Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sandip Basu | -- | 1741 | 2023-07-10 04:44:30 | | | |
| 2 | Rita Xu | Meta information modification | 1741 | 2023-07-10 05:26:46 | | | | |
| 3 | Rita Xu | Meta information modification | 1741 | 2023-07-12 09:55:29 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Adnan, A.; Basu, S. SSTR-based PET-CT in Neuroendocrine Tumors. Encyclopedia. Available online: https://encyclopedia.pub/entry/46586 (accessed on 04 June 2026).
Adnan A, Basu S. SSTR-based PET-CT in Neuroendocrine Tumors. Encyclopedia. Available at: https://encyclopedia.pub/entry/46586. Accessed June 04, 2026.
Adnan, Aadil, Sandip Basu. "SSTR-based PET-CT in Neuroendocrine Tumors" Encyclopedia, https://encyclopedia.pub/entry/46586 (accessed June 04, 2026).
Adnan, A., & Basu, S. (2023, July 10). SSTR-based PET-CT in Neuroendocrine Tumors. In Encyclopedia. https://encyclopedia.pub/entry/46586
Adnan, Aadil and Sandip Basu. "SSTR-based PET-CT in Neuroendocrine Tumors." Encyclopedia. Web. 10 July, 2023.
Copy Citation
Somatostatin receptor (SSTR) agonist-based Positron Emission Tomography-Computed Tomography (PET-CT) imaging is nowadays the mainstay for the assessment and diagnostic imaging of neuroendocrine neoplasms (NEN), especially in well-differentiated neuroendocrine tumors (NET) (World Health Organization (WHO) grade I and II). Major clinical indications for SSTR imaging are primary staging and metastatic workup, especially (a) before surgery, (b) detection of unknown primary in metastatic NET, (c) patient selection for theranostics and appropriate therapy, especially peptide receptor radionuclide therapy (PRRT), while less major indications include treatment response evaluation on and disease prognostication.
SSTR PET-CT
somatostatin receptor
neuroendocrine neoplasm
1. Introduction
Neuroendocrine neoplasms (NENs) are a heterogeneous group of malignant tumors derived from neural and endocrine cells; they can involve many different parts of the body, but most commonly are located in gastrointestinal tract. Gastro-entero-pancreatic neuroendocrine neoplasms (GEP-NEN) are heterogeneous group of malignant tumors, predominantly located in the intestine (small intestine more frequent that the large intestine) and pancreas, with liver being the most common metastatic site [1][2]. Most of the NENs are characterized by slow indolent growth leading to late diagnosis, high prevalence and approximately half of the cases are metastatic at diagnosis [3][4][5][6].
These are classified depending on (a) histopathological grade and proliferative (mitotic) activity, by the World Health Organization (WHO) into (A) well-differentiated grade I, II, and III neuroendocrine tumour (NET) with proliferative index and mitotic activity (< 3%, <2%; 3 to 20%, 2 to 20% and >20%, respectively) and (B) poorly-differentiated neuroendocrine carcinoma (NEC) with mitotic and Ki-67 indices >20% and poorly differentiated histology. The 2019 WHO classification of NET has also divided NENs as NET and NEC based on genetic mutations: mutations in MEN1, DAXX, and ATRX are entity-defining for well-differentiated NETs, whereas NECs usually have TP53 or RB1 mutations [7][8]; (b) functioning status depending upon secretion or non-secretion of peptide hormones as functioning and non-functioning NENs; and (c) embryological origin as foregut, mid-gut, and hind-gut derivatives.
The diagnosis of NET is characterized by detection of immunohistochemical markers—synaptophysin, chromogranin A, and neuron-specific enolase. Over the years, the role of nuclear medicine has grown to play a central role in the diagnosis of NEN after the identification of somatostatin (SST) in 1973 and 5 types of somatostatin receptors (SSTRs 1 to 5) in the early 1990s. The discovery of somatostatin receptors has opened up new avenues for diagnosis, staging, and treatment of NENs, especially the well-differentiated type. Somatostatin-receptor-based imaging using synthetic somatostatin agonists (SSA) was started in 1994 with [111In] In-pentetreotide (Octreoscan) being the first Food and Drug Association (FDA) approved and commercially marketed radiopharmaceutical [9][10][11][12]. Although there was wide acceptance of Octreoscan for NEN imaging, the radiopharmaceutical had many limitations—less favorable tumor-to-background ratio, moderate affinity for receptors, and high gamma energy causing more background noise and high radiation absorbed dose to the patient. To a great extent, these limitations have been alleviated with the advent of the next generation of SSA labelled with positron emitter radio-metal [68Ga] to be used with PET-CT [13][14]. Recent advancements in the detection and mapping of SSTR expression in vivo has opened avenues for targeting the same for therapeutic benefits and personalized management. After securing FDA approval in January 2018, peptide receptor radionuclide therapy (PRRT) using Lutetium-177 DOTA-TATE ([177Lu]Lu-DOTA-TATE) has gained widespread acceptance as one of the frontline treatments in metastatic/inoperable neuroendocrine tumors (NET).
