Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yudong Zhang | -- | 1644 | 2023-06-20 09:40:19 | | | |
| 2 | Conner Chen | Meta information modification | 1644 | 2023-06-25 04:16:31 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Zhou, Q.; Wang, J.; Yu, X.; Wang, S.; Zhang, Y. Deep Learning for Alzheimer’s Disease. Encyclopedia. Available online: https://encyclopedia.pub/entry/45838 (accessed on 12 April 2026).
Zhou Q, Wang J, Yu X, Wang S, Zhang Y. Deep Learning for Alzheimer’s Disease. Encyclopedia. Available at: https://encyclopedia.pub/entry/45838. Accessed April 12, 2026.
Zhou, Qinghua, Jiaji Wang, Xiang Yu, Shuihua Wang, Yudong Zhang. "Deep Learning for Alzheimer’s Disease" Encyclopedia, https://encyclopedia.pub/entry/45838 (accessed April 12, 2026).
Zhou, Q., Wang, J., Yu, X., Wang, S., & Zhang, Y. (2023, June 20). Deep Learning for Alzheimer’s Disease. In Encyclopedia. https://encyclopedia.pub/entry/45838
Zhou, Qinghua, et al. "Deep Learning for Alzheimer’s Disease." Encyclopedia. Web. 20 June, 2023.
Copy Citation
Alzheimer’s and related diseases are significant health issues of this era. The interdisciplinary use of deep learning in this field has shown great promise and gathered considerable interest.
deep learning
Alzheimer’s disease
mild cognitive impairment
1. Alzheimer’s Disease and Mild Cognitive Impairment
Alzheimer’s disease is the most common form of dementia and a significant health issue of this era [1]. Brookmeyer et al. [2] predicted that more than 1% of the world population would be affected by AD or related diseases by 2050, with a significant proportion of this cohort requiring a high level of care. AD usually starts from middle-to-old age as a chronic neurodegenerative disorder, but rare cases of early-onset AD can affect individuals of 45–64 years old [3]. AD leads to cognitive decline symptoms: memory impairment [4], language dysfunction [5], and decline in cognition and judgment [6]. An individual with symptoms may require moderate to constant assistance in day-to-day life, depending on the stage of disease progression. These symptoms severely affect patients’ quality of life (QOL) and their families. Studies into cost-of-illness for dementia and AD reveal that the higher societal need for elderly care significantly increases overall social-economic pressure [7].
The biological process that leads to AD may begin more than 20 years before symptoms appear [8]. The current understanding of AD pathogenesis is based on amyloid peptide deposition and the accumulation and phosphorylation of tau proteins around neurons [9][10][11], which leads to neurodegeneration and eventual brain atrophy. Factors associated with AD include age, genetic predisposition [12], Down’s syndrome [13], brain injuries [14], and cardiorespiratory fitness [15][16][17]. AD-related cognitive impairment can be broadly separated into three stages: (1) preclinical AD, where measurable changes in the brain, cerebral spinal fluid (CSF), and blood plasma can be detected; (2) mild cognitive impairment (MCI) due to AD, where biomarker evidence of AD-related brain change can be present; and (3) dementia due to AD, where changes in the brain are evident and noticeable memory, thinking and behavioural changes appear and impair an individual’s daily function.
The condition most commonly associated with AD is mild cognitive impairment (MCI), the pre-dementia stage of cognitive impairment. However, not all cases of MCI develop into AD. Since no definite pathological description exists, MCI is currently perceived as the level of cognitive impairment above natural age-related cognitive decline [18][19]. Multiple studies have analyzed the demographics and progression of MCI and have found the following: 15–20% of people age 65 or older have MCI from a range of possible causes [20]; 15% of people age 65 or older with MCI developed dementia at two years follow-up [21]; and 32% developed AD and 38% developed dementia at five years follow-up [22][23]. The early diagnosis of MCI and its subtypes can lead to early intervention, which can profoundly impact patient longevity and QOL [24]. Therefore, better understanding the condition and developing effective and accurate diagnostic methods is of great public interest.
