
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Kuo-Hu Chen | -- | 2034 | 2023-09-16 16:30:25 | | | |
| 2 | Jessie Wu | + 5 word(s) | 2039 | 2023-09-18 05:24:50 | | |
Video Upload Options
In a physiological state, glucocorticoids stimulate mature osteoblasts to produce Wnt proteins functioning as signaling molecules, causing a Wnt/β-catenin cascade to be activated. This signaling cascade affects mesenchymal progenitor cells, and has a positive effect on their differentiation into osteoblasts instead of chondrocytes or adipocytes. Glucocorticoids are the most widely used anti-inflammatory drug around the world. Despite their excellent effect in managing many acute inflammatory diseases and autoimmune disorders, the usage of glucocorticoids has been limited due to substantial adverse effects. One of the most well-known side effects is osteoporosis, which further induces bone fracture and other musculoskeletal problems.
1. Mechanism
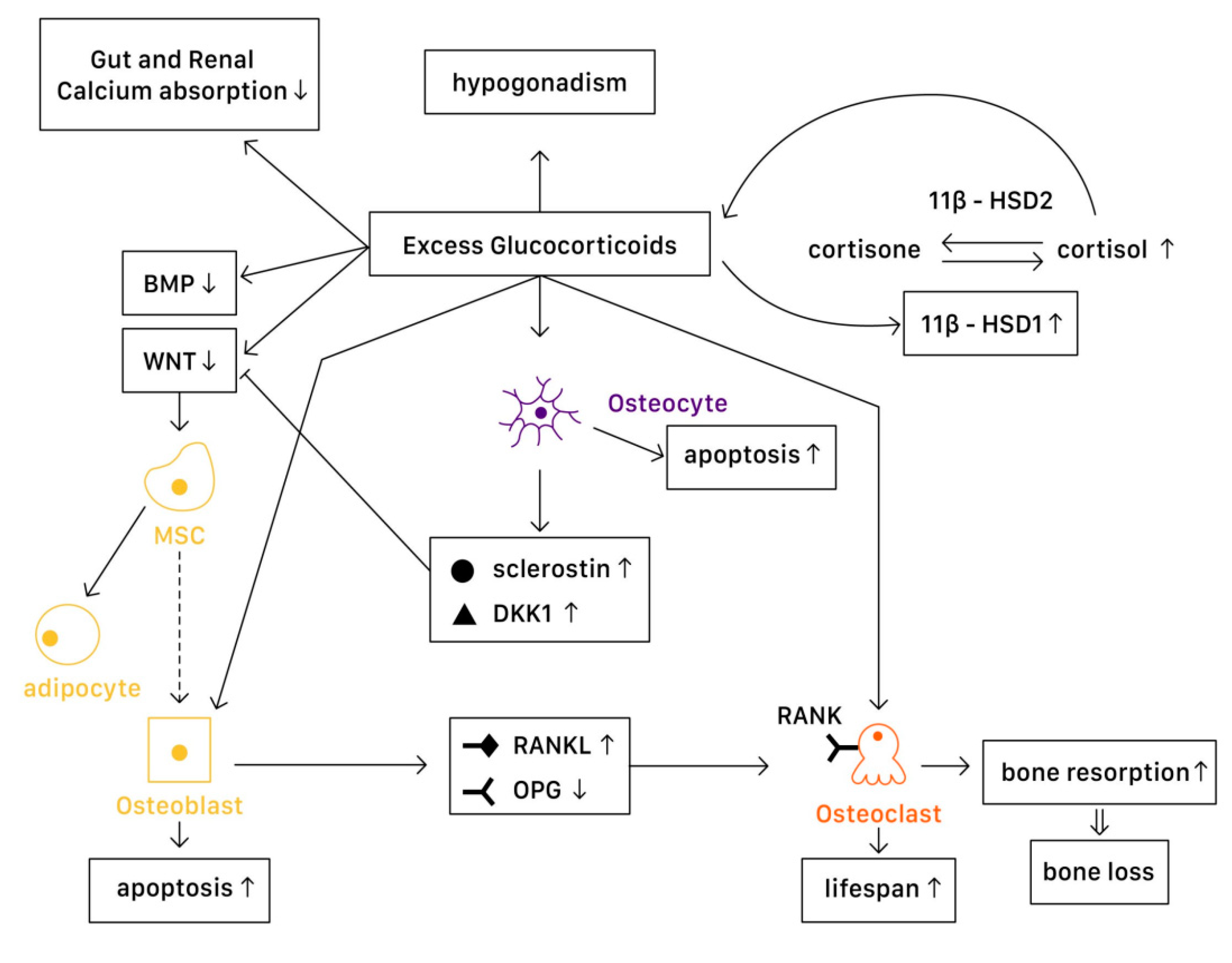
1.1. Physiology
1.2. Pathophysiology
In Osteoblasts
In Osteoclasts
In Osteocytes
Systemic Effects
2. Assessment and Screening
3. Treatment
3.1. Non-Pharmacological Treatment
3.2. Pharmacological Treatments
Bisphosphonate
Calcitonin
Hormone Replacement Therapy (HRT)
Teriparatide
Denosumab
Strontium Ranelate
Future Possible Treatments
References
- Walsh, S.; Jordan, G.R.; Jefferiss, C.; Stewart, K.; Beresford, J.N. High Concentrations of Dexamethasone Suppress the Proliferation but Not the Differentiation or Further Maturation of Human Osteoblast Precursors in Vitro: Relevance to Glucocorticoid-Induced Osteoporosis. Rheumatology 2001, 40, 74–83.