2. Radiopharmaceuticals for SSTR-PET Imaging
2.1. Somatostatin Receptor Agonists
Somatostatin receptors are G-protein coupled receptors (GPCR) binding to somatostatin neuropeptides, a paracrine secreted by gastro-intestinal and brain cells. Presently, various types of somatostatin agonists and few antagonists are available for clinical and/or experimental use. The common radio-pharmaceuticals for clinical use are–[68Ga]Ga-DOTA-Tyr3-Octreotate (DOTA-TATE), [68Ga]Ga-DOTA-Phe1-Tyr3-Octreotide (DOTA-TOC), and [68Ga]Ga-DOTA-NaI3-Octreotide (DOTA-NOC). These three radiopharmaceuticals differ slightly in their pharmacokinetic properties, mainly due to different affinities for SSTR subtypes; while DOTA-TATE is SSTR 2 specific, DOTA-NOC has affinity towards SSTR 2, 3, and 5 and DOTA-TOC has affinity towards SSTR 2 > 5 [15][16][17][18]. Despite different receptor affinity, there is no clinically significant difference and there are ample data to support high accuracy of SSTR PET-CT for detecting lesions as compared to conventional imaging and somatostatin-receptor scintigraphy [19][20][21][22][23][24][25].
[18F]-Fluorine (18F) labelled radio pharmaceuticals are recently being developed with the following advantages: long half-life, no need of in-house generators or cyclotron, and relatively low positron energy which leading to better spatial resolution than [68Ga]. Al [18F]F-NOTA-Octreotide is one recent promising radiopharmaceutical demonstrating high affinity for SSTR2, favorable biodistribution, high tumor uptake, better spatial resolution, and is proven to be safe for clinical applications [5][26][27][28][29][30][31].
[64Cu]Cu-DOTATATE is a cyclotron produced positron emitter that can be manufactured on a large scale, yields similar detection rates as [68Ga]-based SSTR-PET agents and has better pharmacokinetic properties such as (i) longer half-life (~12.7 h), (ii) relatively low positron energy (0.65 vs. 1.9 MeV) leading to shorter positron range (mean −0.56 vs. 3.5 mm), and (iii) higher spatial resolution enabling better detection of smaller lesions. [64Cu] hence permits delayed serial imaging with important implications for personalized dosimetry in peptide receptor radionuclide therapy (PRRT) and radio-guided surgery using a hand-held positron probe [32][33].
2.2. Somatostatin Receptor Antagonists
In opposition to the general belief that agonists will be more suitable as an imaging agent since they are internalized, a recent in vitro study using SSTR-3 antagonist has demonstrated that the antagonist detected 76-fold more sites of binding as against SSTR-3 agonist [34]. Few recent studies have shown that radiolabeled SSTR antagonists produce superior images than radiolabeled SSTR agonists [34][35]. Recently, few studies with small number of NET patients demonstrated radiolabeled SSTR-2 antagonists, e.g., [111In]In-DOTA-BASS and [68Ga]Ga-OPS202 ([68Ga]Ga-NODAGA-JR11) have demonstrated superior images and higher sensitivity compared to radiolabeled SSTR-2 agonists [34][35][36][37][38]. This led to the opinion that [177Lu] labelled antagonist in PRRT may be utilized instead of [177Lu]-labelled agonists. [177Lu]Lu-DOTA-JR11 provided 1.7-to-10.6-fold higher tumor uptake as compared to agonists, resulting in partial remission in half of the enrolled patients [35].
2.3. Appropriate Use Criteria
Appropriateness use criteria (AUC) defines set of scenarios finalized by the representatives of international societies concerned with management of NETs. The process of determining AUC was modelled after RAND/UCLA appropriateness method including a list of common scenarios encountered in NET management, a systematic review of evidence related to these scenarios and development of an appropriateness score for each scenario using a modified Delphi process [39][40]. The workgroup identified 12 scenarios for patients with NETs and scored each scenario as “appropriate”, “may be appropriate”, or “rarely appropriate” on a scale from 1 to 9; where 7 to 9 are appropriate, 4 to 6 are considered may be appropriate, and 1 to 3 indicate that the use is rarely appropriate and is not considered acceptable.
- (a)
-
Appropriate: Initial staging after histologic diagnosis of NET-9; localization of primary tumor in patients with known metastatic NET but unknown primary-9; selection of patients for SSTR targeting PRRT-9; staging NETs before planned surgery-8; evaluation of mass suggestive of NET and not amenable to endoscopic or percutaneous biopsy-8; monitoring of NETs seen predominantly on SSTR PET-8; evaluation of patients with biochemical evidence and symptoms of NET without evidence on conventional imaging (CI) and without prior histological diagnosis of NET-7; restaging at the time of clinical or laboratory progression without progression on CI-7; and new indeterminate lesion on CI with unclear progression-7.
