
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Abbasali Emamjomeh | -- | 3990 | 2023-07-28 18:30:30 | | | |
| 2 | Sirius Huang | Meta information modification | 3990 | 2023-07-31 02:29:41 | | |
Video Upload Options
Herbal chemicals with a long history in medicine have attracted a lot of attention. Flavonolignans and flavonoids are considered as two classes of the above-mentioned compounds with different functional groups which exhibit several therapeutic capabilities such as antimicrobial, anti-inflammatory, antioxidant, antidiabetic, and anticancer activities. Based on the studies, high hydrophobic properties of the aforementioned compounds limit their bioavailability inside the human body and restrict their wide application. Nanoscale formulations such as solid lipid nanoparticles, liposomes, and other types of lipid-based delivery systems have been introduced to overcome the above-mentioned challenges. This approach allows the aforementioned hydrophobic therapeutic compounds to be encapsulated between hydrophobic structures, resulting in improving their bioavailability. The above-mentioned enhanced delivery system improves delivery to the targeted sites and reduces the daily required dosage.
1. Introduction
2. Liposomes
2.1. Flavonoid Liposomes
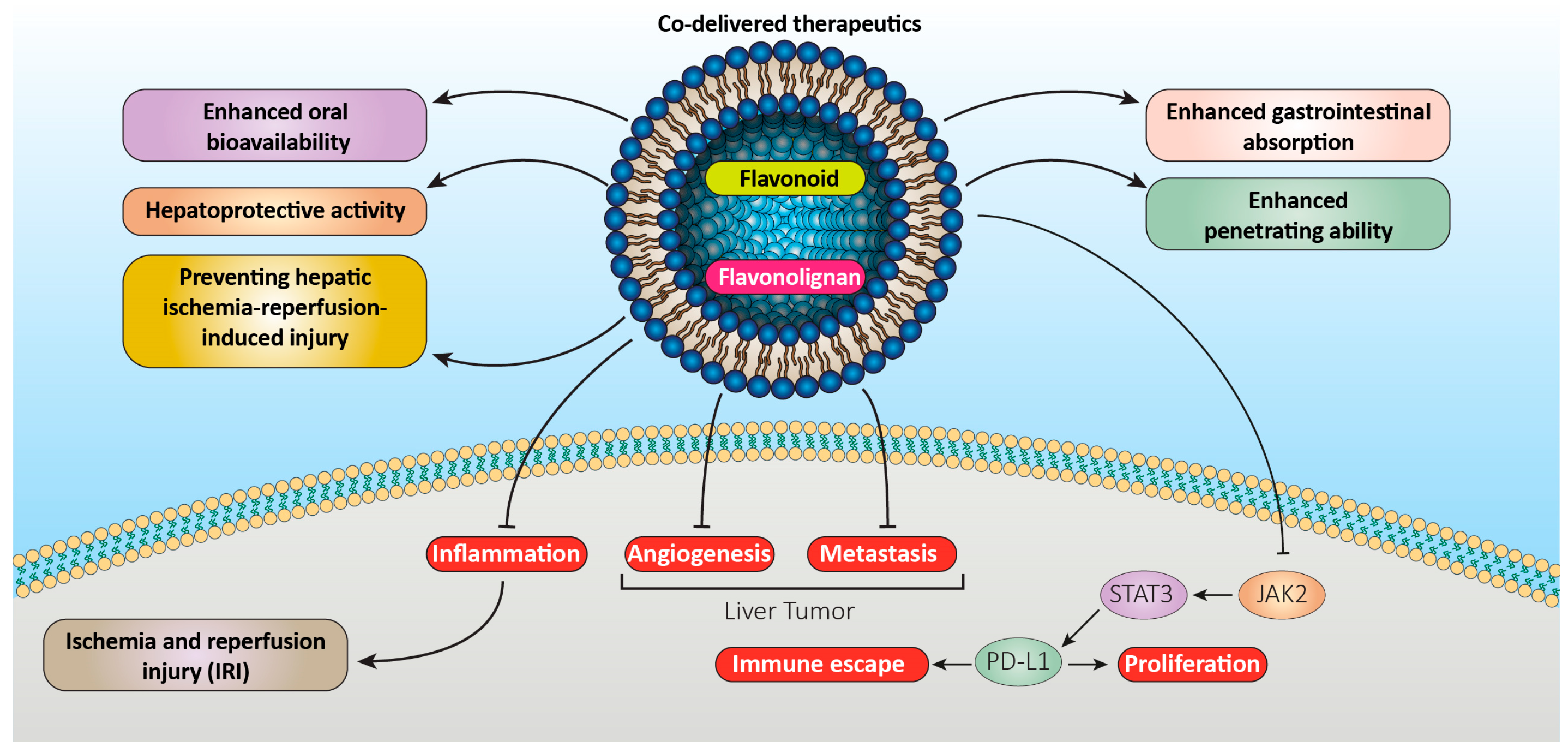
2.2. Flavonolignan Liposomes
3. Micro- and Nanoemulsions
3.1. Flavonoid Nanoemulsions
3.2. Flavonolignan Nanoemulsions
4. Solid Lipid Nanoparticles
4.1. Flavonoid SLNs
4.2. Flavonolignan SLNs
References
- Ashrafizadeh, M.; Ahmadi, Z.; Mohammadinejad, R.; Farkhondeh, T.; Samarghandian, S. Nano-soldiers ameliorate silibinin delivery: A Review Study. Curr. Drug Deliv. 2020, 17, 15–22.
- Iqbal, B.; Ali, J.; Baboota, S. Silymarin loaded nanostructured lipid carrier: From design and dermatokinetic study to mechanistic analysis of epidermal drug deposition enhancement. J. Mol. Liq. 2018, 255, 513–529.
