
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Julie Vinkel Clausen | -- | 2325 | 2023-09-01 09:27:22 | | | |
| 2 | Jessie Wu | + 1 word(s) | 2326 | 2023-09-01 10:03:15 | | |
Video Upload Options
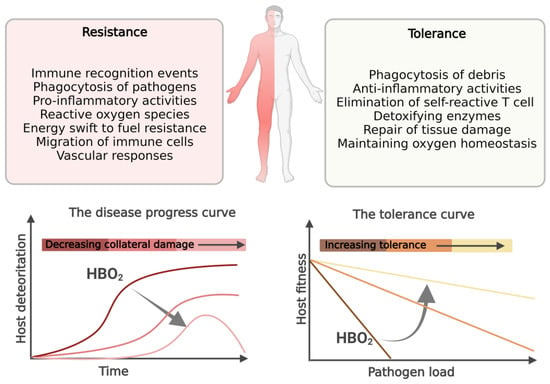
Sepsis and septic shock continue to be a leading cause of death worldwide. Sepsis was previously thought to be an overwhelming, systemic, proinflammatory response to infection, which was followed by a phase of immunosuppression. New paradigms suggest that the proinflammatory and immunosuppression phases occurs simultaneously, and the pathophysiology begind the disease complex is not only explained by the pathogen’s type, load and virulence, but to a large extend also by host’s dysregulated response to infection. Many of these dysregulated host immune responses that occurs in sepsis are also targets of hyperbaric oxygen (HBO2) treatment. HBO2 treatment has been shown to improve survival in clinical studies on patients with necrotizing soft tissue infections as well as experimental sepsis models. Inflammation and oxygen-sensing pathways are connected on the cellular level in a self-reinforcing and detrimental manner in inflammatory conditions, which may be interrupted when intervening with HBO2 treatment. HBO2 treatment acts to maintain homeostasis by protecting the host from collateral tissue damage during resistance to infection by reducing neutrophil extracellular traps, inhibiting neutrophil adhesion to vascular endothelium, reducing proinflammatory cytokines, and halting the Warburg effect, while also aiding the host in tolerance to infection by reducing iron-mediated injury and upregulating anti-inflammatory measures.
1. Neutrophil Mediated Responses in Sepsis
2. Endothelial Dysfunction in Sepsis
3. The Cytokine Mediated Inflammatory Response in Sepsis
4 . Switching Immune Cell Metabolism toward Glycolysis in Sepsis—The Warburg Effect
5. Iron Metabolism in The Pathogenesis of Sepsis
In severe bacterial infections, invading bacteria are frequently transferred from the local infectious site to the bloodstream, where they can cause hemolysis, which results in the oxidation of extracellular hemoglobin and the accumulation of labile heme in plasma [31][32]. To increase their tolerance for infection, the host has defenses against the cytotoxic effects of heme. HO-1 (Heme oxygenase-1) catabolizes heme and breaks it down into biliverdin, iron (Fe), and carbon monoxide [33]. The expression of HO-1 is dependent on the cell type, cellular microenvironment, intensity and duration of stimuli exposure, and is regulated by a panel of redox-sensitive transcription factors, including HIF-1α and NF-κB [34]. In numerous studies and circumstances, HBO2 treatment has been linked to a protective impact of enhanced HO-1 activity [35][36][37][38][39][40]. Most pertinent to the aim of this research is that levels of HO-1 have been shown to increase in a subgroup of NSTI patients with septic shock in response to HBO2 treatment [41]. Other authors have likewise demonstrated an effect of HBO2 treatment on HO-1 with a 30-fold increase in lymphocytes of healthy volunteers 24 h after HBO2 treatment [38]. The beneficial effects of HO-1 stimulation in sepsis have been demonstrated in a lipopolysaccharide-induced model, demonstrating that HO-1 induction reduces acute lung injury [35].
