
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Javier C. Angulo | -- | 3464 | 2023-07-20 14:47:13 | | | |
| 2 | Lindsay Dong | Meta information modification | 3464 | 2023-07-24 02:52:01 | | |
Video Upload Options
Radiation therapy (RT) in the management of pelvic cancers remains a clinical challenge to urologists given the sequelae of urethral stricture disease secondary to fibrosis and vascular insults. The management of post-radiation urethral stricture consists of conservative, endoscopic, and primary reconstructive options. Endoscopic approaches remain an option, but with limited long-term success. Despite concerns with graft take, reconstructive options such as urethroplasties in this population with buccal grafts have shown long-term success rates ranging from 70 to 100%. Robotic reconstruction is augmenting previous options with faster recovery times. Radiation-induced stricture disease is challenging with multiple interventions available, but with successful outcomes demonstrated in various cohorts including urethroplasties with buccal grafts and robotic reconstruction.
1. Introduction
2. Prevalence
3. Stricture Etiology/Physiology
4. Radiation-Induced Changes in Urologic Tissue
5. Pathways of Radiation-Induced Endothelial Cell Death
6. Cellular and Extracellular Components of Fibrosis
7. Radiation-Induced Histologic Changes
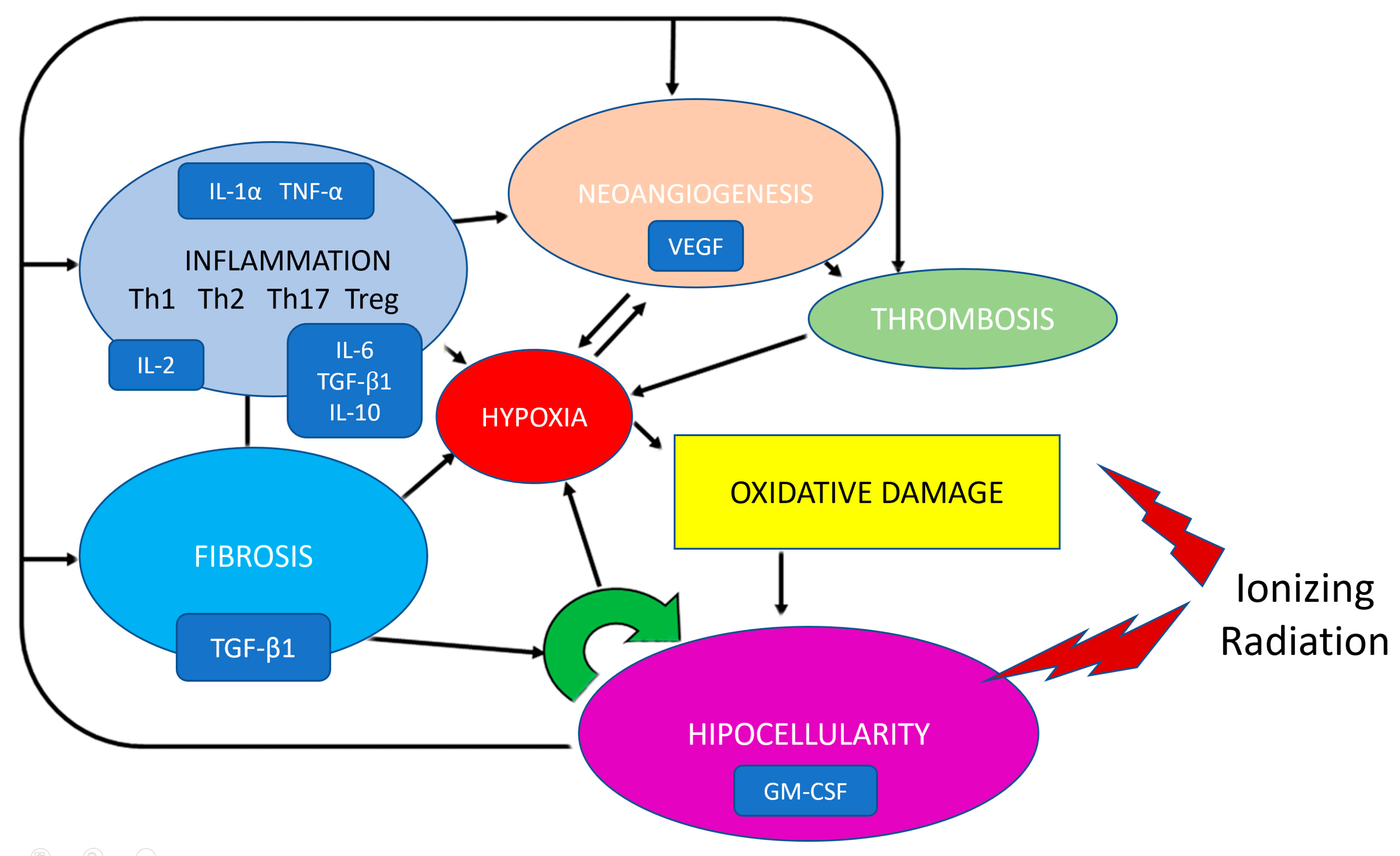
8. Pathogenesis of Radiation Strictures
9. Surgical Pitfalls and Options for Treating Radiation Strictures
10. Conservative Management
11. Endoscopic Management
12. Reconstructive Techniques—Excision and Primary Anastomosis (EPA)
13. Reconstructive Techniques—Buccal Mucosa Graft Urethroplasty
14. Robotic Techniques
References
- CDC. US Cancer Statistics 2019. Available online: cdc.gov/USCS (accessed on 1 February 2023).
