Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Imran Ashraf | -- | 1425 | 2023-07-11 09:32:37 | | | |
| 2 | Camila Xu | Meta information modification | 1425 | 2023-07-11 09:55:11 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Shafi, I.; Fatima, A.; Afzal, H.; Díez, I.D.L.T.; Lipari, V.; Breñosa, J.; Ashraf, I. Imaging Modalities for Dental Disease Diagnosis. Encyclopedia. Available online: https://encyclopedia.pub/entry/46629 (accessed on 06 June 2026).
Shafi I, Fatima A, Afzal H, Díez IDLT, Lipari V, Breñosa J, et al. Imaging Modalities for Dental Disease Diagnosis. Encyclopedia. Available at: https://encyclopedia.pub/entry/46629. Accessed June 06, 2026.
Shafi, Imran, Anum Fatima, Hammad Afzal, Isabel De La Torre Díez, Vivian Lipari, Jose Breñosa, Imran Ashraf. "Imaging Modalities for Dental Disease Diagnosis" Encyclopedia, https://encyclopedia.pub/entry/46629 (accessed June 06, 2026).
Shafi, I., Fatima, A., Afzal, H., Díez, I.D.L.T., Lipari, V., Breñosa, J., & Ashraf, I. (2023, July 11). Imaging Modalities for Dental Disease Diagnosis. In Encyclopedia. https://encyclopedia.pub/entry/46629
Shafi, Imran, et al. "Imaging Modalities for Dental Disease Diagnosis." Encyclopedia. Web. 11 July, 2023.
Copy Citation
Dental radiography (X-ray) is the most used imaging modality by dentists to identify dental issues such as lesions, periapical pathosis, and dental restorations, and evaluate oral health.
healthcare
deep learning
image processing
medical imaging
1. Introduction
Dental radiography (X-ray) is the most used imaging modality by dentists to identify dental issues such as lesions [1][2][3][4][5][6][7], periapical pathosis [8], and dental restorations [9], and evaluate oral health [10][11][12][13][14]. The examples of imaging modalities employed by researchers for dental disease diagnosis are shown in Figure 1. Different imaging modalities have been explored and their differences are outlined in the following subsections.
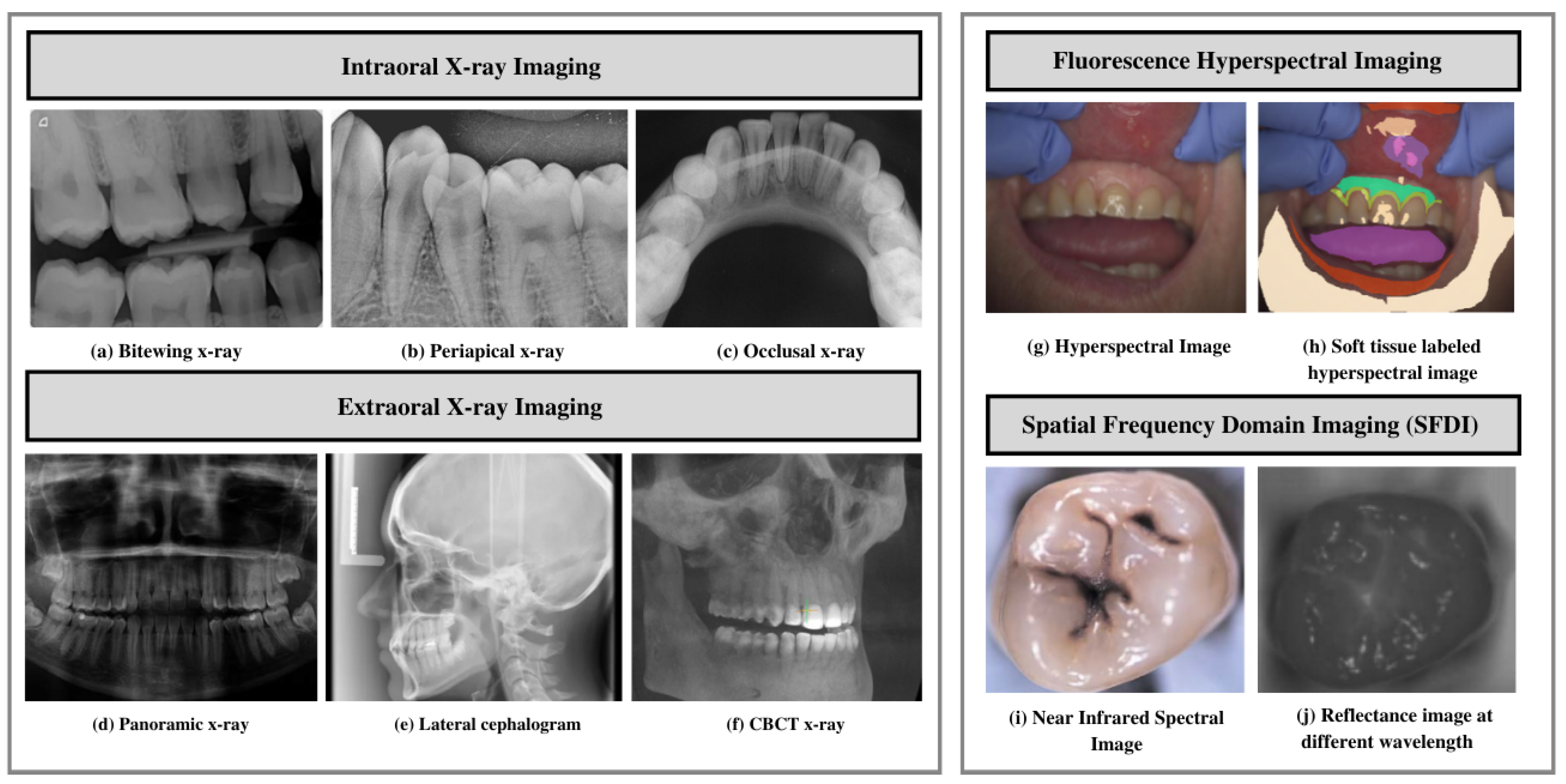
