
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Anudeep Talagavadi Channaiah | -- | 3180 | 2023-08-07 11:22:16 | | | |
| 2 | Jessie Wu | Meta information modification | 3180 | 2023-08-08 04:56:24 | | |
Video Upload Options
Alopecia or baldness is a common diagnosis in clinical practice. Alopecia can be scarring or non-scarring, diffuse or patchy. The most prevalent type of alopecia is non-scarring alopecia, with the majority of cases being androgenetic alopecia (AGA) or alopecia areata (AA). AGA is traditionally treated with minoxidil and finasteride, while AA is treated with immune modulators; however, both treatments have significant downsides.
1. Treatment of Non-Scarring Alopecia
| Stem Cell-Rich | Growth Factor-Rich |
|---|---|
|
I. Adult stem cells A. Adipose derived stem cell (ADSC) 1. Nanofat 2. Stromal vascular fraction (SVF) B. Hair follicular stem cell (HFSCs) 1. Autologous micro grafts [Human intra and extra dermal adipose tissue derived hair follicle stem cells (HD-AFSCs)] 2. Cultured HFSCs i. Hair follicle-derived MSCs (HF-MSC) ii. Hair follicle epidermal stem cells (HF-ESC) C. Bone marrow derived 1. Bone marrow mononuclear cells (BMMC) 2. Bone marrow aspirate concentrate (BMAC) II. Perinatal stem cells A. Umbilical cord blood derived B. Wharton jelly MSCs (WJ-MSC)/Umbilical cord MSCs (UC-MSC) 1. Sub amniotic. 2. Perivascular. 3. Intervascular. C. Amniotic fluid derived D. Placental MSC |
