Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Rafael Resende Assis Silva | -- | 3062 | 2023-06-20 11:19:02 | | | |
| 2 | Conner Chen | Meta information modification | 3062 | 2023-06-25 04:26:05 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Silva, R.R.A.; Marques, C.S.; Arruda, T.R.; Teixeira, S.C.; De Oliveira, T.V. Biodegradable Polymers and Stages of Biodegradation. Encyclopedia. Available online: https://encyclopedia.pub/entry/45852 (accessed on 08 June 2026).
Silva RRA, Marques CS, Arruda TR, Teixeira SC, De Oliveira TV. Biodegradable Polymers and Stages of Biodegradation. Encyclopedia. Available at: https://encyclopedia.pub/entry/45852. Accessed June 08, 2026.
Silva, Rafael Resende Assis, Clara Suprani Marques, Tarsila Rodrigues Arruda, Samiris Cocco Teixeira, Taíla Veloso De Oliveira. "Biodegradable Polymers and Stages of Biodegradation" Encyclopedia, https://encyclopedia.pub/entry/45852 (accessed June 08, 2026).
Silva, R.R.A., Marques, C.S., Arruda, T.R., Teixeira, S.C., & De Oliveira, T.V. (2023, June 20). Biodegradable Polymers and Stages of Biodegradation. In Encyclopedia. https://encyclopedia.pub/entry/45852
Silva, Rafael Resende Assis, et al. "Biodegradable Polymers and Stages of Biodegradation." Encyclopedia. Web. 20 June, 2023.
Copy Citation
Sustainable and biodegradable bioplastics are gaining significant attention due to resource depletion and plastic pollution. An increasing number of environmentally friendly plastics are being introduced to the market with the aim of addressing these concerns. However, many final products still contain additives or mix non-biodegradable polymers to ensure minimum performance, which often undermines their ecological footprint. Moreover, there is a lack of knowledge about all stages of biodegradation and their accuracy in classifying products as biodegradable.
biodegradation
biodegradable plastics
biodegradable polymers
1. Mechanisms of Degradation
Polymer degradation refers to any chemical, physical, or biochemical reaction that involves breaking covalent bonds in the backbone of the polymer, resulting in an irreversible change in its properties due to alterations in the chemical structure and the reduction of molecular weight. The breaking of primary chemical bonds in the main or side chain generates reactive species (free radicals) that are responsible for propagating the degradation process of the polymeric artifact. The initiation of the polymer degradation process is catalyzed by abiotic factors, e.g., heat, light, radiation, humidity, pH of the medium, mechanical stress, and chemical attack. These forms of initiation require activation energy for breaking chemical bonds in the polymer, with the binding energy varying according to the atoms’ connection, i.e., they can have ionic, coordinate, metallic, or covalent primary bonds. Generally, the types of bonds in organic polymers are covalent and usually involve short distances and high energies (1.5 Å and 100 K/mol) [1]. The main covalent bonds found in organic polymers, their binding energy, stability, and binding distance, are discussed in-depth by Canevarolo (2006) [1].
Chain scission or bond breaking occurs when the localized energy in this chemical bond is greater than the energy of the bond. When a more unstable bond is positioned in side groups or short branches, its breakage leads to (i) the loss of that side group or (ii) its modification by the insertion of new atoms (e.g., oxygen), resulting in polymer degradation. This type of degradation can occur both in the solid and molten states. The energy required for bond scission can be provided in different ways, such as heat (thermolysis), water (hydrolysis), oxygen (oxidation), chemistry (solvolysis), light (photolysis), gamma radiation (radiolysis), or shear (mechanical) or weathering (generally UV/ozone degradation), etc.
1.1. Hydrolysis
Hydrolysis is a chemical decomposition process that involves breaking a bond by reacting with water molecules. The hydrolysis process is the most important for initiating the biodegradation of synthetic polymers, especially polyesters. The rate of hydrolytic degradation varies from a few hours to years, depending mainly on the degree of crystallinity, type of functional group, molecular weight, main skeletal structure, morphology, temperature, and pH of the medium. According to Lyu & Untereker (2009) [2], hydrolytic degradation is divided into three levels. The first level involves degradation at the molecular level, in which hydrolysis is controlled only by chemical reactivity. The second level is also molecular but is associated with molecular mobility and water–polymer interactions. The third level is the macroscopic one, where erosion and water diffusion reaction are the governing parameters for degradation.