- Jia, L.; Zhang, D.; Li, Z.; Duan, C.; Wang, Y.; Feng, F.; Wang, F.; Liu, Y.; Zhang, Q. Nanostructured lipid carriers for parenteral delivery of silybin: Biodistribution and pharmacokinetic studies. Colloids Surf. B Biointerfaces 2010, 80, 213–218.
- Piazzini, V.; Lemmi, B.; D’Ambrosio, M.; Cinci, L.; Luceri, C.; Bilia, A.R.; Bergonzi, M.C. Nanostructured lipid carriers as promising delivery systems for plant extracts: The case of silymarin. Appl. Sci. 2018, 8, 1163.
- Ma, Y.; He, H.; Fan, W.; Li, Y.; Zhang, W.; Zhao, W.; Qi, J.; Lu, Y.; Dong, X.; Wu, W. In vivo fate of biomimetic mixed micelles as nanocarriers for bioavailability enhancement of lipid–drug conjugates. ACS Biomater. Sci. Eng. 2017, 3, 2399–2409.
- Kumar, R. Lipid-based nanoparticles for drug-delivery systems. In Nanocarriers for Drug Delivery; Elsevier: Amsterdam, The Netherlands, 2019; pp. 249–284.
- Rawat, M.; Singh, D.; Saraf, S.; Saraf, S. Nanocarriers: Promising vehicle for bioactive drugs. Biol. Pharm. Bull. 2006, 29, 1790–1798.
- Patel, D.; Patel, B.; Thakkar, H. Lipid based nanocarriers: Promising drug delivery system for topical application. Eur. J. Lipid Sci. Technol. 2021, 123, 2000264.
- Müller, R.H.; Radtke, M.; Wissing, S.A. Solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) in cosmetic and dermatological preparations. Adv. Drug Deliv. Rev. 2002, 54, S131–S155.
- Salawi, A. Self-emulsifying drug delivery systems: A novel approach to deliver drugs. Drug Deliv. 2022, 29, 1811–1823.
- Mishra, V.; Nayak, P.; Yadav, N.; Singh, M.; Tambuwala, M.M.; Aljabali, A.A. Orally administered self-emulsifying drug delivery system in disease management: Advancement and patents. Expert Opin. Drug Deliv. 2021, 18, 315–332.
- Khairnar, S.V.; Pagare, P.; Thakre, A.; Nambiar, A.R.; Junnuthula, V.; Abraham, M.C.; Kolimi, P.; Nyavanandi, D.; Dyawanapelly, S. Review on the scale-up methods for the preparation of solid lipid nanoparticles. Pharmaceutics 2022, 14, 1886.