I. Platelet-rich plasma (PRP) A. Autologous activated PRP (AA-PRP) B. Autologous non-activated PRP (A-PRP) II. Autologous growth factor concentrate (GFC) III. Conditioned medium (Secretomes) A. ADSC-CM (AAPE) B. hUCB-MSC-CM C. AF-MSC-CM D. HF-MSC-CM E. BM-MSC-CM (Genetically engineered) IV. Extracellular vesicles A. Exosomes i. New-born foreskin stem cell ii. DPC iii. BM-MSC B. Exosome-like (Ginseng) C. Microvesicles V. Placental extract |
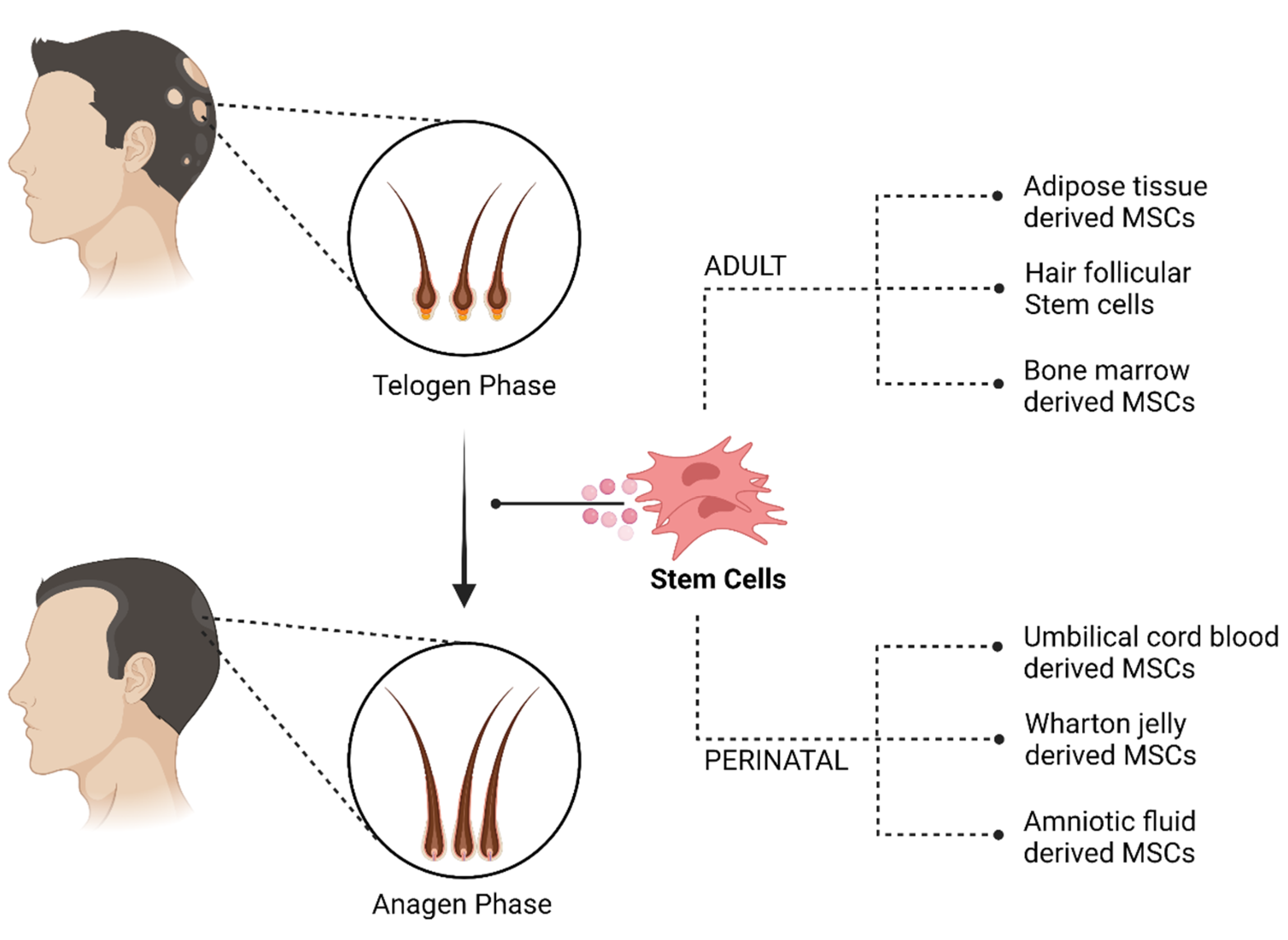
2. Cellular Therapy
2.1. Adult Stem Cells
2.1.1. Adipose Tissue-Derived Cells
2.1.2. Hair Follicular Stem Cells
2.1.3. Bone Marrow-Derived Cells
References
- Kelly, Y.; Blanco, A.; Tosti, A. Androgenetic Alopecia: An Update of Treatment Options. Drugs 2016, 76, 1349–1364.
- Mella, J.M.; Perret, M.C.; Manzotti, M.; Catalano, H.N.; Guyatt, G. Efficacy and Safety of Finasteride Therapy for Androgenetic Alopecia. Arch. Dermatol. 2010, 146, 1141–1150.
- Rahimi-Ardabili, B.; Pourandarjani, R.; Habibollahi, P.; Mualeki, A. Finasteride induced depression: A prospective study. BMC Clin. Pharmacol. 2006, 6, 7.
- D’Amico, A.V.; Roehrborn, C.G. Effect of 1 mg/day finasteride on concentrations of serum prostate-specific antigen in men with androgenic alopecia: A randomised controlled trial. Lancet Oncol. 2007, 8, 21–25.
- Lachgar, S.; Charveron, M.; Gall, Y.; Bonafe, J.L. Minoxidil upregulates the expression of vascular endothelial growth factor in human hair dermal papilla cells. Br. J. Dermatol. 1998, 138, 407–411.
