Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Nelson Villalobos | -- | 1856 | 2023-07-28 00:15:35 | | | |
| 2 | Conner Chen | Meta information modification | 1856 | 2023-07-31 03:19:06 | | | | |
| 3 | Conner Chen | -7 word(s) | 1849 | 2023-08-04 07:15:09 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Acosta-Mejia, M.T.; Villalobos, N. Network Involvement in Motor Symptoms. Encyclopedia. Available online: https://encyclopedia.pub/entry/47371 (accessed on 01 April 2026).
Acosta-Mejia MT, Villalobos N. Network Involvement in Motor Symptoms. Encyclopedia. Available at: https://encyclopedia.pub/entry/47371. Accessed April 01, 2026.
Acosta-Mejia, Martha Teresa, Nelson Villalobos. "Network Involvement in Motor Symptoms" Encyclopedia, https://encyclopedia.pub/entry/47371 (accessed April 01, 2026).
Acosta-Mejia, M.T., & Villalobos, N. (2023, July 27). Network Involvement in Motor Symptoms. In Encyclopedia. https://encyclopedia.pub/entry/47371
Acosta-Mejia, Martha Teresa and Nelson Villalobos. "Network Involvement in Motor Symptoms." Encyclopedia. Web. 27 July, 2023.
Copy Citation
Motor symptoms dominate the clinical expression of PD. Muscular rigidity, akinesia, bradykinesia, gait instability, and resting tremor form the core of the motor symptoms. The concept of “parkinsonism” encompasses all motor impairments. For the clinical diagnosis, parkinsonism is defined as bradykinesia accompanied by resting tremor, rigidity, or both. Dopamine (DA) loss secondary to degeneration of the substantia nigra pars compacta (SNc) initiates parkinsonism by causing impaired modulatory function in the motor network.
network
neurophysiology
oscillation
1. Introduction
Neurodegenerative diseases were one of the first ailments to receive medical attention worldwide [1]. Clinically, these diseases show heterogeneous and overlaying symptoms. Hence, a framework that hones diagnosis will allow better specific treatment and management. Parkinson’s disease (PD) is one of the leading neurodegenerative disorders with heightened prevalence [2][3]. It is considered a movement disorder, although it is accepted that the classic motor symptoms are accompanied by a myriad of nonmotor symptoms, as well as hyposmia, urinary dysfunction, orthostatic hypotension, memory loss, depression, pain, gastrointestinal dysfunction, and sleep disturbances [4]. Gastrointestinal symptoms include drooling, dysphagia, disabled gastric emptying, constipation, and impaired defecation [5][6].
Several cellular mechanisms, including mitochondrial dysfunction, oxidative stress, neuroinflammation, and deficient protein degradation are implicated in the pathogenesis of PD. Nonetheless, the pathological fingerprint consists of neural inclusions of Lewy bodies (LBs) and Lewy neurites, with cell loss in the substantia nigra and other brain areas. The burgeoning of LBs begins from an initial template of alpha-synuclein, which incites the seeding of nearby alpha-synuclein proteins, which triggers the formation of aggregates into a toxic, insoluble pleated sheet structure, to form LBs [7]. At the network neurophysiology level, these pathological fingerprints lead to rearrangement in the electrophysiological and neurophysiological activity that generates the symptoms [8][9][10].
Despite the above, the diagnosis is complicated by the overlap of motor and nonmotor symptoms, in addition to the possibility of other neurodegenerative diseases; as a result, disease management remains suboptimal [4]. In recent years, the criteria for diagnosis have been designed and validated and are dependent on the presence of motor symptoms [4][11][12].
2. Network Involvement in Motor Symptoms
Motor symptoms dominate the clinical expression of PD. Muscular rigidity, akinesia, bradykinesia, gait instability, and resting tremor form the core of the motor symptoms [9][11][12]. The concept of “parkinsonism” encompasses all motor impairments. For the clinical diagnosis, parkinsonism is defined as bradykinesia accompanied by resting tremor, rigidity, or both [7][11][12]. Dopamine (DA) loss secondary to degeneration of the substantia nigra pars compacta (SNc) initiates parkinsonism by causing impaired modulatory function in the motor network [7][9][13].