- Tang, C.; Hoffman, K.E.; Allen, P.K.; Gabel, M.; Schreiber, D.; Choi, S.; Chapin, B.F.; Nguyen, Q.; Davis, J.W.; Corn, P.; et al. Contemporary prostate cancer treatment choices in multidisciplinary clinics referenced to national trends. Cancer 2019, 126, 506–514.
- Del Giudice, F.; Huang, J.; Li, S.; Sorensen, S.; Enemchukwu, E.; Maggi, M.; Salciccia, S.; Ferro, M.; Crocetto, F.; Pandolfo, S.D.; et al. Contemporary trends in the surgical management of urinary incontinence after radical prostatectomy in the United States. Prostate Cancer Prostatic Dis. 2022, 26, 367–373.
- Boorjian, S.A.; Eastham, J.A.; Graefen, M.; Guillonneau, B.; Karnes, R.J.; Moul, J.W.; Schaeffer, E.M.; Stief, C.; Zorn, K.C. A Critical Analysis of the Long-Term Impact of Radical Prostatectomy on Cancer Control and Function Outcomes. Eur. Urol. 2012, 61, 664–675.
- Elliott, S.P.; Meng, M.V.; Elkin, E.P.; McAninch, J.W.; Duchane, J.; Carroll, P.R.; CaPSURE Investigators. Incidence of urethral stricture after primary treatment for prostate cancer: Data from CaPSURE. J. Urol. 2007, 178, 529–534.
- Sterling, J.; Policastro, C.; Nikolavsky, D. Pathophysiology of radiation-induced urethral strictures and therapeutic strategies optimizing outcomes of surgical repair. In Scientific Advances in Reconstructive Urology and Tissue Engineering; Academic Press: Cambridge, MA, USA, 2022; pp. 51–80.
- Liberman, D.; Mehus, B.; Elliott, S.P. Urinary adverse effects of pelvic radiotherapy. Transl. Androl. Urol. 2014, 3, 186–195.
- David, R.V.; Kahokehr, A.A.; Lee, J.; Watson, D.I.; Leung, J.; O’callaghan, M.E. Incidence of genitourinary complications following radiation therapy for localised prostate cancer. World J. Urol. 2022, 40, 2411–2422.
- Muise, A.; Pan, M.M.; Rose, B.; Buckley, J.C. Functional outcomes after prostate cancer treatment: A comparison between single and multiple modalities. Urol. Oncol. Semin. Orig. Investig. 2023, 41, 104.e1–104.e9.
