
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | William Steadman | -- | 3545 | 2023-09-24 08:06:42 | | | |
| 2 | Catherine Yang | Meta information modification | 3545 | 2023-09-25 04:23:50 | | |
Video Upload Options
Prosthetic Joint Infection (PJI) causes significant morbidity and mortality for patients globally. Delivery of antibiotics to the site of infection has potential to improve the treatment outcomes and enhance biofilm eradication. These antibiotics can be delivered using an intra-articular catheter or combined with a carrier substance to enhance pharmacokinetic properties. Use of an antibiotic carrier is an alternative method of maintaining high local concentrations of antibiotic without systemic exposure. These substances incorporate an antimicrobial to prolong its half-life and provide predictable elution characteristics. They may also serve additional roles, such as filling dead space and providing mechanical support for limb alignment. The ideal antibiotic carrier would provide prolonged antibiotic concentrations at an effective level and achieve complete antibiotic release to minimise subtherapeutic elution time. It would also be versatile and have compatibility with the desired antibiotics, and fully resorbable with minimal risk of allergies, and systemic or local adverse effects.
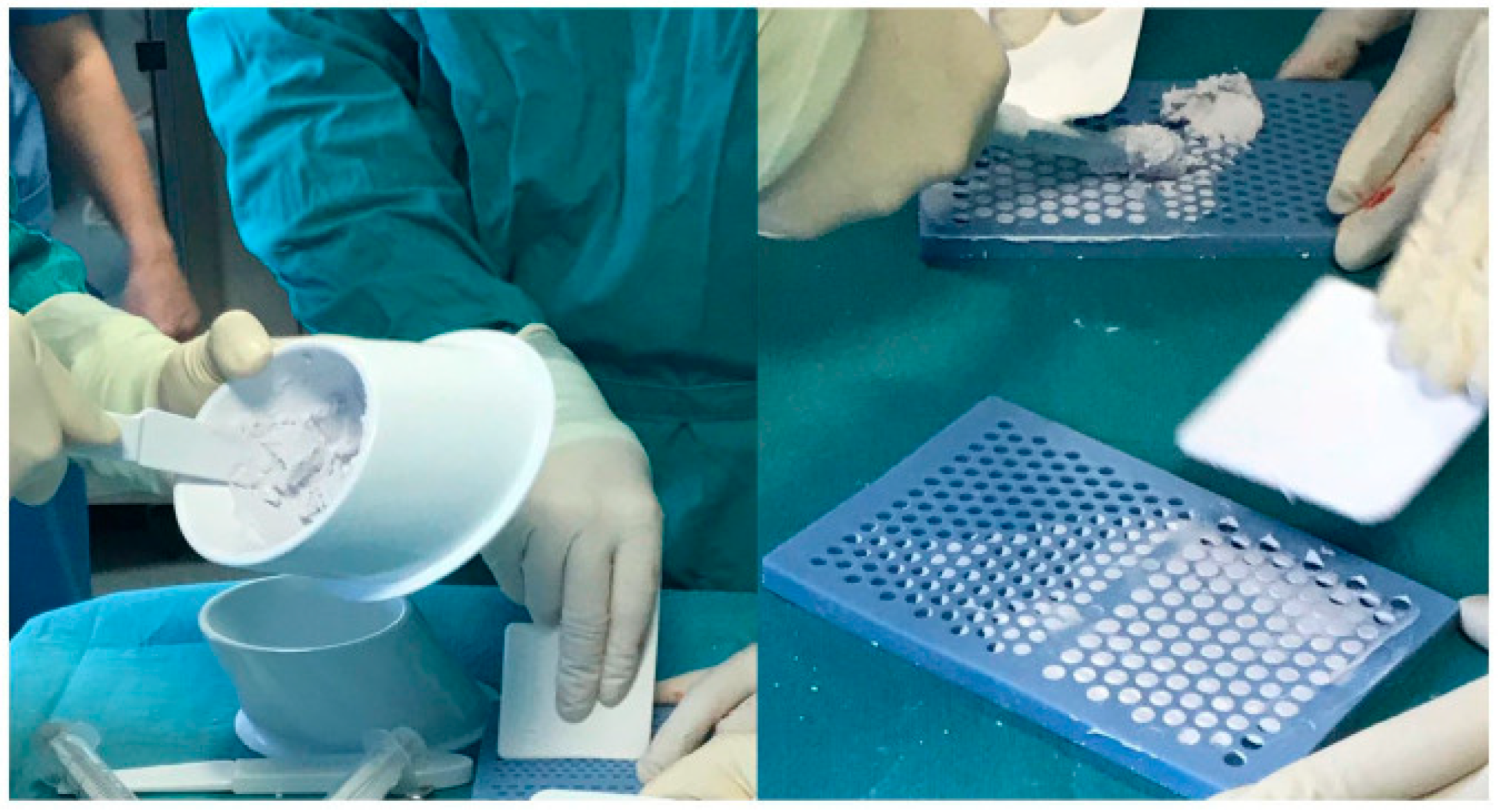
1. PMMA
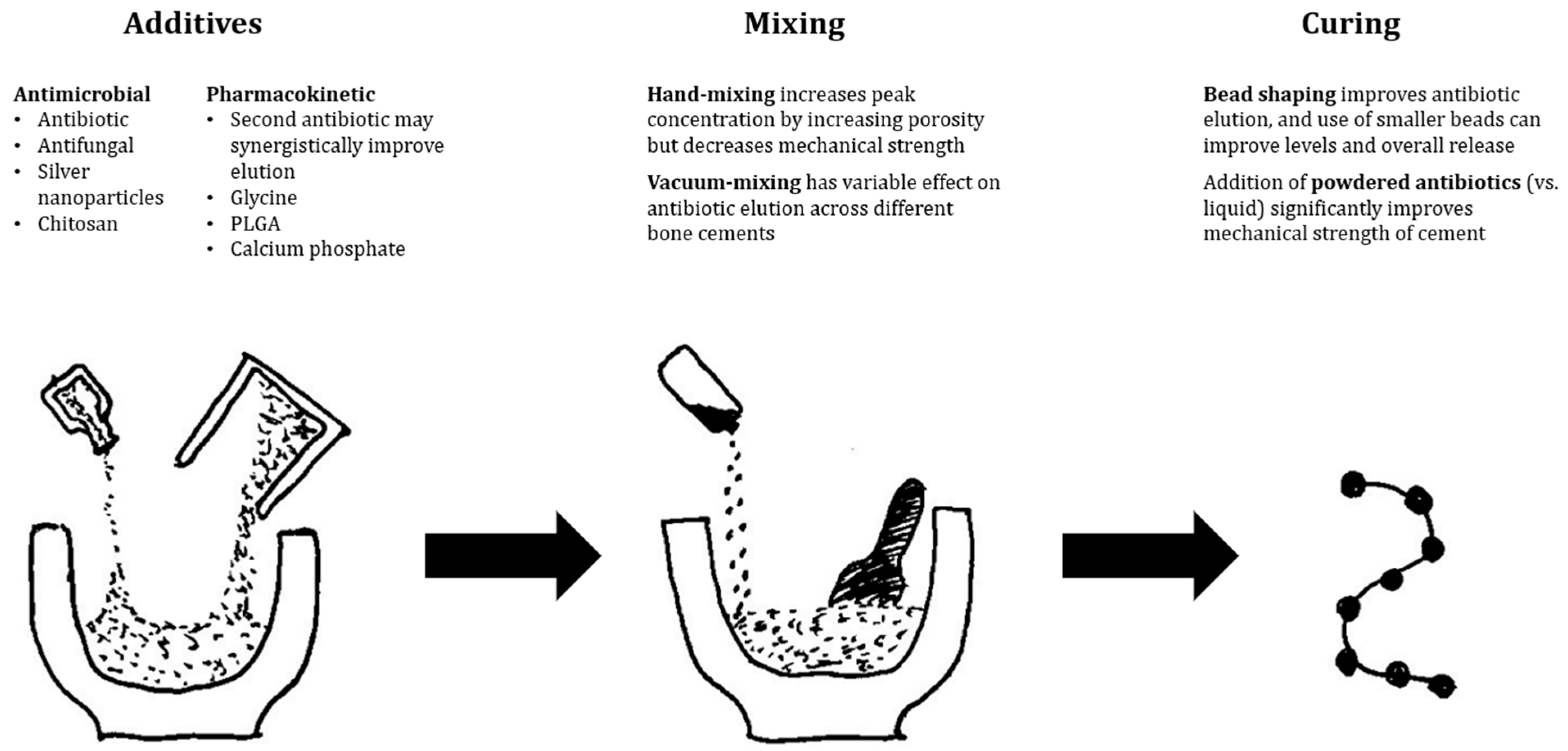
2. Resorbable Carriers
3. Calcium Sulphate
4. Hydroxyapatite
5. Bioactive Glass
6. Hydrogels
7. Nanocarriers

References
- Buchholz, H.W.; Engelbrecht, H. Depot effects of various antibiotics mixed with palacos resins. Chirurg 1970, 41, 511–515.
