
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Raghad Sabaawi Ibrahim | -- | 2409 | 2023-07-15 00:54:11 | | | |
| 2 | Lindsay Dong | Meta information modification | 2409 | 2023-07-18 03:02:48 | | |
Video Upload Options
Glioblastoma’s poor prognosis calls for the discovery of newer, more efficacious management and treatment methods. Metformin and resveratrol exert anticancer effects on major metabolic pathways in glioblastoma cells, resulting in reduced proliferation, increased apoptosis, and reduced tumor growth and volume. The shown effects suggest that metformin and resveratrol can potentially aid in treating glioblastoma.
1. Metformin and Resveratrol in Glioblastoma
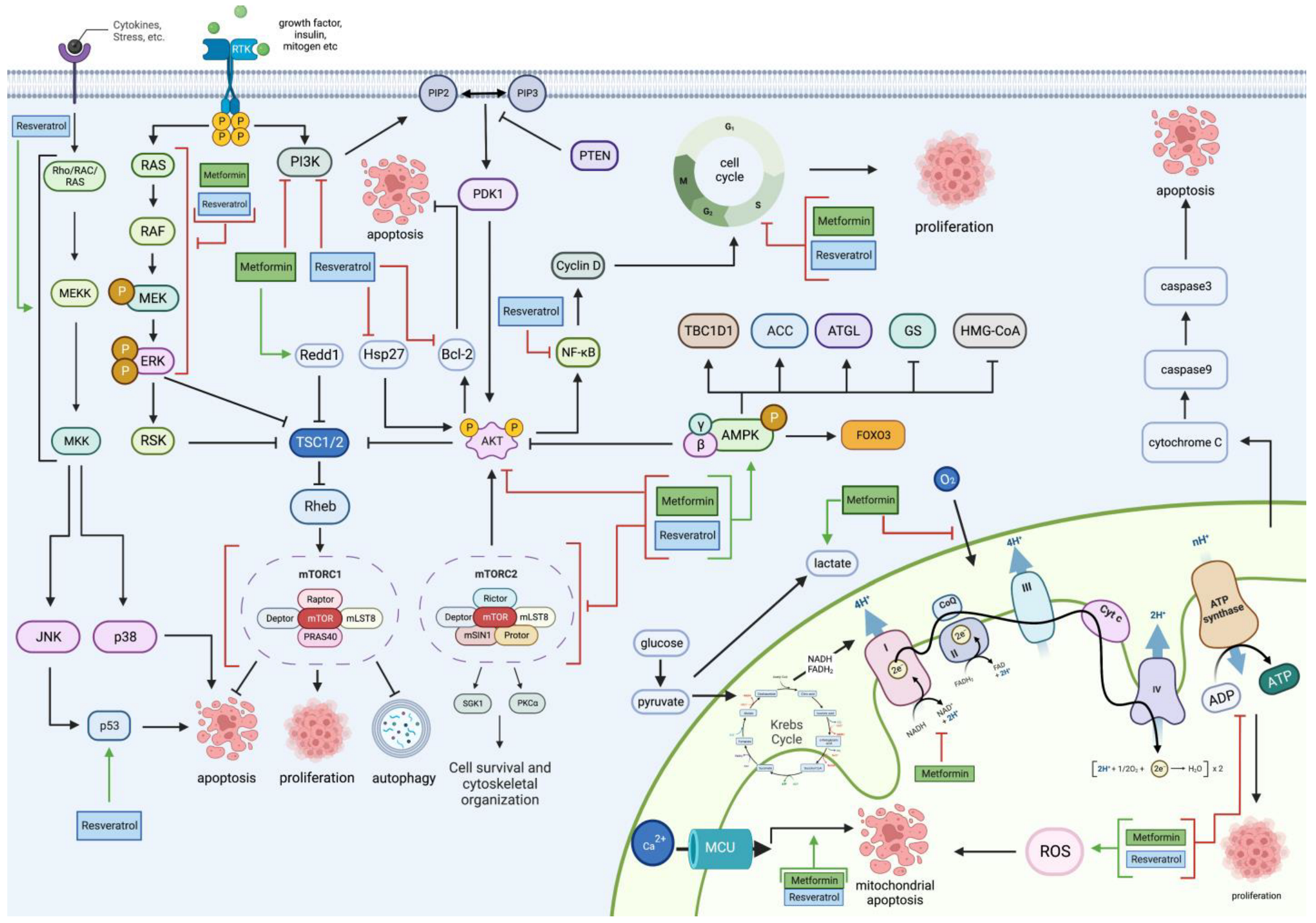
2. Metformin and Resveratrol on Glioblastoma’s Proliferative and Apoptotic Pathways
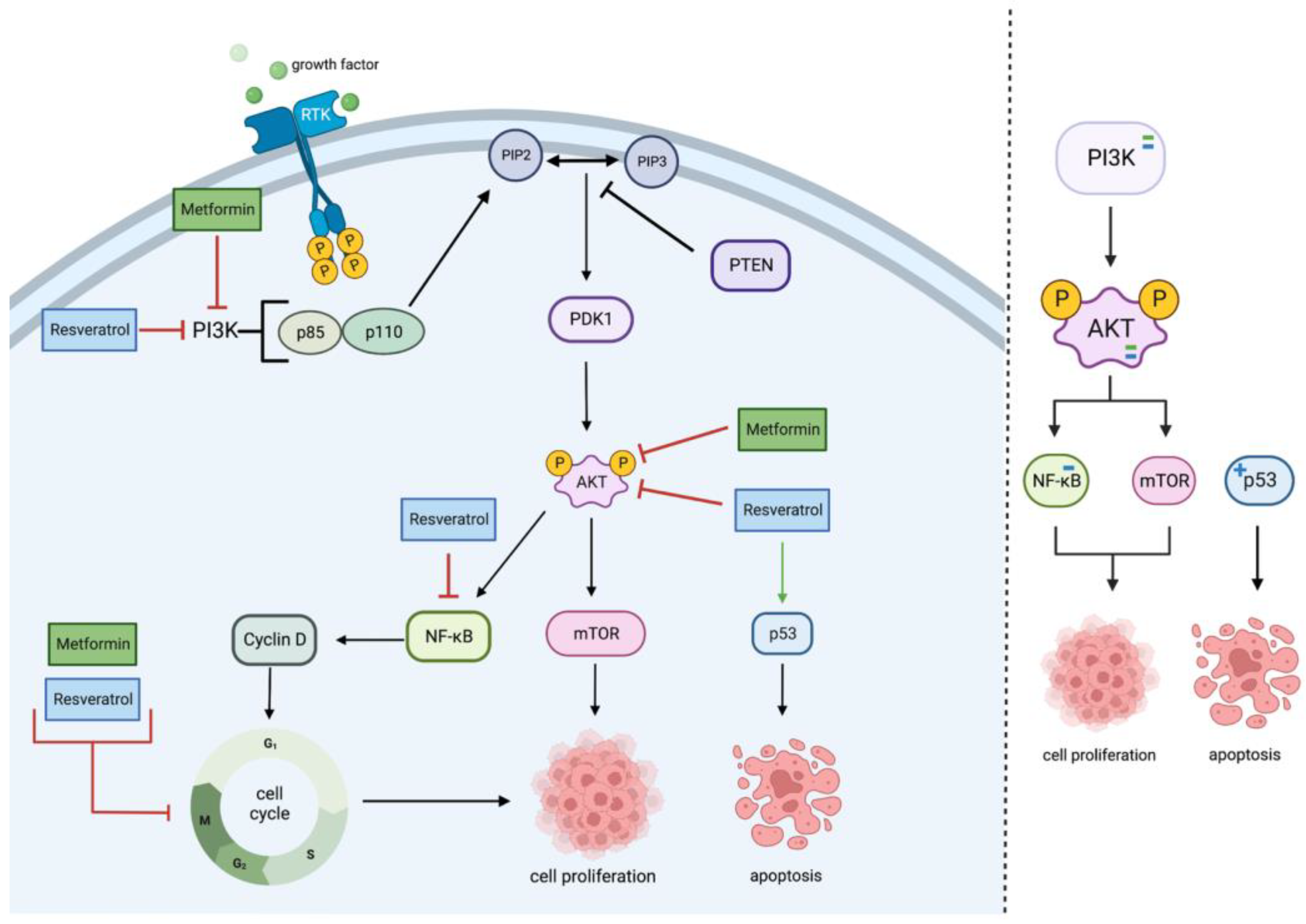
2.1. PI3K/Akt Pathway
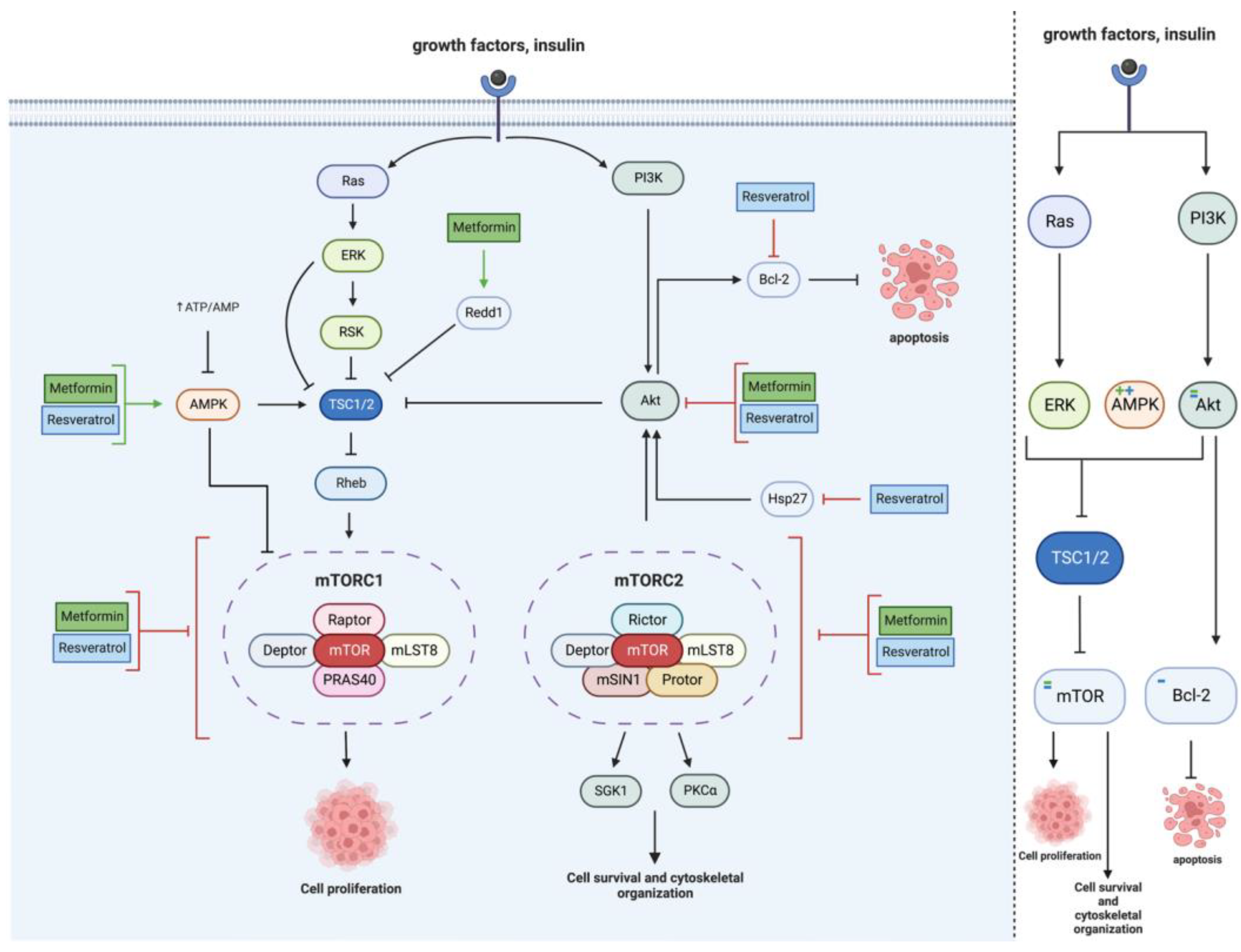
2.2. mTOR Pathway
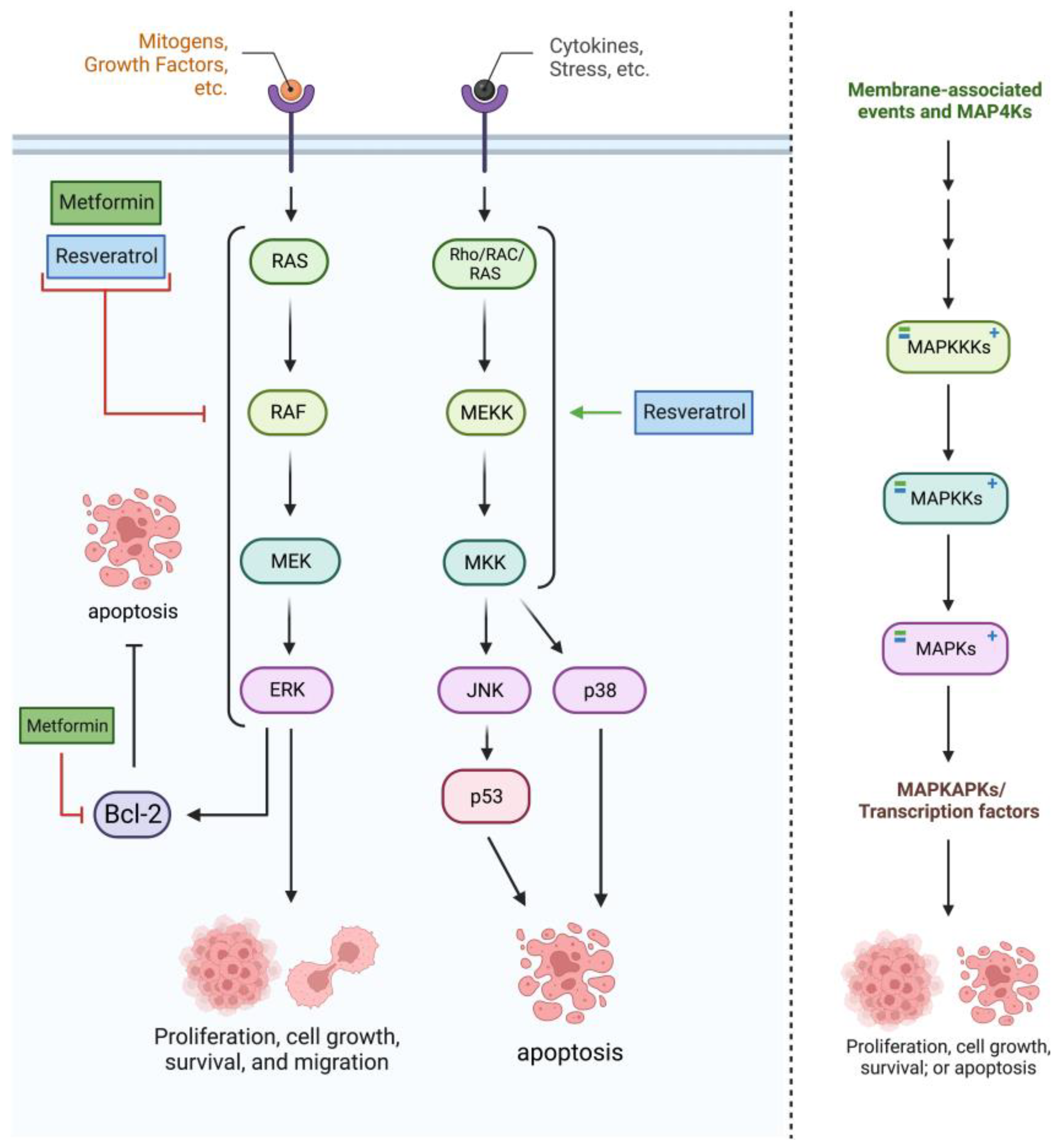
2.3. RAS/RAF/MAPK Pathway
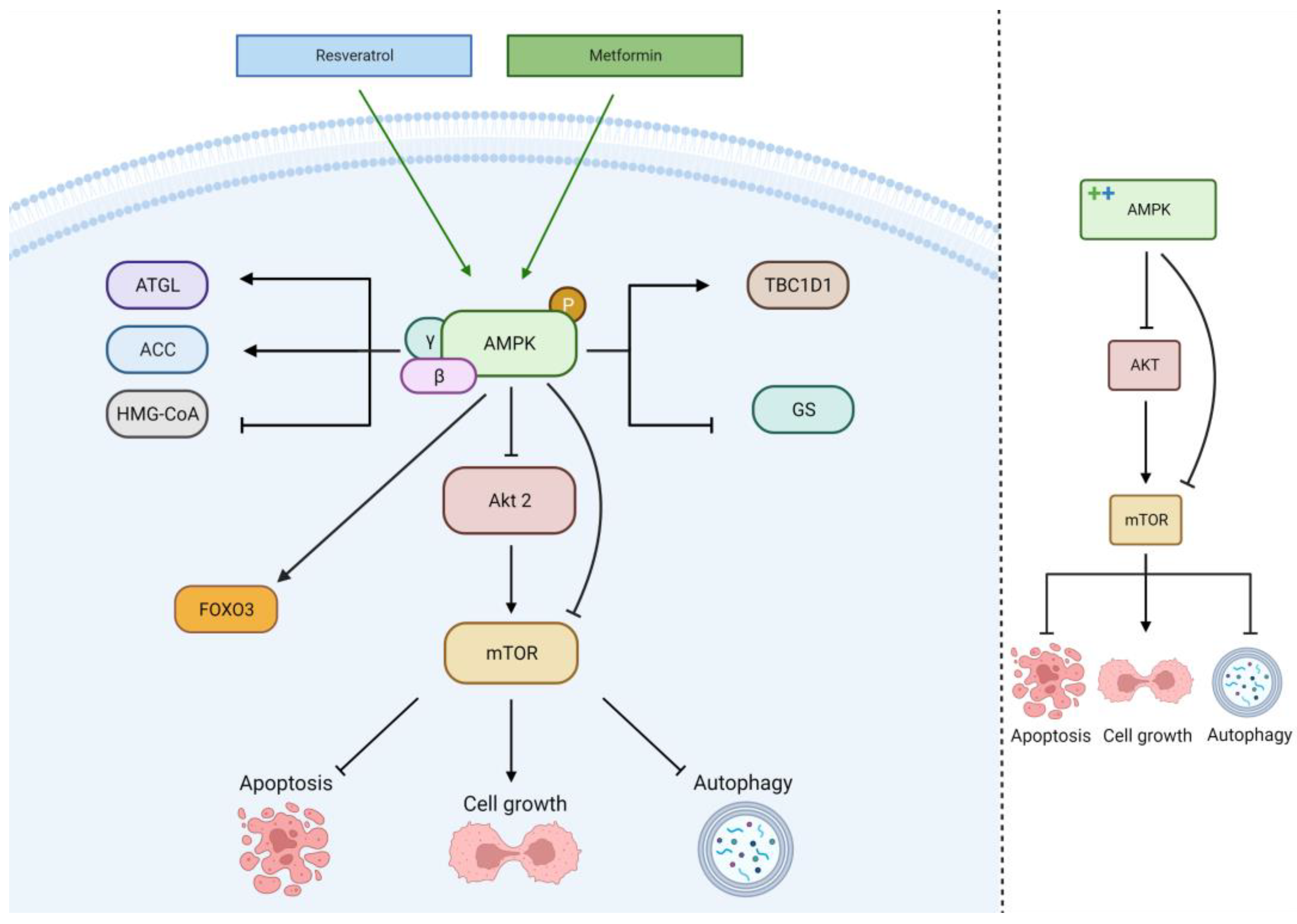
2.4. AMPK Pathway
2.5. Mitochondrial Pathway
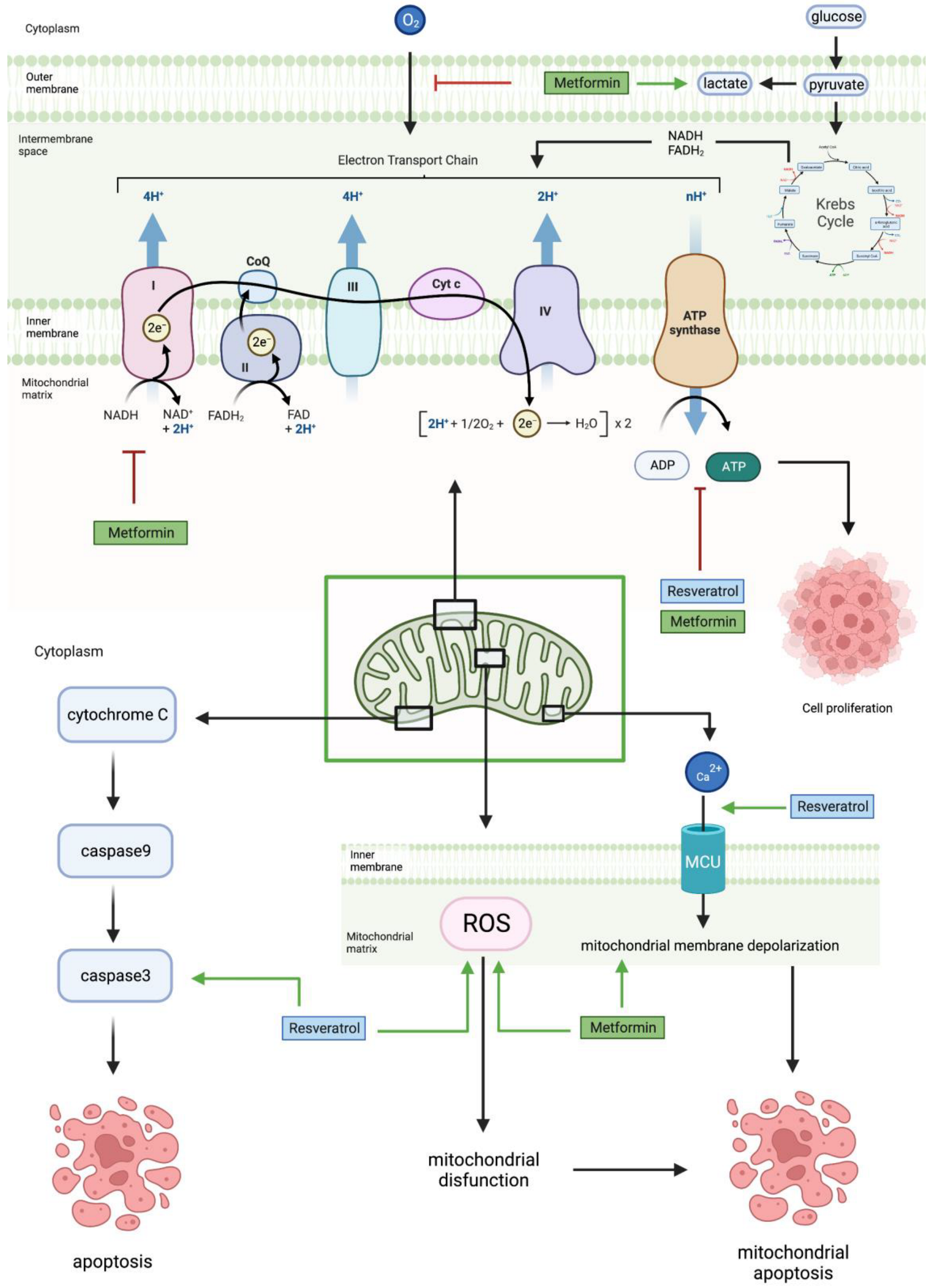
2.6. In Vivo
3. Metformin and Resveratrol on Glucose in Glioblastoma
The interaction of glucose with different pathways, such as the nuclear factor kappa beta (NF-κB) pathway and glycolytic pathway, possibly increased cell proliferation, cell viability, tumorigenesis, NF-κB