2. Diagnostic Methods and Criteria
The current standard diagnosis of AD and MCI is based on a combination of various methods. These methods include cognitive assessments such as the Mini-Mental State Examination [25][26][27], Clinical Dementia Rating [28][29], and Cambridge Cognitive Examination [30][31]. These exams usually take the form of a series of questions and are often performed with physical and neurological examinations. Medical and family history, including psychiatric history and history of cognitive and behavioral changes, are also considered in the diagnosis. Genetic sequencing for particular biomarkers, such as the APOE-e4 allele [32], is used to determine genetic predisposition.
Neuroimaging is commonly used to inspect various signs of brain changes and exclude other potential causes. Structural magnetic resonance and diffusion tensor imaging are widely applied to check for evidence of symptoms of brain atrophy. Various forms of computed tomography (CT) are also used in AD and MCI diagnosis. Regarding positron emission tomography (PET), FDG-PET [33] inspects brain glucose metabolism, while amyloid-PET is applied to measure beta-amyloid levels. Single-photon emission computed tomography [34] (SPECT) is likely to produce false-positive results and is inadequate in clinical use. However, SPECT variants can be potentially used in diagnosis, e.g., 99 mTc-HMPAO SPECT [35][36]. At the same time, FP-CIT SPECT can visualize discrepancies in the nigrostriatal dopaminergic neurons [37]. In neuroimaging, a combination of multiple modalities is commonly used to utilize the functionality of each modality.
New diagnostic factors of CSF and blood plasma biomarkers have been reported in the literature and have been deployed in clinical practice in recent years. There are three main CSF and blood plasma biomarkers: Amyloid-β42, t-tau, and p-tau. Other biomarkers include neurofilament light protein (NFL), and neuron-specific enolase (NSE, and HFABP [38][39]. CSF biomarkers are becoming a critical factor in AD diagnostic criteria in some practices. However, the actual ‘ground truth’ diagnosis of AD can only be made via post-mortem autopsy.
Before this century, the established diagnostic criteria were the NINCDS-ADRDA criteria [40][41]. These criteria were updated by the International Working Group (IWG) in 2007 to include requirements of at least one factor among MRI, PET, and CSF biomarkers [42]. A second revision was introduced in 2010 to include both pre-dementia and dementia phases [43]. This was followed by a third revision to include atypical prodromal Alzheimer’s disease that shows cognition deficits other than memory impairment—IWG-2 [44]. Another independent set of criteria, the NIA-AA criteria, was introduced in 2011. These criteria include measures of brain amyloid, neuronal injury, and degeneration [45]. Individual criteria were introduced for each clinical stage, including pre-clinical [46][47], MCI [48][49], dementia [50][51], and post-mortem autopsy [52].
3. The Deep Learning Approach
Detailed preprocessing with refined extraction of biomarkers combined with statistical analysis is the accepted practice in current medical research. Risacher et al. [53] applied statistical analysis on biomarkers extracted using voxel-based morphometry and parcellation methods from T1-weighted MRI scans of AD, MCI, and HC. The study reveals statistical significance in multiple measures, including hippocampal volume and entorhinal cortex thickness. Qiu et al. [54] further confirmed this significance by analyzing regional volumetric changes through large deformation diffeomorphic metric mapping (LDDMM). Guevremont et al. [55] focused on robustly detecting microRNAs in plasma and used standardized analysis to identify microRNA biomarkers in different phases of Alzheimer’s disease. This study and its statistical analysis yielded useful diagnostic markers reflecting the underlying disease pathology. The different biomarker information extracted was fed into statistical analysis methods with varying numbers of variables to detect changes in biomarkers in disease development [56]. Similar studies also employed other neuroimaging data, genetic data, and CSF biomarkers. These studies supported the use of MRI imaging biomarkers in AD [57] and MCI diagnosis [58], laying the basis for developing automatic diagnostic algorithms.