- Marubayashi, A.; Nakaya, Y.; Fukui, K.; Li, M.; Arase, S. Minoxidil-Induced Hair Growth is Mediated by Adenosine in Cultured Dermal Papilla Cells: Possible Involvement of Sulfonylurea Receptor 2B as a Target of Minoxidil. J. Investig. Dermatol. 2001, 117, 1594–1600.
- FDA. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/97/20834_rogaine%20extra%20strength%20for%20men%205%25_medr.pdf (accessed on 8 January 2022).
- Lucky, A.W.; Piacquadio, D.J.; Ditre, C.M.; Dunlap, F.; Kantor, I.; Pandya, A.G.; Savin, R.C.; Tharp, M.D. A randomized, placebo-controlled trial of 5% and 2% topical minoxidil solutions in the treatment of female pattern hair loss. J. Am. Acad. Dermatol. 2004, 50, 541–553.
- Pratt, C.H.; King, L.E.; Messenger, A.G.; Christiano, A.M.; Sundberg, J.P. Alopecia areata. Nat. Rev. Dis. Prim. 2017, 3, 1–17.
- Walker, S.A.; Rothman, S. A Statistical Study and Consideration of Endocrine Influences. J. Investig. Dermatol. 1950, 14, 403–413.
- Alsantali, A. Alopecia areata: A new treatment plan. Clin. Cosmet. Investig. Dermatol. 2011, 4, 107–115.
- Gentile, P.; Garcovich, S. Autologous activated platelet-rich plasma (AA-PRP) and non-activated (A-PRP) in hair growth: A retrospective, blinded, randomized evaluation in androgenetic alopecia. Expert Opin. Biol. Ther. 2020, 20, 327–337.
- Totey, S.M.; Dhurat, R.S.; Kadam, P.P.; Sevilla, G.P.; Shetty, G. Safety and efficacy of growth factor concentrate in the treatment of nasolabial fold correction: Split face pilot study. Indian J. Dermatol. 2015, 60, 520.
- Fukuoka, H.; Narita, K.; Suga, H. Hair Regeneration Therapy: Application of Adipose-Derived Stem Cells. Curr. Stem Cell Res. Ther. 2017, 12, 531–534.
- Oh, H.A.; Kwak, J.; Kim, B.J.; Jin, H.J.; Park, W.S.; Choi, S.J.; Oh, W.; Um, S. Migration Inhibitory Factor in Conditioned Medium from Human Umbilical Cord Blood-Derived Mesenchymal Stromal Cells Stimulates Hair Growth. Cells 2020, 9, 1344.
- Yoon, B.S.; Moon, J.-H.; Jun, E.K.; Kim, J.; Maeng, I.; Kim, J.S.; Lee, J.H.; Baik, C.S.; Kim, A.; Cho, K.S.; et al. Secretory Profiles and Wound Healing Effects of Human Amniotic Fluid–Derived Mesenchymal Stem Cells. Stem Cells Dev. 2010, 19, 887–902.
- Ou, K.-L.; Kuo, Y.-W.; Wu, C.-Y.; Huang, B.-H.; Pai, F.-T.; Chou, H.-H.; Saito, T.; Ueno, T.; Cho, Y.-C.; Huang, M.-S. The Potential of a Hair Follicle Mesenchymal Stem Cell-Conditioned Medium for Wound Healing and Hair Follicle Regeneration. Appl. Sci. 2020, 10, 2646.
- Payushina, O.V.; Butorina, N.N.; Sheveleva, O.N.; Domaratskaya, E.I. Effect of Mesenchymal Stromal Cells and Conditioned Media on Healing of Skin Wound. Bull. Exp. Biol. Med. 2018, 165, 572–575.
- Carrasco, E.; Soto-Heredero, G.; Mittelbrunn, M. The Role of Extracellular Vesicles in Cutaneous Remodeling and Hair Follicle Dynamics. Int. J. Mol. Sci. 2019, 20, 2758.