- Canalis, E. Mechanisms of Glucocorticoid-Induced Osteoporosis. Curr. Opin. Rheumatol. 2003, 15, 454–457.
- Seibel, M.J.; Cooper, M.S.; Zhou, H. Glucocorticoid-Induced Osteoporosis: Mechanisms, Management, and Future Perspectives. Lancet Diabetes Endocrinol. 2013, 1, 59–70.
- Liu, J.; Lu, C.; Wu, X.; Zhang, Z.; Li, J.; Guo, B.; Li, D.; Liang, C.; Dang, L.; Pan, X.; et al. Targeting Osteoblastic Casein Kinase-2 Interacting Protein-1 to Enhance Smad-Dependent BMP Signaling and Reverse Bone Formation Reduction in Glucocorticoid-Induced Osteoporosis. Sci. Rep. 2017, 7, 41295.
- Liu, K.; Jing, Y.; Zhang, W.; Fu, X.; Zhao, H.; Zhou, X.; Tao, Y.; Yang, H.; Zhang, Y.; Zen, K.; et al. Silencing MiR-106b Accelerates Osteogenesis of Mesenchymal Stem Cells and Rescues against Glucocorticoid-Induced Osteoporosis by Targeting BMP2. Bone 2017, 97, 130–138.
- Jensen, P.R.; Andersen, T.L.; Hauge, E.-M.; Bollerslev, J.; Delaissé, J.-M. A Joined Role of Canopy and Reversal Cells in Bone Remodeling--Lessons from Glucocorticoid-Induced Osteoporosis. Bone 2015, 73, 16–23.
- Aagaard, E.M.; Lin, P.; Modin, G.W.; Lane, N.E. Prevention of Glucocorticoid-Induced Osteoporosis: Provider Practice at an Urban County Hospital. Am. J. Med. 1999, 107, 456–460.
- Lekva, T.; Bollerslev, J.; Kristo, C.; Olstad, O.K.; Ueland, T.; Jemtland, R. The Glucocorticoid-Induced Leucine Zipper Gene (GILZ) Expression Decreases after Successful Treatment of Patients with Endogenous Cushing’s Syndrome and May Play a Role in Glucocorticoid-Induced Osteoporosis. J. Clin. Endocrinol. Metab. 2010, 95, 246–255.
- Reid, I.R. Glucocorticoid Osteoporosis--Mechanisms and Management. Eur. J. Endocrinol. 1997, 137, 209–217.
- Lukert, B.P. Glucocorticoid-Induced Osteoporosis. South. Med. J. 1992, 85, S48–S51.
- Lin, T.; Liu, J.; Yang, S.; Liu, X.; Feng, X.; Fu, D. Relation between the Development of Osteoporosis and Osteonecrosis Following Glucocorticoid in a Rabbit Model. Indian J. Orthop. 2016, 50, 406–413.
- Cabrera, D.; Kruger, M.; Wolber, F.M.; Roy, N.C.; Fraser, K. Effects of Short- and Long-Term Glucocorticoid-Induced Osteoporosis on Plasma Metabolome and Lipidome of Ovariectomized Sheep. BMC Musculoskeletal. Disord. 2020, 21, 349.
- van Bommel, E.J.M.; de Jongh, R.T.; Brands, M.; Heijboer, A.C.; den Heijer, M.; Serlie, M.J.; van Raalte, D.H. The Osteoblast: Linking Glucocorticoid-Induced Osteoporosis and Hyperglycaemia? A Post-Hoc Analysis of a Randomised Clinical Trial. Bone 2018, 112, 173–176.