- Luu, A.; Syed, F.; Raman, G.; Bhalla, A.; Muldoon, E.; Hadley, S.; Smith, E.; Rao, M. Two-Stage Arthroplasty for Prosthetic Joint Infection. J. Arthroplast. 2013, 28, 1490–1498.
- Anagnostakos, K.; Wilmes, P.; Schmitt, E.; Kelm, J. Elution of gentamicin and vancomycin from polymethylmethacrylate beads and hip spacers in vivo. Acta Orthop. 2009, 80, 193–197.
- Wall, V.; Nguyen, T.-H.; Nguyen, N.; Tran, P. Controlling Antibiotic Release from Polymethylmethacrylate Bone Cement. Biomedicines 2021, 9, 26.
- Zalavras, C.G.; Patzakis, M.J.; Holtom, P. Local Antibiotic Therapy in the Treatment of Open Fractures and Osteomyelitis. Clin. Orthop. Relat. Res. 2004, 427, 86–93.
- E Kent, M.; Rapp, R.P.; Smith, K.M. Antibiotic Beads and Osteomyelitis: Here Today, What’s Coming Tomorrow? Orthopedics 2006, 29, 599–603.
- van Vugt, T.A.G.; Arts, J.J.; Geurts, J.A.P. Antibiotic-Loaded Polymethylmethacrylate Beads and Spacers in Treatment of Orthopedic Infections and the Role of Biofilm Formation. Front. Microbiol. 2019, 10, 1626.
- Nelson, C.L.; Evans, R.P.; Blaha, J.D.; Calhoun, J.; Henry, S.L.; Patzakis, M.J. A comparison of gentamicin-impregnated polymethylmethacrylate bead implantation to conventional parenteral antibiotic therapy in infected total hip and knee arthroplasty. Clin. Orthop. Relat. Res. 1993, 295, 96–101.
- Stockley, I.; Mockford, B.J.; Hoad-Reddick, A.; Norman, P. The use of two-stage exchange arthroplasty with depot antibiotics in the absence of long-term antibiotic therapy in infected total hip replacement. J. Bone Jt. Surg. 2008, 90, 145–148.
- Hoad-Reddick, D.A.; Evans, C.R.; Norman, P.; Stockley, I. Is there a role for extended antibiotic therapy in a two-stage revision of the infected knee arthroplasty? J. Bone Jt. Surg. 2005, 87, 171–174.
- Hart, W.J.; Jones, R.S. Two-stage revision of infected total knee replacements using articulating cement spacers and short-term antibiotic therapy. J. Bone Jt. Surg. 2006, 88, 1011–1015.
- Anagnostakos, K.; Meyer, C. Antibiotic Elution from Hip and Knee Acrylic Bone Cement Spacers: A Systematic Review. BioMed Res. Int. 2017, 2017, 4657874.
- Walenkamp, G.H.I.M. Antibiotic Loaded Cement: From Research to Clinical Evidence. In Infection and Local Treatment in Orthopedic Surgery; Springer: Berlin/Heidelberg, Germany, 2007; pp. 170–175.
- Dodds, S.; Smith, T.J.; Akid, R.; Stephenson, J.; Nichol, T.; Banerjee, R.D.; Stockley, I.; Townsend, R. Contrasting Effects of Physical Wear on Elution of Two Antibiotics from Orthopedic Cement. Antimicrob. Agents Chemother. 2012, 56, 1471–1475.
- Roy, M.E.; Peppers, M.P.; Whiteside, L.A.; LaZear, R.M. Vancomycin Concentration in Synovial Fluid: Direct Injection into the Knee vs. Intravenous Infusion. J. Arthroplast. 2014, 29, 564–568.
- Walenkamp, G. Small PMMA beads improve gentamicin release. Acta Orthop. 1989, 60, 668–669.
- Stevens, C.M.; Tetsworth, K.D.; Calhoun, J.H.; Mader, J.T. An articulated antibiotic spacer used for infected total knee arthroplasty: A comparative in vitro elution study of Simplex® and Palacos® bone cements. J. Orthop. Res. 2005, 23, 27–33.
- van De Belt, H.; Neut, D.; Schenk, W.; van Horn, J.R.; van Der Mei, H.C.; Busscher, H.J. Gentamicin release from polymethylmethacrylate bone cements and Staphylococcus aureus biofilm formation. Acta Orthop. 2000, 71, 625–629.
- Neut, D.; van de Belt, H.; van Horn, J.; van der Mei, H.; Busscher, H. The effect of mixing on gentamicin release from polymethylmethacrylate bone cements. Acta Orthop. 2003, 74, 670–676.