phosphorylation, and the expression of Bcl-2, Mcl-1, FPR1, EGFR, VEGF, ERK, EGF, ROS production, STAT3, PDK1, PDK3, ECH, and HADH [11][46][47][48]. These effects arose through multiple signaling intermediates and interactions. For example, high glucose upregulates the expression of a G-protein coupled chemoattractant receptor (GPCR), formyl peptide receptor 1 (FPR1), and epidermal growth factor receptor (EGFR) on GBM cells [11].
A high glucose-induced increase in the Warburg effect is also observed: where cancer cells alter molecular pathways and switch from oxidative phosphorylation to aerobic glycolysis [5][47]. This alteration—the Warburg phenotype—then composes numerous selective molecular advantages for the growth and proliferation of tumor cells.
4. Clinical Considerations and Relevance
4.1. Bioavailibility
4.2. Delivery
4.3. Clinical Trials
5. Conclusions
References
- Thakkar, J.P.; Dolecek, T.A.; Horbinski, C.; Ostrom, Q.T.; Lightner, D.D.; Barnholtz-Sloan, J.S.; Villano, J.L. Epidemiologic and Molecular Prognostic Review of Glioblastoma. Cancer Epidemiol. Biomark. Prev. 2014, 23, 1985–1996.
- Miller, K.D.; Ostrom, Q.T.; Kruchko, C.; Patil, N.; Tihan, T.; Cioffi, G.; Fuchs, H.E.; Waite, K.A.; Jemal, A.; Siegel, R.L.; et al. Brain and Other Central Nervous System Tumor Statistics, 2021. CA Cancer J. Clin. 2021, 71, 381–406.
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.B.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus Concomitant and Adjuvant Temozolomide for Glioblastoma. N. Engl. J. Med. 2005, 352, 987–996.
- Ostrom, Q.T.; Gittleman, H.; Xu, J.; Kromer, C.; Wolinsky, Y.; Kruchko, C.; Barnholtz-Sloan, J.S. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2009–2013. Neuro Oncol. 2016, 18, v1–v75.
- Samec, M.; Liskova, A.; Koklesova, L.; Samuel, S.M.; Zhai, K.; Buhrmann, C.; Varghese, E.; Abotaleb, M.; Qaradakhi, T.; Zulli, A.; et al. Flavonoids against the Warburg Phenotype—Concepts of Predictive, Preventive and Personalised Medicine to Cut the Gordian Knot of Cancer Cell Metabolism. EPMA J. 2020, 11, 377.