- Buckley, L.; Guyatt, G.; Fink, H.A.; Cannon, M.; Grossman, J.; Hansen, K.E.; Humphrey, M.B.; Lane, N.E.; Magrey, M.; Miller, M.; et al. 2017 American College of Rheumatology Guideline for the Prevention and Treatment of Glucocorticoid-Induced Osteoporosis. Arthritis Rheumatol. 2017, 69, 1521–1537.
- Florez, H.; Hernández-Rodríguez, J.; Muxi, A.; Carrasco, J.L.; Prieto-González, S.; Cid, M.C.; Espinosa, G.; Gómez-Puerta, J.A.; Monegal, A.; Guañabens, N.; et al. Trabecular Bone Score Improves Fracture Risk Assessment in Glucocorticoid-Induced Osteoporosis. Rheumatology 2020, 59, 1574–1580.
- Yu, S.-F.; Chen, J.-F.; Chen, Y.-C.; Lai, H.-M.; Ko, C.-H.; Chiu, W.-C.; Su, F.-M.; Hsu, C.-Y.; Su, B.Y.-J.; Wu, C.-H.; et al. Beyond Bone Mineral Density, FRAX-Based Tailor-Made Intervention Thresholds for Therapeutic Decision in Subjects on Glucocorticoid: A Nationwide Osteoporosis Survey. Medicine 2017, 96, e5959.
- Kaji, H.; Yamauchi, M.; Chihara, K.; Sugimoto, T. The Threshold of Bone Mineral Density for Vertebral Fracture in Female Patients with Glucocorticoid-Induced Osteoporosis. Endocr. J. 2006, 53, 27–34.
- Graeff, C.; Marin, F.; Petto, H.; Kayser, O.; Reisinger, A.; Peña, J.; Zysset, P.; Glüer, C.-C. High Resolution Quantitative Computed Tomography-Based Assessment of Trabecular Microstructure and Strength Estimates by Finite-Element Analysis of the Spine, but Not DXA, Reflects Vertebral Fracture Status in Men with Glucocorticoid-Induced Osteoporosis. Bone 2013, 52, 568–577.
- Kalpakcioglu, B.B.; Engelke, K.; Genant, H.K. Advanced Imaging Assessment of Bone Fragility in Glucocorticoid-Induced Osteoporosis. Bone 2011, 48, 1221–1231.
- Solomon, D.H.; Katz, J.N.; Jacobs, J.P.; La Tourette, A.M.; Coblyn, J. Management of Glucocorticoid-Induced Osteoporosis in Patients with Rheumatoid Arthritis: Rates and Predictors of Care in an Academic Rheumatology Practice. Arthritis Rheum. 2002, 46, 3136–3142.
- McKeown, E.; Bykerk, V.P.; De Leon, F.; Bonner, A.; Thorne, C.; Hitchon, C.A.; Boire, G.; Haraoui, B.; Ferland, D.S.; Keystone, E.C.; et al. CATCH Investigators. Quality Assurance Study of the Use of Preventative Therapies in Glucocorticoid-Induced Osteoporosis in Early Inflammatory Arthritis: Results from the CATCH Cohort. Rheumatology 2012, 51, 1662–1669.
- Yood, R.A.; Harrold, L.R.; Fish, L.; Cernieux, J.; Emani, S.; Conboy, E.; Gurwitz, J.H. Prevention of Glucocorticoid-Induced Osteoporosis: Experience in a Managed Care Setting. Arch. Intern. Med. 2001, 161, 1322–1327.
- Sadat-Ali, M.; Alelq, A.H.; Alshafei, B.A.; Al-Turki, H.A.; Abujubara, M.A. Osteoporosis Prophylaxis in Patients Receiving Chronic Glucocorticoid Therapy. Ann. Saudi Med. 2009, 29, 215–218.
- Caballero-Uribe, C.V.; Acosta-Velez, J.G.; Hernandez-Ruiz, E.A.; Andrade Rodado, F.M.; Peñuela-Epalza, M. Prevention, Diagnosis, and Treatment of Glucocorticoid Induced Osteoporosis by Rheumatologists in a Latin-American City. J. Clin. Rheumatol. 2008, 14, 153–155.
- Werth, V.P. Glucocorticoid-Induced Osteoporosis Evaluation, Prevention, and Treatment. J. Clin. Rheumatol. 1997, 3 (Suppl. S2), 69–73.
- Hsu, E.; Nanes, M. Advances in Treatment of Glucocorticoid-Induced Osteoporosis. Curr. Opin. Endocrinol. Diabetes Obes. 2017, 24, 411–417.