Therefore, hydrolysis can cause biopolymers to degrade either through surface erosion or bulk erosion. During surface erosion, the outer layer of the polymer degrades first, while the inner material is degraded last. In contrast, bulk erosion occurs when water molecules quickly diffuse into the amorphous regions of the polymer, causing a rapid loss of strength and structural properties [3].
Hydrolysis occurs mainly in hygroscopic polymers and those with water-sensitive groups in the polymeric backbone. During hydrolysis, the polymer is always split into two components; otherwise, it will not be considered hydrolysis (hydro = water; lysis = breakdown). If the products are not ionized, one part gains a hydrogen atom (H+), and the other gains a hydroxyl group (OH-) from the broken water molecule. Figure 1 shows the hydrolysis rate ranking of the main polymers that undergo degradation when exposed to moisture, e.g., polyanhydrides, polyesters, polyethers, polyamides, polycarbonates, etc. Furthermore, it is shown that hydrolysis also depends on the polymer’s polarity and degree of crystallinity. More hydrophobic polymers have a lower reaction rate because the water content in the polymer and the water permeability decrease with decreasing polymer polarity. Therefore, hydrolytic stability increases in the same order as hydrophobicity. In turn, an increase in crystalline phases in polymers inhibits the plasticization of the polymer by the water in these regions since the steric effect and strong intermolecular interactions impede water penetration in the ordered regions, i.e., crystalline.
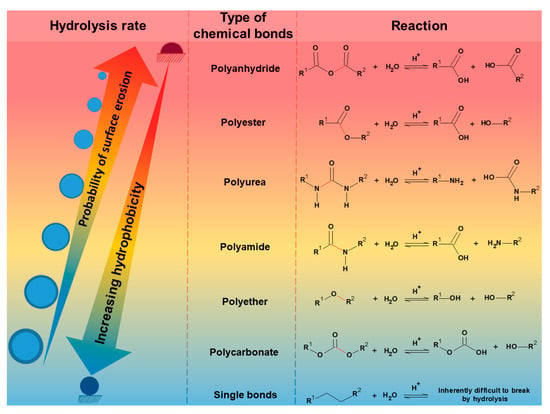
Figure 1. Ranking of the hydrolysis rate of the main polymers that suffer degradation when exposed to humidity.
In turn, if the polymers become ionized after separation, one part will receive two hydrogen atoms with a localized positive charge, while the other part will have an oxygen atom with a negative charge. For example, amino acids are released from protein chains by hydrolysis (Figure 2a). Silva et al. (2021) [4] showed that the absorption of water in the polymeric matrix could reduce both the temperature of decomposition of the polymers and act as a plasticizing effect, i.e., reducing the glass transition temperature of the polymers. The reduction of glass transition temperature (Tg) by water absorption is one of the most significant effects in modifying the properties of plastics, as water reduces intermolecular interactions between polymeric chains. As a result, plastics have reduced stiffness (Young’s modulus), tensile strength, and decomposition temperature [4]. Therefore, the reduction in the performance of these properties and the increase in water vapor permeability, catalyzed by water as a plasticizer, are critical parameters that make the use of plastics for food packaging unfeasible.
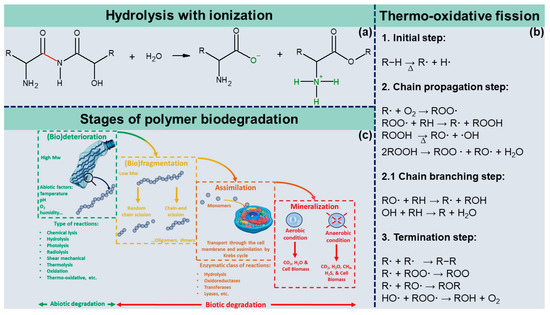
Figure 2. Ionization of an amino acid after hydrolysis (a); steps of thermo-oxidative degradation reactions (b); stages of polymer biodegradation (c).