- Somuncu Özge, S.; Taşlı, P.N.; Şişli, H.B.; Somuncu, S.; Şahin, F. Characterization and Differentiation of Stem Cells Isolated from Human Newborn Foreskin Tissue. Appl. Biochem. Biotechnol. 2015, 177, 1040–1054.
- Yan, H.; Gao, Y.; Ding, Q.; Liu, J.; Li, Y.; Jin, M.; Xu, H.; Ma, S.; Wang, X.; Zeng, W.; et al. Exosomal Micro RNAs Derived from Dermal Papilla Cells Mediate Hair Follicle Stem Cell Proliferation and Differentiation. Int. J. Biol. Sci. 2019, 15, 1368–1382.
- Rajendran, R.L.; Gangadaran, P.; Bak, S.S.; Oh, J.M.; Kalimuthu, S.; Lee, H.W.; Baek, S.H.; Zhu, L.; Sung, Y.K.; Jeong, S.Y.; et al. Extracellular vesicles derived from MSCs activates dermal papilla cell in vitro and promotes hair follicle conversion from telogen to anagen in mice. Sci. Rep. 2017, 7, 15560.
- Choi, B.Y. Hair-Growth Potential of Ginseng and Its Major Metabolites: A Review on Its Molecular Mechanisms. Int. J. Mol. Sci. 2018, 19, 2703.
- Kwon, T.-R.; Oh, C.T.; Choi, E.J.; Park, H.M.; Han, H.J.; Ji, H.J.; Kim, B.J. Human placental extract exerts hair growth-promoting effects through the GSK-3β signaling pathway in human dermal papilla cells. Int. J. Mol. Med. 2015, 36, 1088–1096.
- Vestita, M.; Filoni, A.; Bonamonte, D.; Elia, R.; Giudice, G. Abstract: The Use of Nanofat in Androgenic Alopecia. a Prospective Blinded Study. Plast. Reconstr. Surg.—Glob. Open 2017, 5, 90.
- Epstein, G.K.; Epstein, J.S. Mesenchymal Stem Cells and Stromal Vascular Fraction for Hair Loss: Current Status. Facial Plast. Surg. Clin. N. Am. 2018, 26, 503–511.
- Gentile, P. Autologous Cellular Method Using Micrografts of Human Adipose Tissue Derived Follicle Stem Cells in Androgenic Alopecia. Int. J. Mol. Sci. 2019, 20, 3446.
- Wang, B.; Liu, X.-M.; Liu, Z.-N.; Wang, Y.; Han, X.; Lian, A.-B.; Mu, Y.; Jin, M.-H.; Liu, J.-Y. Human hair follicle-derived mesenchymal stem cells: Isolation, expansion, and differentiation. World J. Stem Cells 2020, 12, 462–470.
- Cheng, C.-C.; Tsutsui, K.; Taguchi, T.; Sanzen, N.; Nakagawa, A.; Kakiguchi, K.; Yonemura, S.; Tanegashima, C.; Keeley, S.D.; Kiyonari, H.; et al. Hair follicle epidermal stem cells define a niche for tactile sensation. eLife 2018, 7, e38883.
- Chacón-Martínez, C.A.; Klose, M.; Niemann, C.; Glauche, I.; Wickström, S. Hair follicle stem cell cultures reveal self-organizing plasticity of stem cells and their progeny. EMBO J. 2016, 36, 151–164.
- Elmaadawi, I.H.; Mohamed, B.M.; Ibrahim, Z.A.S.; Abdou, S.M.; El Attar, Y.A.; Youssef, A.; Shamloula, M.M.; Taha, A.; Metwally, H.G.; El Afandy, M.M.; et al. Stem cell therapy as a novel therapeutic intervention for resistant cases of alopecia areata and androgenetic alopecia. J. Dermatol. Treat. 2018, 29, 431–440.