- Mirchandani, Y.; Patravale, V.B.; Brijesh, S. Solid lipid nanoparticles for hydrophilic drugs. J. Control. Release 2021, 335, 457–464.
- Izza, N.m.; Watanabe, N.; Okamoto, Y.; Suga, K.; Wibisono, Y.; Kajimura, N.; Mitsuoka, K.; Umakoshi, H. Dependence of the Core–Shell Structure on the Lipid Composition of Nanostructured Lipid Carriers: Implications for Drug Carrier Design. ACS Appl. Nano Mater. 2022, 5, 9958–9969.
- Wang, J.; Gong, J.; Wei, Z. Strategies for liposome drug delivery systems to improve tumor treatment efficacy. AAPS PharmSciTech 2022, 23, 27.
- Sharma, V.K.; Agrawal, M.K. A historical perspective of liposomes-a bio nanomaterial. Mater. Today Proc. 2021, 45, 2963–2966.
- Hwang, J.Y.; Li, Z.; Loh, X.J. Small molecule therapeutic-loaded liposomes as therapeutic carriers: From development to clinical applications. RSC Adv. 2016, 6, 70592–70615.
- Danaei, M.; Dehghankhold, M.; Ataei, S.; Hasanzadeh Davarani, F.; Javanmard, R.; Dokhani, A.; Khorasani, S.; Mozafari, M. Impact of particle size and polydispersity index on the clinical applications of lipidic nanocarrier systems. Pharmaceutics 2018, 10, 57.
- Wang, T.; Suita, Y.; Miriyala, S.; Dean, J.; Tapinos, N.; Shen, J. Advances in lipid-based nanoparticles for cancer chemoimmunotherapy. Pharmaceutics 2021, 13, 520.
- Elmowafy, M.; Viitala, T.; Ibrahim, H.M.; Abu-Elyazid, S.K.; Samy, A.; Kassem, A.; Yliperttula, M. Silymarin loaded liposomes for hepatic targeting: In vitro evaluation and HepG2 drug uptake. Eur. J. Pharm. Sci. 2013, 50, 161–171.
- El-Samaligy, M.S.; Afifi, N.N.; Mahmoud, E.A. Evaluation of hybrid liposomes-encapsulated silymarin regarding physical stability and in vivo performance. Int. J. Pharm. 2006, 319, 121–129.
- El-Samaligy, M.; Afifi, N.; Mahmoud, E. Increasing bioavailability of silymarin using a buccal liposomal delivery system: Preparation and experimental design investigation. Int. J. Pharm. 2006, 308, 140–148.
- Chu, C.; Tong, S.-S.; Xu, Y.; Wang, L.; Fu, M.; Ge, Y.-R.; Yu, J.-N.; Xu, X.-M. Proliposomes for oral delivery of dehydrosilymarin: Preparation and evaluation in vitro and in vivo. Acta Pharmacol. Sin. 2011, 32, 973–980.
- Furneri, P.M.; Petronio, G.P.; Fuochi, V.; Cupri, S.; Pignatello, R. Nanosized devices as antibiotics and antifungals delivery: Past, news, and outlook. In Nanostructures for Drug Delivery; Elsevier: Amsterdam, The Netherlands, 2017; pp. 697–748.
- Khorasani, S.; Danaei, M.; Mozafari, M. Nanoliposome technology for the food and nutraceutical industries. Trends Food Sci. Technol. 2018, 79, 106–115.
- Mozafari, R.M. Nanoliposomes: From Fundamentals to Recent Developments; Trafford Publishing Co. Ltd.: Oxford, UK, 2005.
- Szoka, F., Jr.; Papahadjopoulos, D. Procedure for preparation of liposomes with large internal aqueous space and high capture by reverse-phase evaporation. Proc. Natl. Acad. Sci. USA 1978, 75, 4194–4198.
- Liao, A.-M.; Cai, B.; Huang, J.-H.; Hui, M.; Lee, K.-K.; Lee, K.Y.; Chun, C. Synthesis, anticancer activity and potential application of diosgenin modified cancer chemotherapeutic agent cytarabine. Food Chem. Toxicol. 2021, 148, 111920.
- Yu, J.Y.; Chuesiang, P.; Shin, G.H.; Park, H.J. Post-processing techniques for the improvement of liposome stability. Pharmaceutics 2021, 13, 1023.