Lyu & Untereker (2009) [2] demonstrated that water can dissolve in many polymers at a level of approximately 1% wt., which can increase the rate of degradation by hydrolysis. However, in other polymers, the rate of water penetration is much slower than the rate of reaction to break the polymer chains into soluble fragments, indicating that the polymer will degrade by surface erosion. Additionally, Silva et al. (2022) [5] showed that incorporating certain additives, such as LiCl, into polymers to create active antimicrobial packaging may have unintended side effects, such as increased water absorption due to the hygroscopic nature of the added filler. Therefore, a current challenge in the physicochemical and biodegradation of polymers is to investigate ways to synthesize or combine polymers that are water-resistant and can degrade rapidly at the end of their life cycle.
1.2. Thermolysis
Thermal decomposition, also known as thermolysis, is a chemical reaction in which a reacting substance decomposes into at least two new substances upon heating. In the case of polymers, thermal decomposition generates molecules and atoms that are different from the precursor without the simultaneous involvement of other reagents such as oxygen. Since the heat received breaks the bonds of the molecules of the reactants, thermal decomposition is generally an endothermic process. If the chemical energy of the reactants is greater than that of the products, the decomposition reaction will be exothermic (ΔH), indicating that the reactants are highly reactive and the products are stable. An exothermic decomposition reaction releases heat and may be accompanied by an explosion or another chemical reaction.
The results should be conceptualized with the term “decomposition temperature” instead of “degradation temperature”. Unfortunately, the latter term is treated as a synonym for the former, which is incorrect. Degradation temperature refers to the temperature at which loss of some function or property of the material being studied occurs. For example, protein denaturation, inactivation of active antimicrobial agents, change in color or transparency, and reduction in mechanical or barrier performance to gases. On the other hand, the decomposition temperature (TDT) should be used to discuss TGA results because it refers to the decomposition of the polymer into smaller molecules, constituent atoms, and/or the release of gases such as CO2, CH4, CO, etc. In this sense, the degradation temperature often occurs before the decomposition temperature because most properties and functions of materials are thermosensitive, and some properties depend on secondary (intermolecular) bonds that break at mild temperatures. Therefore, when the TGA detects mass loss, it is crucial to describe the event as thermal decomposition as it necessarily involves the breaking of primary bonds, confirming the occurrence of material thermolysis.
1.3. Oxidation and Thermo-Oxidative Fission
In oxidation reactions, a reduction in the average molar mass of the polymer is not necessarily observed, but a marked change in its physical and chemical properties, e.g., a color change of the material, may occur. Regardless of the atmosphere’s composition, polymers will start to decompose if heated enough. However, thermal oxidation differs from thermal decomposition in that it generally catalyzes oxidation reactions culminating in material decomposition at milder temperatures.
Thermo-oxidative fission of polymers is a self-catalytic process that occurs in three stages: initiation, propagation, and termination. The oxygen molecule is considered a highly reactive chemical species, as it reacts quickly with any environmental free radicals. In the first step, heat-catalyzed degradation is initiated when polymer chains form radicals (R*) either by hydrogen abstraction or by homolytic scission of the C-C bond. Next, the propagation of degradation involves a series of intermediate reactions. The first intermediate step is the reaction of a free radical (R·) with an oxygen molecule (O2), forming a peroxy radical (ROO·) that abstracts a hydrogen atom from another polymeric chain, producing a hydroperoxide (ROOH). Hydroperoxides are highly unstable; therefore, they decompose into two new free radicals, (RO·) + (·OH), which attack the polymer chain, abstracting labile hydrogens and introducing new radicals [6]. The thermo-oxidative reaction ends by recombining two radicals, forming stable products, or abstracting hydrogen or π bonds. Figure 2b shows the thermo-oxidative degradation reactions elucidated above.
2. Abiotic and Biotic Degradation
The degradation process of a polymer depends on its intrinsic properties and the extrinsic conditions to which it is exposed, such as the biodiversity and occurrence of microorganisms, which vary locally. Therefore, the degradation of materials can generally be classified as abiotic (heat, radiation, oxygen, humidity, solvents/chemicals) or biotic (bacteria, fungi, algae). Abiotic degradation is usually the first stage after the end of the useful life of the plastic, during which physical and chemical changes occur, but not biological actions, resulting in the modification of at least one property or characteristic of the material. Some of these alterations are visible to the naked eye, such as changes in color, dimensions, cracks, and weight, while others require tools for characterization, such as mechanical and rheological properties, degree of crystallinity, oxidation state, and molecular weight distribution.