- (b)
-
May be appropriate: Restaging of patients with NETs at initial follow up after resection with curative intent-6; selection of patients with non-functional NETs for SSA treatment-6; and monitoring in patients with NETs seen on both CI and SSTR PET with active disease and no clinical evidence of progression-6.
2.4. Clinical Utility of SSTR-PET
Detection and Initial Staging: When compared with conventional imaging, SSTR-PET offers multiple advantages in terms of detection rate of primary and metastatic disease and change in management (observed in 44% upfront and in 9% of patients who have undergone previous somatostatin receptor scintigraphy with Octreoscan) ([41], Figure 1). Bauckneht et al., in their review and meta-analysis including 1143 patients of pancreatic NET, demonstrated a pooled sensitivity and specificity of 79.6% (95% CI—71 to 87%) and 95% (95% CI—75 to 100%), respectively; heterogeneity of 59.6% and 51.5%, and on a per patient and per lesion basis, the pooled detection rates of primary lesion were 81% (95% CI—65 to 90%) and 92% (95% CI—80 to 97%), respectively [42]. Geijer and Breimer, in their meta-analysis on 2015 patients, demonstrated pooled sensitivity of 93% (95% CI—91 to 94%) [43]. Despite theoretical advantage, conflicting results were observed in detection with contrast enhanced vs. non-enhanced PET-CT for the detection of primary and metastatic disease. While Kazmierczak et al. showed 50% and 30% improvement in sensitivity and accuracy, Mayerhoefer et al. found only moderate improvement in sensitivity and hardly any change in specificity [44][45]. Similarly, combining PET and MRI has theoretical advantage of high soft tissue contrast for MRI and metabolic data from PET.
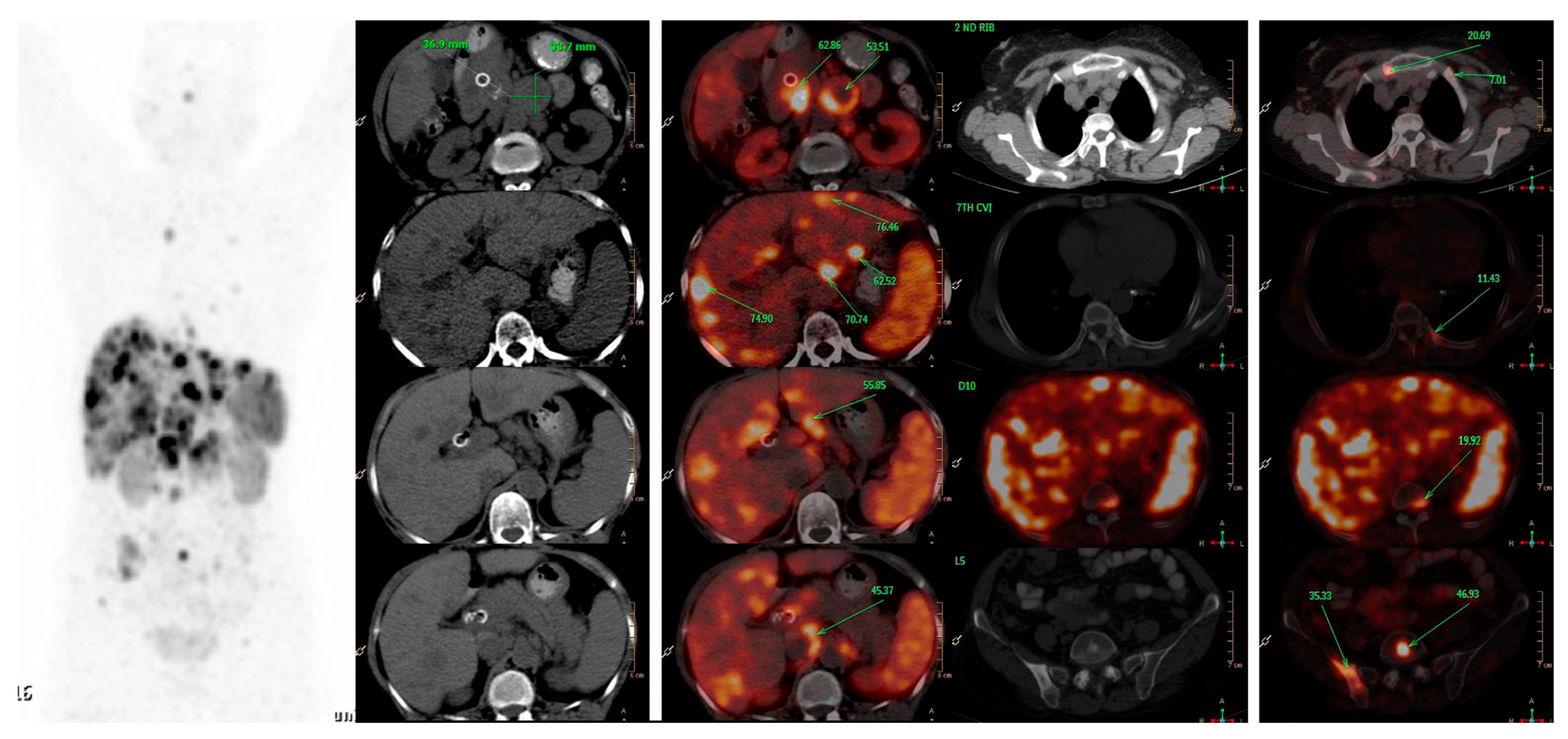
Figure 1. A 66-years-old female patient, presented with features of obstructive jaundice. Triphasic CECT scan abdomen showed large hypodense SOL in head of pancreas encasing CBD with significant upstream dilatation of biliary tree with multiple hypodense bilobar hepatic metastases and multiple abdominal & retroperitoneal lymph nodes. Patient was referred for PRRT and 68Ga-DOTATATE PET-CT was done which confirmed above-mentioned triphasic CE-CT scan findings and showed many new skeletal and marrow lesions at multiple skeletal sites. Hence is SSTR-based PET-CT represents a better modality for metastatic workup than conventional imaging.