- Alavi, M.; Rai, M.; Varma, R.S.; Hamidi, M.; Mozafari, M.R. Conventional and novel methods for the preparation of micro and nanoliposomes. Micro Nano Bio Asp. 2022, 1, 18–29.
- Has, C.; Sunthar, P. A comprehensive review on recent preparation techniques of liposomes. J. Liposome Res. 2020, 30, 336–365.
- Di Costanzo, A.; Angelico, R. Formulation strategies for enhancing the bioavailability of silymarin: The state of the art. Molecules 2019, 24, 2155.
- Jing, D.; Wu, W.; Chen, X.; Xiao, H.; Zhang, Z.; Chen, F.; Zhang, Z.; Liu, J.; Shao, Z.; Pu, F. Quercetin encapsulated in Folic Acid-Modified Liposomes is therapeutic against osteosarcoma by non-Covalent binding to the JH2 Domain of JAK2 via the JAK2-STAT3-PDL1. Pharmacol. Res. 2022, 182, 106287.
- Tang, L.; Li, K.; Zhang, Y.; Li, H.; Li, A.; Xu, Y.; Wei, B. Quercetin liposomes ameliorate streptozotocin-induced diabetic nephropathy in diabetic rats. Sci. Rep. 2020, 10, 2440.
- Ferreira-Silva, M.; Faria-Silva, C.; Carvalheiro, M.C.; Simões, S.; Marinho, H.S.; Marcelino, P.; Campos, M.C.; Metselaar, J.M.; Fernandes, E.; Baptista, P.V. Quercetin Liposomal Nanoformulation for Ischemia and Reperfusion Injury Treatment. Pharmaceutics 2022, 14, 104.
- Li, J.; Li, Z.; Gao, Y.; Liu, S.; Li, K.; Wang, S.; Gao, L.; Shi, M.; Liu, Z.; Han, Z. Effect of a drug delivery system made of quercetin formulated into PEGylation liposomes on cervical carcinoma in vitro and in vivo. J. Nanomater. 2021, 2021, 9389934.
- Zhang, Y.; Guan, R.; Huang, H. Anti-Allergic Effects of Quercetin and Quercetin Liposomes in RBL-2H3 Cells. Endocr. Metab. Immune Disord. Drug Targets 2022, 23, 692–701.
- Li, H.; Chen, P.; Wang, M.; Wang, W.; Li, F.; Han, X.; Ren, J.; Duan, X. Liposome quercetin enhances the ablation effects of microwave ablation in treating the rabbit VX2 liver tumor model. Int. J. Hyperth. 2022, 39, 162–172.
- Renault-Mahieux, M.; Vieillard, V.; Seguin, J.; Espeau, P.; Le, D.T.; Lai-Kuen, R.; Mignet, N.; Paul, M.; Andrieux, K. Co-Encapsulation of Fisetin and Cisplatin into Liposomes for Glioma Therapy: From Formulation to Cell Evaluation. Pharmaceutics 2021, 13, 970.
- Altamimi, M.A.; Hussain, A.; AlRajhi, M.; Alshehri, S.; Imam, S.S.; Qamar, W. Luteolin-loaded elastic liposomes for transdermal delivery to control breast cancer: In vitro and ex vivo evaluations. Pharmaceuticals 2021, 14, 1143.
- Deshmukh, P.K.; Mutha, R.E.; Surana, S.J. Electrostatic deposition assisted preparation, characterization and evaluation of chrysin liposomes for breast cancer treatment. Drug Dev. Ind. Pharm. 2021, 47, 809–819.
- Huang, R.; Zhao, Z.; Jiang, X.; Li, W.; Zhang, L.; Wang, B.; Tie, H. Liposomal chrysin attenuates hepatic ischaemia-reperfusion injury: Possible mechanism via inhibiting NLRP3 inflammasome. J. Pharm. Pharmacol. 2022, 74, 216–226.
- Tian, J.-Y.; Chi, C.-L.; Bian, G.; Xing, D.; Guo, F.-J.; Wang, X.-Q. PSMA conjugated combinatorial liposomal formulation encapsulating genistein and plumbagin to induce apoptosis in prostate cancer cells. Colloids Surf. B Biointerfaces 2021, 203, 111723.