- Cavalcanti, A.G.; Yucel, S.; Deng, D.Y.; McANINCH, J.W.; Baskin, L.S. The Distribution of Neuronal and Inducible Nitric Oxide Synthase in Urethral Stricture Formation. J. Urol. 2004, 171, 1943–1947.
- Simsek, A.; Aldamanhori, R.; Chapple, C.R.; MacNeil, S. Overcoming scarring in the urethra: Challenges for tissue engineering. Asian J. Urol. 2018, 5, 69–77.
- Mundy, A.R.; Andrich, D.E. Urethral strictures. BJU Int. 2011, 107, 6–26.
- Hofer, M.D.; Cheng, E.Y.; Bury, M.I.; Park, E.; Xu, W.; Hong, S.J.; Kaplan, W.E.; Sharma, A.K. Analysis of Primary Urethral Wound Healing in the Rat. Urology 2014, 84, 246.e1–246.e7.
- Singh, M.; Blandy, J. The Pathology of Urethral Stricture. J. Urol. 1976, 115, 673–676.
- Baskin, L.S.; Constantinescu, S.C.; Howard, P.S.; McAninch, J.W.; Ewalt, D.H.; Duckett, J.W.; Snyder, H.M.; Macarak, E.J. Biochemical Characterization and Quantitation of the Collagenous Components of Urethral Stricture Tissue. J. Urol. 1993, 150, 642–647.
- Rodemann, H.P.; Blaese, M.A. Responses of Normal Cells to Ionizing Radiation. Semin. Radiat. Oncol. 2007, 17, 81–88.
- Fajardo, L.F. The pathology of ionizing radiation as defined by morphologic patterns. Acta Oncol. 2005, 44, 13–22.
- Fajardo, L.F. Is the pathology of radiation injury different in small vs large blood vessels? Cardiovasc. Radiat. Med. 1999, 1, 108–110.
- Peña, L.A.; Fuks, Z.; Kolesnick, R.N. Radiation-induced apoptosis of endothelial cells in the murine central nervous system: Protection by fibroblast growth factor and sphingomyelinase deficiency. Cancer Res. 2000, 60, 321–327.
- Reyes, J.G.; Robayna, I.G.; Delgado, P.S.; González, I.H.; Aguiar, J.Q.; Rosas, F.E.; Fanjul, L.F.; de Galarreta, C.M.R. c-Jun Is a Downstream Target for Ceramide-activated Protein Phosphatase in A431 Cells. J. Biol. Chem. 1996, 271, 21375–21380.
- Dressler, K.A.; Mathias, S.; Kolesnick, R.N. Tumor Necrosis Factor-α Activates the Sphingomyelin Signal Transduction Pathway in a Cell-Free System. Science 1992, 255, 1715–1718.
- Müller, G.; Ayoub, M.; Storz, P.; Rennecke, J.; Fabbro, D.; Pfizenmaier, K. PKC zeta is a molecular switch in signal transduction of TNF-alpha, bifunctionally regulated by ceramide and arachidonic acid. EMBO J. 1995, 14, 1961–1969.
- Haimovitz-Friedman, A.; Kan, C.C.; Ehleiter, D.; Persaud, R.S.; McLoughlin, M.; Fuks, Z.; Kolesnick, R.N. Ionizing radiation acts on cellular membranes to generate ceramide and initiate apoptosis. J. Exp. Med. 1994, 180, 525–535.
- Haimovitz-Friedman, A.; Balaban, N.; McLoughlin, M.; Ehleiter, D.; Michaeli, J.; Vlodavsky, I.; Fuks, Z. Protein kinase C mediates basic fibroblast growth factor protection of endothelial cells against radiation-induced apoptosis. Cancer Res. 1994, 54, 2591–2597.
- Basu, S.; Bayoumy, S.; Zhang, Y.; Lozano, J.; Kolesnick, R. BAD Enables Ceramide to Signal Apoptosis via Ras and Raf-1. J. Biol. Chem. 1998, 273, 30419–30426.