- Koklesova, L.; Liskova, A.; Samec, M.; Qaradakhi, T.; Zulli, A.; Smejkal, K.; Kajo, K.; Jakubikova, J.; Behzadi, P.; Pec, M.; et al. Genoprotective Activities of Plant Natural Substances in Cancer and Chemopreventive Strategies in the Context of 3P Medicine. EPMA J. 2020, 11, 261–287.
- Samec, M.; Liskova, A.; Kubatka, P.; Uramova, S.; Zubor, P.; Samuel, S.M.; Zulli, A.; Pec, M.; Bielik, T.; Biringer, K.; et al. The Role of Dietary Phytochemicals in the Carcinogenesis via the Modulation of MiRNA Expression. J. Cancer Res. Clin. Oncol. 2019, 145, 1665–1679.
- Hardie, D.G. A New Understanding of Metformin. In Comprehensive Pharmacology; Elsevier: Amsterdam, The Netherlands, 2022; pp. 280–300.
- Takhwifa, F.; Aninditha, T.; Setiawan, H.; Sauriasari, R. The Potential of Metformin as an Antineoplastic in Brain Tumors: A Systematic Review. Heliyon 2021, 7, E06558.
- Rauf, A.; Imran, M.; Butt, M.S.; Nadeem, M.; Peters, D.G.; Mubarak, M.S. Resveratrol as an Anticancer Agent: A Review. Crit. Rev. Food Sci. Nutr. 2017, 58, 1428–1447.
- Bao, Z.; Chen, K.; Krepel, S.; Tang, P.; Gong, W.; Zhang, M.; Liang, W.; Trivett, A.; Zhou, M.; Wang, J.M. High Glucose Promotes Human Glioblastoma Cell Growth by Increasing the Expression and Function of Chemoattractant and Growth Factor Receptors. Transl. Oncol. 2019, 12, 1155–1163.
- Tseng, H.W.; Li, S.C.; Tsai, K.W. Metformin Treatment Suppresses Melanoma Cell Growth and Motility through Modulation of MicroRNA Expression. Cancers 2019, 11, 209.
- Chen, K.; Qian, W.; Jiang, Z.; Cheng, L.; Li, J.; Sun, L.; Zhou, C.; Gao, L.; Lei, M.; Yan, B.; et al. Metformin Suppresses Cancer Initiation and Progression in Genetic Mouse Models of Pancreatic Cancer. Mol. Cancer 2017, 16, 131.
- Wu, X.P.; Xiong, M.; Xu, C.S.; Duan, L.N.; Dong, Y.Q.; Luo, Y.; Niu, T.H.; Lu, C.R. Resveratrol Induces Apoptosis of Human Chronic Myelogenous Leukemia Cells in Vitro through P38 and JNK-Regulated H2AX Phosphorylation. Acta Pharmacol. Sin. 2015, 36, 353–361.
- Kueck, A.; Opipari, A.W.; Griffith, K.A.; Tan, L.; Choi, M.; Huang, J.; Wahl, H.; Liu, J.R. Resveratrol Inhibits Glucose Metabolism in Human Ovarian Cancer Cells. Gynecol. Oncol. 2007, 107, 450–457.
- Würth, R.; Pattarozzi, A.; Gatti, M.; Bajetto, A.; Corsaro, A.; Parodi, A.; Sirito, R.; Massollo, M.; Marini, C.; Zona, G.; et al. Metformin Selectively Affects Human Glioblastoma Tumor-Initiating Cell Viability. Cell Cycle 2012, 12, 145–156.
- Hassan, M.A.; Fakhoury, I.; El Masri, Z.; Ghazale, N.; Dennaoui, R.; Atat, O.E.; Kanaan, A.; El-Sibai, M. Metformin Treatment Inhibits Motility and Invasion of Glioblastoma Cancer Cells. Anal. Cell. Pathol. 2018, 2018, 5917470.
- Sesen, J.; Dahan, P.; Scotland, S.J.; Saland, E.; Dang, V.T.; Lemarié, A.; Tyler, B.M.; Brem, H.; Toulas, C.; Moyal, E.C.J.; et al. Metformin Inhibits Growth of Human Glioblastoma Cells and Enhances Therapeutic Response. PLoS ONE 2015, 10, e0123721.
- Liu, L.; Zhang, Y.; Zhu, K.; Song, L.; Tao, M.; Huang, P.; Pan, Y. Resveratrol Inhibits Glioma Cell Growth via Targeting LRIG1. JBUON 2018, 23, 403–409.
- Jiao, Y.; Li, H.; Liu, Y.; Guo, A.; Xu, X.; Qu, X.; Wang, S.; Zhao, J.; Li, Y.; Cao, Y. Resveratrol Inhibits the Invasion of Glioblastoma-Initiating Cells via Down-Regulation of the PI3K/Akt/NF-ΚB Signaling Pathway. Nutrients 2015, 7, 4383–4402.
- Clark, P.A.; Bhattacharya, S.; Elmayan, A.; Darjatmoko, S.R.; Thuro, B.A.; Yan, M.B.; van Ginkel, P.R.; Polans, A.S.; Kuo, J.S. Resveratrol Targeting of AKT and P53 in Glioblastoma and Glioblastoma Stem-like Cells to Suppress Growth and Infiltration. J. Neurosurg. 2017, 126, 1448–1460.
- Önay Uçar, E.; Şengelen, A. Resveratrol and SiRNA in Combination Reduces Hsp27 Expression and Induces Caspase-3 Activity in Human Glioblastoma Cells. Cell Stress Chaperones 2019, 24, 763–775.