- Maheshwari, H.; Agarwal, R.; Patil, C.; Katare, O.P. Preparation and pharmacological evaluation of silibinin liposomes. Arzneimittelforschung 2003, 53, 420–427.
- Ochi, M.M.; Amoabediny, G.; Rezayat, S.M.; Akbarzadeh, A.; Ebrahimi, B. In vitro co-delivery evaluation of novel pegylated nano-liposomal herbal drugs of silibinin and glycyrrhizic acid (nano-phytosome) to hepatocellular carcinoma cells. Cell J. (Yakhteh) 2016, 18, 135.
- Ripoli, M.; Angelico, R.; Sacco, P.; Ceglie, A.; Mangia, A. Phytoliposome-based silibinin delivery system as a promising strategy to prevent hepatitis C virus infection. J. Biomed. Nanotechnol. 2016, 12, 770–780.
- Yang, G.; Zhao, Y.; Zhang, Y.; Dang, B.; Liu, Y.; Feng, N. Enhanced oral bioavailability of silymarin using liposomes containing a bile salt: Preparation by supercritical fluid technology and evaluation in vitro and in vivo. Int. J. Nanomed. 2015, 10, 6633.
- Mohsen, A.M.; Asfour, M.H.; Salama, A.A. Improved hepatoprotective activity of silymarin via encapsulation in the novel vesicular nanosystem bilosomes. Drug Dev. Ind. Pharm. 2017, 43, 2043–2054.
- Mady, F.M.; Essa, H.; El-Ammawi, T.; Abdelkader, H.; Hussein, A.K. Formulation and clinical evaluation of silymarin pluronic-lecithin organogels for treatment of atopic dermatitis. Drug Des. Dev. Ther. 2016, 10, 1101.
- Singh, I.R.; Pulikkal, A.K. Preparation, stability and biological activity of essential oil-based nano emulsions: A comprehensive review. OpenNano 2022, 8, 100066.
- Shaker, D.S.; Ishak, R.A.; Ghoneim, A.; Elhuoni, M.A. Nanoemulsion: A review on mechanisms for the transdermal delivery of hydrophobic and hydrophilic drugs. Sci. Pharm. 2019, 87, 17.
- Souto, E.B.; Cano, A.; Martins-Gomes, C.; Coutinho, T.E.; Zielińska, A.; Silva, A.M. Microemulsions and nanoemulsions in skin drug delivery. Bioengineering 2022, 9, 158.
- McClements, D.J. Nanoemulsions versus microemulsions: Terminology, differences, and similarities. Soft Matter 2012, 8, 1719–1729.
- Pavoni, L.; Perinelli, D.R.; Bonacucina, G.; Cespi, M.; Palmieri, G.F. An overview of micro-and nanoemulsions as vehicles for essential oils: Formulation, preparation and stability. Nanomaterials 2020, 10, 135.
- Md, S.; Gan, S.Y.; Haw, Y.H.; Ho, C.L.; Wong, S.; Choudhury, H. In vitro neuroprotective effects of naringenin nanoemulsion against β-amyloid toxicity through the regulation of amyloidogenesis and tau phosphorylation. Int. J. Biol. Macromol. 2018, 118, 1211–1219.
- Kaplan, A.B.U.; Cetin, M.; Orgul, D.; Taghizadehghalehjoughi, A.; Hacımuftuoglu, A.; Hekimoglu, S. Formulation and in vitro evaluation of topical nanoemulsion and nanoemulsion-based gels containing daidzein. J. Drug Deliv. Sci. Technol. 2019, 52, 189–203.
- Hussein, J.; El-Naggar, M.E. Synthesis of an environmentally quercetin nanoemulsion to ameliorate diabetic-induced cardiotoxicity. Biocatal. Agric. Biotechnol. 2021, 33, 101983.
- Mahadev, M.; Nandini, H.S.; Ramu, R.; Gowda, D.V.; Almarhoon, Z.M.; Al-Ghorbani, M.; Mabkhot, Y.N. Fabrication and evaluation of quercetin nanoemulsion: A delivery system with improved bioavailability and therapeutic efficacy in diabetes mellitus. Pharmaceuticals 2022, 15, 70.