In nature, biotic and abiotic factors can act together to decompose organic matter. This is because some microbes excrete extracellular enzymes that act directly on plastics, and prior fragmentation and reduction of molar mass are not necessary to make the microorganisms available. An example of this is the degradation of polyhydroxybutyrate (PHB) by the action of intracellular and extracellular depolymerase of bacteria and fungi [7]. However, abiotic factors weaken the polymer structure, producing smaller polymer fragments that can pass through cell membranes and are biodegraded within microbial cells by cellular enzymes, catalyzing the biological stage of biodegradation. Most plastics degrade first at the polymer surface, as it is the most exposed and vulnerable to chemical (abiotic) or bacterial/enzyme (biotic) attack. The Table 1 presents a list of enzymes and bacteria involved in the biodegradation of various types of polymers, including the type of polymer, biodegradation mechanism, mode of action and mechanisms.
Table 1. Enzymes and bacteria involved in biodegradation of polymers.
| Type of Enzyme/Bacteria | Polymer Type | Biodegradation Mechanism | Mode of Action and Mechanisms |
|---|---|---|---|
| Proteases | Proteins | Hydrolysis | Catalyze the cleavage of peptide bonds in proteins, breaking them down into smaller peptides and eventually amino acids. |
| Lipases | Lipids | Hydrolysis | Break down ester bonds in lipids, producing free fatty acids and glycerol. |
| Amylases | Starch | Hydrolysis | Break down the α-1,4-glycosidic bonds in starch, producing glucose. |
| Cellulases | Cellulose | Hydrolysis | Break down the β-1,4-glycosidic bonds in cellulose, producing glucose. |
| Chitinases | Chitin | Hydrolysis | Break down the β-1,4-glycosidic bonds in chitin, producing N-acetylglucosamine. |
| Laccases | Lignin | Oxidation | Oxidize the phenolic and non-phenolic structures in lignin, breaking down the polymer into smaller fragments. |
| Peroxidases | Lignin | Oxidation | Catalyze the oxidation of lignin by hydrogen peroxide or oxygen, breaking it down into smaller fragments. |
| Kosakonia sp. | Polyethylene | Anaerobic metabolism | Production of extracellular enzymes to break down polyethylene into smaller fragments for cellular uptake and utilization as carbon and energy sources. |
| Aspergillus sp. | Various | Aerobic metabolism | Produce reactive oxygen species and a range of extracellular enzymes, e.g., cellulases, hemicellulases, and ligninases. |
On the other hand, biotic degradation is classified as the biodegradation caused by the action of microorganisms that modify and consume the polymer or polymeric monomers, producing molecules of low molar mass (acids, aldehydes, terpenes, and H2O) and gases (CO2, CH4, and N2). According to Oliveira et al. (2020), the main biodegradation mechanism is the adhesion of microorganisms to the polymer surface, followed by the colonization of the exposed surface. After colonization, enzymatic degradation of the polymer occurs by hydrolytic cleavage, producing molecules of low molecular weight until the final mineralization in CO2 and H2O [10][11].
3. Stages of Biodegradation
Biodegradation can occur over different periods (as long as it meets the established standards, typically around 6 months) in various circumstances and environments. Ideally, it should happen naturally, without human intervention. The stages of biodegradation of polymeric materials are categorized into four stages: (bio)deterioration, (bio)fragmentation, assimilation, and mineralization (Figure 2c). The process can stop at any stage; however, plastic biodegradation is only confirmed after verifying mineralization [11][12].
3.1. (Bio)Deterioration
The first indication of biodegradation is (bio)deterioration, in which the cooperative action of different microorganisms and/or abiotic factors fragments macro materials into small fractions (micro, sub-micro). Deterioration is a superficial degradation that can be identified with the naked eye and is responsible for modifying the material’s mechanical, physical, and chemical properties. The big difference between biodegradation and deterioration is that the former is only confirmed by deterioration, while the latter is already observed by weight loss and macro-deformations (cracks, roughness, scratches, holes).