Figure 1. Imaging modalities. (a–c) Examples of different intraoral imaging. (d–f) Extraoral imaging modalities. (g–j) Near-infrared imaging. (a) Bitewing X-ray images. (b) Periapical X-rays. (c) Occlusal X-ray. (d) Panoramic X-ray. (e) Lateral cephalograms. (f) CBCT X-ray. (g,h) Fluorescence hyperspectral imaging. (i,j) Spatial frequency domain imaging.
2. X-ray Imaging Systems
To reinstate traditional photographic X-ray films, digital X-ray imaging is employed. X-ray images rely on sensors to produce enhanced images of oral structures [15]. In traditional dental disease diagnosis, the images are evaluated by dentists to identify issues such as tooth lesions and cavities, and devise treatments accordingly [16]. Dentists take several types of dental X-rays to record different mouth views. For example, for the detection of dental cavities, and to monitor mouth and teeth health, intraoral radiographs are used. In addition, dentists use extraoral radiographs to detect impacted teeth, monitor the development and growth of jaws, and identify potential problems in jaws, facial bones, and teeth.
2.1. Intraoral X-ray Imaging
Intraoral radiographs remain one of dentistry’s most widely used imaging modalities. These radiographs provide high spatial resolution images that can be used to identify dental and jawbone diseases [17]. Furthermore, these radiographs provide helpful information on bone structure and density. Paralleling and bisecting angles are two techniques to obtain an intraoral radiograph. The sensor is placed on the tooth in parallel planes, leveraging the parallel technique, exposing the radiation. The latter technique involves placing the receptor as close as possible to the tooth and exposing it to a central X-ray beam. The beam is directed perpendicularly to the imaginary line. This line allows bisecting the angle forming a long axis on the tooth and receptor plane [18]. The following are the types of intraoral radiographs used widely by dentists for dental diagnosis and treatment planning.
-
Bitewing X-ray provides a detailed account of maxillary and mandibular dental arches in a certain region of supporting bone. Bitewing radiographs aid in detecting tooth decay variations, finding dental decay, and identifying restorations.