6. The Anti-Inflammatory Response in Sepsis
Specific anti-inflammatory cytokines balance out the strong proinflammatory response to restore immunological balance and increase host resistance to infection [42]. Proinflammatory cytokines, anti-inflammatory cytokines, and soluble inhibitors of proinflammatory cytokines make up the cytokine network, and a tightly controlled balance between these three groups of cytokines is essential for preventing excessive, tissue-damaging inflammation during the thorough removal of invading pathogens [43][44]. IL-10 (interleukin-10), IL-4 (interleukin-4), and TGF-β (tumor growth factor beta) are the three best described anti-inflammatory regulators operating in sepsis [45]. There have been few investigations on the influence of HBO2 medication on the production of anti-inflammatory cytokines in sepsis, with inconsistent results. One clinical study found no effect of HBO2 treatment on IL-10 in septic patients after one or three treatment sessions [20]. In a cecal ligation and puncture-induced sepsis model macrophages isolated from HBO2-treated mice demonstrated enhanced IL-10 secretion as compared with controls, and IL-10 deficiency mice were not protected from sepsis mortality by IL-10 expression [46]. A protective effect of IL-10 elevation in response to HBO2 treatment has also been demonstrated in another model [47]. Animal studies have found that HBO2 treatment decreases the level of TGF-β messengers and proteins in several conditions [48][49][50][51], while IL-4 is upregulated in response to HBO2 treatment [89-91]. This upregulation of IL-4 was confirmed in a systematic review on cytokine responses to HBO2 in human tissue, whereas the authors found no effect on IL-10 and a possible reducing effect on TGF-β [18].
7. Hypoxia in The Pathogenesis of Sepsis
During an infection, tissue hypoxia is caused by vascular dysfunction and increased oxygen consumption due to increased metabolic activity of immune cells. Hypoxia acts as a stressor in cells, activating NF-κB, which then upregulates hypoxia-inducible factors (HIFs) and the cellular hypoxia response in a positive feedback loop [92, 93]. This might culminate in reinforcing pathophysiological effects in which both hypoxia and inflammation contribute to disease [52]. It has been revealed that HIF-1 may be a critical determinant of the sepsis phenotype due to its association with the production of pro-inflammatory cytokines, which results in the clinical manifestation of sepsis symptoms such as tachycardia, hypotension, and hypothermia [53][54]. Changes in oxygen availability, rather than constant hypoxia or hyperoxia, have a more dominant effect on HIF-1α expression, making HBO2 treatment a potent regulator of HIF expression [55][56][57]. The effect of HBO2 treatment on HIF is multi-faceted and may occur both at the transcriptional and post-transcriptional level mediated by the actions of oxygen and reactive oxygen species. The treatment-mediated oxygen supply will result in the O2-sensitive hydroxylation of HIF-1α proteins and its inactivation via the actions of PHD and FIH. The exact mechanism by which oxygen sensing regulates the NF-κB pathway is still a matter of debate [58][59][60]. Both PHDs and FIH have been identified as playing a role in the control of NF-κB activity in hypoxia, either by directly impacting IKKβ (inhibitor of nuclear factor kappa B kinase subunit beta) activity, or through hydroxylase activity, ultimately leading to hindered tyrosine phosphorylation of IκBα (NF-kappa-B inhibitor alpha), and the continuous binding of this inhibitory protein [59]. When HBO2 treatment is administered in hypoxic inflammatory conditions, the regulation of HIF-1α will primarily be driven by increased oxygen delivery, which causes HIF-1α to be hydroxylated and degraded via the actions of PHD, thereby decreasing the expression of HIF-1α proteins [95]. This is in line with the previous observation that HBO2 administrated to non-inflamed well-oxygenated tissue causes an increase in HIF-1α protein levels. However, when given to inflamed and ischemic tissue that overexpresses HIF-1α, the overexpressed HIF-1α is reduced [61]. The oxygen-mediated activation of PHD and HIF will leave NF-κB bound to its inhibitory IκBα preventing its target genes to be transcribed [59]. As a result, in septic conditions, HBO2 treatment may break the positive feedback loop mechanism by which hypoxia induces inflammation and inflammation induces hypoxia, thereby preventing the damage caused by this cycle (Figure 2) [52].
References
- Elsbach, P.; Weiss, J. Oxygen-dependent and oxygen-independent mechanisms of microbicidal activity of neutrophils. Immunol. Lett. 1985, 11, 159–163.
- Nguyen, G.T.; Green, E.R.; Mecsas, J. Neutrophils to the ROScue: Mechanisms of NADPH Oxidase Activation and Bacterial Resistance. Front. Cell Infect. Microbiol. 2017, 7, 373.
