The internet of things (IoT) plays a vital role in smart drug delivery systems. It is an emerging field that offers promising solutions for improving the efficacy, safety, and patient compliance of drug therapies. IoT-based drug delivery systems leverage advanced devices, sophisticated sensors, and smart tools to monitor and analyse the health matrices of the patient in real-time, allowing for personalised and targeted drug delivery. This technology is implemented through various types of devices, including wearable and implantable devices such as infusion pumps, smart pens, inhalers, and auto-injectors. However, the development and implementation of IoT-based drug delivery systems pose several challenges, such as ensuring data security and privacy, regulatory compliance, compatibility, and reliability.
1. Introduction
A drug delivery system refers to the method and route by which a drug is delivered to the site where it will act in the body to show its treatment efficacy. Effective drug delivery requires a multidisciplinary approach and a comprehensive understanding of the drug’s properties and the targeted site. However, the lack of targeted delivery limits its efficacy and bioavailability
[1]. A collaborative effort between pharmacologists, biologists, chemists, and engineers is required to optimise drug delivery and disease detection
[2]. Drug delivery systems comprise various technologies and strategies that are formulated with the objective of conveying therapeutic agents to their targeted site of action within the human body. These systems employ diverse strategies and pathways of administration that are customised to suit the distinctive requirements of the medication and the targeted therapeutic region. Oral delivery, through tablets, capsules, or liquids, is the most common and convenient method. Injectable delivery allows for precise dosage control and rapid absorption when administered subcutaneously, intramuscularly, intravenously, or intra-arterially. Topical delivery involves applying medications to the skin or mucous membranes, while inhalation delivery is through aerosols, gases, or powders for direct respiratory system delivery. Transdermal delivery uses patches or devices for consistent drug release. Implantable delivery involves surgical placement for long-term release. Targeted delivery systems accumulate drugs at specific sites, such as tumours or the brain. Smart drug delivery is an innovative approach that amalgamates advanced devices, state-of-the-art sensors, and sophisticated software into drug delivery systems, aiming to optimise drug efficacy, safety, and patient compliance
[3]. Such systems are designed explicitly to release medication automatically based on a patient’s health status, thus mitigating human error and ensuring optimal dosing at the right time
[4]. It enables personalised medicine by monitoring the patient’s health status, allowing for tailored drug dosage and release. Systems can improve medication compliance and reduce the burden of managing complex regimens, a challenge for many patients
[5].
2. Internet of Things and Healthcare IoT
IoT, an acronym for “internet of things”, is a network of physical technologies that use in-built sensors, software, and connectivity to exchange data. It allows devices to communicate and perform tasks without human intervention. IoT can revolutionise industries by improving efficiency, automating tasks, and providing novel data analysis. IoT-based drug delivery systems optimise efficiency, accuracy, and safety through smart devices, remote control, and real-time data analysis
[6]. Wireless drug delivery, also known as wireless drug administration, refers to a technology-based method for delivering drugs or medications to a patient without the need for physical connections or wires. This can be accomplished through the use of implantable devices, such as pumps or ports, that can receive commands from a remote control or other device to dispense the appropriate amount of medication
[7].