Machine learning has amassed great popularity among current automated diagnostic algorithms due to its adaptivity to data and the ability to generalize knowledge with lower requirements of expert experience. The study by Klöppel et al. [59] proved the validity of applying machine learning algorithms in diagnosing dementia through a performance comparison between the Support Vector Machine (SVM) classification of local grey matter volumes and human diagnosis by professional radiologists. Janousova et al. [60] proposed penalized regression with resampling to search for discriminative regions to aid Gaussian kernel SVM classification. The regions found by the study coincide with the previous morphological studies. These breakthroughs led to the development of many machine-learning algorithms for AD and MCI detection. Zhang et al. [61] proposed a kernel combination method for the fusion of heterogeneous biomarkers for classification with linear SVM. Liu et al. [62] proposed the Multifold Bayesian Kernelization (MBK) algorithm, where a Bayesian framework derives kernel weights and synthesis analysis provides the diagnostic probabilities of each biomarker. Zhang et al. [63] proposed the extraction of the eigenbrain using Welch’s t-test (WTT) [64] combined with a polynomial kernel SVM [65] and particle swarm optimization (PSO) [66].
There has also been considerable interest in applying deep learning (DL), a branch of machine learning, to the field of AD and related diseases. Deep learning integrates the two-step feature extraction and classification process into neural networks, universal approximators based on backpropagation parameter training. [67]. Deep learning has made considerable advances in the domain of medical data, e.g., breast cancer [68], tuberculosis [69], and glioma [70]. Instead of hand-crafting features, models, and optimizers, deep learning leverages the layered structure of neural networks for the automated abstraction of various levels of features. For example, Feng et al. [71] used the proposed deep learning model to extract biomarkers for MRI in neuroimaging. The study demonstrates that the deep learning approach outperformed other neuroimaging biomarkers of amyloid and tau pathology and neurodegeneration in prodromal AD. A visualization of the field of this survey is shown in Figure 1.
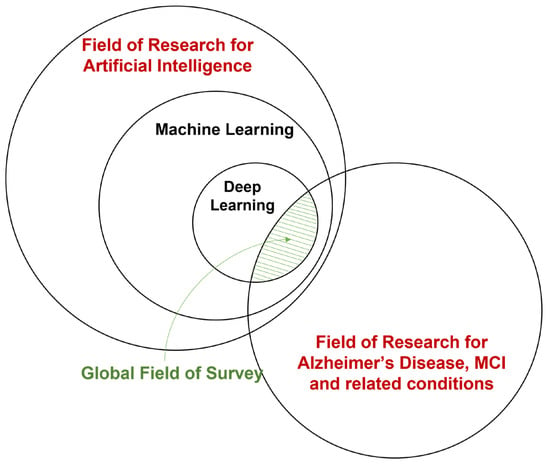
Figure 1. A broad overview of the field of this survey.
4. Areas of Interest
The primary aim of the surveyed deep learning studies in Alzheimer’s and related diseases is detecting and predicting neurodegeneration to provide early detection and accurate prognosis to support treatment and intervention. The main interests of this interdisciplinary field can be roughly categorized into three areas:
- Classification of various stages of AD. This area targets diagnosis or efficient progression monitoring. Current studies mostly focus on AD, MCI subtypes, and normal cognitive controls (NC). A few studies contain the subjective cognitive decline (SCD) stage before MCI.
- Predicting MCI conversion. This area is mainly approached by formulating prediction as a classification problem, which usually involves defining MCI converters and non-converters based on a time threshold from the initial diagnosis. Some studies also aim at the prediction of time-to-conversion for MCI to AD.
- Prediction of clinical measures. This area aims at producing surrogate biomarkers to reduce cost or invasivity, e.g., neuroimaging to replace lumbar puncture. Prediction of clinical measures, e.g., ADAS-Cog13 [72] and ventricular volume [73], is also used for longitudinal studies and attempts to achieve a more comprehensive evaluation of disease progression and model performance benchmarking.
There are also other areas of interest, including knowledge discovery, where studies attempt to understand AD through data [74]. Another area of interest is phenotyping and sample enrichment for clinical trials of treatments [75], where DL models are used to select patients that will likely respond to treatment and prevent ineffective or unnecessary treatment [76]. Interest also lies in segmentation and preprocessing, where DL models are applied to achieve higher performance or efficiency than conventional pipelines [77].
References
- Tatulian, S.A. Challenges and hopes for Alzheimer’s disease. Drug Discov. Today 2022, 27, 1027–1043.
- Brookmeyer, R.; Johnson, E.; Ziegler-Graham, K.; Arrighi, H.M. Forecasting the global burden of Alzheimer’s disease. Alzheimer’s Dement. 2007, 3, 186–191.