- van der Poll, T.; van de Veerdonk, F.L.; Scicluna, B.P.; Netea, M.G. The immunopathology of sepsis and potential therapeutic targets. Nat. Rev. Immunol. 2017, 17, 407–420.
- Grimberg-Peters, D.; Buren, C.; Windolf, J.; Wahlers, T.; Paunel-Gorgulu, A. Hyperbaric Oxygen Reduces Production of Reactive Oxygen Species in Neutrophils from Polytraumatized Patients Yielding in the Inhibition of p38 MAP Kinase and Downstream Pathways. PLoS ONE 2016, 11, e0161343.
- de Wolde, S.D.; Hulskes, R.H.; de Jonge, S.W.; Hollmann, M.W.; van Hulst, R.A.; Weenink, R.P.; Kox, M. The Effect of Hyperbaric Oxygen Therapy on Markers of Oxidative Stress and the Immune Response in Healthy Volunteers. Front. Physiol. 2022, 13, 826163.
- Ince, C.; Mayeux, P.R.; Nguyen, T.; Gomez, H.; Kellum, J.A.; Ospina-Tascon, G.A.; Hernandez, G.; Murray, P.; De Backer, D.; Workgroup, A.X. The Endothelium in Sepsis. Shock 2016, 45, 259–270.
- Zamboni, W.A.; Roth, A.C.; Russell, R.C.; Graham, B.; Suchy, H.; Kucan, J.O. Morphologic analysis of the microcirculation during reperfusion of ischemic skeletal muscle and the effect of hyperbaric oxygen. Plast. Reconstr. Surg. 1993, 91, 1110–1123.
- Baiula, M.; Greco, R.; Ferrazzano, L.; Caligiana, A.; Hoxha, K.; Bandini, D.; Longobardi, P.; Spampinato, S.; Tolomelli, A. Integrin-mediated adhesive properties of neutrophils are reduced by hyperbaric oxygen therapy in patients with chronic non-healing wound. PLoS ONE 2020, 15, e0237746.
- Kalns, J.; Lane, J.; Delgado, A.; Scruggs, J.; Ayala, E.; Gutierrez, E.; Warren, D.; Niemeyer, D.; George Wolf, E.; Bowden, R.A. Hyperbaric oxygen exposure temporarily reduces Mac-1 mediated functions of human neutrophils. Immunol. Lett. 2002, 83, 125–131.
- Thom, S.R.; Bhopale, V.M.; Mancini, D.J.; Milovanova, T.N. Actin S-nitrosylation inhibits neutrophil beta2 integrin function. J. Biol. Chem. 2008, 283, 10822–10834.
- Hedetoft, M.; Moser, C.; Jensen, P.O.; Vinkel, J.; Hyldegaard, O. Soluble ICAM-1 is modulated by hyperbaric oxygen treatment and correlates with disease severity and mortality in patients with necrotizing soft-tissue infection. J. Appl. Physiol. (1985) 2021, 130, 729–736.
- Buras, J.A.; Stahl, G.L.; Svoboda, K.K.; Reenstra, W.R. Hyperbaric oxygen downregulates ICAM-1 expression induced by hypoxia and hypoglycemia: The role of NOS. Am. J. Physiol. Cell Physiol. 2000, 278, C292–C302.
- Macdonald, J.; Galley, H.F.; Webster, N.R. Oxidative stress and gene expression in sepsis. Br. J. Anaesth. 2003, 90, 221–232.
- Bubici, C.; Papa, S.; Dean, K.; Franzoso, G. Mutual cross-talk between reactive oxygen species and nuclear factor-kappa B: Molecular basis and biological significance. Oncogene 2006, 25, 6731–6748.
- Liu, S.; Lu, C.; Liu, Y.; Zhou, X.; Sun, L.; Gu, Q.; Shen, G.; Guo, A. Hyperbaric Oxygen Alleviates the Inflammatory Response Induced by LPS Through Inhibition of NF-kappaB/MAPKs-CCL2/CXCL1 Signaling Pathway in Cultured Astrocytes. Inflammation 2018, 41, 2003–2011.
- Sakoda, M.; Ueno, S.; Kihara, K.; Arikawa, K.; Dogomori, H.; Nuruki, K.; Takao, S.; Aikou, T. A potential role of hyperbaric oxygen exposure through intestinal nuclear factor-kappaB. Crit. Care Med. 2004, 32, 1722–1729.