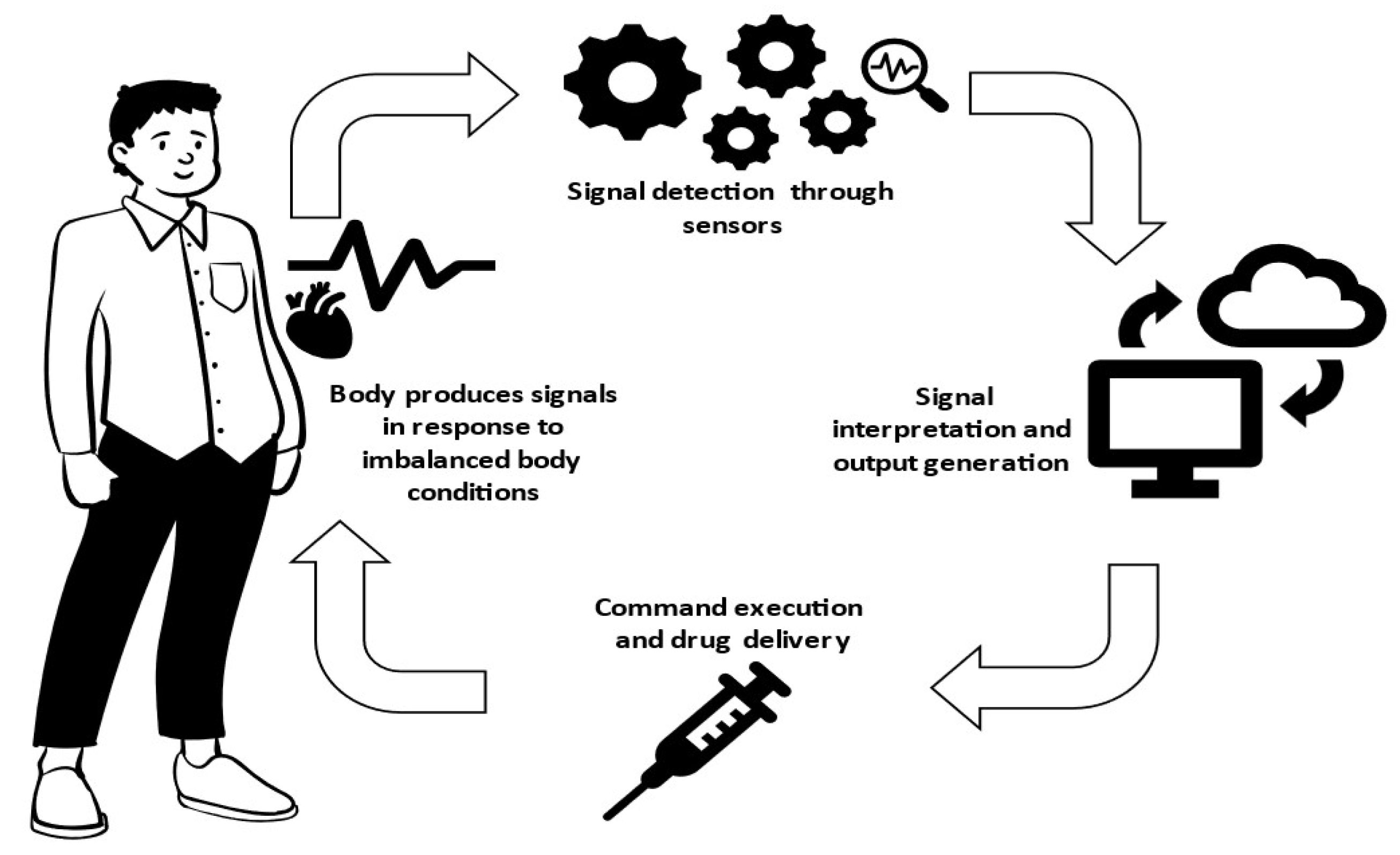
Figure 1 shows the process of drug delivery on demand based on IoT. Such devices offer a promising solution to enhance healthcare facilities’ operations by ensuring accurate and timely drug administration, continuous monitoring of patients, and improving patient outcomes
[8]. They enable physicians to focus on other critical tasks, provide valuable data for informed decisions, and enhance the quality of healthcare.
Figure 1. Process of drug delivery on demand based on IoT.
2.1. Healthcare IoT
Healthcare IoT, or the internet of things in healthcare, is the integration of connected technology in the medical industry to collect and exchange data for improved patient care and better decision making. Examples of healthcare IoT devices include wearable fitness trackers, smart pills, and remote supervision devices. The data collected is used by healthcare providers to diagnose the patient’s health and plan their care, leading to more personalised, effective, and efficient treatment. Additionally, healthcare IoT can reduce costs and increase efficiency within the healthcare system by reducing the need for in-person appointments.
Figure 2 shows different elements in an IoT-based system
[9].
Figure 2. Elements of IoT-based system.