- Bak, D.H.; Choi, M.J.; Kim, S.R.; Lee, B.C.; Kim, J.M.; Jeon, E.S.; Oh, W.; Lim, E.S.; Park, B.C.; Kim, M.J.; et al. Human umbilical cord blood mesenchymal stem cells engineered to overexpress growth factors accelerate outcomes in hair growth. Korean J. Physiol. Pharmacol. 2018, 22, 555–566.
- Czarnecka, A.; Odziomek, A.; Murzyn, M.; Dubis, J.; Bagłaj-Oleszczuk, M.; Anita, H.-G. Wharton’s jelly-derived mesenchymal stem cells in the treatment of four patients with alopecia areata. Adv. Clin. Exp. Med. 2021, 30, 211–218.
- Cananzi, M.; Atala, A.; De Coppi, P. Stem cells derived from amniotic fluid: New potentials in regenerative medicine. Reprod. Biomed. Online 2009, 18 (Suppl. 1), 17–27.
- Abd-Allah, S.H.; El-Shal, A.S.; Shalaby, S.M.; Abd-Elbary, E.; Mazen, N.F.; Kader, R.R.A. The role of placenta-derived mesenchymal stem cells in healing of induced full-thickness skin wound in a mouse model. IUBMB Life 2015, 67, 701–709.
- Williams, A.R.; Hare, J.M. Mesenchymal Stem Cells: Biology, Pathophysiology, Translational Findings, and Therapeutic Implications for Cardiac Disease. Circ. Res. 2011, 109, 923–940.
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Krause, D.S.; Deans, R.J.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317.
- Qin, X.; Jiang, Q.; Nagano, K.; Moriishi, T.; Miyazaki, T.; Komori, H.; Ito, K.; Von Der Mark, K.; Sakane, C.; Kaneko, H.; et al. Runx2 is essential for the transdifferentiation of chondrocytes into osteoblasts. PLoS Genet. 2020, 16, e1009169.
- Caplan, H.; Olson, S.D.; Kumar, A.; George, M.; Prabhakara, K.S.; Wenzel, P.; Bedi, S.; Toledano-Furman, N.E.; Triolo, F.; Kamhieh-Milz, J.; et al. Mesenchymal Stromal Cell Therapeutic Delivery: Translational Challenges to Clinical Application. Front. Immunol. 2019, 10, 1645.
- Asakawa, K.; Toyoshima, K.-E.; Ishibashi, N.; Tobe, H.; Iwadate, A.; Kanayama, T.; Hasegawa, T.; Nakao, K.; Toki, H.; Noguchi, S.; et al. Hair organ regeneration via the bioengineered hair follicular unit transplantation. Sci. Rep. 2012, 2, 424.
- Balañá, M.E. Epidermal stem cells and skin tissue engineering in hair follicle regeneration. World J. Stem Cells 2015, 7, 711–727.
- Gentile, P.; Scioli, M.G.; Bielli, A.; Orlandi, A.; Cervelli, V. Stem cells from human hair follicles: First mechanical isolation for immediate autologous clinical use in androgenetic alopecia and hair loss. Stem Cell Investig. 2017, 4, 58.
- Marquez-Curtis, L.A.; Janowska-Wieczorek, A.; McGann, L.E.; Elliott, J.A. Mesenchymal stromal cells derived from various tissues: Biological, clinical and cryopreservation aspects. Cryobiology 2015, 71, 181–197.
- Sheykhhasan, M.; Qomi, R.T.; Ghiasi, M. Fibrin Scaffolds Designing in order to Human Adipose-derived Mesenchymal Stem Cells Differentiation to Chondrocytes in the Presence of TGF-β. Int. J. Stem Cells 2015, 8, 219–227.
- Suh, A.; Pham, A.; Cress, M.J.; Pincelli, T.; TerKonda, S.P.; Bruce, A.J.; Zubair, A.C.; Wolfram, J.; Shapiro, S.A. Adipose-derived cellular and cell-derived regenerative therapies in dermatology and aesthetic rejuvenation. Ageing Res. Rev. 2019, 54, 100933.
