Approval |
|---|---|---|---|---|---|---|
| Gemtuzumab ozogamicin | Humanised IgG4 | CD33 | Acid–labile hydrazone | Calicheamicin | Relapse or refractory AML and newly diagnosed CD33+ AML | Approved in 2000, withdrawn in 2010 but then reapproved in 2017 for relapsed/refractory malignancies FDA approved for newly diagnosed CD33+ AML in ≥1-month paediatric patient |
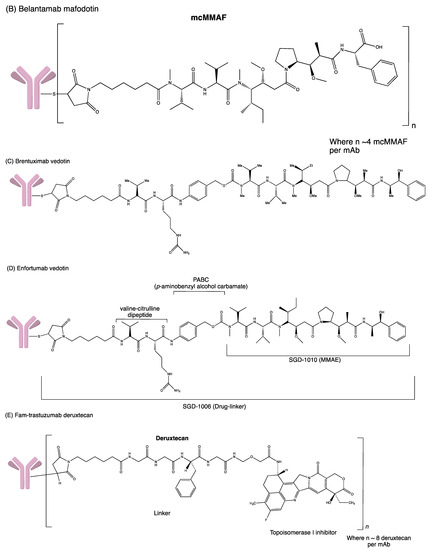
| Brentuximab vedotin | Chimeric IgG1 | CD30 | Protease-cleavable dipeptide | MMAE | HL, ALCL and different subtypes of T-cell lymphomas | FDA accelerated approval in 2011 In 2018, FDA approved the treatment of previously untreated stage III-IV HL and previously untreated ALCL and other CD30+ peripheral T-cell lymphomas. |
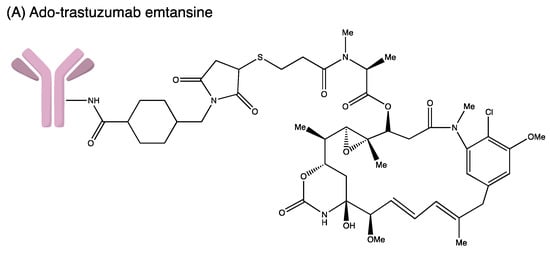
| Ado-trastuzumab emtansine | Humanised IgG1 | HER2 | SMCC | DM1 | Metastatic HER2+ breast cancer, previously treated with trastuzumab and a taxane and as adjuvant treatment for HER2+ early breast cancer with the residual invasive disease after neoadjuvant taxane and trastuzumab | FDA approved in 2013 FDA approved for adjuvant treatment in 2019 |
| Inotuzumab Ozogamicin | Humanised IgG4 | CD22 | Acid–labile hydrazone | Calicheamicin | Relapsed or refractory B-cell precursor ALL | FDA approved in February 2017 |
| Monotherapy treatment of relapsed or refractory CD22-positive B-cell precursor | EMA approved in June 2017 | |||||
| Polatuzumab vedotin | Humanised IgG1 | CD79b | Cleavable dipeptide | MMAE | Relapsed or refractory diffuse large B-cell lymphoma | FDA accelerated approval in 2019 |
| Enfortumab vedotin | Fully human IgG1 | Nectin 4 | Protease-cleavable dipeptide (Val–Cit) linker | MMAE | Locally advanced or metastatic urothelial cancer in adult patients who received prior treatment with a PD-1/L1 inhibitor and platinum-based chemotherapy in neoadjuvant/adjuvant setting | FDA accelerated approved in 2019 |
| Trastuzumab deruxtecan | Humanised IgG1 | HER2/ERB2 | Protease-cleavable tetra-peptide (Gly–Gly–Phe–Gly) linker | DXd | Unresectable locally advanced or metastatic HER2+ breast cancer, previously treated with trastuzumab and a taxane, adjuvant treatment for HER2+ early breast cancer with the residual invasive disease after neoadjuvant taxane and trastuzumab | FDA accelerated approved in 2019 |
| Sacituzumab govitecan | Humanised IgG1 | TROP-2 | Acid–labile ester (CL2 linker) | SN-38 | Triple-negative breast cancer, urothelial and other cancers | FDA accelerated approval in April 2020 for mTNBC |
| FDA regular approval for in April 2021 TNBC | ||||||
| FDA accelerated approval in April 2021 for mUC | ||||||
| Belantamab mafodotin | Humanised IgG1 | BCMA | Protease- resistant maleimidohexanoic linker |
MMAF | Relapsed or refractory multiple myeloma in adults who have received at least four prior therapies | FDA accelerated approval in 2020 |
| Loncastuximab tesirine- lpyl | Humanised IgG1 | CD19 | Valine–alanine dipeptide | PDB dimer | Relapsed or refractory large B-cell lymphoma | FDA accelerated approval in April 2021 |