- Yang, J.; Liu, X.; Bhalla, K.; Kim, C.N.; Ibrado, A.M.; Cai, J.; Peng, T.-I.; Jones, D.P.; Wang, X. Prevention of Apoptosis by Bcl-2: Release of Cytochrome c from Mitochondria Blocked. Science 1997, 275, 1129–1132.
- Belka, C.; Budach, W. Anti-apoptotic Bcl-2 proteins: Structure, function and relevance for radiation biology. Int. J. Radiat. Biol. 2002, 78, 643–658.
- Bayreuther, K.; Francz, P.; Rodemann, H. Fibroblasts in normal and pathological terminal differentiation, aging, apoptosis and transformation. Arch. Gerontol. Geriatr. 1992, 15, 47–74.
- Rodemann, H.P.; Binder, A.; Burger, A.; Güven, N.; Löffler, H.; Bamberg, M. The underlying cellular mechanism of fibrosis. Kidney Int. Suppl. 1996, 54, S32–S36.
- Burger, A.; Löffler, H.; Bamberg, M.; Rodemann, H.P. Molecular and cellular basis of radiation fibrosis. Int. J. Radiat. Biol. 1998, 73, 401–408.
- Rodemann, H.; Bamberg, M. Cellular basis of radiation-induced fibrosis. Radiother. Oncol. 1995, 35, 83–90.
- Paun, A.; Kunwar, A.; Haston, C.K. Acute adaptive immune response correlates with late radiation-induced pulmonary fibrosis in mice. Radiat. Oncol. 2015, 10, 45.
- Takigawa, N.; Segawa, Y.; Saeki, T.; Kataoka, M.; Ida, M.; Kishino, D.; Fujiwara, K.; Ohsumi, S.; Eguchi, K.; Takashima, S. Bronchiolitis obliterans organizing pneumonia syndrome in breast-conserving therapy for early breast cancer: Radiation-induced lung toxicity. Int. J. Radiat. Oncol. Biol. Phys. 2000, 48, 751–755.
- Bessout, R.; Demarquay, C.; Moussa, L.; René, A.; Doix, B.; Benderitter, M.; Sémont, A.; Mathieu, N. TH17 predominant T-cell responses in radiation-induced bowel disease are modulated by treatment with adipose-derived mesenchymal stromal cells. J. Pathol. 2015, 237, 435–446.
- Garrido-Mesa, N.; Algieri, F.; Rodriguez Nogales, A.; Galvez, J. Functional plasticity of Th17 cells: Implications in gastrointestinal tract function. Int. Rev. Immunol. 2013, 32, 493–510.
- Martin, M.; Lefaix, J.; Delanian, S. TGF-beta1 and radiation fibrosis: A master switch and a specific therapeutic target? Int. J. Radiat. Oncol. Biol. Phys. 2000, 47, 277–290.
- Mayer, M. Biochemical and biological aspects of the plasminogen activation system. Clin. Biochem. 1990, 23, 197–211.
- Hughes, M.; Caza, T.; Li, G.; Daugherty, M.; Blakley, S.; Nikolavsky, D. Histologic characterization of the post-radiation urethral stenosis in men treated for prostate cancer. World J. Urol. 2019, 38, 2269–2277.
- Hanin, L.; Zaider, M. A mechanistic description of radiation-induced damage to normal tissue and its healing kinetics. Phys. Med. Biol. 2013, 58, 825–839.
- Martin, J.M.; Richardson, M.; Siva, S.; Cardoso, M.; Handmer, M.; Sidhom, M. Mechanisms, mitigation, and management of urinary toxicity from prostate radiotherapy. Lancet Oncol. 2022, 23, e534–e543.
- Rodda, S.; Tyldesley, S.; Morris, W.J.; Keyes, M.; Halperin, R.; Pai, H.; McKenzie, M.; Duncan, G.; Morton, G.; Hamm, J.; et al. ASCENDE-RT: An Analysis of Treatment-Related Morbidity for a Randomized Trial Comparing a Low-Dose-Rate Brachytherapy Boost with a Dose-Escalated External Beam Boost for High- and Intermediate-Risk Prostate Cancer. Int. J. Radiat. Oncol. 2017, 98, 286–295.