Detection of unknown primary: Carcinoma of unknown primary (CUP) accounts for 3 to 5% of all malignancies and is divided into following subtypes based on histological subtypes: adenocarcinoma (80 to 85%), squamous cell carcinoma (5 to 10%), and neuroendocrine tumor (2 to 4%) [46]. Neuroendocrine tumors of unknown primary (CUP-NETs) are primary tumors with undetermined origin among metastatic NETs and accounts for 11 to 22% of NETs ([47][48]; Figure 2). Ma et al., in their meta-analysis of 484 patients, demonstrated pooled sensitivity and specificity of SSTR imaging in identifying CUP-NETs as 82% and 55%, respectively [49]. The area under the receiver operating curve (ROC) was 69% and pooled detection rate for CUP-NETs was 61%. SSTR PET-CT identified most metastases in liver (57.9%) followed by lymph nodes (22.8%), bones (12.8%), lung (2.8%), and others (1.7%). Sampathirao et al., in their study on 51 CUP-NET patients using “dual tracer” PET-CT with [68Ga]GA-DOTATATE and [18F]-FDG, demonstrated sensitivity of 60.78%, whereas overall lesion detection sensitivity was 96.87% [50]. Delpassand et al., in their study, found [64Cu]Cu-DOTATATE to be an effective radiopharmaceutical in detecting NET lesions with sensitivity and specificity of 90.9% and 96.6%, respectively [32].
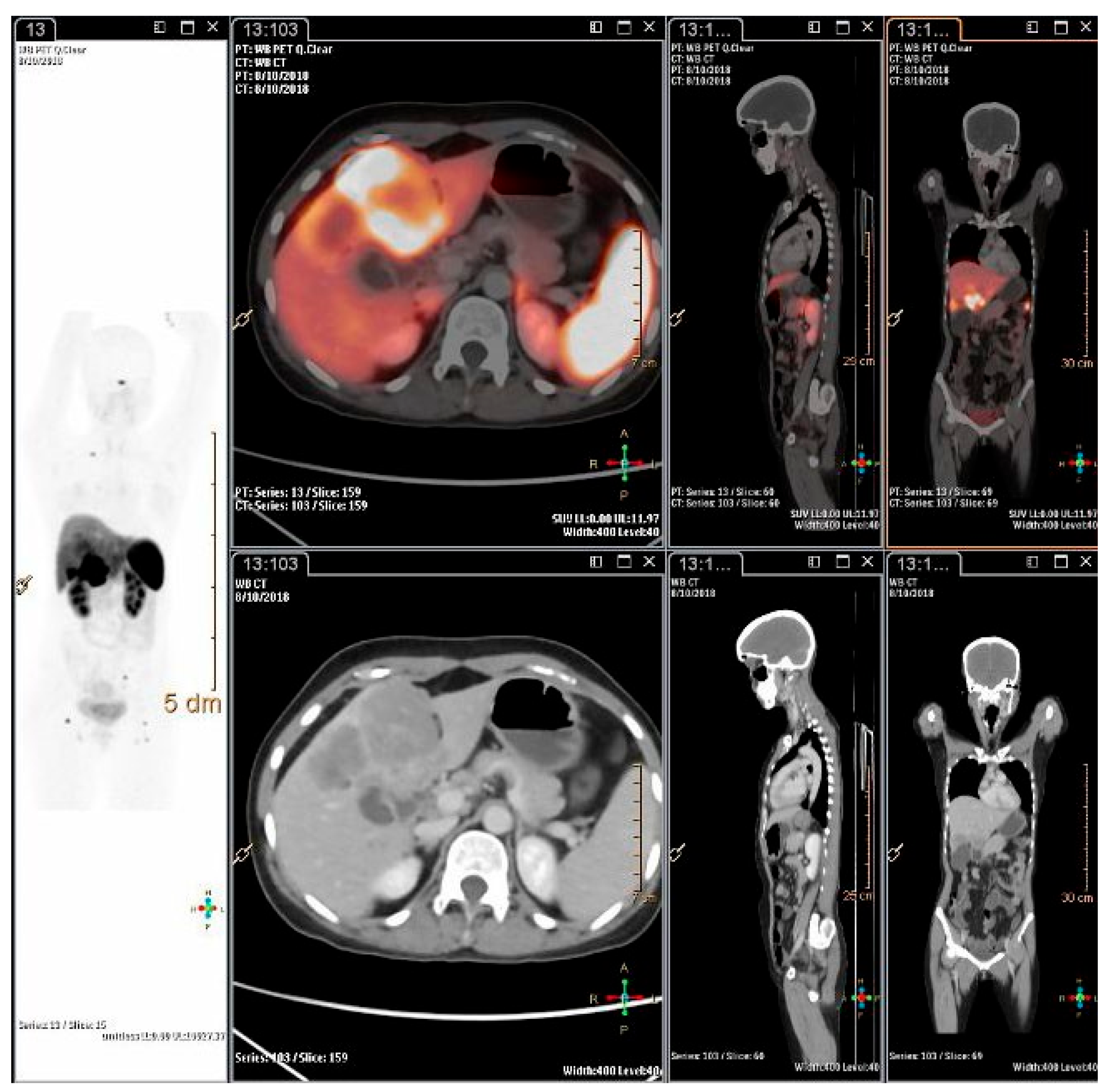
Figure 2. A 26-years-old lady, presented with pain abdomen and was evaluated for the same. CECT abdomen showed multiple liver lesions and abdominal lymph nodes. Biopsy and IHC from liver lesion showed high grade neuroendocrine carcinoma, large cell variant (Ki-67 index 80%). She was referred for opinion for PRRT. 68Ga-DOTATATE PET-CT scan showed intensely SSTR expressing polypoidal gall bladder mass infiltrating into inferior surface of liver with SSTR expressing multiple liver lesions, abdomino-pelvic lymph nodes and SSTR expressing lesion in head of right femur.
References