| Tisotumab vedotin | IgG1 | Tissue factor (TF) | mc–val–cit–PABC | MMAE | Recurrent or metastatic cervical cancer in patients with disease progression during or following chemotherapy | FDA accelerated approval in September 2021 |
| Moxetumomab Pasudotox | - | CD22 | Recombinant covalently fused | Pseudotox | Relapsed or refractory HCL who received at least two prior systemic therapies | FDA approval in September 2018 |
4. Antibody–Drug Conjugates under Development
4.1. Potential ADCs to Be Approved
4.1.1. AGS67E
4.1.2. Denintuzumab Mafodotin (SGN-CD19A)
4.1.3. MEDI4276
4.1.4. Mirvetuximab Soravtansine (IMGN853)
4.1.5. Patritumab Deruxtecan (U3-1402)
4.1.6. Telisotuzumab Vedotin (ABBV-399)
4.2. Discontinued/Terminated Clinical Trials
4.2.1. AGS-16C3F
4.2.2. Coltuximab Ravtansine (SAR3419)
4.2.3. Glembatumumab Vedotin (CDX-011)
4.2.4. Lorvotuzumab Mertansine (IMGN-901)
4.2.5. Rovalpituzumab Tesirine (Rova-T)
5. Challenges of the Use of Antibody–Drug Conjugate
5.1. Toxicity of ADC
5.2. Antibody & Antigen Specificity
5.3. Immunogenicity of Antibody
5.4. Stability of Linkers
5.5. Target Features of Successful ADCs
The target features of successful ADCs can be identified from several aspects. First, the toxicity of a successful ADC must be acceptable, with lower bystander killing effects to protect normal cells from being destroyed unintentionally, and the DAR should be optimized for each ADC. Second, the interaction between antigen expressed in tumours and the antibody must be highly specified to achieve a favourable on-target effect. Moreover, since ADCs consist of engineered antibodies, they may trigger an immunogenic response. Thus, a successful ADC must be safe to introduce into the human body. Finally, the stability of the linker is one of the key features of successful ADC which should only release the drug when it interacts with antigen to promise the safety and efficacy of ADC.
6. Conclusions
References
- Neu, H.C. Paul Ehrlich: Scientist for Life. JAMA 1985, 254, 121.
- Chau, C.H.; Steeg, P.S.; Figg, W.D. Antibody–drug conjugates for cancer. Lancet 2019, 394, 793–804.
- Carter, P.J.; Lazar, G.A. Next generation antibody drugs: Pursuit of the’high-hanging fruit’. Nat. Rev. Drug Discov. 2018, 17, 197–223.
- Mullard, A. Maturing antibody-drug conjugate pipeline hits 30. Nat. Rev. Drug Discov. 2013, 12, 329–333.
- Drago, J.Z.; Modi, S.; Chandarlapaty, S. Unlocking the potential of antibody–drug conjugates for cancer therapy. Nat. Rev. Clin. Oncol. 2021, 18, 327–344.
- Birrer, M.J.; Moore, K.N.; Betella, I.; Bates, R. Antibody-drug conjugate-based therapeutics: State of the science. J. Natl. Cancer Inst. 2019, 111, 538–549.
- Hoffmann, R.M.; Coumbe, B.G.; Josephs, D.H.; Mele, S.; Ilieva, K.M.; Cheung, A.; Tutt, A.N.; Spicer, J.F.; Thurston, D.E.; Crescioli, S.; et al. Antibody structure and engineering considerations for the design and function of Antibody Drug Conjugates (ADCs). Oncoimmunology 2018, 7, e1395127.