3.2. (Bio)Fragmentation
The second stage is biofragmentation, a step in which catalytic agents (e.g., enzymes) are excreted by the microorganisms, progressively reducing the molecular weight of the polymers. At this moment, the polymers are cleaved until the production of small molecules (dimers and monomers). The term biofragmentation or, in some cases the depolymerization, should be used for situations where macromolecular size reduction occurs without changing the chemical composition or the monomer unit’s structure. Some enzymatic tests can be used to estimate the propensity for biofragmentation of polymers, such as tests of enzymatic mixtures for solid-wet reaction in polyethylene terephthalate (PET) [13].
3.3. Assimilation
Assimilation is the third stage and occurs in the cytoplasm when small molecules produced in depolymerization integrate with the microbial metabolism to produce energy, biomass, and other metabolites. Therefore, assimilation happens when microorganisms use polymers as their carbon/nitrogen sources, converting CO2 or CH4/NH3 or nitrate into cell building blocks [14][15]. This assimilation can occur through the three classic catabolic pathways: aerobic respiration, anaerobic respiration, and/or fermentation, and it is the only event in which fragments of polymeric materials are absorbed inside microbial cells [11]. This absorption is responsible for producing energy, via the production of adenosine triphosphate (ATP), aiming to form structural elements of cells. This allows microorganisms to grow, proliferate, and consume new energy packages (substrates) from the environment [11].
3.4. Mineralization
The final stage, mineralization, occurs concurrently with assimilation, during which organic material is converted into minerals through the excretion of metabolites and simple molecules that can be absorbed by both the environment and microorganisms [7][11]. The biodegradation process typically involves different microorganisms with complex interactions and symbiosis, making it difficult to simulate degradation in a natural environment in the laboratory. For instance, some microorganisms mainly break down polymers and produce CO2 (mineralization), while others reduce the polymer into its constituent monomers, and some use these monomers and excrete simpler residual compounds that serve as substrates, while others use the excreted residues as a source of energy. With the metabolic routes’ complexity and generation of new products, it is noteworthy that CO2 and H2O gases are produced during aerobic biodegradation, which can be used to monitor activity at this stage. In contrast, to aerobic processes, which produce CO2, the anaerobic process results in the generation of both CO2 and CH4 [15][16]. Therefore, mineralization is the only stage capable of indicating the material’s biodegradation and must be estimated through standardized respirometric methods, such as measuring the evolution of the gases mentioned above for anaerobic environments or oxygen consumption for aerobic environments, as in the ISO 14852.
4. Greenwashing Concept
Despite efforts to assess biodegradation and reduce environmental damage related to the improper disposal of plastic artifacts, in recent years, there has been an increase in the number of corporations that adopt green marketing strategies and label products with more environmental benefits than they actually have, e.g., biodegradability and sustainability. Greenwashing consists of deceiving consumers regarding the environmental conduct of a company or the environmental benefits that a product or service can offer. In this context, it is common to find packages with green parts, drawings of leaves, and words such as “green”, “bio”, and “eco”, without explaining exactly what they refer to, but conveying the idea of a product that is less harmful to the environment. Such practices have been used to attract consumers who are aware and committed to sustainable actions, unduly influencing their purchasing decisions. Therefore, in addition to adopting norms for evaluating the biodegradation of plastics, inspections of commercialized products must be implemented to combat this practice.
As a result, some technical standards of biodegradability were developed to regulate the correct labeling of these materials and serve as references for assessing the level of degradation of plastics. These standards use a set of instruments and techniques to simulate biodegradation conditions in the laboratory as closely as possible to real environmental conditions, indicating the level of degradation for each stage of biodegradation. An example of this is the weatherometer (accelerated aging test), an instrument that subjects samples to different temperatures, UV radiation, and humidity for a specific time, making it possible to expedite the comparison between the properties of plastics. In this sense, the main standards that track the biodegradation of plastics, along with the techniques and interpretations employed, will be presented below.
References
- Canevarolo, S.V., Jr. Ciência dos Polímeros; Artliber: São Paulo, Brazil, 2006; p. 277.