- Hofland, J.; Kaltsas, G.; de Herder, W.W. Advances in the diagnosis and management of well-differentiated neuroendocrine neoplasms. Endocr. Rev. 2019, 41, 371–403.
- Mignon, M. Natural history of neuroendocrine entero-pancreatic tumors. Digestion 2000, 62, 51–58.
- Yao, J.C.; Hassan, M.; Phan, A.; Dagohoy, C.; Leary, C.; Mares, J.E.; Abdalla, E.K.; Fleming, J.B.; Vauthey, J.-N.; Rashid, A.; et al. One hundred years after “carcinoid”: Epidemiology of and prognostic factors for neuroen-docrine tumors in 35, 825 cases in the United States. J. Clin. Oncol. 2008, 26, 3063–3072.
- Modlin, I.M.; Oberg, K.; Chung, D.C.; Jensen, R.T.; de Herder, W.W.; Thakker, P.R.V.; Caplin, M.; Fave, P.G.D.; Kaltsas, G.A.; Krenning, P.E.P.; et al. Gastroenteropancreatic neuroendocrine tumours. Lancet Oncol. 2008, 9, 61–72.
- Hallet, J.; Law, C.H.L.; Cukier, M.; Saskin, R.; Liu, N.; Singh, S. Exploring the rising incidence of neuroendocrine tumors: A population-based analysis of epidemiology, metastatic presentation, and outcomes. Cancer 2014, 121, 589–597.
- Sackstein, P.E.; O’Neil, D.S.; Neugut, A.I.; Chabot, J.; Fojo, T. Epidemiologic trends in neuroendocrine tumors: An examination of incidence rates and survival of specific patient subgroups over the past 20 years. Semin. Oncol. 2018, 45, 249–258.
- Rindi, G.; Klimstra, D.S.; Abedi-Ardekani, B.; Asa, S.L.; Bosman, F.T.; Brambilla, E.; Busam, K.J.; de Krijger, R.R.; Dietel, M.; El-Naggar, A.K.; et al. A common classification framework for neuroendocrine neoplasms: An Interna-tional Agency for Research on Cancer (IARC) and World Health Organization (WHO) expert consensus proposal. Mod. Pathol. 2018, 31, 1770–1786.
- Klimstra, D.S.; Klöppel, G.; La Rosa, S.; Rindi, G. Classification of neuroendocrine neoplasms of the digestive system. In WHO Classification of Tumours, 5th ed.; Digestive System Tumours; IARC: Lyon, France, 2019; pp. 16–19.
- Patel, Y.C. Somatostatin and Its Receptor Family. Front. Neuroendocr. 1999, 20, 157–198.