- Lu, Z.; Ma, J.; Liu, B.; Dai, C.; Xie, T.; Ma, X.; Li, M.; Dong, J.; Lan, Q.; Huang, Q. Hyperbaric oxygen therapy sensitizes nimustine treatment for glioma in mice. Cancer Med. 2016, 5, 3147–3155.
- De Wolde, S.D.; Hulskes, R.H.; Weenink, R.P.; Hollmann, M.W.; Van Hulst, R.A. The Effects of Hyperbaric Oxygenation on Oxidative Stress, Inflammation and Angiogenesis. Biomolecules 2021, 11, 1210.
- Weisz, G.; Lavy, A.; Adir, Y.; Melamed, Y.; Rubin, D.; Eidelman, S.; Pollack, S. Modification of in vivo and in vitro TNF-alpha, IL-1, and IL-6 secretion by circulating monocytes during hyperbaric oxygen treatment in patients with perianal Crohn’s disease. J. Clin. Immunol. 1997, 17, 154–159.
- Hedetoft, M.; Garred, P.; Madsen, M.B.; Hyldegaard, O. Hyperbaric oxygen treatment is associated with a decrease in cytokine levels in patients with necrotizing soft-tissue infection. Physiol. Rep. 2021, 9, e14757.
- Warburg, O.; Wind, F.; Negelein, E. The Metabolism of Tumors in the Body. J. Gen. Physiol. 1927, 8, 519–530.
- Srivastava, A.; Mannam, P. Warburg revisited: Lessons for innate immunity and sepsis. Front. Physiol. 2015, 6, 70.
- Cheng, S.C.; Quintin, J.; Cramer, R.A.; Shepardson, K.M.; Saeed, S.; Kumar, V.; Giamarellos-Bourboulis, E.J.; Martens, J.H.; Rao, N.A.; Aghajanirefah, A.; et al. mTOR- and HIF-1alpha-mediated aerobic glycolysis as metabolic basis for trained immunity. Science 2014, 345, 1250684.
- Bar-Or, D.; Carrick, M.; Tanner, A., 2nd; Lieser, M.J.; Rael, L.T.; Brody, E. Overcoming the Warburg Effect: Is it the key to survival in sepsis? J. Crit. Care 2018, 43, 197–201.
- Yang, L.; Xie, M.; Yang, M.; Yu, Y.; Zhu, S.; Hou, W.; Kang, R.; Lotze, M.T.; Billiar, T.R.; Wang, H.; et al. PKM2 regulates the Warburg effect and promotes HMGB1 release in sepsis. Nat. Commun. 2014, 5, 4436.
- Wang, R.; Green, D.R. Metabolic checkpoints in activated T cells. Nat. Immunol. 2012, 13, 907–915.
- Tezgin, D.; Giardina, C.; Perdrizet, G.A.; Hightower, L.E. The effect of hyperbaric oxygen on mitochondrial and glycolytic energy metabolism: The caloristasis concept. Cell Stress Chaperones 2020, 25, 667–677.
- Zhang, L.; Ke, J.; Min, S.; Wu, N.; Liu, F.; Qu, Z.; Li, W.; Wang, H.; Qian, Z.; Wang, X. Hyperbaric Oxygen Therapy Represses the Warburg Effect and Epithelial-Mesenchymal Transition in Hypoxic NSCLC Cells via the HIF-1alpha/PFKP Axis. Front. Oncol. 2021, 11, 691762.
- Poff, A.M.; Ward, N.; Seyfried, T.N.; Arnold, P.; D’Agostino, D.P. Non-Toxic Metabolic Management of Metastatic Cancer in VM Mice: Novel Combination of Ketogenic Diet, Ketone Supplementation, and Hyperbaric Oxygen Therapy. PLoS ONE 2015, 10, e0127407.
- Poff, A.M.; Ari, C.; Seyfried, T.N.; D’Agostino, D.P. The ketogenic diet and hyperbaric oxygen therapy prolong survival in mice with systemic metastatic cancer. PLoS ONE 2013, 8, e65522.