- Ceramella, J.; Groo, A.-C.; Iacopetta, D.; Séguy, L.; Mariconda, A.; Puoci, F.; Saturnino, C.; Leroy, F.; Since, M.; Longo, P. A winning strategy to improve the anticancer properties of Cisplatin and Quercetin based on the nanoemulsions formulation. J. Drug Deliv. Sci. Technol. 2021, 66, 102907.
- Son, H.-Y.; Lee, M.-S.; Chang, E.; Kim, S.-Y.; Kang, B.; Ko, H.; Kim, I.-H.; Zhong, Q.; Jo, Y.-H.; Kim, C.-T. Formulation and characterization of quercetin-loaded oil in water nanoemulsion and evaluation of hypocholesterolemic activity in rats. Nutrients 2019, 11, 244.
- Ahmadi Oskooei, F.; Mehrzad, J.; Asoodeh, A.; Motavalizadehkakhky, A. Olive oil-based quercetin nanoemulsion (QuNE)’s interactions with human serum proteins (HSA and HTF) and its anticancer activity. J. Biomol. Struct. Dyn. 2021, 41, 778–791.
- Marques, M.B.; Machado, A.P.; Santos, P.A.; Carrett-Dias, M.; Araújo, G.S.; da Silva Alves, B.; de Oliveira, B.S.; da Silva Júnior, F.M.; Dora, C.L.; Cañedo, A.D. Anti-MDR effects of quercetin and its Nanoemulsion in multidrug-resistant human Leukemia cells. Anti-Cancer Agents Med. Chem. (Former. Curr. Med. Chem.-Anti-Cancer Agents) 2021, 21, 1911–1920.
- Magura, J.; Hassan, D.; Moodley, R.; Mackraj, I. Hesperidin-loaded nanoemulsions improve cytotoxicity, induce apoptosis, and downregulate miR-21 and miR-155 expression in MCF-7. J. Microencapsul. 2021, 38, 486–495.
- Colombo, M.; Figueiró, F.; de Fraga Dias, A.; Teixeira, H.F.; Battastini, A.M.O.; Koester, L.S. Kaempferol-loaded mucoadhesive nanoemulsion for intranasal administration reduces glioma growth in vitro. Int. J. Pharm. 2018, 543, 214–223.
- Singh, B.; Bandopadhyay, S.; Kapil, R.; Singh, R.; Katare, O.P. Self-emulsifying drug delivery systems (SEDDS): Formulation development, characterization, and applications. Crit. Rev. Ther. Drug Carr. Syst. 2009, 26, 427–451.
- Zanchetta, B.; Chaud, M.; Santana, M. Self-emulsifying drug delivery systems (SEDDS) in pharmaceutical development. J. Adv. Chem. Eng. 2015, 5, 1000130.
- Li, X.; Yuan, Q.; Huang, Y.; Zhou, Y.; Liu, Y. Development of silymarin self-microemulsifying drug delivery system with enhanced oral bioavailability. Aaps Pharmscitech 2010, 11, 672–678.
- Panapisal, V.; Charoensri, S.; Tantituvanont, A. Formulation of microemulsion systems for dermal delivery of silymarin. Aaps Pharmscitech 2012, 13, 389–399.
- Wu, W.; Wang, Y.; Que, L. Enhanced bioavailability of silymarin by self-microemulsifying drug delivery system. Eur. J. Pharm. Biopharm. 2006, 63, 288–294.
- Deng, M.; Chen, H.; Xie, L.; Liu, K.; Zhang, X.; Li, X. Tea saponins as natural emulsifiers and cryoprotectants to prepare silymarin nanoemulsion. LWT 2022, 156, 113042.
- Wei, Y.; Ye, X.; Shang, X.; Peng, X.; Bao, Q.; Liu, M.; Guo, M.; Li, F. Enhanced oral bioavailability of silybin by a supersaturatable self-emulsifying drug delivery system (S-SEDDS). Colloids Surf. A Physicochem. Eng. Asp. 2012, 396, 22–28.
- Kumar, S.; Randhawa, J.K. High melting lipid based approach for drug delivery: Solid lipid nanoparticles. Mater. Sci. Eng. C 2013, 33, 1842–1852.
- Mehnert, W.; Mäder, K. Solid lipid nanoparticles: Production, characterization and applications. Adv. Drug Deliv. Rev. 2012, 64, 83–101.