Motor output is modulated by the basal ganglia (BG). The BG comprises several nuclei: the striatum (Str), the external (GPe) and internal (GPi) segments of the globus pallidus (GP), the subthalamic nucleus (STN), the substantia nigra compacta (SNc) and the reticulata (SNr). Functionally, the cortex (Cx) sends motor information by excitatory axons to the Str, STN, and thalamus (Th). In this way, the information reaches the circuit through the Str and emerges through the output nuclei, the GPi/SNr, which then send the information to Th.The Str dually organizes the circuits according to the projection neurons that send their axons to the output nuclei. Thus, the connection between the Str and GPi/SNr forms the “direct” pathway. At the same time, the Str establishes a connection, before reaching the output nuclei, with the GPe and the STN, giving rise to the “indirect” pathway (Str-GPe-STN-GPi/SNr; Figure 1) [14][15][16]. In turn, this network forms larger parallel circuits that include the frontal Cx, ventral Th, and two nuclei of the brainstem: the superior colliculus [17] and the pedunculopontine nucleus (PPN) [18]. Based on the functions of the cortical area of origin, the BG-Th-Cx network is designated as “motor”, “associative/cognitive”, and “limbic” circuits [8][19]. In our context, parkinsonism arises from abnormal activity patterns in the motor circuit (Figure 1).
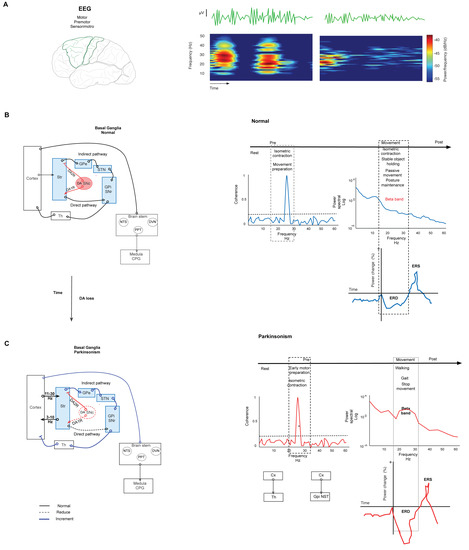
Figure 1. Basal ganglia network in normal and parkinsonism conditions. (A) Spectrogram representation of EEG recording of power in beta-band activity in motor and premotor cortex. The spectrogram shows changes in power at the frequency of the beta band. (B) Left. Simplified schematic of the BG network in normal conditions. The BG modulate the motor output through the projection neurons of the Str that send their axons to the output nuclei: GPi/SNr in two pathways. The direct path connects the Str with GPi/SNr. In the indirect path, the Str connects with the GPe, the STN, and GPi/SNr. Synthesized by SNc, DA is the network modulator through two types of receptors expressed in both pathways. D1 receptors are expressed in the direct path, and D2 receptors are expressed in the indirect path. Under normal conditions, DA release in the Str reduces the combination of these effects, modulating the GPi and SNr activity, and thus it reduces the inhibition of thalamocortical neurons. Right. Graph representation of changes in the parameters of EEG in normal conditions in several stages of movement. The coherence increments are during isometric contraction and training preparation. Beta power declines during movement performance, postural maintenance, and stable object holding. The same movement events show the desynchronization of beta power (ERD). (C) Simplified schematic representation of the BG network in parkinsonism. Left. Degeneration of the nigrostriatal pathway causes DAloss-induced aberrant transmission of the sensorimotor Str and reduces both the direct pathway’s tonic excitation and the indirect pathway’s tonic inhibition; consequently, the BG overinhibit their thalamic and brainstem targets. This causes motor symptoms and aberrant oscillation in beta-band frequency. Right. Changes in beta-band frequency are shown in coherence, power, and ERD during motor performance. The coherence increment is during isometric contraction and movement preparation between CxM-Th and CxM-Gpi/NST. The power density of an ERD shows increment during gait, walking, and movement cessation. Continuous lines represent physiological connections. The dashed line represents a decrease. The bold line represents increased activity. SNc. Substantia nigra compacta. GPi. Globus pallidus internus. GPe. Globus pallidus external. STN. Subthalamic nucleus. Str. Striatum. The graft was elaborated based on the data reported in the main text and supported by the respective references.