- Awad, M.A.; Gaither, T.W.; Osterberg, E.C.; Murphy, G.P.; Baradaran, N.; Breyer, B.N. Prostate cancer radiation and urethral strictures: A systematic review and meta-analysis. Prostate Cancer Prostatic Dis. 2018, 21, 168–174.
- Hindson, B.R.; Millar, J.L.; Matheson, B. Urethral strictures following high-dose-rate brachytherapy for prostate cancer: Analysis of risk factors. Brachytherapy 2012, 12, 50–55.
- Sullivan, L.; Williams, S.G.; Tai, K.H.; Foroudi, F.; Cleeve, L.; Duchesne, G. Urethral stricture following high dose rate brachytherapy for prostate cancer. Radiother. Oncol. 2009, 91, 232–236.
- Merrick, G.S.; Butler, W.M.; Wallner, K.E.; Galbreath, R.W.; Anderson, R.L.; Allen, Z.A.; Adamovich, E. Risk Factors for the Development of Prostate Brachytherapy Related Urethral Strictures. J. Urol. 2006, 175, 1376–1381.
- Sowerby, R.J.; Gani, J.; Yim, H.; Radomski, S.B.; Catton, C. Long-term complications in men who have early or late radio-therapy after radical prostatectomy. Can. Urol. Assoc. J. 2014, 8, 253–258.
- Herschorn, S.; Elliott, S.; Coburn, M.; Wessells, H.; Zinman, L. SIU/ICUD Consultation on Urethral Strictures: Posterior Urethral Stenosis After Treatment of Prostate Cancer. Urology 2014, 83, S59–S70.
- Merrick, G.S.; Butler, W.M.; Wallner, K.E.; Galbreath, R.W.; Lief, J.H. Long-term urinary quality of life after permanent prostate brachytherapy. Int. J. Radiat. Oncol. 2003, 56, 454–461.
- Allen, Z.A.; Merrick, G.S.; Butler, W.M.; Wallner, K.E.; Kurko, B.; Anderson, R.L.; Murray, B.C.; Galbreath, R.W. Detailed urethral dosimetry in the evaluation of prostate brachytherapy-related urinary morbidity. Int. J. Radiat. Oncol. 2005, 62, 981–987.
- Zaffuto, E.; Gandaglia, G.; Fossati, N.; Dell’Oglio, P.; Moschini, M.; Cucchiara, V.; Suardi, N.; Mirone, V.; Bandini, M.; Shariat, S.F.; et al. Early Postoperative radiotherapy is assocciated with worse functional outcomes in patients with prostate cancer. J. Urol. 2017, 197, 669–675.
- Tibbs, M.K. Wound healing following radiation therapy: A review. Radiother. Oncol. 1997, 42, 99–106.
- Barry, J.M. Visual urethrotomy in the management of the obliterated membranous urethra. Urol. Clin. N. Am. 1989, 16, 319–324.
- Stein, D.M.; Santucci, R.A. Pro: Endoscopic realignment for pelvic fracture urethral injuries. Transl. Androl. Urol. 2015, 4, 72–78.
- Yasuda, K.; Yamanishi, T.; Isaka, S.; Okano, T.; Masai, M.; Shimazaki, J. Endoscopic Re-Establishment of Membranous Urethral Disruption. J. Urol. 1991, 145, 977–979.
- Rozanski, A.T.; Moynihan, M.J.; Zhang, L.T.; Muise, A.C.; Holst, D.D.; Copacino, S.A.; Zinman, L.N.; Buckley, J.C.; Vanni, A.J. The Efficacy and Safety of a Conservative Management Approach to Radiation-Induced Male Urethral Strictures in Elderly Patients With Comorbidities. Société Int. d’Urologie J. 2022, 3, 14–20.