- Bourin, P.; Bunnell, B.A.; Casteilla, L.; Dominici, M.; Katz, A.J.; March, K.L.; Redl, H.; Rubin, J.P.; Yoshimura, K.; Gimble, J.M. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: A joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT). Cytotherapy 2013, 15, 641–648.
- Suzuki, E.; Fujita, D.; Takahashi, M.; Oba, S.; Nishimatsu, H. Adipose tissue-derived stem cells as a therapeutic tool for cardiovascular disease. World J. Cardiol. 2015, 7, 454–465.
- Traktuev, D.O.; Prater, D.N.; Merfeld-Clauss, S.; Sanjeevaiah, A.R.; Saadatzadeh, M.R.; Murphy, M.; Johnstone, B.H.; Ingram, D.A.; March, K.L. Robust functional vascular network formation in vivo by cooperation of adipose progenitor and endothelial cells. Circ. Res. 2009, 104, 1410–1420.
- Traktuev, D.O.; Merfeld-Clauss, S.; Li, J.; Kolonin, M.; Arap, W.; Pasqualini, R.; Johnstone, B.H.; March, K.L. A Population of Multipotent CD34-Positive Adipose Stromal Cells Share Pericyte and Mesenchymal Surface Markers, Reside in a Periendothelial Location, and Stabilize Endothelial Networks. Circ. Res. 2008, 102, 77–85.
- Rehman, J.; Traktuev, D.; Li, J.; Merfeld-Clauss, S.; Temm-Grove, C.J.; Bovenkerk, J.E.; Pell, C.L.; Johnstone, B.H.; Considine, R.V.; March, K.L. Secretion of Angiogenic and Antiapoptotic Factors by Human Adipose Stromal Cells. Circulation 2004, 109, 1292–1298.
- Kinnaird, T.; Stabile, E.; Burnett, M.; Lee, C.; Barr, S.; Fuchs, S.; Epstein, S. Marrow-Derived Stromal Cells Express Genes Encoding a Broad Spectrum of Arteriogenic Cytokines and Promote In Vitro and In Vivo Arteriogenesis Through Paracrine Mechanisms. Circ. Res. 2004, 94, 678–685.
- Whiting, D.A. Scalp Biopsy as a Diagnostic and Prognostic Tool in Androgenetic Alopecia. Dermatol. Ther. 1998, 8, 24–33.
- Festa, E.; Fretz, J.; Berry, R.; Schmidt, B.; Rodeheffer, M.; Horowitz, M.; Horsley, V. Adipocyte Lineage Cells Contribute to the Skin Stem Cell Niche to Drive Hair Cycling. Cell 2011, 146, 761–771.
- Hausman, G.J.; Martin, R.J. The Development of Adipocytes Located around Hair Follicles in the Fetal Pig. J. Anim. Sci. 1982, 54, 1286–1296.
- Szöke, K.; Brinchmann, J.E. Concise Review: Therapeutic Potential of Adipose Tissue-Derived Angiogenic Cells. Stem Cells Transl. Med. 2012, 1, 658–667.
- Crisan, M.; Yap, S.; Casteilla, L.; Chen, C.-W.; Corselli, M.; Park, T.S.; Andriolo, G.; Sun, B.; Zheng, B.; Zhang, L.; et al. A Perivascular Origin for Mesenchymal Stem Cells in Multiple Human Organs. Cell Stem Cell 2008, 3, 301–313.
- Perez-Meza, D.; Ziering, C.; Sforza, M.; Krishnan, G.; Ball, E.; Daniels, E. Hair follicle growth by stromal vascular fraction-enhanced adipose transplantation in baldness. Stem Cells Cloning Adv. Appl. 2017, 10, 1–10.