- Filippi-Chiela, E.C.; Villodre, E.S.; Zamin, L.L.; Lenz, G. Autophagy Interplay with Apoptosis and Cell Cycle Regulation in the Growth Inhibiting Effect of Resveratrol in Glioma Cells. PLoS ONE 2011, 6, e20849.
- Wang, G.; Dai, F.; Yu, K.; Jia, Z.; Zhang, A.; Huang, Q.; Kang, C.; Jiang, H.; Pu, P. Resveratrol Inhibits Glioma Cell Growth via Targeting Oncogenic MicroRNAs and Multiple Signaling Pathways. Int. J. Oncol. 2015, 46, 1739–1747.
- Xia, C.; Liu, C.; He, Z.; Cai, Y.; Chen, J. Metformin Inhibits Cervical Cancer Cell Proliferation by Modulating PI3K/Akt-Induced Major Histocompatibility Complex Class I-Related Chain A Gene Expression. J. Exp. Clin. Cancer Res. 2020, 39, 127.
- Kim, T.S.; Lee, M.; Park, M.; Kim, S.Y.; Shim, M.S.; Lee, C.Y.; Choi, D.H.; Cho, Y. Metformin and Dichloroacetate Suppress Proliferation of Liver Cancer Cells by Inhibiting MTOR Complex 1. Int. J. Mol. Sci. 2021, 22, 10027.
- Liu, Y.; Zhang, Y.; Jia, K.; Dong, Y.; Ma, W. Metformin Inhibits the Proliferation of A431 Cells by Modulating the PI3K/Akt Signaling Pathway. Exp. Ther. Med. 2022, 24, 445.
- Innets, B.; Thongsom, S.; Petsri, K.; Racha, S.; Yokoya, M.; Moriue, S.; Chaotham, C.; Chanvorachote, P. Akt/MTOR Targeting Activity of Resveratrol Derivatives in Non-Small Lung Cancer. Molecules 2022, 27, 8268.
- Wang, J.; Li, J.; Cao, N.; Li, Z.; Han, J.; Li, L. Resveratrol, an Activator of SIRT1, Induces Protective Autophagy in Non-Small-Cell Lung Cancer via Inhibiting Akt/MTOR and Activating P38-MAPK. Onco. Targets Ther. 2018, 11, 7777–7786.
- Zou, Z.; Tao, T.; Li, H.; Zhu, X. MTOR Signaling Pathway and MTOR Inhibitors in Cancer: Progress and Challenges. Cell Biosci. 2020, 10, 31.
- Xiong, Z.S.; Gong, S.F.; Si, W.; Jiang, T.; Li, Q.L.; Wang, T.J.; Wang, W.J.; Wu, R.Y.; Jiang, K. Effect of Metformin on Cell Proliferation, Apoptosis, Migration and Invasion in A172 Glioma Cells and Its Mechanisms. Mol. Med. Rep. 2019, 20, 887–894.
- Yuan, Y.; Xue, X.; Guo, R.B.; Sun, X.L.; Hu, G. Resveratrol Enhances the Antitumor Effects of Temozolomide in Glioblastoma via ROS-Dependent AMPK-TSC-MTOR Signaling Pathway. CNS Neurosci. Ther. 2012, 18, 536–546.
- Guarnaccia, L.; Navone, S.E.; Masseroli, M.M.; Balsamo, M.; Caroli, M.; Valtorta, S.; Moresco, R.M.; Campanella, R.; Schisano, L.; Fiore, G.; et al. Effects of Metformin as Add-On Therapy against Glioblastoma: An Old Medicine for Novel Oncology Therapeutics. Cancers 2022, 14, 1412.
- Carmignani, M.; Volpe, A.R.; Aldea, M.; Soritau, O.; Irimie, A.; Florian, I.S.; Tomuleasa, C.; Baritchii, A.; Petrushev, B.; Crisan, G.; et al. Glioblastoma Stem Cells: A New Target for Metformin and Arsenic Trioxide. J. Biol. Regul. Homeost. Agents 2014, 28, 1–15.
- Jung, J.S.; Woo, J.S. Resveratrol Induces Cell Death through ROS-Dependent MAPK Activation in A172 Human Glioma Cells. J. Life Sci. 2016, 26, 212–219.
- Mihaylova, M.M.; Shaw, R.J. The AMPK Signalling Pathway Coordinates Cell Growth, Autophagy and Metabolism. Nat. Cell Biol. 2011, 13, 1016–1023.
- Mazurek, M.; Litak, J.; Kamieniak, P.; Kulesza, B.; Jonak, K.; Baj, J.; Grochowski, C. Metformin as Potential Therapy for High-Grade Glioma. Cancers 2020, 12, 210.
- Sato, A.; Sunayama, J.; Okada, M.; Watanabe, E.; Seino, S.; Shibuya, K.; Suzuki, K.; Narita, Y.; Shibui, S.; Kayama, T.; et al. Glioma-Initiating Cell Elimination by Metformin Activation of FOXO3 via AMPK. Stem Cells Transl. Med. 2012, 1, 811.
- Foretz, M.; Carling, D.; Guichard, C.; Ferre, P.; Foufelle, F. AMP-Activated Protein Kinase Inhibits the Glucose-Activated Expression of Fatty Acid Synthase Gene in Rat Hepatocytes. J. Biol. Chem. 1998, 273, 14767–14771.