- Larsen, R.; Gozzelino, R.; Jeney, V.; Tokaji, L.; Bozza, F.A.; Japiassu, A.M.; Bonaparte, D.; Cavalcante, M.M.; Chora, A.; Ferreira, A.; et al. A central role for free heme in the pathogenesis of severe sepsis. Sci. Transl. Med. 2010, 2, 51ra71.
- Weis, S.; Carlos, A.R.; Moita, M.R.; Singh, S.; Blankenhaus, B.; Cardoso, S.; Larsen, R.; Rebelo, S.; Schauble, S.; Del Barrio, L.; et al. Metabolic Adaptation Establishes Disease Tolerance to Sepsis. Cell 2017, 169, 1263–1275.e14.
- Tenhunen, R.; Marver, H.S.; Schmid, R. The enzymatic conversion of heme to bilirubin by microsomal heme oxygenase. Proc. Natl. Acad. Sci. USA 1968, 61, 748–755.
- Prawan, A.; Kundu, J.K.; Surh, Y.J. Molecular basis of heme oxygenase-1 induction: Implications for chemoprevention and chemoprotection. Antioxid. Redox Signal. 2005, 7, 1688–1703.
- Huang, T.Y.; Tsai, P.S.; Wang, T.Y.; Huang, C.L.; Huang, C.J. Hyperbaric oxygen attenuation of lipopolysaccharide-induced acute lung injury involves heme oxygenase-1. Acta Anaesthesiol. Scand. 2005, 49, 1293–1301.
- Li, Q.; Li, J.; Zhang, L.; Wang, B.; Xiong, L. Preconditioning with hyperbaric oxygen induces tolerance against oxidative injury via increased expression of heme oxygenase-1 in primary cultured spinal cord neurons. Life Sci. 2007, 80, 1087–1093.
- He, X.; Xu, X.; Fan, M.; Chen, X.; Sun, X.; Luo, G.; Chen, L.; Mu, Q.; Feng, Y.; Mao, Q.; et al. Preconditioning with hyperbaric oxygen induces tolerance against renal ischemia-reperfusion injury via increased expression of heme oxygenase-1. J. Surg. Res. 2011, 170, e271–e277.
- Speit, G.; Dennog, C.; Eichhorn, U.; Rothfuss, A.; Kaina, B. Induction of heme oxygenase-1 and adaptive protection against the induction of DNA damage after hyperbaric oxygen treatment. Carcinogenesis 2000, 21, 1795–1799.
- Rothfuss, A.; Radermacher, P.; Speit, G. Involvement of heme oxygenase-1 (HO-1) in the adaptive protection of human lymphocytes after hyperbaric oxygen (HBO) treatment. Carcinogenesis 2001, 22, 1979–1985.
- Yin, X.; Wang, X.; Fan, Z.; Peng, C.; Ren, Z.; Huang, L.; Liu, Z.; Zhao, K. Hyperbaric Oxygen Preconditioning Attenuates Myocardium Ischemia-Reperfusion Injury Through Upregulation of Heme Oxygenase 1 Expression: PI3K/Akt/Nrf2 Pathway Involved. J. Cardiovasc. Pharmacol. Ther. 2015, 20, 428–438.
- Hedetoft, M.; Jensen, P.O.; Moser, C.; Vinkel, J.; Hyldegaard, O. Hyperbaric oxygen treatment impacts oxidative stress markers in patients with necrotizing soft-tissue infection. J. Investig. Med. 2021, 69, 1330–1338.
- Arina, P.; Singer, M. Pathophysiology of sepsis. Curr. Opin. Anaesthesiol. 2021, 34, 77–84.
- van der Poll, T.; van Deventer, S.J. Cytokines and anticytokines in the pathogenesis of sepsis. Infect. Dis. Clin. N. Am. 1999, 13, 413–426.
- Hack, C.E.; Aarden, L.A.; Thijs, L.G. Role of cytokines in sepsis. Adv. Immunol. 1997, 66, 101–195.
- Schulte, W.; Bernhagen, J.; Bucala, R. Cytokines in sepsis: Potent immunoregulators and potential therapeutic targets—An updated view. Mediators Inflamm. 2013, 2013, 165974.
- Buras, J.A.; Holt, D.; Orlow, D.; Belikoff, B.; Pavlides, S.; Reenstra, W.R. Hyperbaric oxygen protects from sepsis mortality via an interleukin-10-dependent mechanism. Crit. Care Med. 2006, 34, 2624–2629.