-
Periapical X-ray portrays teeth in a full-dimensional view of one of either dental arches. The radiograph allows for detecting issues in a specific set of teeth and identifying root structure abnormalities, and detecting the surrounding bone structure.
-
Occlusal X-ray shows tooth positioning and their subsequent development in the dental arches of either the maxilla or mandible.
2.2. Extraoral X-ray Imaging
Extraoral imaging focuses on detecting dental issues in the jaw and skull. These are generally used to identify problems between the teeth, jaws, and temporomandibular joint.
-
On a single radiograph, a panoramic X-ray gives a two-dimensional view of the oral cavity including both the maxilla and mandible. These types of X-rays help identify impacted teeth and diagnose dental tumors [19].
-
Cone-beam computed tomography (CBCT) offers a substantial solution to the conventional radiography demerits. CBCT imaging is used. This type of imaging shows the interior body structures as (three-dimensional) 3-D images and enables identifying fractures and tumors in face bones. This imaging aids surgeons in avoiding after-surgery complications [22].
3. Near Infrared Imaging Systems
Near-infrared imaging is a nonionizing photo-optical method leveraged for caries detection. This imaging employs long wave radiation against tooth sides [23]. It penetrates objects deeper, thus acquiring good contrast between health and carious tissues [24][25]. This type of imaging offers certain advantages over conventional detection methods, including less radiation exposure. Furthermore, this method provides improved quality images using DIAGNOCAM [23] that transmit light through the alveolar process, including:
-
Fluorescence hyperspectral imaging system is a non-contact approach to dental tissue diagnostics. It helps degenerate raw data in a sizeable amount making it suitable for computer vision processing [26]. This imaging system combines spatial and spectral information, enabling dentists to obtain a precise optical characterization of dental issues, including dental plaque. The images are captured using a line scanning camera with 400–1000 nm spectral direction with a 5 nm sampling interval and spatial resolution of 22 μm. In addition, the hyperspectral imaging modality helps assess dental caries severity [27].
-
Spatial frequency domain imaging (SFDI) is a quantitative imaging technique [28] that enables the separation of components that are scattered and the optical absorption of a sample. This imaging modality relies on modulating project fringe patterns’ depth at varying frequencies and phases.
4. Spectral Ranges
There are different spectral bands that have been explored in dental applications.
-
Near-infrared, mid-infrared, and long-infrared: These spectral ranges provide valuable information about the chemical composition and molecular structure of dental tissues; this helps in the detection and characterization of dental lesions. Infrared is divided into three spectral regions, mainly near infrared ranging between 4000 and 14,000 cm−1, mid-infrared (MIR) ranging between 400 and 4000 cm−1, and far infrared, ranging between 25 and 400 cm−1 [29].
-
Radio frequency (RF) range: Non-ionizing radio frequency pulse with a range of frequencies is used in the presence of a controlled magnetic field for generating MRI [33]. The MRIs generated have found applications in implant dentistry, providing more precise information related to bone density, contour, and bone height [34].
5. Challenges in Automated Dental Disease Diagnosis
AI-based models have recently gained immense popularity for predicting, detecting, and diagnosing dental diseases. However, specific issues include limited data availability, accessibility, generality, lacking methodological standards, and practical issues revolving around the usefulness and standards in developing such solutions [35]. Therefore, the prime research focus remains on developing efficient and accurate systems to overcome these issues, and help dental practitioners plan treatment and prognosis. Disease classification has been researched extensively. However, certain challenges limit researchers from accomplishing similar levels of achievement in disease segmentation and treatment planning. A few of the open challenges include:
-
Limited Data Availability and Comprehensiveness: Due to data protection concerns, medical, especially dental, data is not readily accessible. Moreover, certain challenges including lack in terms of structure and relatively smaller size hinder applications of artificial intelligence techniques [35]. Thus, data availability affects the extent to which deep-learning-based approaches can be employed in this field.
-
Data Annotation: Medical data annotation requires specialized knowledge from healthcare professionals. Moreover, data labeling requires an adequate workforce and the process is cost intensive. In the absence of progressive flow and accurately annotated data, deep-learning algorithms cannot make correct interpretations and accurate predictions [36].