- Mishra, V.; Bansal, K.K.; Verma, A.; Yadav, N.; Thakur, S.; Sudhakar, K.; Rosenholm, J.M. Solid lipid nanoparticles: Emerging colloidal nano drug delivery systems. Pharmaceutics 2018, 10, 191.
- Müller, R.H.; MaÈder, K.; Gohla, S. Solid lipid nanoparticles (SLN) for controlled drug delivery–a review of the state of the art. Eur. J. Pharm. Biopharm. 2000, 50, 161–177.
- Rostami, E.; Kashanian, S.; Azandaryani, A.H.; Faramarzi, H.; Dolatabadi, J.E.N.; Omidfar, K. Drug targeting using solid lipid nanoparticles. Chem. Phys. Lipids 2014, 181, 56–61.
- Shangguan, M.; Qi, J.; Lu, Y.; Wu, W. Comparison of the oral bioavailability of silymarin-loaded lipid nanoparticles with their artificial lipolysate counterparts: Implications on the contribution of integral structure. Int. J. Pharm. 2015, 489, 195–202.
- Xu, P.; Yin, Q.; Shen, J.; Chen, L.; Yu, H.; Zhang, Z.; Li, Y. Synergistic inhibition of breast cancer metastasis by silibinin-loaded lipid nanoparticles containing TPGS. Int. J. Pharm. 2013, 454, 21–30.
- He, J.; Hou, S.; Lu, W.; Zhu, L.; Feng, J. Preparation, pharmacokinetics and body distribution of silymarin-loaded solid lipid nanoparticles after oral administration. J. Biomed. Nanotechnol. 2007, 3, 195–202.
- Gill, B.; Singh, J.; Sharma, V.; Kumar, S.H. Emulsomes: An emerging vesicular drug delivery system. Asian J. Pharm. (AJP) 2014, 6, 133–142.
- Khan, S.; Baboota, S.; Ali, J.; Khan, S.; Narang, R.S.; Narang, J.K. Nanostructured lipid carriers: An emerging platform for improving oral bioavailability of lipophilic drugs. Int. J. Pharm. Investig. 2015, 5, 182.
- Rishitha, N.; Muthuraman, A. Therapeutic evaluation of solid lipid nanoparticle of quercetin in pentylenetetrazole induced cognitive impairment of zebrafish. Life Sci. 2018, 199, 80–87.
- Vijayakumar, A.; Baskaran, R.; Jang, Y.S.; Oh, S.H.; Yoo, B.K. Quercetin-loaded solid lipid nanoparticle dispersion with improved physicochemical properties and cellular uptake. Aaps Pharmscitech 2017, 18, 875–883.
- Ahmad, N.; Banala, V.T.; Kushwaha, P.; Karvande, A.; Sharma, S.; Tripathi, A.K.; Verma, A.; Trivedi, R.; Mishra, P.R. Quercetin-loaded solid lipid nanoparticles improve osteoprotective activity in an ovariectomized rat model: A preventive strategy for post-menopausal osteoporosis. Rsc Adv. 2016, 6, 97613–97628.
- Talarico, L.; Consumi, M.; Leone, G.; Tamasi, G.; Magnani, A. Solid lipid nanoparticles produced via a coacervation method as promising carriers for controlled release of quercetin. Molecules 2021, 26, 2694.
- Azizi, M.; Li, Y.; Kaul, N.; Abbaspourrad, A. Study of the physicochemical properties of fish oil solid lipid nanoparticle in the presence of palmitic acid and quercetin. J. Agric. Food Chem. 2019, 67, 671–679.
- Shtay, R.; Keppler, J.K.; Schrader, K.; Schwarz, K. Encapsulation of (-)-epigallocatechin-3-gallate (EGCG) in solid lipid nanoparticles for food applications. J. Food Eng. 2019, 244, 91–100.
- Cengiz, M.; Kutlu, H.M.; Burukoglu, D.D.; Ayhancı, A. A comparative study on the therapeutic effects of silymarin and silymarin-loaded solid lipid nanoparticles on D-GaIN/TNF-α-induced liver damage in Balb/c mice. Food Chem. Toxicol. 2015, 77, 93–100.