Wireless sensor network (WSN): Wireless sensor networks (WSNs) collect data from the environment using small, low-power sensors with wireless communication. They are used in industrial, agricultural, and environmental monitoring systems and measure physical parameters such as temperature, humidity, pressure, motion, and sound. The data are wirelessly transmitted to a central unit for analysis, making it efficient. WSNs serve as sensors, providing data to IoT platforms. Continuous monitoring through WBANs can detect emergencies and diseases early, improving healthcare for people with disabilities and children. Data are wirelessly sent to a monitoring device, which analyses and controls drug delivery, ensuring patients receive the required medication. WBANs benefit drug delivery by improving patient compliance, reducing costs, and increasing healthcare accessibility
[10]. Wireless body area networks (WBANs) are a type of wireless sensor network (WSN) system that employs wearable or implanted sensors to collect real-time data on a patient’s physiological parameters and clinical metrics
[10]. These data are wirelessly transmitted to a central monitoring device that analyses it and remotely controls the drug delivery system, ensuring patients receive the required medication in the right dosage at the right time. WBANs provide numerous benefits in drug delivery, including improved patient compliance, reduced healthcare costs, and increased accessibility to healthcare services
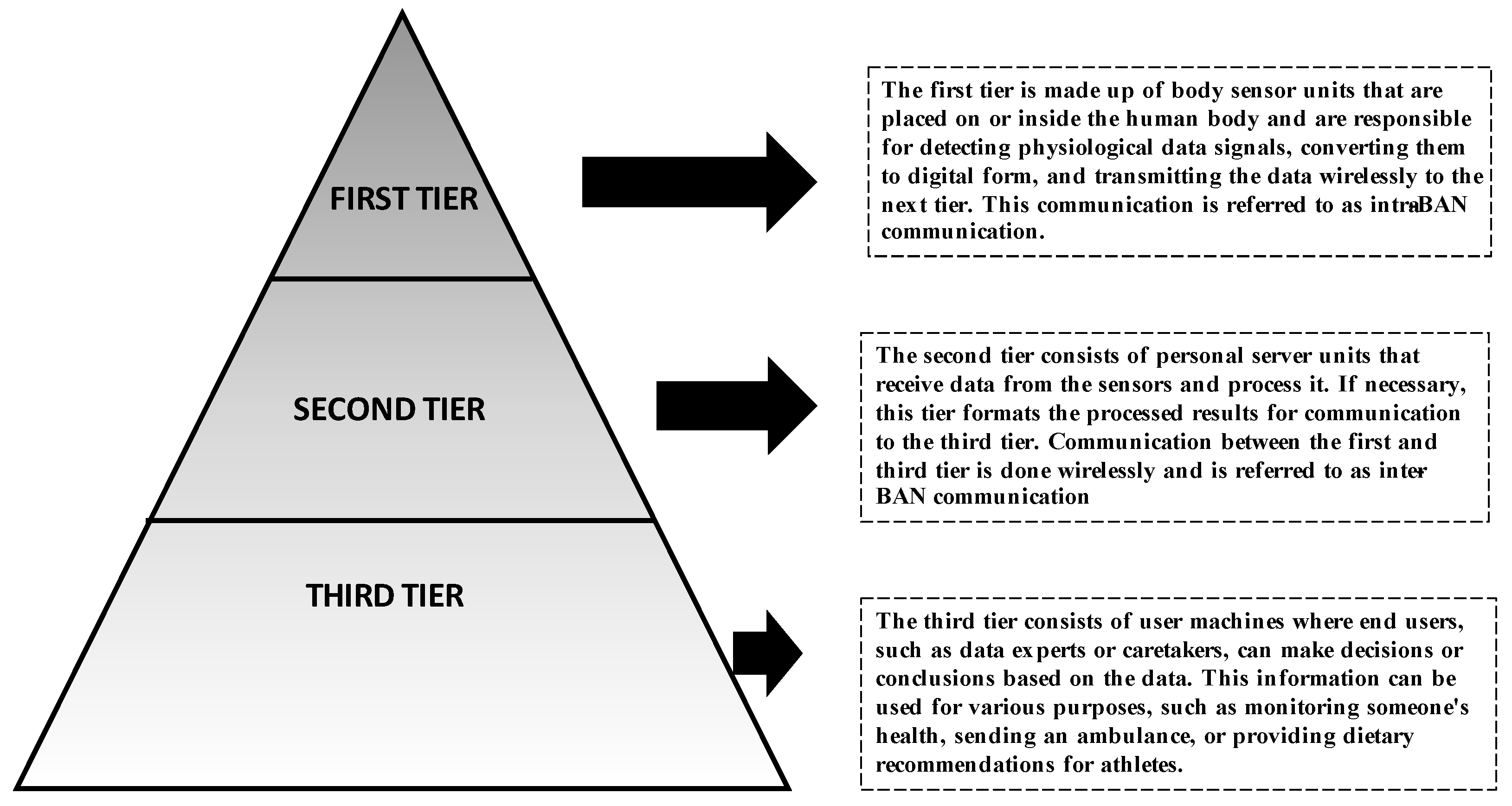