- Hardie, D.G.; Ross, F.A.; Hawley, S.A. AMPK: A Nutrient and Energy Sensor That Maintains Energy Homeostasis. Nat. Rev. Mol. Cell Biol. 2012, 13, 251–262.
- Wu, Z.; Ho, W.S.; Lu, R. Targeting Mitochondrial Oxidative Phosphorylation in Glioblastoma Therapy. Neuromol. Med. 2022, 24, 18–22.
- Feng, S.W.; Chang, P.C.; Chen, H.Y.; Hueng, D.Y.; Li, Y.F.; Huang, S.M. Exploring the Mechanism of Adjuvant Treatment of Glioblastoma Using Temozolomide and Metformin. Int. J. Mol. Sci. 2022, 23, 8171.
- Öztürk, Y.; Günaydın, C.; Yalçın, F.; Nazıroğlu, M.; Braidy, N. Resveratrol Enhances Apoptotic and Oxidant Effects of Paclitaxel through TRPM2 Channel Activation in DBTRG Glioblastoma Cells. Oxid. Med. Cell. Longev. 2019, 2019, 4619865.
- Li, J.; Qin, Z.; Liang, Z. The Prosurvival Role of Autophagy in Resveratrol-Induced Cytotoxicity in Human U251 Glioma Cells. BMC Cancer 2009, 9, 215.
- Wang, L.; Long, L.; Wang, W.; Liang, Z. Resveratrol, a Potential Radiation Sensitizer for Glioma Stem Cells Both in Vitro and in Vivo. J. Pharmacol. Sci. 2015, 129, 216–225.
- McKelvey, K.J.; Wilson, E.B.; Short, S.; Melcher, A.A.; Biggs, M.; Diakos, C.I.; Howell, V.M. Glycolysis and Fatty Acid Oxidation Inhibition Improves Survival in Glioblastoma. Front. Oncol. 2021, 11, 570.
- Supabphol, S.; Seubwai, W.; Wongkham, S.; Saengboonmee, C. High Glucose: An Emerging Association between Diabetes Mellitus and Cancer Progression. J. Mol. Med. 2021, 99, 1175–1193.
- Bielecka-Wajdman, A.M.; Ludyga, T.; Smyk, D.; Smyk, W.; Mularska, M.; Świderek, P.; Majewski, W.; Mullins, C.S.; Linnebacher, M.; Obuchowicz, E. Glucose Influences the Response of Glioblastoma Cells to Temozolomide and Dexamethasone. Cancer Control 2022, 29, 1–15.
- Kenechukwu, F.C.; Isaac, G.T.; Nnamani, D.O.; Momoh, M.A.; Attama, A.A. Enhanced Circulation Longevity and Pharmacodynamics of Metformin from Surface-Modified Nanostructured Lipid Carriers Based on Solidified Reverse Micellar Solutions. Heliyon 2022, 8, e09100.
- Graham, G.G.; Punt, J.; Arora, M.; Day, R.O.; Doogue, M.P.; Duong, J.K.; Furlong, T.J.; Greenfield, J.R.; Greenup, L.C.; Kirkpatrick, C.M.; et al. Clinical Pharmacokinetics of Metformin. Clin. Pharm. 2011, 50, 81–99.
- Zhou, G.; Myers, R.; Li, Y.; Chen, Y.; Shen, X.; Fenyk-Melody, J.; Wu, M.; Ventre, J.; Doebber, T.; Fujii, N.; et al. Role of AMP-Activated Protein Kinase in Mechanism of Metformin Action. J. Clin. Investig. 2001, 108, 1167.
- Li, C.; Wang, Z.; Lei, H.; Zhang, D. Recent Progress in Nanotechnology-Based Drug Carriers for Resveratrol Delivery. Drug Deliv. 2023, 30, 2174206.
- Walle, T.; Hsieh, F.; DeLegge, M.H.; Oatis, J.E.; Walle, U.K. High Absorption but Very Low Bioavailability of Oral Resveratrol in Humans. Drug Metab. Dispos. 2004, 32, 1377–1382.
- Kafoud, A.; Salahuddin, Z.; Ibrahim, R.S.; Al-Janahi, R.; Mazurakova, A.; Kubatka, P.; Büsselberg, D. Potential Treatment Options for Neuroblastoma with Polyphenols through Anti-Proliferative and Apoptotic Mechanisms. Biomolecules 2023, 13, 563.
- Poonia, N.; Lather, V.; Narang, J.K.; Beg, S.; Pandita, D. Resveratrol-Loaded Folate Targeted Lipoprotein-Mimetic Nanoparticles with Improved Cytotoxicity, Antioxidant Activity and Pharmacokinetic Profile. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 114, 111016.
- Argenziano, M.; Ansari, I.A.; Muntoni, E.; Spagnolo, R.; Scomparin, A.; Cavalli, R. Lipid-Coated Nanocrystals as a Tool for Improving the Antioxidant Activity of Resveratrol. Antioxidants 2022, 11, 1007.
- Ohno, M.; Kitanaka, C.; Miyakita, Y.; Tanaka, S.; Sonoda, Y.; Mishima, K.; Ishikawa, E.; Takahashi, M.; Yanagisawa, S.; Ohashi, K.; et al. Metformin with Temozolomide for Newly Diagnosed Glioblastoma: Results of Phase I Study and a Brief Review of Relevant Studies. Cancers 2022, 14, 4222.