- Cerretani, D.; Giorgi, G.; Fornara, P.; Bocchi, L.; Neri, L.; Ceffa, R.; Ghisellini, F.; Ritter, M.A. The in vitro elution characteristics of vancomycin combined with imipenem-cilastatin in acrylic bone–cements: A pharmacokinetic study. J. Arthroplast. 2002, 17, 619–626.
- Bitsch, R.; Kretzer, J.; Vogt, S.; Büchner, H.; Thomsen, M.; Lehner, B. Increased antibiotic release and equivalent biomechanics of a spacer cement without hard radio contrast agents. Diagn. Microbiol. Infect. Dis. 2015, 83, 203–209.
- Penner, M.J.; Duncan, C.P.; Masri, B.A. The in vitro elution characteristics of antibiotic-loaded CMW and Palacos-R bone cements. J. Arthroplast. 1999, 14, 209–214.
- McLaren, A.C.; Nelson, C.L.; McLaren, S.G.; DeClerk, G.R. The Effect of Glycine Filler on the Elution Rate of Gentamicin from Acrylic Bone Cement. Clin. Orthop. Relat. Res. 2004, 427, 25–27.
- Frew, N.M.; Cannon, T.; Nichol, T.; Smith, T.; Stockley, I. Comparison of the elution properties of commercially available gentamicin and bone cement containing vancomycin with ‘home-made’ preparations. Bone Jt. J. 2017, 99-B, 73–77.
- Meyer, J.; Piller, G.; Spiegel, C.A.; Hetzel, S.; Squire, M. Vacuum-Mixing Significantly Changes Antibiotic Elution Characteristics of Commercially Available Antibiotic-Impregnated Bone Cements. J. Bone Jt. Surg. 2011, 93, 2049–2056.
- Minelli, E.B.; Della Bora, T.; Benini, A. Different microbial biofilm formation on polymethylmethacrylate (PMMA) bone cement loaded with gentamicin and vancomycin. Anaerobe 2011, 17, 380–383.
- McLaren, A.C.; Nugent, M.; Economopoulos, K.; Kaul, H.; Vernon, B.L.; McLemore, R. Hand-mixed and Premixed Antibiotic-loaded Bone Cement Have Similar Homogeneity. Clin. Orthop. Relat. Res. 2009, 467, 1693–1698.
- Samara, E.; Moriarty, T.F.; Decosterd, L.; Richards, R.G.; Gautier, E.; Wahl, P. Antibiotic stability over six weeks in aqueous solution at body temperature with and without heat treatment that mimics the curing of bone cement. Bone Jt. Res. 2017, 6, 296–306.
- Edelstein, A.I.; Okroj, K.T.; Rogers, T.; Della Valle, C.J.; Sporer, S.M. Systemic Absorption of Antibiotics From Antibiotic-Loaded Cement Spacers for the Treatment of Periprosthetic Joint Infection. J. Arthroplast. 2018, 33, 835–839.
- Dagneaux, L.; Limberg, A.K.; Osmon, D.R.; Leung, N.; Berry, D.J.; Abdel, M.P. Acute Kidney Injury When Treating Periprosthetic Joint Infections After Total Knee Arthroplasties with Antibiotic-Loaded Spacers. J. Bone Jt. Surg. 2021, 103, 754–760.
- Jafari, S.M.; Huang, R.; Joshi, A.; Parvizi, J.; Hozack, W.J. Renal Impairment Following Total Joint Arthroplasty: Who Is at Risk? J. Arthroplast. 2010, 25, 49–53.
- Duey, R.E.; Chong, A.C.M.; McQueen, D.A.; Womack, J.L.; Song, Z.; Steinberger, T.A.; Wooley, P.H. Mechanical properties and elution characteristics of polymethylmethacrylate bone cement impregnated with antibiotics for various surface area and volume constructs. Iowa Orthop. J. 2012, 32, 104–115.
- Van de Belt, H.; Neut, D.; Schenk, W.; van Horn, J.R.; van der Mei, H.C.; Busscher, H.J. Infection of orthopedic implants and the use of antibiotic-loaded bone cements: A review. Acta Orthop. Scand. 2001, 72, 557–571.
- Buttaro, M.; Valentini, R.; Piccaluga, F. Persistent infection associated with residual cement after resection arthroplasty of the hip. Acta Orthop. 2004, 75, 427–429.