[11]. However, their implementation presents challenges such as security, privacy, and interoperability. WBANs can be implemented in wearable or implanted drug delivery systems, with the former delivering medication through a patch or infusion and the latter delivering medication directly to the target area. The communication in a WBAN typically consists of three tiers
[12], as given in
Figure 3.
Figure 3. Different tiers of communication in WBAN.
2.2. Body Sensors
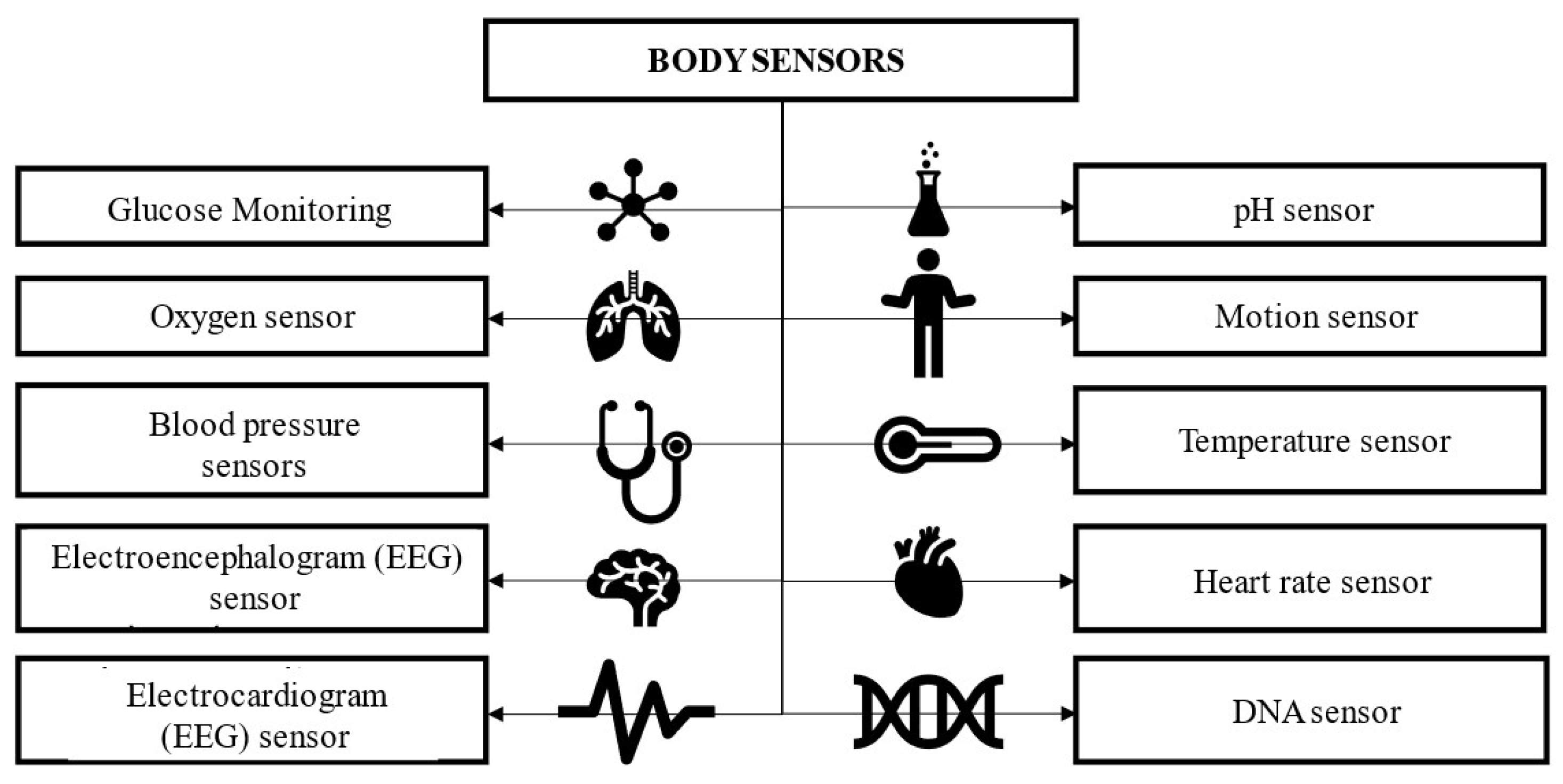
Body sensors are the devices that collect the data and transmit them to the network. They can be small and compact and can be placed in various locations to gather data. Figure 4 shows body sensors that are commonly used in health management settings.
Figure 4. Body sensors commonly used in healthcare applications.
Sensors integrated into drug delivery systems enable real-time monitoring of various physiological parameters, drug levels, or biomarkers. This data can be used to adjust drug dosing or personalise treatment regimens. For instance, glucose sensors can be combined with insulin pumps to create closed-loop systems (also known as artificial pancreas systems), where blood glucose levels are continuously monitored and insulin delivery is automatically adjusted based on the real-time measurements
[13].
3. Applications of Smart Drug Delivery Devices
IoT-based drug delivery systems are becoming increasingly popular in the healthcare sector due to their potential to enhance the accuracy, efficiency, and effectiveness of drug delivery. In smart drug delivery systems, various algorithms are involved to analyse data, make intelligent decisions, and optimise treatment. Closed-loop control algorithms are used in systems such as closed-loop