- Weckbecker, G.; Lewis, I.; Albert, R.; Schmid, H.A.; Hoyer, D.; Bruns, C. Opportunities in somatostatin research: Biological, chemical and therapeutic aspects. Nat. Rev. Drug Discov. 2003, 2, 999–1017.
- Krenning, E.; Breeman, W.; Kooij, P.; Lameris, J.; Bakker, W.; Koper, J.; Ausema, L.; Reubi, J.; Lamberts, S. Localisation of Endocrine-Related Tumours with Radioiodinated Analogue of Somatostatin. Lancet 1989, 333, 242–244.
- Heiman, M.L.; Murphy, W.A.; Coy, D.H. Differential binding of somatostatin agonists to somatostatin receptors in brain and ade-nohypophysis. Neuroendocrinology 1987, 45, 429–436.
- Eychenne, R.; Bouvry, C.; Bourgeois, M.; Loyer, P.; Benoist, E.; Lepareur, N. Overview of Radiolabeled Somatostatin Analogs for Cancer Imaging and Therapy. Molecules 2020, 25, 4012.
- Mikołajczak, R.; Maecke, H.R. Radiopharmaceuticals for somatostatin receptor imaging. Nucl. Med. Rev. 2016, 19, 126–132.
- Virgolini, I.; Ambrosini, V.; Bomanji, J.B.; Baum, R.P.; Fanti, S.; Gabriel, M.; Papathanasiou, N.D.; Pepe, G.; Oyen, W.; De Cristoforo, C.; et al. Procedure guidelines for PET/CT tumour imaging with 68Ga-DOTA-conjugated peptides: 68Ga-DOTA-TOC, 68Ga-DOTA-NOC, 68Ga-DOTA-TATE. Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 2004–2010.
- Hofmann, M.; Maecke, H.; Börner, A.; Weckesser, E.; Schöffski, P.; Oei, M.; Schumacher, J.; Henze, M.; Heppeler, A.; Meyer, J.; et al. Biokinetics and imaging with the somatostatin receptor PET radioligand 68Ga-DOTATOC: Preliminary data. Eur. J. Nucl. Med. 2001, 28, 1751–1757.
- Hofman, M.S.; Lau, W.F.; Hicks, R.J. Somatostatin receptor imaging with 68Ga DOTATATE PET/CT: Clinical utility, normal patterns, pearls, and pitfalls in interpretation. Radiographics 2015, 35, 500–516.
- Wild, D.; Mäcke, H.R.; Waser, B.; Reubi, J.C.; Ginj, M.; Rasch, H.; Müller-Brand, J.; Hofmann, M. 68Ga-DOTANOC: A first compound for PET imaging with high affinity for somatostatin receptor subtypes 2 and 5. Eur. J. Nucl. Med. Mol. Imaging 2004, 32, 724.
- Poeppel, T.D.; Binse, I.; Petersenn, S.; Lahner, H.; Schott, M.; Antoch, G.; Brandau, W.; Bockisch, A.; Boy, C. 68Ga-DOTATOC versus 68Ga-DOTATATE PET/CT in functional imaging of neuroendocrine tumors. J. Nucl. Med. 2011, 52, 1864–1870.
- Bodei, L.; Ambrosini, V.; Herrmann, K.; Modlin, I. Current Concepts in (68)Ga-DOTATATE Imaging of Neuroendocrine Neoplasms: Interpretation, Biodistribution, Dosimetry, and Molecular Strategies. J. Nucl. Med. 2017, 58, 1718–1726.
- Bozkurt, M.F.; Virgolini, I.; Balogova, S.; Beheshti, M.; Rubello, D.; Decristoforo, C.; Ambrosini, V.; Kjaer, A.; Delgado-Bolton, R.; Kunikowska, J.; et al. Guideline for PET/CT imaging of neuroendocrine neoplasms with 68Ga-DOTA-conjugated somatostatin receptor targeting peptides and 18F-DOPA. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1588–1601.
- Treglia, G.; Sadeghi, R.; Giovinazzo, F.; Galiandro, F.; Annunziata, S.; Muoio, B.; Kroiss, A.S. PET with Different Radiopharmaceuticals in Neuroendocrine Neoplasms: An Umbrella Review of Published Meta-Analyses. Cancers 2021, 13, 5172.
- Graham, M.M.; Gu, X.; Ginader, T.; Breheny, P.; Sunderland, J.J. 68Ga-DOTATOC Imaging of Neuroendocrine Tumors: A Systematic Review and Metaanalysis. J. Nucl. Med. 2017, 58, 1452–1458.
- Gabriel, M.; Decristoforo, C.; Kendler, D.; Dobrozemsky, G.; Heute, D.; Uprimny, C.; Kovacs, P.; Von Guggenberg, E.; Bale, R.; Virgolini, I.J. 68Ga-DOTA-Tyr3-Octreotide PET in Neuroendocrine Tumors: Comparison with Somatostatin Receptor Scintigraphy and CT. J. Nucl. Med. 2007, 48, 508–518.