- Ravier, E.; Fassi-Fehri, H.; Crouzet, S.; Gelet, A.; Abid, N.; Martin, X. Complications after artificial urinary sphincter implantation in patients with or without prior radiotherapy. BJU Int. 2014, 115, 300–307.
- Eltahawy, E.; Gur, U.; Virasoro, R.; Schlossberg, S.M.; Jordan, G.H. Management of recurrent anastomotic stenosis following radical prostatectomy using holmium laser and steroid injection. BJU Int. 2008, 102, 796–798.
- Kravchick, S.; Lobik, L.; Peled, R.; Cytron, S. Transrectal Ultrasonography-Guided Injection of Long-Acting Steroids in the Treatment of Recurrent/Resistant Anastomotic Stenosis After Radical Prostatectomy. J. Endourol. 2013, 27, 875–879.
- Redshaw, J.D.; Broghammer, J.A.; Smith, T.G., 3rd; Voelzke, B.B.; Erickson, B.A.; McClung, C.D.; Elliott, S.P.; Alsikafi, N.F.; Presson, A.P.; Aberger, M.E.; et al. Intralesional injection of mitomycin-C at transurethral incision of bladder neck contracture may offer limited benefit: TURNS Study Group. J. Urol. 2015, 193, 587–592.
- Rozanski, A.T.; Zhang, L.T.; Holst, D.D.; Copacino, S.A.; Vanni, A.J.; Buckley, J.C. The Effect of Radiation Therapy on the Efficacy of Internal Urethrotomy With Intralesional Mitomycin C for Recurrent Vesicourethral Anastomotic Stenoses and Bladder Neck Contractures: A Multi-Institutional Experience. Urology 2020, 147, 294–298.
- Vanni, A.J.; Zinman, L.N.; Buckley, J.C. Radial Urethrotomy and Intralesional Mitomycin C for the Management of Recurrent Bladder Neck Contractures. J. Urol. 2011, 186, 156–160.
- Meeks, J.J.; Brandes, S.B.; Morey, A.F.; Thom, M.; Mehdiratta, N.; Valadez, C.; Granieri, M.A.; Gonzalez, C.M. Urethroplasty for Radiotherapy Induced Bulbomembranous Strictures: A Multi-Institutional Experience. J. Urol. 2011, 185, 1761–1765.
- Hofer, M.D.; Zhao, L.C.; Morey, A.F.; Scott, J.F.; Chang, A.J.; Brandes, S.B.; Gonzalez, C.M. Outcomes after Urethroplasty for Radiotherapy Induced Bulbomembranous Urethral Stricture Disease. J. Urol. 2014, 191, 1307–1312.
- Rourke, K.; Kinnaird, A.; Zorn, J. Observations and outcomes of urethroplasty for bulbomembranous stenosis after radiation therapy for prostate cancer. World J. Urol. 2015, 34, 377–382.
- Fuchs, J.S.; Hofer, M.D.; Sheth, K.R.; Cordon, B.H.; Scott, J.M.; Morey, A.F. Improving Outcomes of Bulbomembranous Urethroplasty for Radiation-induced Urethral Strictures in Post-Urolume Era. Urology 2016, 99, 240–245.
- Glass, A.S.; McAninch, J.W.; Zaid, U.B.; Cinman, N.M.; Breyer, B.N. Urethroplasty After Radiation Therapy for Prostate Cancer. Urology 2012, 79, 1402–1406.
- Voelzke, B.B.; Leddy, L.S.; Myers, J.B.; Breyer, B.N.; Alsikafi, N.F.; Broghammer, J.A.; Elliott, S.P.; Vanni, A.J.; Erickson, B.A.; Buckley, J.C.; et al. Multi-institutional outcomes and associations after excision and primary anastomosis for radiotherapy-associated bulbomembranous urethral stenoses following prostate cancer treatment. Urology 2021, 152, 117–122.
- Blakely, S.; Caza, T.; Landas, S.; Nikolavsky, D. Dorsal Onlay Urethroplasty for Membranous Urethral Strictures: Urinary and Erectile Functional Outcomes. J. Urol. 2015, 195, 1501–1507.