insulin delivery for diabetes management, where real-time sensor data are continuously monitored and analysed to determine the appropriate drug dosage or delivery rate. Pharmacokinetic and pharmacodynamic algorithms utilise mathematical models to optimise dosing regimens and predict drug responses based on factors such as drug absorption, distribution, metabolism, and excretion. Machine learning algorithms can analyse large datasets to identify patterns or correlations that inform drug dosing decisions and optimise treatment outcomes. These algorithms enable personalised dosing recommendations, adaptive dosing adjustments, and real-time alerts, enhancing the effectiveness and safety of drug delivery systems
[14]. Smart drug delivery systems hold significant potential in improving medication compliance and reducing the burden of complex regimens. IoT-based systems provide unique advantages for smart drug delivery, leveraging their connectivity and seamless data exchange capabilities. With real-time monitoring and data analytics, IoT sensors continuously track drug delivery parameters, patient responses, and environmental factors, enabling prompt intervention and optimised treatment protocols
[3]. Remote access and connectivity allow healthcare providers to remotely monitor patient progress, making them particularly valuable for chronic disease management and telemedicine services. Enhanced patient engagement through mobile apps, wearables, and interactive interfaces empowers patients to actively participate in their treatment, leading to improved self-management and treatment outcomes
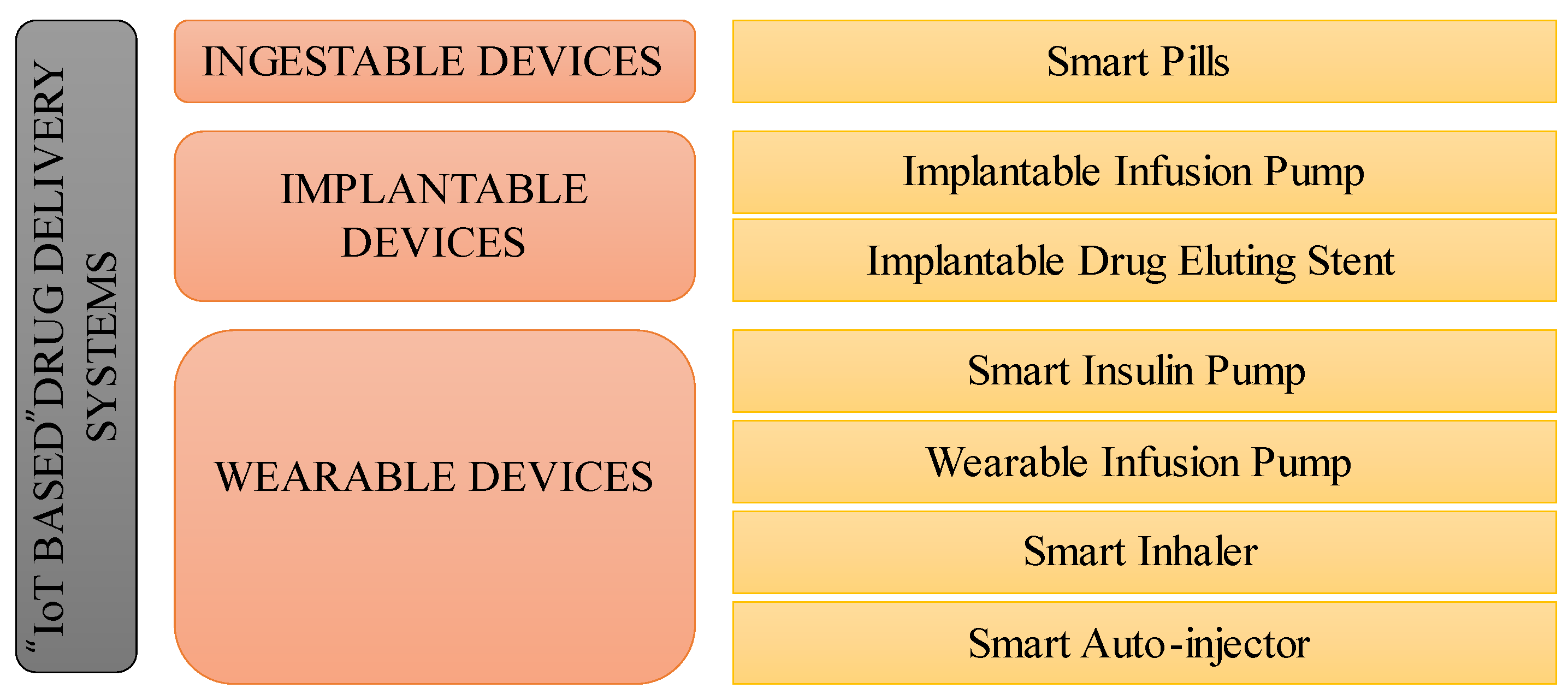