- Shi, X.; Wu, Y.; Ni, H.; Li, M.; Zhang, C.; Qi, B.; Wei, M.; Wang, T.; Xu, Y. Antibiotic-loaded calcium sulfate in clinical treatment of chronic osteomyelitis: A systematic review and meta-analysis. J. Orthop. Surg. Res. 2022, 17, 104.
- Tarar, M.Y.; Khalid, A.; Usman, M.; Javed, K.; Shah, N.; Abbas, M.W. Wound Leakage With the Use of Calcium Sulphate Beads in Prosthetic Joint Surgeries: A Systematic Review. Cureus 2021, 13, e19650.
- Abosala, A.; Ali, M. The Use of Calcium Sulphate beads in Periprosthetic Joint Infection, a systematic review. J. Bone Jt. Infect. 2020, 5, 43–49.
- Howlin, R.P.; Brayford, M.J.; Webb, J.S.; Cooper, J.J.; Aiken, S.S.; Stoodley, P. Antibiotic-Loaded Synthetic Calcium Sulfate Beads for Prevention of Bacterial Colonization and Biofilm Formation in Periprosthetic Infections. Antimicrob. Agents Chemother. 2015, 59, 111–120.
- Heuberger, R.; Wahl, P.; Krieg, J.; Gautier, E. Low in vitro third-body wear on total hip prostheses induced by calcium sulphate used for local antibiotic therapy. Eur. Cells Mater. 2014, 28, 246–257.
- McPherson, F.E.; Dipane, B.M.; Sherif, S. Dissolvable Antibiotic Beads in Treatment of Periprosthetic Joint Infection and Revision Arthroplasty—The Use of Synthetic Pure Calcium Sulfate (Stimulan®) Impregnated with Vancomycin & Tobramycin. Reconstr. Rev. 2013, 3, 32–43.
- Kallala, R.; Harris, W.E.; Ibrahim, M.; Dipane, M.; McPherson, E. Use of Stimulan absorbable calcium sulphate beads in revision lower limb arthroplasty. Bone Jt. Res. 2018, 7, 570–579.
- Tarar, M.Y.; Toe, K.K.Z.; Javed, K.; Shah, N.; Khalid, A. The Risk of Iatrogenic Hypercalcemia in Patients Undergoing Calcium Sulphate Beads Implantation in Prosthetic Joint Surgery: A Systematic Review. Cureus 2021, 13, e18777.
- Tarity, T.D.; Xiang, W.; Jones, C.W.; Gkiatas, I.; Nocon, A.; Selemon, N.A.; Carli, A.; Sculco, P.K. Do Antibiotic-Loaded Calcium Sulfate Beads Improve Outcomes After Debridement, Antibiotics, and Implant Retention? A Matched Cohort Study. Arthroplast. Today 2022, 14, 90–95.
- Flierl, M.A.; Culp, B.M.; Okroj, K.T.; Springer, B.D.; Levine, B.R.; Della Valle, C.J. Poor Outcomes of Irrigation and Debridement in Acute Periprosthetic Joint Infection With Antibiotic-Impregnated Calcium Sulfate Beads. J. Arthroplast. 2017, 32, 2505–2507.
- Reinisch, K.; Schläppi, M.; Meier, C.; Wahl, P. Local antibiotic treatment with calcium sulfate as carrier material improves the outcome of debridement, antibiotics, and implant retention procedures for periprosthetic joint infections after hip arthroplasty—A retrospective study. J. Bone Jt. Infect. 2022, 7, 11–21.
- Lum, Z.; Pereira, G.C. Local bio-absorbable antibiotic delivery in calcium sulfate beads in hip and knee arthroplasty. J. Orthop. 2018, 15, 676–678.
- Ene, R.; Nica, M.; Ene, D.; Cursaru, A.; Cirstoiu, C. Review of calcium-sulphate-based ceramics and synthetic bone substitutes used for antibiotic delivery in PJI and osteomyelitis treatment. EFORT Open Rev. 2021, 6, 297–304.
- Wassif, R.K.; Elkayal, M.; Shamma, R.N.; Elkheshen, S.A. Recent advances in the local antibiotics delivery systems for management of osteomyelitis. Drug Deliv. 2021, 28, 2392–2414.
- Hasegawa, M.; Tone, S.; Naito, Y.; Wakabayashi, H.; Sudo, A. Use of antibiotic-impregnated hydroxyapatite for infection following total knee arthroplasty. Mod. Rheumatol. 2021, 31, 1073–1077.
- McNally, M.A.; Ferguson, J.Y.; Lau, A.C.K.; Diefenbeck, M.; Scarborough, M.; Ramsden, A.J.; Atkins, B.L. Single-stage treatment of chronic osteomyelitis with a new absorbable, gentamicin-loaded, calcium sulphate/hydroxyapatite biocomposite. Bone Jt. J. 2016, 98-B, 1289–1296.