Synthesized in the SNc, DA is a critical modulator in the network. Expressed in both direct and indirect pathways, DA receptors are coupled to different second messenger systems (through Gs or Golf for D1-like receptors and Gi or Go for D2-like receptors). In the direct pathway, DA acts on D1 receptors to inhibit the BG output nucleus. DA acts on D2 receptors in the indirect pathway to suppress activity. Under normal conditions, DA release in the Str reduces the combination of these effects under GPi and SNr activity, reducing the inhibition of thalamocortical neurons that receive the input from the output nucleus (Figure 1) [20][21]. Thus, the loss of DA by nigrostriatal pathway degeneration induces aberrant transmission of the sensorimotor striatum [21] (more strongly than transmission to the associative and limbic regions); consequently, this pathway degeneration allows GPe-STN activity to go into overdrive, thus raising the inhibition of STN neurons and their projections to output nuclei [22][23]. This change causes parkinsonism (Figure 1).
Neuronal activity patterns play an essential role in determining the integrative functions of the BG. In neural ensembles, information is transmitted through temporal patterns of action potentials [24]. Therefore, it is accepted that the information is encoded in the firing rate of individual neurons [25]. In this context, changes in the firing rate of individual neurons in some specific nuclei of the BG induced by DA loss explain the pathophysiology of parkinsonism. In the network, the loss of DA reduces the direct pathway’s tonic excitation and the indirect pathway’s tonic inhibition [13][21]. Both changes increase the mean firing rates of output nuclei. Consequently, the BG overinhibits their thalamic and brainstem targets [26]. This causes decreased activity in Th and Cx, resulting in akinesia.
Secondary to the loss of DA, the Str and the Th show changes in their firing rate. In the Str, the neurons projecting to the direct pathway show decreased spontaneous activity, while those of the indirect pathway show increased spontaneous activity [8]. In the Th, neurons show a slowed firing rate [27], and their firing is modulated during reaching movements [8]. The GPe shows a decreased firing rate after DA depletion [28], and at the same time, the local levels of GABA are increased [29][30]. Reports of changes in the motor cortex (CxM) in parkinsonism are scarce. Recently, the decreased firing of neurons projecting to the pyramidal tract was shown, but did not affect those projecting to the Str [31]. These results suggest that transmission from cortical neurons to pyramidal tract neurons might be involved in motor symptoms associated with parkinsonism [8]. It has been proposed that cortical firing activity could be secondary to dysfunction in the BG and Th and possible changes in the Cx secondary to the loss of DA and other neurotransmitters [32]. However, other studies show findings related to the alterations associated with parkinsonism, both in firing frequency and patterns and network synchrony.
Firing Pattern Implication in the Neurophysiology of the BG Network
Physiologically, the action potential is the canonical form of information transmission in the brain. Nevertheless, it is known that specific neurons, based on their intrinsic electrical properties, can show increased mean firing frequency over a short time; this type of activity is described as the burst pattern [13][33].
In the BG normal network, the firing pattern of the GPi, GPe, and STN neurons is random, although action potentials do not occur in bursts. In this condition, the GPi fires action potentials continuously at high frequency. In the same way, the GPe fires action potentials at high frequency, although with pauses, and the STN also fires action potentials continuously but in a medium range of frequencies [13]. These characteristics change considerably secondary to DA depletion. Extensive evidence allows us to accept that DA depletion modifies the intrinsic properties of the neurons of different nuclei of the circuit [34][35][36][37]. Similarly, the GPe shows an increase in the range in which burst firing occurs, adding to the diminished firing rate [38][39]. Similarly, the STN shows a modified firing pattern similar to bursting and increased firing frequency [40]. Notably, burst activity in the STN has been resolutely correlated with clinically severe parkinsonism in patients with PD [41].