- Mukhtar, E.; Adhami, V.M.; Mukhtar, H. Targeting microtubules by natural agents for cancer therapymicrotubule-targeting agents for cancer chemotherapy. Mol. Cancer Ther. 2014, 13, 275–284.
- Tsuchikama, K.; An, Z. Antibody-drug conjugates: Recent advances in conjugation and linker chemistries. Protein Cell 2018, 9, 33–46.
- Lu, J.; Jiang, F.; Lu, A.; Zhang, G. Linkers having a crucial role in antibody–drug conjugates. Int. J. Mol. Sci. 2016, 17, 561.
- Frigerio, M.; Kyle, A.F. The chemical design and synthesis of linkers used in antibody drug conjugates. Curr. Top. Med. Chem. 2017, 17, 3393–3424.
- Gorovits, B.; Krinos-Fiorotti, C. Proposed mechanism of off-target toxicity for antibody–drug conjugates driven by mannose receptor uptake. Cancer Immunol. Immunother. 2013, 62, 217–223.
- Peters, C.; Brown, S. Antibody–drug conjugates as novel anti-cancer chemotherapeutics. Biosci. Rep. 2015, 35, e00225.
- Li, F.; Emmerton, K.K.; Jonas, M.; Zhang, X.; Miyamoto, J.B.; Setter, J.R.; Nicholas, N.D.; Okeley, N.M.; Lyon, R.P.; Benjamin, D.R.; et al. Intracellular Released Payload Influences Potency and Bystander-Killing Effects of Antibody-Drug Conjugates in Preclinical ModelsReleased Payload Impacts ADC Activity and Bystander Killing. Cancer Res. 2016, 76, 2710–2719.
- Khera, E.; Thurber, G.M. Pharmacokinetic and immunological considerations for expanding the therapeutic window of next-generation antibody–drug conjugates. BioDrugs 2018, 32, 465–480.
- Chu, Y.; Zhou, X.; Wang, X. Antibody-drug conjugates for the treatment of lymphoma: Clinical advances and latest progress. J. Hematol. Oncol. 2021, 14, 88.
- PubChem. Compound Summary of Denintuzumab Mafodotin. 2014. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Denintuzumab-mafodotin (accessed on 1 November 2022).
- Seagen, Inc. A Safety Study of SGN-CD19A for B-Cell Lymphoma. NCT01786135; 2013. Available online: https://clinicaltrials.gov/ct2/show/NCT01786135 (accessed on 1 October 2022).
- Rinnerthaler, G.; Gampenrieder, S.P.; Greil, R. HER2 Directed Antibody-Drug-Conjugates beyond T-DM1 in Breast Cancer. Int. J. Mol. Sci. 2019, 20, 1115.
- PubChem. Compound Summary for CID 91810695, Mirvetuximab Soravtansine. 2015. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Mirvetuximab-soravtansine (accessed on 1 November 2022).
- Moore, K.N.; Vergote, I.; Oaknin, A.; Colombo, N.; Banerjee, S.; Oza, A.; Pautier, P.; Malek, K.; Birrer, M.J. FORWARD I: A Phase III study of mirvetuximab soravtansine versus chemotherapy in platinum-resistant ovarian cancer. Future Oncol. 2018, 14, 1669–1678.
- Hashimoto, Y.; Koyama, K.; Kamai, Y.; Hirotani, K.; Ogitani, Y.; Zembutsu, A.; Abe, M.; Kaneda, Y.; Maeda, N.; Shiose, Y.; et al. A Novel HER3-Targeting Antibody–Drug Conjugate, U3-1402, Exhibits Potent Therapeutic Efficacy through the Delivery of Cytotoxic Payload by Efficient InternalizationPreclinical Evaluation of U3-1402, a HER3-Targeting ADC. Clin. Cancer Res. 2019, 25, 7151–7161.