- Bidossi, A.; Bottagisio, M.; Logoluso, N.; De Vecchi, E. In Vitro Evaluation of Gentamicin or Vancomycin Containing Bone Graft Substitute in the Prevention of Orthopedic Implant-Related Infections. Int. J. Mol. Sci. 2020, 21, 9250.
- Zampelis, V.; Tägil, M.; Lidgren, L.; Isaksson, H.; Atroshi, I.; Wang, J.-S. The effect of a biphasic injectable bone substitute on the interface strength in a rabbit knee prosthesis model. J. Orthop. Surg. Res. 2013, 8, 25.
- Colding-Rasmussen, T.; Horstmann, P.; Petersen, M.M.; Hettwer, W. Antibiotic Elution Characteristics and Pharmacokinetics of Gentamicin and Vancomycin from a Mineral Antibiotic Carrier: An in vivo Evaluation of 32 Clinical Cases. J. Bone Jt. Infect. 2018, 3, 234–240.
- Logoluso, N.; Drago, L.; Gallazzi, E.; George, D.A.; Morelli, I.; Romanò, C.L. Calcium-Based, Antibiotic-Loaded Bone Substitute as an Implant Coating: A Pilot Clinical Study. J. Bone Jt. Infect. 2016, 1, 59–64.
- McNally, M.A.; Ferguson, J.Y.; Scarborough, M.; Ramsden, A.; Stubbs, D.A.; Atkins, B.L. Mid- to long-term results of single-stage surgery for patients with chronic osteomyelitis using a bioabsorbable gentamicin-loaded ceramic carrier. Bone Jt. J. 2022, 104-B, 1095–1100.
- Sakellariou, V.I.; Savvidou, O.; Markopoulos, C.; Drakou, A.; Mavrogenis, A.F.; Papagelopoulos, P.J. Combination of Calcium Hydroxyapatite Antibiotic Carrier with Cement Spacers in Peri-Prosthetic Knee Infections. Surg. Infect. 2015, 16, 748–754.
- Lindfors, N.; Hyvönen, P.; Nyyssönen, M.; Kirjavainen, M.; Kankare, J.; Gullichsen, E.; Salo, J. Bioactive glass S53P4 as bone graft substitute in treatment of osteomyelitis. Bone 2010, 47, 212–218.
- Rahaman, M.N.; Bal, B.S.; Huang, W. Review: Emerging developments in the use of bioactive glasses for treating infected prosthetic joints. Mater. Sci. Eng. C 2014, 41, 224–231.
- Drago, L.; Toscano, M.; Bottagisio, M. Recent Evidence on Bioactive Glass Antimicrobial and Antibiofilm Activity: A Mini-Review. Materials 2018, 11, 326.
- Bortolin, M.; De Vecchi, E.; Romanò, C.L.; Toscano, M.; Mattina, R.; Drago, L. Antibiofilm agents against MDR bacterial strains: Is bioactive glass BAG-S53P4 also effective? J. Antimicrob. Chemother. 2015, 71, 123–127.
- Drago, L.; Romanò, D.; De Vecchi, E.; Vassena, C.; Logoluso, N.; Mattina, R.; Romano, C.L. Bioactive glass BAG-S53P4 for the adjunctive treatment of chronic osteomyelitis of the long bones: An in vitroand prospective clinical study. BMC Infect. Dis. 2013, 13, 584.
- Pérez-Tanoira, R.; García-Pedrazuela, M.; Hyyrynen, T.; Soininen, A.; Aarnisalo, A.; Nieminen, M.; Tiainen, V.-M.; Konttinen, Y.T.; Kinnari, T. Effect of S53P4 bone substitute on staphylococcal adhesion and biofilm formation on other implant materials in normal and hypoxic conditions. J. Mater. Sci. Mater. Med. 2015, 26, 239.
- Cunha, M.T.; Murça, M.A.; Nigro, S.; Klautau, G.B.; Salles, M.J.C. In vitro antibacterial activity of bioactive glass S53P4 on multiresistant pathogens causing osteomyelitis and prosthetic joint infection. BMC Infect. Dis. 2018, 18, 157.
- Gupta, S.; Majumdar, S.; Krishnamurthy, S. Bioactive glass: A multifunctional delivery system. J. Control Release 2021, 335, 481–497.