[13]. The scalability and interoperability of IoT infrastructure enable seamless integration with existing healthcare systems, allowing smart drug delivery systems to adapt to evolving needs. Predictive maintenance and data-driven insights optimise system reliability, resource allocation, and workflow efficiency. The classification of IoT-based drug delivery systems is shown in
Figure 5.
Figure 5. Types of IoT-based drug delivery system.
3.1. Wearable Smart Drug Delivery Devices
IoT-based wearable drug delivery devices are innovative solutions for healthcare, leveraging technology to improve delivery accuracy and efficiency. Worn on the body, like a patch or wristband, health devices track vital indicators and administer medication based on collected data. The devices aid medication adherence for patients with chronic conditions, reducing the risk of adverse events occurring.
3.1.1. Smart Insulin Pens
Smart insulin pens use IoT technology to track and manage insulin delivery for diabetes patients. They transmit dosage and schedule information wirelessly, helping providers monitor treatment. Smart insulin pens provide precise dosing based on blood sugar levels, time, and activity, preventing health complications. Smart insulin pens can help diabetics manage their condition by providing insightful data about their insulin usage
[15]. By tracking doses and blood sugar levels, users can detect patterns and receive tailored recommendations for treatment and lifestyle modifications with the associated mobile app. The branded smart insulin pens that are available commercially, and their limitations, are listed in
Table 1.