-
Limited Generalizability: Varying imaging characteristics lead to limited deep-learning model generalizability [37]. The underlying possible generalizability deficits must be elucidated to facilitate the development of improved modeling strategies.
-
Class Imbalance: The predominant occurrence of standard samples as compared to abnormal samples leads to class imbalance [38]. The imbalanced data lead to learning bias in the majority class.
-
External Validation: Lack of external validation leads to issues in the replication and transparency of AI-based models within dentistry. The community standards for model sharing, benchmarking, and reproducibility must be adhered to [39].
-
Interpretability: Lack in terms of interpretability and transparency makes it challenging to predict failures. Interpretability must be ensured to build a proper rapport between technology and humans, and generalize algorithms for specific tasks [40].
-
Expertise Gap: The ability to make accurate diagnoses and treatment plans relies on expertise derived from the extensive knowledge and practical experience. AI may not be able to fully replicate the nuanced decision-making that experienced clinicians possess. Bridging the gap between human expertise and AI capabilities poses a significant challenge in automated dental disease diagnosis.
-
Sensitivity and Specificity Limitations: Due to variations in image quality and anatomical structures, AI models may have limitations in achieving high sensitivity and specificity.
-
Image Interpretation Issues: The overlapping structures and presence of artifacts make interpreting dental images a daunting task. AI models should overcome these challenges to ensure accurate and reliable interpretation of dental images.
-
Variations in Pathology Presentation: Dental diseases manifest in different ways. These variations can be in terms of size, shape, or appearance. AI models are required to be able to take into account these variations accurately to provide accurate detection and classification of different pathologies.
References
- Lin, X.; Hong, D.; Zhang, D.; Huang, M.; Yu, H. Detecting Proximal Caries on Periapical Radiographs Using Convolutional Neural Networks with Different Training Strategies on Small Datasets. Diagnostics 2022, 12, 1047.
- Sornam, M.; Prabhakaran, M. A new linear adaptive swarm intelligence approach using back propagation neural network for dental caries classification. In Proceedings of the 2017 IEEE International Conference on Power, Control, Signals and Instrumentation Engineering (ICPCSI), Chennai, India, 21–22 September 2017.
- Ekert, T.; Krois, J.; Meinhold, L.; Elhennawy, K.; Emara, R.; Golla, T.; Schwendicke, F. Deep Learning for the Radiographic Detection of Apical Lesions. J. Endod. 2019, 45, 917–922.e5.
- Okada, K.; Rysavy, S.; Flores, A.; Linguraru, M.G. Noninvasive differential diagnosis of dental periapical lesions in cone-beam CT scans. Med. Phys. 2015, 42, 1653–1665.
- Chang, J.; Chang, M.F.; Angelov, N.; Hsu, C.Y.; Meng, H.W.; Sheng, S.; Glick, A.; Chang, K.; He, Y.R.; Lin, Y.B.; et al. Application of deep machine learning for the radiographic diagnosis of periodontitis. Clin. Oral Investig. 2022, 26, 6629–6637.
- Bayraktar, Y.; Ayan, E. Diagnosis of interproximal caries lesions with deep convolutional neural network in digital bitewing radiographs. Clin. Oral Investig. 2021, 26, 623–632.
- Nishitani, Y.; Nakayama, R.; Hayashi, D.; Hizukuri, A.; Murata, K. Segmentation of teeth in panoramic dental X-ray images using U-Net with a loss function weighted on the tooth edge. Radiol. Phys. Technol. 2021, 14, 64–69.
- Miki, Y.; Muramatsu, C.; Hayashi, T.; Zhou, X.; Hara, T.; Katsumata, A.; Fujita, H. Classification of teeth in cone-beam CT using deep convolutional neural network. Comput. Biol. Med. 2017, 80, 24–29.
- Abdalla-Aslan, R.; Yeshua, T.; Kabla, D.; Leichter, I.; Nadler, C. An artificial intelligence system using machine-learning for automatic detection and classification of dental restorations in panoramic radiography. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2020, 130, 593–602.