- Meseguer-Olmo, L.; Ros-Nicolás, M.J.; Clavel-Sainz, M.; Vicente-Ortega, V.; Alcaraz-Baños, M.; Lax-Pérez, A.; Arcos, D.; Ragel, C.V.; Vallet-Regí, M. Biocompatibility andin vivogentamicin release from bioactive sol-gel glass implants. J. BioMed Mater. Res. 2002, 61, 458–465.
- Xie, Z.; Liu, X.; Jia, W.; Zhang, C.; Huang, W.; Wang, J. Treatment of osteomyelitis and repair of bone defect by degradable bioactive borate glass releasing vancomycin. J. Control Release 2009, 139, 118–126.
- Jia, W.-T.; Zhang, X.; Luo, S.-H.; Liu, X.; Huang, W.-H.; Rahaman, M.N.; Day, D.E.; Zhang, C.-Q.; Xie, Z.-P.; Wang, J.-Q. Novel borate glass/chitosan composite as a delivery vehicle for teicoplanin in the treatment of chronic osteomyelitis. Acta Biomater. 2010, 6, 812–819.
- Zhang, X.; Jia, W.; Gu, Y.; Xiao, W.; Liu, X.; Wang, D.; Zhang, C.; Huang, W.; Rahaman, M.N.; Day, D.E.; et al. Teicoplanin-loaded borate bioactive glass implants for treating chronic bone infection in a rabbit tibia osteomyelitis model. Biomaterials 2010, 31, 5865–5874.
- Domingues, R.; Cortés, M.; Gomes, T.; Diniz, H.; Freitas, C.; Gomes, J.; Faria, A.M.; Sinisterra, R. Bioactive glass as a drug delivery system of tetracycline and tetracycline associated with β-cyclodextrin. Biomaterials 2004, 25, 327–333.
- Ding, H.; Zhao, C.-J.; Cui, X.; Gu, Y.-F.; Jia, W.-T.; Rahaman, M.N.; Wang, Y.; Huang, W.-H.; Zhang, C.-Q. A Novel Injectable Borate Bioactive Glass Cement as an Antibiotic Delivery Vehicle for Treating Osteomyelitis. PLoS ONE 2014, 9, e85472.
- Jia, W.-T.; Fu, Q.; Huang, W.-H.; Zhang, C.-Q.; Rahaman, M.N. Comparison of Borate Bioactive Glass and Calcium Sulfate as Implants for the Local Delivery of Teicoplanin in the Treatment of Methicillin-Resistant Staphylococcus aureus-Induced Osteomyelitis in a Rabbit Model. Antimicrob. Agents Chemother. 2015, 59, 7571–7580.
- Lindfors, N.; Geurts, J.; Drago, L.; Arts, J.J.; Juutilainen, V.; Hyvönen, P.; Suda, A.J.; Domenico, A.; Artiaco, S.; Alizadeh, C.; et al. Antibacterial Bioactive Glass, S53P4, for Chronic Bone Infections—A Multinational Study. In A Modern Approach to Biofilm-Related Orthopaedic Implant Infections: Advances in Microbiology, Infectious Diseases and Public Health Volume 5; Drago, L., Ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 81–92.
- Romanò, C.L.; Logoluso, N.; Meani, E.; Romanò, D.; De Vecchi, E.; Vassena, C.; Drago, L. A comparative study of the use of bioactive glass S53P4 and antibiotic-loaded calcium-based bone substitutes in the treatment of chronic osteomyelitis. Bone Jt. J. 2014, 96-B, 845–850.
- McAndrew, J.; Efrimescu, C.; Sheehan, E.; Niall, D. Through the looking glass; bioactive glass S53P4 (BonAlive®) in the treatment of chronic osteomyelitis. Ir. J. Med Sci. 2013, 182, 509–511.
- Overstreet, D.J.; Huynh, R.; Jarbo, K.; McLemore, R.Y.; Vernon, B.L. In situforming, resorbable graft copolymer hydrogels providing controlled drug release. J. BioMed Mater. Res. Part A 2012, 101A, 1437–1446.
- Censi, R.; Casadidio, C.; Dubbini, A.; Cortese, M.; Scuri, S.; Grappasonni, I.; Golob, S.; Vojnovic, D.; Sabbieti, M.G.; Agas, D.; et al. Thermosensitive hybrid hydrogels for the controlled release of bioactive vancomycin in the treatment of orthopaedic implant infections. Eur. J. Pharm. Biopharm. 2019, 142, 322–333.
- Lai, P.-L.; Hong, D.-W.; Ku, K.-L.; Lai, Z.-T.; Chu, I.-M. Novel thermosensitive hydrogels based on methoxy polyethylene glycol-co-poly(lactic acid-co-aromatic anhydride) for cefazolin delivery. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 553–560.