- Anderi, R.; Makdissy, N.; Azar, A.; Rizk, F.; Hamade, A. Cellular therapy with human autologous adipose-derived adult cells of stromal vascular fraction for alopecia areata. Stem Cell Res. Ther. 2018, 9, 1–9.
- Aronowitz, J.A.; Lockhart, R.A.; Birnbaum, Z.E.; Hakakian, C.S.; Daniels, E.; Washenik, K. Abstract: Stromal Vascular Fraction Enhanced Adipose Transplantation in Hair Loss Early Experience & Active Phase II FDA Investigation. Plast. Reconstr. Surg. Glob. Open 2016, 4, 50–55.
- Shin, H.; Ryu, H.H.; Kwon, O.; Park, B.-S.; Jo, S.J. Clinical use of conditioned media of adipose tissue-derived stem cells in female pattern hair loss: A retrospective case series study. Int. J. Dermatol. 2015, 54, 730–735.
- Shin, H.; Won, C.H.; Chung, W.-K.; Park, B.-S. Up-to-date Clinical Trials of Hair Regeneration Using Conditioned Media of Adipose-Derived Stem Cells in Male and Female Pattern Hair Loss. Curr. Stem Cell Res. Ther. 2017, 12, 524–530.
- Fukuoka, H.; Suga, H. Hair Regeneration Treatment Using Adipose-Derived Stem Cell Conditioned Medium: Follow-up with Trichograms. Eplasty 2015, 15, e10.
- Kim, S.J.; Kim, M.J.; Lee, Y.J.; Lee, J.C.; Kim, J.H.; Kim, D.H.; Do, Y.H.; Choi, J.W.; Chung, S.I.; Do, B.-R. Innovative method of alopecia treatment by autologous adipose-derived SVF. Stem Cell Res. Ther. 2021, 12, 486.
- Stevens, H.P.; Donners, B.S.; De Bruijn, B.J. Introducing Platelet-Rich Stroma: Platelet-Rich Plasma (PRP) and Stromal Vascular Fraction (SVF) Combined for the Treatment of Androgenetic Alopecia. Aesthetic Surg. J. 2018, 38, 811–822.
- Cho, I.J.; Lui, P.; Obajdin, J.; Riccio, F.; Stroukov, W.; Willis, T.; Spagnoli, F.; Watt, F.M. Mechanisms, Hallmarks, and Implications of Stem Cell Quiescence. Stem Cell Rep. 2019, 12, 1190–1200.
- Ji, J.; Qian, G.; Bigliardi, P.L.; Ho, B.S.-Y.; Xie, X.-M.; Bigliardi-Qi, M. Aging in hair follicle stem cells and niche microenvironment. J. Dermatol. 2017, 44, 1097–1104.
- Rompolas, P.; Greco, V. Stem cell dynamics in the hair follicle niche. Semin. Cell Dev. Biol. 2014, 25–26, 34–42.
- Watt, F.M.; Hogan, B.L.M. Out of Eden: Stem Cells and Their Niches. Science 2000, 287, 1427–1430.
- Yu, H.; Fang, D.; Kumar, S.M.; Li, L.; Nguyen, T.K.; Acs, G.; Herlyn, M.; Xu, X. Isolation of a Novel Population of Multipotent Adult Stem Cells from Human Hair Follicles. Am. J. Pathol. 2006, 168, 1879–1888.
- Kang, S.; Chovatiya, G.; Tumbar, T. Epigenetic control in skin development, homeostasis and injury repair. Exp. Dermatol. 2019, 28, 453–463.
- Waghmare, S.K.; Bansal, R.; Lee, J.; Zhang, Y.V.; McDermitt, D.J.; Tumbar, T. Quantitative proliferation dynamics and random chromosome segregation of hair follicle stem cells. EMBO J. 2008, 27, 1309–1320.
- Zhang, Y.V.; Cheong, J.; Ciapurin, N.; McDermitt, D.J.; Tumbar, T. Distinct Self-Renewal and Differentiation Phases in the Niche of Infrequently Dividing Hair Follicle Stem Cells. Cell Stem Cell 2009, 5, 267–278.