3.1.2. Wearable Infusion Pumps
Wearable infusion pumps deliver medications through a catheter under the skin. They are portable, discreet, and programmed to administer precise doses at specific times, providing continuous medication throughout the day. Wearable infusion pumps offer convenient and mobile delivery of medication while minimising infection risk by avoiding disturbance to the infusion site. Wearable infusion pumps offer precise dosing and can adjust medication based on real-time glucose levels for personalised treatment. The wearable infusion pumps for smart drug delivery, and their limitations, are listed in Table 2.
3.1.3. Smart Inhalers
Smart inhalers are a type of wearable smart drug delivery device that assists individuals with breathing disorders such as asthma and COPD to better manage their symptoms. These devices can monitor a patient’s inhaler usage, provide reminders to take medication, and track medication adherence over time. Some smart inhalers are also able to measure lung function and provide feedback to patients and their healthcare providers. The information gathered by intelligent inhalers can be utilised to enhance treatment strategies, enhance patient results, and decrease healthcare expenditure. The smart inhalation devices for drug delivery, and their limitations, are listed in Table 3.
Table 3. List of smart inhalation devices for drug delivery.
| Ref. |
Brand Name |
Device Name |
Key Features |
Drug Delivered |
Limitations |
| [28] |
Teva Pharmaceuticals |
ProAir® Digihaler™ |
Smart inhaler records usage data, automates dosing. |
Albuterol for the treatment of asthma and COPD |
Age restriction, limited emergency use, maintenance required. |
| [29] |
ResMed |
Propeller Health |
Inhaler usage tracker and reminder with app integration and customization. |
Various inhaler types for the treatment of asthma and COPD |
Limited compatibility, needs patients’ engagement, limited clinical evidence. |
| [30] |
Adherium |
Smartinhaler™ |
Smart inhaler sensor with app integration, tracking usage, reminders, and notifications. |
Various inhaler types for the treatment of asthma and COPD |
Inhaler compatibility, limited feedback, dependence. |
| [31] |
CoheroHealth |
HeroTracker® sensors |
Wireless sensors track inhaler medication usage automatically and support most inhaler types. |
Various inhaler types for the treatment of asthma and COPD |
Limited battery life, limited data sharing. |
3.1.4. Smart Auto-Injectors
Smart auto-injectors are programmable medical devices that are designed to automatically inject a specific dose of medication. They are also equipped with IoT technology which enables them to connect to other devices or networks and share data in real-time. The IoT-enabled smart auto-injectors are used to administer various types of medication, such as insulin, epinephrine, and other types of drugs. They can be pre-programmed to deliver medication at a specific time or when certain physiological parameters are detected, such as glucose levels in the case of diabetes patients. Table 4 shows some examples of smart auto-injectors for drug delivery.
Table 4. List of smart auto-injectors for drug delivery.
| Ref. |
Brand Name |
Device Name |
Key Features |
Drug Delivered |
Limitations |
| [32] |
Amgen |
Neulasta Onpro Kit |
Smart auto-injector treats neutropenia during chemotherapy with companion app control. |
Pegfilgrastim for the treatment of neutropenia |
SubQ only, cost, reliability issues. |
| [33] |
Teva Pharmaceuticals |
Ajovy Autoinjector |
Smart auto-injector for migraine treatment, controlled by companion app. |
Fremanezumab for the treatment of migraines |
Potential side effects, proper usage required. |
| [34] |
AbbVie |
Humira Citrate-Free Pen |
Auto-injector for treating autoimmune conditions, controlled by companion app. |
Adalimumab for the treatment of autoimmune conditions |
Not compatible with other drug treatment, expensive, side effects experienced. |
| [35] |
Eli Lilly and Company |
Trulicity Pen |
A smart auto-injector for type 2 diabetes, with medication tracking and reminders. |
Dulaglutide for the treatment of type 2 diabetes |
Drug-specific use, side effects. |
3.2. Implatable Smart Drug Delivery Devices
IoT (internet of things)-based implantable drug delivery devices are a growing trend in the healthcare industry, offering new and innovative solutions for drug delivery. These devices leverage IoT technology to provide real-time monitoring and control of drug delivery, improving the accuracy and efficiency of the delivery process.