- Romanò, C.L.; Scarponi, S.; Gallazzi, E.; Romanò, D.; Drago, L. Antibacterial coating of implants in orthopaedics and trauma: A classification proposal in an evolving panorama. J. Orthop. Surg. Res. 2015, 10, 157.
- Tsikopoulos, K.; Bidossi, A.; Drago, L.; Petrenyov, D.; Givissis, P.; Mavridis, D.; Papaioannidou, P. Is Implant Coating With Tyrosol- and Antibiotic-loaded Hydrogel Effective in Reducing Cutibacterium (Propionibacterium) acnes Biofilm Formation? A Preliminary In Vitro Study. Clin. Orthop. Relat. Res. 2019, 477, 1736–1746.
- Kim, H.; Chang, R.; Morales, S.; Chan, H.-K. Bacteriophage-Delivering Hydrogels: Current Progress in Combating Antibiotic Resistant Bacterial Infection. Antibiotics 2021, 10, 130.
- Overstreet, D.J.; Badha, V.S.; Heffernan, J.M.; Childers, E.P.; Moore, R.C.; Vernon, B.L.; McLaren, A.C. Temperature-responsive PNDJ hydrogels provide high and sustained antimicrobial concentrations in surgical sites. Drug Deliv. Transl. Res. 2019, 9, 802–815.
- Hsu, Y.-H.; Yu, Y.-H.; Lee, D.; Chou, Y.-C.; Wu, C.-K.; Lu, C.-J.; Liu, S.-J. Pharmaceutical-eluting hybrid degradable hydrogel/microparticle loaded sacs for finger joint interpositional arthroplasty. Biomater. Adv. 2022, 137, 212846.
- De Meo, D.; Ceccarelli, G.; Iaiani, G.; Torto, F.L.; Ribuffo, D.; Persiani, P.; Villani, C. Clinical Application of Antibacterial Hydrogel and Coating in Orthopaedic and Traumatology Surgery. Gels 2021, 7, 126.
- Zagra, L.; Gallazzi, E.; Romanò, D.; Scarponi, S.; Romanò, C. Two-stage cementless hip revision for peri-prosthetic infection with an antibacterial hydrogel coating: Results of a comparative series. Int. Orthop. 2018, 43, 111–115.
- Capuano, N.; Logoluso, N.; Gallazzi, E.; Drago, L.; Romanò, C.L. One-stage exchange with antibacterial hydrogel coated implants provides similar results to two-stage revision, without the coating, for the treatment of peri-prosthetic infection. Knee Surg. Sport. Traumatol. Arthrosc. 2018, 26, 3362–3367.
- Thakrar, R.R.; Horriat, S.; Kayani, B.; Haddad, F.S. Indications for a single-stage exchange arthroplasty for chronic prosthetic joint infection. Bone Jt. J. 2019, 101-B, 19–24.
- Romanò, C.L.; Malizos, K.; Capuano, N.; Mezzoprete, R.; D’Arienzo, M.; Van Der Straeten, C.; Scarponi, S.; Drago, L. Does an Antibiotic-Loaded Hydrogel Coating Reduce Early Post-Surgical Infection After Joint Arthroplasty? J. Bone Jt. Infect. 2016, 1, 34–41.
- Wang, D.-Y.; Su, L.; Yang, G.; Ren, Y.; Zhang, M.; Jing, H.; Zhang, X.; Bayston, R.; van der Mei, H.C.; Busscher, H.J.; et al. Self-targeting of zwitterion-based platforms for nano-antimicrobials and nanocarriers. J. Mater. Chem. B 2022, 10, 2316–2322.
- Huang, B.; Wang, L.; Tang, K.; Chen, S.; Xu, Y.; Liao, H.; Niu, C. IR780 Based Sonotherapeutic Nanoparticles to Combat Multidrug-Resistant Bacterial Infections. Front. Chem. 2022, 10, 840598.
- Yu, Q.; Deng, T.; Lin, F.-C.; Zhang, B.; Zink, J.I. Supramolecular Assemblies of Heterogeneous Mesoporous Silica Nanoparticles to Co-deliver Antimicrobial Peptides and Antibiotics for Synergistic Eradication of Pathogenic Biofilms. ACS Nano 2020, 14, 5926–5937.
- Cui, T.; Wu, S.; Sun, Y.; Ren, J.; Qu, X. Self-Propelled Active Photothermal Nanoswimmer for Deep-Layered Elimination of Biofilm In Vivo. Nano Lett. 2020, 20, 7350–7358.