- Wang, Y.-K.; Zhang, Y.-M.; Qin, S.-Q.; Wang, X.; Ma, T.; Guo, J.-B.; Zhu, C.; Luo, Z.-J. Effects of Alendronate for Treatment of Glucocorticoid-Induced Osteoporosis: A Meta-Analysis of Randomized Controlled Trials. Medicine 2018, 97, e12691.
- Kan, S.-L.; Yuan, Z.-F.; Li, Y.; Ai, J.; Xu, H.; Sun, J.-C.; Feng, S.-Q. Alendronate Prevents Glucocorticoid-Induced Osteoporosis in Patients with Rheumatic Diseases: A Meta-Analysis. Medicine 2016, 95, e3990.
- Berris, K.K.; Repp, A.L.; Kleerekoper, M. Glucocorticoid-Induced Osteoporosis. Curr. Opin. Endocrinol. Diabetes Obes. 2007, 14, 446–450.
- Adler, R.A.; Hochberg, M.C. Sugge.ested Guidelines for Evaluation and Treatment of Glucocorticoid-Induced Osteoporosis for the Department of Veterans Affairs. Arch. Intern. Med. 2003, 163, 2619–2624.
- Uchida, K.; Nakajima, H.; Miyazaki, T.; Yayama, T.; Kawahara, H.; Kobayashi, S.; Tsuchida, T.; Okazawa, H.; Fujibayashi, Y.; Baba, H. Effects of Alendronate on Bone Metabolism in Glucocorticoid-Induced Osteoporosis Measured by 18F-Fluoride PET: A Prospective Study. J. Nucl. Med. 2009, 50, 1808–1814.
- Furukawa, F.; Kaminaka, C.; Ikeda, T.; Kanazawa, N.; Yamamoto, Y.; Ohta, C.; Nishide, T.; Tsujioka, K.; Hattori, M.; Uede, K.; et al. Preliminary Study of Etidronate for Prevention of Corticosteroid-Induced Osteoporosis Caused by Oral Glucocorticoid Therapy. Clin. Exp. Dermatol. 2011, 36, 165–168.
- Yamada, S.; Takagi, H.; Tsuchiya, H.; Nakajima, T.; Ochiai, H.; Ichimura, A.; Iwata, H.; Toriyama, T. Comparative Studies on Effect of Risedronate and Alfacalcidol against Glucocorticoid-Induced Osteoporosis in Rheumatoid Arthritic Patients. Yakugaku Zasshi 2007, 127, 1491–1496.
- Sambrook, P.N.; Roux, C.; Devogelaer, J.-P.; Saag, K.; Lau, C.-S.; Reginster, J.-Y.; Bucci-Rechtweg, C.; Su, G.; Reid, D.M. Bisphosphonates and Glucocorticoid Osteoporosis in Men: Results of a Randomized Controlled Trial Comparing Zoledronic Acid with Risedronate. Bone 2012, 50, 289–295.
- Ren, H.; Shen, G.; Tang, J.; Qiu, T.; Zhang, Z.; Zhao, W.; Yu, X.; Huang, J.; Liang, D.; Yao, Z.; et al. Promotion Effect of Extracts from Plastrum Testudinis on Alendronate against Glucocorticoid-Induced Osteoporosis in Rat Spine. Sci. Rep. 2017, 7, 10617.
- Woolf, A.D. An Update on Glucocorticoid-Induced Osteoporosis. Curr. Opin. Rheumatol. 2007, 19, 370–375.
- Saag, K.G. Prevention of Glucocorticoid-Induced Osteoporosis. South. Med. J. 2004, 97, 555–559.
- Pennisi, P.; Trombetti, A.; Rizzoli, R. Glucocorticoid-Induced Osteoporosis and Its Treatment. Clin. Orthop Relat. Res. 2006, 443, 39–47.
- Kumar, R. Glucocorticoid-Induced Osteoporosis. Curr. Opin. Nephrol. Hypertens. 2001, 10, 589–595.
- Clowes, J.A.; Peel, N.; Eastell, R. Glucocorticoid-Induced Osteoporosis. Curr. Opin. Rheumatol. 2001, 13, 326–332.
- Wang, T.; Han, C.; Tian, P.; Li, P.-F.; Ma, X.-L. Role of Teriparatide in Glucocorticoid-Induced Osteoporosis through Regulating Cellular Reactive Oxygen Species. Orthop. Surg. 2018, 10, 152–159.