- Lee, J.H.; Han, S.S.; Kim, Y.H.; Lee, C.; Kim, I. Application of a fully deep convolutional neural network to the automation of tooth segmentation on panoramic radiographs. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2020, 129, 635–642.
- Leite, A.F.; Gerven, A.V.; Willems, H.; Beznik, T.; Lahoud, P.; Gaêta-Araujo, H.; Vranckx, M.; Jacobs, R. Artificial intelligence-driven novel tool for tooth detection and segmentation on panoramic radiographs. Clin. Oral Investig. 2020, 25, 2257–2267.
- Cantu, A.G.; Gehrung, S.; Krois, J.; Chaurasia, A.; Rossi, J.G.; Gaudin, R.; Elhennawy, K.; Schwendicke, F. Detecting caries lesions of different radiographic extension on bitewings using deep learning. J. Dent. 2020, 100, 103425.
- Chang, H.J.; Lee, S.J.; Yong, T.H.; Shin, N.Y.; Jang, B.G.; Kim, J.E.; Huh, K.H.; Lee, S.S.; Heo, M.S.; Choi, S.C.; et al. Deep Learning Hybrid Method to Automatically Diagnose Periodontal Bone Loss and Stage Periodontitis. Sci. Rep. 2020, 10, 7531.
- Lee, C.T.; Kabir, T.; Nelson, J.; Sheng, S.; Meng, H.W.; Dyke, T.E.V.; Walji, M.F.; Jiang, X.; Shams, S. Use of the deep learning approach to measure alveolar bone level. J. Clin. Periodontol. 2021, 49, 260–269.
- Dental Imaging Systems–Cameras | X-rays | Scanners-Eclipse Dental Engineering Ltd. Available online: https://eclipse-dental.com/dental-blog-articles/dental-imaging-systems-\T1\textendash–-cameras-x-rays-scanners/ (accessed on 4 August 2022).
- Dental X-Rays: Purpose, Procedure, and Risks. Available online: https://www.healthline.com/health/dental-x-rays (accessed on 4 August 2022).
- Intraoral Radiographs | FOR.org. Available online: https://www.for.org/en/treat/treatment-guidelines/edentulous/diagnostics/diagnostic-imaging/intra-oral-radiographs (accessed on 4 August 2022).
- Dental X-rays | WebMD. Available online: https://www.webmd.com/oral-health/dental-x-rays/ (accessed on 4 August 2022).
- Kim, J.; Lee, H.S.; Song, I.S.; Jung, K.H. DeNTNet: Deep Neural Transfer Network for the detection of periodontal bone loss using panoramic dental radiographs. Sci. Rep. 2019, 9, 17615.
- Helal, N.M.; Basri, O.A.; Baeshen, H.A. Significance of cephalometric radiograph in orthodontic treatment plan decision. J. Contemp. Dent. Pract. 2019, 20, 789–7793.
- Rischen, R.J.; Breuning, K.H.; Bronkhorst, E.M.; Kuijpers-Jagtman, A.M. Records needed for orthodontic diagnosis and treatment planning: A systematic review. PLoS ONE 2013, 8, e74186.
- Patel, S.; Kanagasingam, S.; Mannocci, F. Cone Beam Computed Tomography (CBCT) in Endodontics. Dent. Update 2010, 37, 373–379.
- Kühnisch, J.; Söchtig, F.; Pitchika, V.; Laubender, R.; Neuhaus, K.W.; Lussi, A.; Hickel, R. In vivo validation of near-infrared light transillumination for interproximal dentin caries detection. Clin. Oral Investig. 2015, 20, 821–829.
- Simon, J.C.; Lucas, S.A.; Staninec, M.; Tom, H.; Chan, K.H.; Darling, C.L.; Fried, D. Transillumination and reflectance probes for in vivo near-IR imaging of dental caries. In Proceedings of the Lasers in Dentistry XX, San Francisco, CA, USA, 1–6 February 2014; Rechmann, P., Fried, D., Eds.; SPIE: Bellingham, WA, USA, 2014.