- Putzer, D.; Gabriel, M.; Henninger, B.; Kendler, D.; Uprimny, C.; Dobrozemsky, G.; Decristoforo, C.; Bale, R.J.; Jaschke, W.; Virgolini, I.J. Bone Metastases in Patients with Neuroendocrine Tumor: 68Ga-DOTA-Tyr3-Octreotide PET in Comparison to CT and Bone Scintigraphy. J. Nucl. Med. 2009, 50, 1214–1221.
- Hou, J.; Long, T.; He, Z.; Zhou, M.; Yang, N.; Chen, D.; Zeng, S.; Hu, S. Evaluation of (18)F-AlF-NOTA octreotide for imaging neuro-endocrine neoplasms: Comparison with (68)Ga-DOTATATE PET/CT. EJNMMI Res. 2021, 11, 55.
- Long, T.; Yang, N.; Zhou, M.; Chen, D.; Li, Y.; Li, J.; Tang, Y.; Liu, Z.; Li, Z.; Hu, S. Clinical Application of 18F-AlF-NOTA-Octreotide PET/CT in Combination With 18F-FDG PET/CT for Imaging Neuroendocrine Neoplasms. Clin. Nucl. Med. 2019, 44, 452–458.
- Waldmann, C.M.; Stuparu, A.D.; van Dam, R.M.; Slavik, R. The Search for an Alternative to Ga-DOTA-TATE in Neuroendocrine Tumor Theranostics: Current State of 18F-labeled Somatostatin Analog Development. Theranostics 2019, 9, 1336–1347.
- Laverman, P.; McBride, W.J.; Sharkey, R.M.; Eek, A.; Joosten, L.; Oyen, W.J.; Goldenberg, D.M.; Boerman, O.C. A Novel Facile Method of Labeling Octreotide with 18F-Fluorine. J. Nucl. Med. 2010, 51, 454–461.
- Laverman, P.; D’souza, C.A.; Eek, A.; McBride, W.J.; Sharkey, R.M.; Oyen, W.J.G.; Goldenberg, D.M.; Boerman, O.C. Optimized labeling of NOTA-conjugated octreotide with F-18. Tumor Biol. 2011, 33, 427–434.
- Leyton, J.; Iddon, L.; Perumal, M.; Indrevoll, B.; Glaser, M.; Robins, E.; George, A.J.T.; Cuthbertson, A.; Luthra, S.K.; Aboagye, E.O. Targeting somatostatin receptors: Preclinical evaluation of novel 18F fluoroethyltria-zole-Tyr3-octreotate analogs for PET. J. Nucl. Med. 2011, 52, 1441–1448.
- Delpassand, E.S.; Ranganathan, D.; Wagh, N.; Shafie, A.; Gaber, A.; Abbasi, A.; Kjaer, A.; Tworowska, I.; Núñez, R. 64Cu-DOTATATE PET/CT for Imaging Patients with Known or Suspected Somatostatin Receptor-Positive Neuroendocrine Tumors: Results of the First U.S. Prospective, Reader-Masked Clinical Trial. J. Nucl. Med. 2020, 61, 890–896.
- Sadowski, S.M.; Millo, C.; Neychev, V.; Aufforth, R.; Keutgen, X.; Glanville, J.; Alimchandani, M.; Nilubol, N.; Herscovitch, P.; Quezado, M.; et al. Feasibility of radio-guided surgery with 68gallium DOTATATE in patients with gas-tro-entero-pancreatic neuroendocrine tumors. Ann. Surg. Oncol. 2015, 22, 676–682.
- Wild, D.; Fani, M.; Fischer, R.; Del Pozzo, L.; Kaul, F.; Krebs, S.; Fischer, R.; Rivier, J.E.F.; Reubi, J.C.; Maecke, H.R.; et al. Comparison of Somatostatin Receptor Agonist and Antagonist for Peptide Receptor Radionuclide Therapy: A Pilot Study. J. Nucl. Med. 2014, 55, 1248–1252.
- Wild, D.; Fani, M.; Behe, M.; Brink, I.; Rivier, J.E.; Reubi, J.C.; Maecke, H.R.; Weber, W.A. First Clinical Evidence That Imaging with Somatostatin Receptor Antagonists Is Feasible. J. Nucl. Med. 2011, 52, 1412–1417.
- Fani, M.; Nicolas, G.P.; Wild, D. Somatostatin Receptor Antagonists for Imaging and Therapy. J. Nucl. Med. 2017, 58 (Suppl. 2), 61S–66S.
- Nicolas, G.P.; Schreiter, N.; Kaul, F.; Uiters, J.; Bouterfa, H.; Kaufmann, J.; Erlanger, T.E.; Cathomas, R.; Christ, E.; Fani, M.; et al. Sensitivity Comparison of Ga OPS202 and Ga-DOTATOC PET/CT in Patients with Gastroenteropancreatic Neuroendocrine Tumors: A Prospective Phase II Imaging Study. J. Nucl. Med. 2018, 59, 915–921.