- Saag, K.G.; Zanchetta, J.R.; Devogelaer, J.-P.; Adler, R.A.; Eastell, R.; See, K.; Krege, J.H.; Krohn, K.; Warner, M.R. Effects of Teriparatide versus Alendronate for Treating Glucocorticoid-Induced Osteoporosis: Thirty-Six-Month Results of a Randomized, Double-Blind, Controlled Trial. Arthritis Rheum. 2009, 60, 3346–3355.
- Burshell, A.L.; Möricke, R.; Correa-Rotter, R.; Chen, P.; Warner, M.R.; Dalsky, G.P.; Taylor, K.A.; Krege, J.H. Correlations between Biochemical Markers of Bone Turnover and Bone Density Responses in Patients with Glucocorticoid-Induced Osteoporosis Treated with Teriparatide or Alendronate. Bone 2010, 46, 935–939.
- Eastell, R.; Chen, P.; Saag, K.G.; Burshell, A.L.; Wong, M.; Warner, M.R.; Krege, J.H. Bone Formation Markers in Patients with Glucocorticoid-Induced Osteoporosis Treated with Teriparatide or Alendronate. Bone 2010, 46, 929–934.
- Mazziotti, G.; Maffezzoni, F.; Doga, M.M.; Hofbauer, L.C.; Adler, R.A.; Giustina, A. Outcome of Glucose Homeostasis in Patients with Glucocorticoid-Induced Osteoporosis Undergoing Treatment with Bone Active-Drugs. Bone 2014, 67, 175–180.
- Murphy, D.R.; Smolen, L.J.; Klein, T.M.; Klein, R.W. The Cost Effectiveness of Teriparatide as a First-Line Treatment for Glucocorticoid-Induced and Postmenopausal Osteoporosis Patients in Sweden. BMC Musculoskelet. Disord. 2012, 13, 213.
- Saag, K.G.; Wagman, R.B.; Geusens, P.; Adachi, J.D.; Messina, O.D.; Emkey, R.; Chapurlat, R.; Wang, A.; Pannacciulli, N.; Lems, W.F. Denosumab versus Risedronate in Glucocorticoid-Induced Osteoporosis: A Multicentre, Randomised, Double-Blind, Active-Controlled, Double-Dummy, Non-Inferiority Study. Lancet Diabetes Endocrinol. 2018, 6, 445–454.
- Etani, Y.; Ebina, K.; Hirao, M.; Kitaguchi, K.; Kashii, M.; Ishimoto, T.; Nakano, T.; Okamura, G.; Miyama, A.; Takami, K.; et al. Combined Effect of Teriparatide and an Anti-RANKL Monoclonal Antibody on Bone Defect Regeneration in Mice with Glucocorticoid-Induced Osteoporosis. Bone 2020, 139, 115525.
- Lu, S.-Y.; Wang, C.-Y.; Jin, Y.; Meng, Q.; Liu, Q.; Liu, Z.; Liu, K.-X.; Sun, H.-J.; Liu, M.-Z. The Osteogenesis-Promoting Effects of Alpha-Lipoic Acid against Glucocorticoid-Induced Osteoporosis through the NOX4, NF-KappaB, JNK and PI3K/AKT Pathways. Sci. Rep. 2017, 7, 3331.
- Sato, D.; Takahata, M.; Ota, M.; Fukuda, C.; Hasegawa, T.; Yamamoto, T.; Amizuka, N.; Tsuda, E.; Okada, A.; Hiruma, Y.; et al. Siglec-15-Targeting Therapy Protects against Glucocorticoid-Induced Osteoporosis of Growing Skeleton in Juvenile Rats. Bone 2020, 135, 115331.
- Hozayen, W.G.; El-Desouky, M.A.; Soliman, H.A.; Ahmed, R.R.; Khaliefa, A.K. Antiosteoporotic Effect of Petroselinum Crispum, Ocimum Basilicum and Cichorium Intybus L. in Glucocorticoid-Induced Osteoporosis in Rats. BMC Complement. Altern. Med. 2016, 16, 165.
- Chen, Z.; Xue, J.; Shen, T.; Ba, G.; Yu, D.; Fu, Q. Curcumin Alleviates Glucocorticoid-Induced Osteoporosis by Protecting Osteoblasts from Apoptosis in Vivo and in Vitro. Clin. Exp. Pharmacol. Physiol. 2016, 43, 268–276.
- Kim, J.; Lee, H.; Kang, K.S.; Chun, K.-H.; Hwang, G.S. Protective Effect of Korean Red Ginseng against Glucocorticoid-Induced Osteoporosis in Vitro and in Vivo. J. Ginseng Res. 2015, 39, 46–53.