3.2.1. Implantable Infusion Pumps
These devices are implantable pumps that can deliver drugs, such as pain medications or chemotherapy, directly to the site of action. The pumps can be controlled wirelessly using a smartphone or other device, allowing healthcare providers to adjust the delivery rate as needed.
Implantable sensor-based infusion pumps are medical devices that are surgically implanted into the body to deliver precise doses of medication, such as insulin for the treatment of diabetes or pain medication for the management of chronic pain. These devices are equipped with sensors that can monitor the patient’s physiological parameters and adjust the delivery of medication accordingly. Some examples of implantable sensor-based infusion pumps are given in Table 5.
Table 5. Implantable sensor-based infusion pumps for drug delivery.
| Ref. |
Brand Name |
Device Name |
Key Features |
Drug Delivered |
Limitations |
| [36] |
Abbott |
Infinity™ Deep Brain Stimulation (DBS) |
Implantable infusion pump for deep brain stimulation therapy in Parkinson’s. |
Various medications, such as levodopa for the treatment of Parkinson’s disease |
Invasive procedure, possible neurological side effects, frequent follow-up. |
| [37] |
B. Braun Medical |
Infusomat Space |
Implanted pump delivers medication to specific body areas for various conditions. |
Various medications, such as opioids for pain management |
Compatibility limitations, requires specialised training, susceptible to error. |
3.2.2. Implantable Drug-Eluting Stents
These are stents that are implanted in blood vessels to deliver drugs, such as anti-inflammatory or anti-proliferative agents, directly to the site of action. The stents can be controlled wirelessly using a smartphone or other device, allowing healthcare providers to adjust the delivery rate as needed. A list of some examples of implantable drug-eluting stents is given in Table 6.
Table 6. List of implantable drug-eluting stent.
| Ref. |
Brand Name |
Device Name |
Key Features |
Drug Delivered |
Limitations |
| [38] |
Abbott |
Xience Sierra™ |
Implantable stent with drug coating and remote monitoring for artery treatment. |
Everolimus, an immunosuppressant medication, for the prevention of restenosis after coronary artery angioplasty |
Complications and potential re-treatment needed. |
| [39] |
Medtronic |
Resolute Onyx™ |
Implantable stent with polymer coating and remote monitoring to prevent re-narrowing. |
Zotarolimus, an immunosuppressant medication, for the prevention of restenosis after coronary artery angioplasty |
Expertise required, risk of complications with some medications. |
| [40] |
Boston Scientific |
Promus PREMIER™ |
Remote monitoring, drug-eluting, artery stent for preventing re-narrowing with app. |
Everolimus, an immunosuppressant medication, for the prevention of restenosis after coronary artery angioplasty |
Late complications, limited options. |
3.3. Ingestible Smart Drug Delivery Devices
IoT-based ingestible smart drug delivery devices are another growing trend in the healthcare industry. These devices leverage IoT technology to provide real-time monitoring and control of drug delivery, improving the accuracy and efficiency of the delivery process. Some examples of IoT-based ingestible smart drug delivery devices are given in Table 7.
Table 7. Smart pills for drug delivery and vitals monitoring.
| Ref. |
Brand Name |
Device Name |
Key Features |
Drug Delivered |
Limitations |
| [41] |
Proteus Digital Health |
Abilify MyCite® |
Ingestible pill with sensor and wearable patch for medication adherence monitoring and health data tracking. |
Aripiprazole, an antipsychotic medication |
Limited availability, privacy, cost. |
| [42] |
etectRx |
ID-Cap™ System |
Smart pill with sensor sends data to wearable and smartphone, tracking medication adherence, heart rate, and temperature. |
Various medications, including antibiotics, painkillers, and anticoagulants |
Limited battery life, the transmission distance between the reader and the sensor is limited, patient non-compliance. |