- Lyu, S.; Untereker, D. Degradability of Polymers for Implantable Biomedical Devices. Int. J. Mol. Sci. 2009, 10, 4033–4065.
- Kabir, E.; Kaur, R.; Lee, J.; Kim, K.-H.; Kwon, E.E. Prospects of biopolymer technology as an alternative option for non-degradable plastics and sustainable management of plastic wastes. J. Clean. Prod. 2020, 258, 120536.
- Silva, R.R.A.; de Freitas, P.A.V.; Teixeira, S.C.; de Oliveira, T.V.; Marques, C.S.; Stringheta, P.C.; dos Santos Pires, A.C.; Ferreira, S.O.; Soares, N.F.F. Plasticizer Effect and Ionic Cross-Linking: The Impact of Incorporating Divalent Salts in Methylcellulose Films for Colorimetric Detection of Volatile Ammonia. Food Biophys. 2021, 17, 59–74.
- Silva, R.R.A.; Marques, C.S.; Arruda, T.R.; Teixeira, S.C.; de Oliveira, T.V.; Stringheta, P.C.; dos Santos, A.C.P.; Soares, N.F.F. Ionic Strength of Methylcellulose-Based Films: An Alternative for Modulating Mechanical Performance and Hydrophobicity for Potential Food Packaging Application. Polysaccharides 2022, 3, 426–440.
- Gryn’ova, G.; Hodgson, J.L.; Coote, M.L. Revising the mechanism of polymer autooxidation. Org. Biomol. Chem. 2011, 9, 480–490.
- Shah, A.A.; Hasan, F.; Hameed, A.; Ahmed, S. Biological degradation of plastics: A comprehensive review. Biotechnol. Adv. 2008, 26, 246–265.
- Mohanan, N.; Montazer, Z.; Sharma, P.K.; Levin, D.B. Microbial and Enzymatic Degradation of Synthetic Plastics. Front Microbiol. 2020, 11, 580709.
- Ghatge, S.; Yang, Y.; Ahn, J.-H.; Hur, H.-G. Biodegradation of polyethylene: A brief review. Appl. Biol. Chem. 2020, 63, 27.
- Lucas, N.; Bienaime, C.; Belloy, C.; Queneudec, M.; Silvestre, F.; Nava-Saucedo, J.-E. Polymer biodegradation: Mechanisms and estimation techniques—A review. Chemosphere 2008, 73, 429–442.
- Oliveira, J.; Belchior, A.; da Silva, V.D.; Rotter, A.; Petrovski, Ž.; Almeida, P.L.; Lourenço, N.D.; Gaudêncio, S.P. Marine Environmental Plastic Pollution: Mitigation by Microorganism Degradation and Recycling Valorization. Front. Mar. Sci. 2020, 7, 567126.
- Tokiwa, Y.; Calabia, B.; Ugwu, C.; Aiba, S. Biodegradability of Plastics. Int. J. Mol. Sci. 2009, 10, 3722–3742.
- Kaabel, S.; Therien, J.P.; Dschênes, C.; Dustin, D.D.; Friščić, T.; Auclair, K. Enzymatic depolymerization of highly crystalline polyethylene terephthalate enabled in moist-solid reaction mixtures. Proc. Natl. Acad. Sci. USA 2021, 118, e2026452118.
- Elahi, A.; Bukhari, D.A.; Shamim, S.; Rehman, A. Plastics degradation by microbes: A sustainable approach. J. King Saud Univ. Sci. 2021, 33, 101538.
- Kawai, F. Biodegradation of Polymers (Bioassimilation, Biomineralization, Biodisintegration, Compost), Overview. Encycl. Polym. Nanomater. 2015, 1, 155–160.
- Gu, J.D.; Ford, T.E.; Mitton, D.B.; Mitchell, R. Microbial Corrosion of Metals. In The Uhlig Corrosion Handbook, 2nd ed.; Revie, W., Ed.; Wiley: New York, NY, USA, 2000; pp. 915–927.
More
Information
Subjects:
Others
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
2.8K
Revisions:
2 times
(View History)
Update Date:
25 Jun 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