- Chung, P.H.; Esposito, P.; Wessells, H.; Voelzke, B.B. Incidence of Stress Urinary Incontinence After Posterior Urethroplasty for Radiation-induced Urethral Strictures. Urology 2018, 114, 188–192.
- Ahyai, S.A.; Schmid, M.; Kuhl, M.; Kluth, L.A.; Soave, A.; Riechardt, S.; Chun, F.K.-H.; Engel, O.; Fisch, M.; Dahlem, R. Outcomes of Ventral Onlay Buccal Mucosa Graft Urethroplasty in Patients after Radiotherapy. J. Urol. 2015, 194, 441–446.
- Policastro, C.G.; Simhan, J.; Martins, F.E.; Lumen, N.; Venkatesan, K.; Angulo, J.C.; Gupta, S.; Rusilko, P.; Pérez, E.A.R.; Redger, K.; et al. A multi-institutional critical assessment of dorsal onlay urethroplasty for post-radiation urethral stenosis. World J. Urol. 2020, 39, 2669–2675.
- Elbakry, A.A.; Pan, M.M.; Buckley, J.C. Frontiers in post-radiation urologic reconstruction; robotic surgery and near-infrared fluorescence imaging: A Narrative Review. AME Med. J. 2022, 7, 7.
- Toia, B.; Seth, J.; Ecclestone, H.; Pakzad, M.; Hamid, R.; Greenwell, T.; Ockrim, J. Outcomes of reconstructive urinary tract surgery after pelvic radiotherapy. Scand. J. Urol. 2019, 53, 156–160.
- Vetterlein, M.W.; Kluth, L.A.; Zumstein, V.; Meyer, C.P.; Ludwig, T.A.; Soave, A.; Riechardt, S.; Engel, O.; Dahlem, R.; Fisch, M.; et al. Buccal mucosal graft urethroplasty for radiation-induced urethral strictures: An evaluation using the extended Urethral Stricture Surgery Patient-Reported Outcome Measure (USS PROM). World J. Urol. 2020, 38, 2863–2872.
- Flamiatos, J.F.; Chen, Y.; Lambert, W.E.; Martinez Acevedo, A.; Becker, T.M.; Bash, J.C.; Amling, C.L. Open versus robot-assisted radical cystectomy: 30-day perioperative comparison and predictors for cost-to patient, complication, and read-mission. J. Robot. Surg. 2019, 13, 129–140.
- Khalil, M.I.; Tourchi, A.; Langford, B.T.; Bhandari, N.R.; Payakachat, N.; Davis, R.; Safaan, A.; Raheem, O.A.; Kamel, M.H. Early Postoperative Morbidity of Robotic Versus Open Radical Cystectomy in Obese Patients. J. Endourol. 2020, 34, 461–468.
- Kim, S.; Buckley, J.C. Robotic Lower Urinary Tract Reconstruction. Urol. Clin. N. Am. 2020, 48, 103–112.
- Unterberg, S.H.; Patel, S.H.; Fuller, T.W.; Buckley, J.C. Robotic assisted proximal perineal urethroplasty: Improving visualization and ergonomics. Urology 2019, 125, 230–233.
- Bearrick, E.N.; Findlay, B.L.; Maciejko, L.A.; Hebert, K.J.; Anderson, K.T.; Viers, B.R. Robotic urethral reconstruction out-comes in men with posterior urethral stenosis. Urology 2022, 161, 118–124.
- Kirshenbaum, E.J.; Zhao, L.C.; Myers, J.B.; Elliott, S.P.; Vanni, A.J.; Baradaran, N.; Erickson, B.A.; Buckley, J.C.; Voelzke, B.B.; Granieri, M.A.; et al. Patency and Incontinence Rates After Robotic Bladder Neck Reconstruction for Vesicourethral Anastomotic Stenosis and Recalcitrant Bladder Neck Contractures: The Trauma and Urologic Reconstructive Network of Surgeons Experience. Urology 2018, 118, 227–233.