- Bai, X.; Song, Z.; Zhou, Y.; Pan, S.; Wang, F.; Guo, Z.; Jiang, M.; Wang, G.; Kong, R.; Sun, B. The apoptosis of peripheral blood lymphocytes promoted by hyperbaric oxygen treatment contributes to attenuate the severity of early stage acute pancreatitis in rats. Apoptosis 2014, 19, 58–75.
- Yoshinoya, Y.; Bocker, A.H.; Ruhl, T.; Siekmann, U.; Pallua, N.; Beier, J.P.; Kim, B.S. The Effect of Hyperbaric Oxygen Therapy on Human Adipose-Derived Stem Cells. Plast. Reconstr. Surg. 2020, 146, 309–320.
- Romero-Valdovinos, M.; Cardenas-Mejia, A.; Gutierrez-Gomez, C.; Flisser, A.; Kawa-Karasik, S.; Ortiz-Monasterio, F. Keloid skin scars: The influence of hyperbaric oxygenation on fibroblast growth and on the expression of messenger RNA for insulin like growth factor and for transforming growth factor. In Vitro Cell Dev. Biol. Anim. 2011, 47, 421–424.
- Oscarsson, N.; Ny, L.; Molne, J.; Lind, F.; Ricksten, S.E.; Seeman-Lodding, H.; Giglio, D. Hyperbaric oxygen treatment reverses radiation induced pro-fibrotic and oxidative stress responses in a rat model. Free Radic. Biol. Med. 2017, 103, 248–255.
- Wee, H.Y.; Lim, S.W.; Chio, C.C.; Niu, K.C.; Wang, C.C.; Kuo, J.R. Hyperbaric oxygen effects on neuronal apoptosis associations in a traumatic brain injury rat model. J. Surg. Res. 2015, 197, 382–389.
- Choudhury, R. Hypoxia and hyperbaric oxygen therapy: A review. Int. J. Gen. Med. 2018, 11, 431–442.
- Fitzpatrick, S.F.; Gojkovic, M.; Macias, D.; Tegnebratt, T.; Lu, L.; Samen, E.; Rundqvist, H.; Johnson, R.S. Glycolytic Response to Inflammation Over Time: Role of Myeloid HIF-1alpha. Front. Physiol. 2018, 9, 1624.
- Peyssonnaux, C.; Cejudo-Martin, P.; Doedens, A.; Zinkernagel, A.S.; Johnson, R.S.; Nizet, V. Cutting edge: Essential role of hypoxia inducible factor-1alpha in development of lipopolysaccharide-induced sepsis. J. Immunol. 2007, 178, 7516–7519.
- Cimino, F.; Balestra, C.; Germonpre, P.; De Bels, D.; Tillmans, F.; Saija, A.; Speciale, A.; Virgili, F. Pulsed high oxygen induces a hypoxic-like response in human umbilical endothelial cells and in humans. J. Appl. Physiol. (1985) 2012, 113, 1684–1689.
- Balestra, C.; Germonpre, P. Hypoxia, a multifaceted phenomenon: The example of the “normobaric oxygen paradox”. Eur. J. Appl. Physiol. 2012, 112, 4173–4175.
- Rocco, M.; D’Itri, L.; De Bels, D.; Corazza, F.; Balestra, C. The "normobaric oxygen paradox”: A new tool for the anesthetist? Minerva Anestesiol. 2014, 80, 366–372.
- D’Ignazio, L.; Bandarra, D.; Rocha, S. NF-kappaB and HIF crosstalk in immune responses. FEBS J. 2016, 283, 413–424.
- D’Ignazio, L.; Rocha, S. Hypoxia Induced NF-kappaB. Cells 2016, 5, 10.
- Eltzschig, H.K.; Carmeliet, P. Hypoxia and inflammation. N. Engl. J. Med. 2011, 364, 656–665.
- Hadanny, A.; Efrati, S. The Hyperoxic-Hypoxic Paradox. Biomolecules 2020, 10, 958.
- Schneider, D.S.; Ayres, J.S. Two ways to survive infection: What resistance and tolerance can teach us about treating infectious diseases. Nat. Rev. Immunol. 2008, 8, 889–895.