- Nicolas, G.P.; Beykan, S.; Bouterfa, H.; Kaufmann, J.; Bauman, A.; Lassmann, M.; Reubi, J.C.; Rivier, J.E.F.; Maecke, H.R.; Fani, M.; et al. Safety, Biodistribution, and Radiation Dosimetry of Ga-OPS202 in Patients with Gastroenteropancreatic Neuroendocrine Tumors: A Prospective Phase I Imaging Study. J. Nucl. Med. 2018, 59, 909–914.
- Hope, T.A.; Bergsland, E.K.; Bozkurt, M.F.; Graham, M.; Heaney, A.P.; Herrmann, K.; Howe, J.; Kulke, M.H.; Kunz, P.L.; Mailman, J.; et al. Appropriate Use Criteria for Somatostatin Receptor PET Imaging in Neuroendocrine Tumors. J. Nucl. Med. 2017, 59, 66–74.
- Hendel, R.C.; Patel, M.R.; Allen, J.M.; Min, J.K.; Shaw, L.J.; Wolk, M.J.; Douglas, P.S.; Kramer, C.M.; Stainback, R.F.; Bailey, S.R.; et al. Appropriate use of cardiovascular technology: 2013 ACCF appropriate use criteria methodology update: A report of the American College of Cardiology Foundation appropriate use criteria task force. J. Am. Coll. Cardiol. 2013, 61, 1305–1317.
- Barrio, M.; Czernin, J.; Fanti, S.; Ambrosini, V.; Binse, I.; Du, L.; Eiber, M.; Herrmann, K.; Fendler, W.P. The Impact of Somatostatin Receptor–Directed PET/CT on the Management of Patients with Neuroendocrine Tumor: A Systematic Review and Meta-Analysis. J. Nucl. Med. 2017, 58, 756–761.
- Bauckneht, M.; Albano, D.; Annunziata, S.; Santo, G.; Guglielmo, P.; Frantellizzi, V.; Branca, A.; Ferrari, C.; Vento, A.; Mirabile, A.; et al. Somatostatin Receptor PET/CT Imaging for the Detection and Staging of Pancreatic NET: A Systematic Review and Meta-Analysis. Diagnostics 2020, 10, 598.
- Geijer, H.; Breimer, L.H. Somatostatin receptor PET/CT in neuroendocrine tumours: Update on systematic review and me-ta-analysis. Eur. J. Nucl. Med. Mol. Imaging 2013, 40, 1770–1780.
- Kazmierczak, P.M.; Rominger, A.; Wenter, V.; Spitzweg, C.; Auernhammer, C.; Angele, M.K.; Rist, C.; Cyran, C.C. The added value of 68Ga-DOTA-TATE-PET to contrast-enhanced CT for primary site detection in CUP of neuroendocrine origin. Eur. Radiol. 2016, 27, 1676–1684.
- Mayerhoefer, M.E.; Schuetz, M.; Magnaldi, S.; Weber, M.; Trattnig, S.; Karanikas, G. Are contrast media required for (68)Ga-DOTATOC PET/CT in patients with neuroendocrine tumours of the abdomen? Eur. Radiol. 2012, 22, 938–946.
- Oien, K.A. Pathologic Evaluation of Unknown Primary Cancer. Semin. Oncol. 2009, 36, 8–37.
- Pavel, M.; O’Toole, D.; Costa, F.; Capdevila, J.; Gross, D.; Kianmanesh, R.; Krenning, E.; Knigge, U.; Salazar, R.; Pape, U.-F.; et al. ENETS Consensus Guidelines Update for the Management of Distant Metastatic Disease of Intestinal, Pancreatic, Bronchial Neuroendocrine Neoplasms (NEN) and NEN of Unknown Primary Site. Neuroendocrinology 2016, 103, 172–185.
- Alexandraki, K.; Angelousi, A.; Boutzios, G.; Kyriakopoulos, G.; Rontogianni, D.; Kaltsas, G. Management of neuroendocrine tumors of unknown primary. Rev. Endocr. Metab. Disord. 2017, 18, 423–431.
- Ma, H.; Kan, Y.; Yang, J.-G. Clinical value of 68Ga-DOTA-SSTR PET/CT in the diagnosis and detection of neuroendocrine tumors of unknown primary origin: A systematic review and meta-analysis. Acta Radiol. 2020, 62, 1217–1228.
- Sampathirao, N.; Basu, S. MIB-1 Index–Stratified Assessment of Dual-Tracer PET/CT with 68Ga-DOTATATE and 18F-FDG and Multimodality Anatomic Imaging in Metastatic Neuroendocrine Tumors of Unknown Primary in a PRRT Workup Setting. J. Nucl. Med. Technol. 2017, 45, 34–41.
More
Information
Subjects:
Neuroimaging
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
689
Revisions:
3 times
(View History)
Update Date:
12 Jul 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