- Angevin, E.; Strickler, J.H.; Weekes, C.D.; Heist, R.S.; Morgensztern, D.; Nemunaitis, J.J.; Fan, X.; Beaulieu, J.; Motwani, M.; Afar, D.E.; et al. Phase I study of ABBV-399, a c-Met antibody-drug conjugate (ADC), as monotherapy and in combination with erlotinib in patients (pts) with non-small cell lung cancer (NSCLC). J. Clin. Oncol. 2017, 35, 2509.
- Kollmannsberger, C.; Choueiri, T.K.; Heng, D.Y.; George, S.; Jie, F.; Croitoru, R.; Poondru, S.; Thompson, J.A. A Randomized Phase II Study of AGS-16C3F Versus Axitinib in Previously Treated Patients with Metastatic Renal Cell Carcinoma. Oncologist 2021, 26, 182-e361.
- Kantarjian, H.M.; Lioure, B.; Kim, S.K.; Atallah, E.; Leguay, T.; Kelly, K.; Marolleau, J.-P.; Escoffre-Barbe, M.; Thomas, X.G.; Cortes, J.; et al. A phase II study of coltuximab ravtansine (SAR3419) monotherapy in patients with relapsed or refractory acute lymphoblastic leukemia. Clin. Lymphoma Myeloma Leuk. 2016, 16, 139–145.
- Bristol-Myers Squibb. A Study of BMS-986148 in Patients with Select Advanced Solid Tumors. NCT02341625; 2015. Available online: https://clinicaltrials.gov/ct2/show/NCT02341625 (accessed on 1 October 2022).
- Ott, P.A.; Pavlick, A.C.; Johnson, D.B.; Hart, L.L.; Infante, J.R.; Luke, J.J.; Lutzky, J.; Rothschild, N.E.; Spitler, L.E.; Cowey, C.L.; et al. A phase 2 study of glembatumumab vedotin, an antibody-drug conjugate targeting glycoprotein NMB, in patients with advanced melanoma. Cancer Cell Int. 2019, 125, 1113–1123.
- Socinski, M.A.; Kaye, F.J.; Spigel, D.R.; Kudrik, F.J.; Ponce, S.; Ellis, P.M.; Majem, M.; Lorigan, P.; Gandhi, L.; Gutierrez, M.E.; et al. Phase 1/2 study of the CD56-targeting antibody-drug conjugate lorvotuzumab mertansine (IMGN901) in combination with carboplatin/etoposide in small-cell lung cancer patients with extensive-stage disease. Clin. Lung Cancer 2017, 18, 68–76.
- PubChem. Compound Summary for CID 131954443, Rovalpituzumab Tesirine. 2018. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/131954443 (accessed on 2 November 2022).
- AbbVie. AbbVie Discontinues Rovalpituzumab Tesirine (Rova-T) Research and Development Program. 2019. Available online: https://news.abbvie.com/news/press-releases/abbvie-discontinues-rovalpituzumab-tesirine-rova-t-research-and-development-program.htm (accessed on 1 November 2022).
- Zhao, P.; Zhang, Y.; Li, W.; Jeanty, C.; Xiang, G.; Dong, Y. Recent advances of antibody drug conjugates for clinical applications. Acta Pharm. Sin. B 2020, 10, 1589–1600.
- Plenderleith, I.H. Treating the treatment: Toxicity of cancer chemotherapy. Can. Fam. Physician 1990, 36, 1827.
- Nejadmoghaddam, M.-R.; Minai-Tehrani, A.; Ghahremanzadeh, R.; Mahmoudi, M.; Dinarvand, R.; Zarnani, A.-H. Antibody-drug conjugates: Possibilities and challenges. Avicenna J. Med. Biotechnol. 2019, 11, 3.
- Farràs, M.; Miret, J.; Camps, M.; Román, R.; Martínez, Ó.; Pujol, X.; Erb, S.; Ehkirch, A.; Cianferani, S.; Casablancas, A. Homogeneous antibody-drug conjugates: DAR 2 anti-HER2 obtained by conjugation on isolated light chain followed by mAb assembly. MAbs 2020, 12, 1702262.
- Teicher, B.A.; Chari, R.V. Antibody conjugate therapeutics: Challenges and potential. Clin. Cancer Res. 2011, 17, 6389–6397.