- Lee, J.; Tumbar, T. Hairy tale of signaling in hair follicle development and cycling. Semin. Cell Dev. Biol. 2012, 23, 906–916.
- Gentile, P.; Scioli, M.G.; Cervelli, V.; Orlandi, A.; Garcovich, S. Autologous Micrografts from Scalp Tissue: Trichoscopic and Long-Term Clinical Evaluation in Male and Female Androgenetic Alopecia. BioMed Res. Int. 2020, 2020, 7397162.
- Ito, M.; Liu, Y.; Yang, Z.; Nguyen, J.; Liang, F.; Morris, R.J.; Cotsarelis, G. Stem cells in the hair follicle bulge contribute to wound repair but not to homeostasis of the epidermis. Nat. Med. 2005, 11, 1351–1354.
- Plikus, M.V.; Mayer, J.A.; De La Cruz, D.; Baker, R.E.; Maini, P.K.; Maxson, R.; Chuong, C.-M. Cyclic dermal BMP signalling regulates stem cell activation during hair regeneration. Nature 2008, 451, 340–344.
- Garza, L.A.; Yang, C.-C.; Zhao, T.; Blatt, H.B.; Lee, M.; He, H.; Stanton, D.C.; Carrasco, L.; Spiegel, J.H.; Tobias, J.W.; et al. Bald scalp in men with androgenetic alopecia retains hair follicle stem cells but lacks CD200-rich and CD34-positive hair follicle progenitor cells. J. Clin. Investig. 2011, 121, 613–622.
- Gentile, P.; Scioli, M.G.; Bielli, A.; De Angelis, B.; De Sio, C.; De Fazio, D.; Ceccarelli, G.; Trivisonno, A.; Orlandi, A.; Cervelli, V.; et al. Platelet-Rich Plasma and Micrografts Enriched with Autologous Human Follicle Mesenchymal Stem Cells Improve Hair Re-Growth in Androgenetic Alopecia. Biomolecular Pathway Analysis and Clinical Evaluation. Biomedicines 2019, 7, 27.
- Dong, L.; Hao, H.; Xia, L.; Liu, J.; Ti, D.; Tong, C.; Hou, Q.; Han, Q.; Zhao, Y.; Liu, H.; et al. Treatment of MSCs with Wnt1a-conditioned medium activates DP cells and promotes hair follicle regrowth. Sci. Rep. 2015, 4, 5432.
- Im, G.-I. Bone marrow-derived stem/stromal cells and adipose tissue-derived stem/stromal cells: Their comparative efficacies and synergistic effects. J. Biomed. Mater. Res. Part A 2017, 105, 2640–2648.
- Chahla, J.; Mannava, S.; Cinque, M.E.; Geeslin, A.G.; Codina, D.; LaPrade, R.F. Bone Marrow Aspirate Concentrate Harvesting and Processing Technique. Arthrosc. Tech. 2017, 6, e441–e445.
- Imam, M.A.; Mahmoud, S.S.; Holton, J.; Abouelmaati, D.; Elsherbini, Y.; Snow, M. A systematic review of the concept and clinical applications of Bone Marrow Aspirate Concentrate in Orthopaedics. SICOT J. 2017, 3, 17.
- Kim, G.B.; Seo, M.-S.; Park, W.T.; Lee, G.W. Bone Marrow Aspirate Concentrate: Its Uses in Osteoarthritis. Int. J. Mol. Sci. 2020, 21, 3224.
- Yoo, B.-Y.; Shin, Y.-H.; Yoon, H.-H.; Seo, Y.-K.; Song, K.-Y.; Park, J.-K. Application of mesenchymal stem cells derived from bone marrow and umbilical cord in human hair multiplication. J. Dermatol. Sci. 2010, 60, 74–83.