- Bussaneli, D.G.; Restrepo, M.; Boldieri, T.; Pretel, H.; Mancini, M.W.; Santos-Pinto, L.; Cordeiro, R.C.L. Assessment of a new infrared laser transillumination technology (808 nm) for the detection of occlusal caries—An in vitro study. Lasers Med. Sci. 2014, 30, 1873–1879.
- Thiem, D.G.; Römer, P.; Gielisch, M.; Al-Nawas, B.; Schlüter, M.; Plaß, B.; Kämmerer, P.W. Hyperspectral imaging and artificial intelligence to detect oral malignancy–part 1-automated tissue classification of oral muscle, fat and mucosa using a light-weight 6-layer deep neural network. Head Face Med. 2021, 17, 1–9.
- Bounds, A.D.; Girkin, J.M. Early stage dental caries detection using near infrared spatial frequency domain imaging. Sci. Rep. 2021, 11, 2433.
- Urban, B.E.; Subhash, H.M. Multimodal hyperspectral fluorescence and spatial frequency domain imaging for tissue health diagnostics of the oral cavity. Biomed. Opt. Express 2021, 12, 6954.
- Moraes, L.G.P.; Rocha, R.S.F.; Menegazzo, L.M.; Araújo, E.B.d.; Yukimito, K.; Moraes, J.C.S. Infrared spectroscopy: A tool for determination of the degree of conversion in dental composites. J. Appl. Oral Sci. 2008, 16, 145–149.
- Alasiri, R.A.; Algarni, H.A.; Alasiri, R.A. Ocular hazards of curing light units used in dental practice—A systematic review. Saudi Dent. J. 2019, 31, 173–180.
- Angmar-Mansson, B.; Ten Bosh, J. Advances in methods for diagnosing coronal caries—A review. Adv. Dent. Res. 1993, 7, 70–79.
- Sadasiva, K.; Kumar, K.S.; Rayar, S.; Shamini, S.; Unnikrishnan, M.; Kandaswamy, D. Evaluation of the efficacy of visual, tactile method, caries detector dye, and laser fluorescence in removal of dental caries and confirmation by culture and polymerase chain reaction: An in vivo study. J. Pharm. Bioallied Sci. 2019, 11, S146.
- Idiyatullin, D.; Corum, C.; Moeller, S.; Prasad, H.S.; Garwood, M.; Nixdorf, D.R. Dental magnetic resonance imaging: Making the invisible visible. J. Endod. 2011, 37, 745–752.
- Niraj, L.K.; Patthi, B.; Singla, A.; Gupta, R.; Ali, I.; Dhama, K.; Kumar, J.K.; Prasad, M. MRI in dentistry-A future towards radiation free imaging–systematic review. J. Clin. Diagn. Res. 2016, 10, ZE14.
- Schwendicke, F.; Samek, W.; Krois, J. Artificial Intelligence in Dentistry: Chances and Challenges. J. Dent. Res. 2020, 99, 769–774.
- Rasmussen, C.B.; Kirk, K.; Moeslund, T.B. The Challenge of Data Annotation in Deep Learning—A Case Study on Whole Plant Corn Silage. Sensors 2022, 22, 1596.
- Krois, J.; Cantu, A.G.; Chaurasia, A.; Patil, R.; Chaudhari, P.K.; Gaudin, R.; Gehrung, S.; Schwendicke, F. Generalizability of deep learning models for dental image analysis. Sci. Rep. 2021, 11, 6102.
- Li, D.C.; Liu, C.W.; Hu, S.C. A learning method for the class imbalance problem with medical data sets. Comput. Biol. Med. 2010, 40, 509–518.
- Aittokallio, T. What are the current challenges for machine learning in drug discovery and repurposing? Expert Opin. Drug Discov. 2022, 17, 423–425.
- Shan, T.; Tay, F.; Gu, L. Application of Artificial Intelligence in Dentistry. J. Dent. Res. 2020, 100, 232–244.
More
Information
Subjects:
Primary Health Care
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.1K
Revisions:
2 times
(View History)
Update Date:
11 Jul 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