The reciprocal connection between the GPe and STN inside the network is physiologically transcendental. Both cores are considered the pacemaker of the network [42], and this notion is given particular importance in the development of bursts [23]. Higher activity from the indirect pathway onto the GPe that guides rhythmicity is demonstrated after DA loss by increased density in the synaptic link between both cores; consequently, the GPe increases inhibition that causes a hyperpolarization-induced higher burst pattern in STN neurons [23][42]. In addition, during parkinsonism, neurons of the GPe and GPi increase synchronization, and the GPi tends to fire in a burst pattern [43].
Thalamic neurons have intrinsic properties that allow them to burst under normal conditions or exhibit tonic bursting depending on the physiological state [44]; the burst pattern trends for the BG nuclei are similar to those in the Th [45], especially in the motor region [40][45]. Notably, burst activity results from a convergence of axons from the cerebellum in the Th motor regions [8]. In this sense, a connection between the cerebellum and BG was described recently and suggested implications for parkinsonism symptoms [8][46]. Therefore, the cerebellum-Th-Cx network contributes to parkinsonism [47].
References
- GBD 2016 Neurology Collaborators. Global, regional, and national burden of neurological disorders, 1990–2016: A systematic analysis for the Global Burden of Disease Study. Lancet Neurol. 2019, 18, 459–480.
- Balestrino, R.; Schapira, A.H.V. Parkinson disease. Eur. J. Neurol. 2020, 27, 27–42.
- Armstrong, M.J.; Okun, M.S. Diagnosis and Treatment of Parkinson Disease: A Review. JAMA 2020, 323, 548–560.
- Tolosa, E.; Garrido, A.; Scholz, S.W.; Poewe, W. Challenges in the diagnosis of Parkinson’s disease. Lancet Neurol. 2021, 20, 385–397.
- Schapira, A.H.V.; Chaudhuri, K.R.; Jenner, P. Non-motor features of Parkinson disease. Nat. Rev. Neurosci. 2017, 18, 435–450.
- Camacho, M.; Greenland, J.C.; Williams-Gray, C.H. The Gastrointestinal Dysfunction Scale for Parkinson’s Disease. Mov. Disord. 2021, 36, 2358–2366.
- Kalia, L.V.; Lang, A.E. Parkinson’s disease. Lancet 2015, 386, 896–912.
- Galvan, A.; Devergnas, A.; Wichmann, T. Alterations in neuronal activity in basal ganglia-thalamocortical circuits in the parkinsonian state. Front. Neuroanat. 2015, 9, 5.
- McGregor, M.M.; Nelson, A.B. Circuit Mechanisms of Parkinson’s Disease. Neuron 2019, 101, 1042–1056.
- Bove, C.; Travagli, R.A. Neurophysiology of the brain stem in Parkinson’s disease. J. Neurophysiol. 2019, 121, 1856–1864.
- Postuma, R.B.; Berg, D.; Stern, M.; Poewe, W.; Olanow, C.W.; Oertel, W.; Obeso, J.; Marek, K.; Litvan, I.; Lang, A.E.; et al. MDS clinical diagnostic criteria for Parkinson’s disease. Movement disorders. Mov. Disord. 2015, 30, 1591–1601.
- Homayoun, H. Parkinson Disease. Ann. Intern. Med. 2018, 169, ITC33–ITC48.
- Nambu, A.; Tachibana, Y.; Chiken, S. Cause of parkinsonian symptoms: Firing rate, firing pattern or dynamic activity changes? Basal Ganglia 2015, 5, 1–6.
- Albin, R.L.; Young, A.B.; Penney, J.B. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989, 12, 366–375.
- DeLong, M.R. Primate models of movement disorders of basal ganglia origin. Trends Neurosci. 1990, 13, 281–285.
- Parent, A.; Hazrati, L.N. Functional anatomy of the basal ganglia. I. The cortico-basal ganglia-thalamo-cortical loop. Brain research. Brain Res. Rev. 1995, 20, 91–127.
- Kim, H.F.; Hikosaka, O. Parallel basal ganglia circuits for voluntary and automatic behaviour to reach rewards. Brain A J. Neurol. 2015, 138, 1776–1800.
- Meoni, S.; Cury, R.G.; Moro, E. New players in basal ganglia dysfunction in Parkinson’s disease. Prog. Brain Res. 2020, 252, 307–327.
- Alexander, G.E.; Crutcher, M.D.; DeLong, M.R. Basal ganglia-thalamocortical circuits: Parallel substrates for motor, oculomotor, “prefrontal” and “limbic” functions. Prog. Brain Res. 1990, 85, 119–146.
- Gerfen, C.R.; Engber, T.M.; Mahan, L.C.; Susel, Z.; Chase, T.N.; Monsma, F.J., Jr.; Sibley, D.R. D1 and D2 dopamine receptor-regulated gene expression of striatonigral and striatopallidal neurons. Science 1990, 250, 1429–1432.
- Surmeier, D.J.; Graves, S.M.; Shen, W. Dopaminergic modulation of striatal networks in health and Parkinson’s disease. Curr. Opin. Neurobiol. 2014, 29, 109–117.
- Galvan, A.; Wichmann, T. Pathophysiology of parkinsonism. Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 2008, 119, 1459–1474.
- Bevan, M.D.; Magill, P.J.; Terman, D.; Bolam, J.P.; Wilson, C.J. Move to the rhythm: Oscillations in the subthalamic nucleus-external globus pallidus network. Trends Neurosci. 2002, 25, 525–531.
- Stanley, G.B. Reading and writing the neural code. Nat. Neurosci. 2013, 16, 259–263.
- Gerstein, G.L. Analysis of Firing Pafferns in Single Neurons. Science 1960, 131, 1811–1812.
- Wichmann, T. Changing views of the pathophysiology of Parkinsonism. Mov. Disord. Off. J. Mov. Disord. Soc. 2019, 34, 1130–1143.
- Schneider, J.S.; Rothblat, D.S. Alterations in intralaminar and motor thalamic physiology following nigrostriatal dopamine depletion. Brain Res. 1996, 742, 25–33.
- Nishibayashi, H.; Ogura, M.; Kakishita, K.; Tanaka, S.; Tachibana, Y.; Nambu, A.; Kita, H.; Itakura, T. Cortically evoked responses of human pallidal neurons recorded during stereotactic neurosurgery. Mov. Disord. Off. J. Mov. Disord. Soc. 2011, 26, 469–476.
- Schroeder, J.A.; Schneider, J.S. GABA-opioid interactions in the globus pallidus: -Met-enkephalinamide attenuates potassium-evoked GABA release after nigrostriatal lesion. J. Neurochem. 2002, 82, 666–673.
- Galvan, A.; Hu, X.; Smith, Y.; Wichmann, T. Localization and function of GABA transporters in the globus pallidus of parkinsonian monkeys. Exp. Neurol. 2010, 223, 505–515.
- Pasquereau, B.; Turner, R.S. Primary motor cortex of the parkinsonian monkey: Differential effects on the spontaneous activity of pyramidal tract-type neurons. Cereb. Cortex 2011, 21, 1362–1378.
- Lindenbach, D.; Bishop, C. Critical involvement of the motor cortex in the pathophysiology and treatment of Parkinson’s disease. Neurosci. Biobehav. Rev. 2013, 37, 2737–2750.
- Llinás, R.R. Intrinsic electrical properties of mammalian neurons and CNS function: A historical perspective. Front. Cell. Neurosci. 2014, 8, 320.
- Chan, C.S.; Glajch, K.E.; Gertler, T.S.; Guzman, J.N.; Mercer, J.N.; Lewis, A.S.; Goldberg, A.B.; Tkatch, T.; Shigemoto, R.; Fleming, S.M.; et al. HCN channelopathy in external globus pallidus neurons in models of Parkinson’s disease. Nat. Neurosci. 2011, 14, 85–92.
- Choi, S.J.; Ma, T.C.; Ding, Y.; Cheung, T.; Joshi, N.; Sulzer, D.; Mosharov, E.V.; Kang, U.J. Alterations in the intrinsic properties of striatal cholinergic interneurons after dopamine lesion and chronic L-DOPA. eLife 2020, 9, e56920.
- Day, M.; Wokosin, D.; Plotkin, J.L.; Tian, X.; Surmeier, D.J. Differential excitability and modulation of striatal medium spiny neuron dendrites. J. Neurosci. Off. J. Soc. Neurosci. 2008, 28, 11603–11614.
- Ünal, B.; Shah, F.; Kothari, J.; Tepper, J.M. Anatomical and electrophysiological changes in striatal TH interneurons after loss of the nigrostriatal dopaminergic pathway. Brain Struct. Funct. 2015, 220, 331–349.
- Wichmann, T.; Soares, J. Neuronal firing before and after burst discharges in the monkey basal ganglia is predictably patterned in the normal state and altered in parkinsonism. J. Neurophysiol. 2006, 95, 2120–2133.
- Hutchison, W.D.; Lozano, A.M.; Davis, K.D.; Saint-Cyr, J.A.; Lang, A.E.; Dostrovsky, J.O. Differential neuronal activity in segments of globus pallidus in Parkinson’s disease patients. Neuroreport 1994, 5, 1533–1537.
- Magnin, M.; Morel, A.; Jeanmonod, D. Single-unit analysis of the pallidum, thalamus and subthalamic nucleus in parkinsonian patients. Neuroscience 2000, 96, 549–564.
- Sharott, A.; Gulberti, A.; Zittel, S.; Tudor Jones, A.A.; Fickel, U.; Münchau, A.; Köppen, J.A.; Gerloff, C.; Westphal, M.; Buhmann, C.; et al. Activity parameters of subthalamic nucleus neurons selectively predict motor symptom severity in Parkinson’s disease. J. Neurosci. Off. J. Soc. Neurosci. 2014, 34, 6273–6285.
- Plenz, D.; Kital, S.T. A basal ganglia pacemaker formed by the subthalamic nucleus and external globus pallidus. Nature 1999, 400, 677–682.
- Tachibana, Y.; Iwamuro, H.; Kita, H.; Takada, M.; Nambu, A. Subthalamo-pallidal interactions underlying parkinsonian neuronal oscillations in the primate basal ganglia. Eur. J. Neurosci. 2011, 34, 1470–1484.
- Roy, D.S.; Zhang, Y.; Halassa, M.M.; Feng, G. Thalamic subnetworks as units of function. Nat. Neurosci. 2022, 25, 140–153.
- Bosch-Bouju, C.; Smither, R.A.; Hyland, B.I.; Parr-Brownlie, L.C. Reduced reach-related modulation of motor thalamus neural activity in a rat model of Parkinsn’s disease. J. Neurosci. 2014, 34, 15836–15850.
- Bostan, A.C.; Dum, R.P.; Strick, P.L. Functional Anatomy of Basal Ganglia Circuits with the Cerebral Cortex and the Cerebellum. Prog. Neurol. Surg. 2018, 33, 50–61.
- Milosevic, L.; Kalia, S.K.; Hodaie, M.; Lozano, A.M.; Popovic, M.R.; Hutchison, W.D. Physiological mechanisms of thalamic ventral intermediate nucleus stimulation for tremor suppression. Brain A J. Neurol. 2018, 141, 2142–2155.
More
Information
Subjects:
Neurosciences
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
527
Revisions:
3 times
(View History)
Update Date:
04 Aug 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