- Loi, S.M.; Pijnenberg, Y.; Velakoulis, D. Recent research advances in young-onset dementia. Curr. Opin. Psychiatry 2023, 36, 126–133.
- Zhang, W.; Xu, C.; Sun, J.; Shen, H.-M.; Wang, J.; Yang, C. Impairment of the autophagy–lysosomal pathway in Alzheimer’s diseases: Pathogenic mechanisms and therapeutic potential. Acta Pharm. Sin. B 2022, 12, 1019–1040.
- Boeve, B.F.; Boxer, A.L.; Kumfor, F.; Pijnenburg, Y.; Rohrer, J.D. Advances and controversies in frontotemporal dementia: Diagnosis, biomarkers, and therapeutic considerations. Lancet Neurol. 2022, 21, 258–272.
- Sügis, E.; Dauvillier, J.; Leontjeva, A.; Adler, P.; Hindie, V.; Moncion, T.; Collura, V.; Daudin, R.; Loe-Mie, Y.; Herault, Y.; et al. HENA, heterogeneous network-based data set for Alzheimer’s disease. Sci. Data 2019, 6, 151.
- Wimo, A.; Jönsson, L.; Bond, J.; Prince, M.; Winblad, B.; Alzheimer Disease International. The worldwide economic impact of dementia 2010. Alzheimer’s Dement. 2013, 9, 1–11.e3.
- López-Cuenca, I.; Nebreda, A.; García-Colomo, A.; Salobrar-García, E.; de Frutos-Lucas, J.; Bruña, R.; Ramírez, A.I.; Ramirez-Toraño, F.; Salazar, J.J.; Barabash, A.; et al. Early visual alterations in individuals at-risk of Alzheimer’s disease: A multidisciplinary approach. Alzheimer’s Res. Ther. 2023, 15, 19.
- Toschi, N.; Baldacci, F.; Zetterberg, H.; Blennow, K.; Kilimann, I.; Teipel, S.J.; Cavedo, E.; dos Santos, A.M.; Epelbaum, S.; Lamari, F. Alzheimer’s disease biomarker-guided diagnostic workflow using the added value of six combined cerebrospinal fluid candidates: Ab1–42, total-tau, phosphorylated-tau, NFL, neurogranin, and YKL-40. Alzheimer’s Dement. 2017, 1, 10.
- Scheltens, P.; Blennow, K.; Breteler, M.M.B.; de Strooper, B.; Frisoni, G.B.; Salloway, S.; Van der Flier, W.M. Alzheimer’s disease. Lancet 2016, 388, 505–517.
- Vogt, A.-C.S.; Jennings, G.T.; Mohsen, M.O.; Vogel, M.; Bachmann, M.F. Alzheimer’s Disease: A Brief History of Immunotherapies Targeting Amyloid β. Int. J. Mol. Sci. 2023, 24, 3895.
- Van der Lee, S.J.; Wolters, F.J.; Ikram, M.K.; Hofman, A.; Ikram, M.A.; Amin, N.; van Duijn, C.M. The effect of APOE and other common genetic variants on the onset of Alzheimer’s disease and dementia: A community-based cohort study. Lancet Neurol. 2018, 17, 434–444.
- Fortea, J.; Vilaplana, E.; Carmona-Iragui, M.; Benejam, B.; Videla, L.; Barroeta, I.; Fernández, S.; Altuna, M.; Pegueroles, J.; Montal, V. Clinical and biomarker changes of Alzheimer’s disease in adults with Down syndrome: A cross-sectional study. Lancet 2020, 395, 1988–1997.
- Brett, B.L.; Gardner, R.C.; Godbout, J.; Dams-O’connor, K.; Keene, C.D. Traumatic Brain Injury and Risk of Neurodegenerative Disorder. Biol. Psychiatry 2021, 91, 498–507.
- Letnes, J.M.; Nes, B.M.; Wisløff, U. Age-related decline in peak oxygen uptake: Cross-sectional vs. longitudinal findings. A review. Int. J. Cardiol. Cardiovasc. Risk Prev. 2023, 16, 200171.
- Tari, A.R.; Nauman, J.; Zisko, N.; Skjellegrind, H.K.; Bosnes, I.; Bergh, S.; Stensvold, D.; Selbæk, G.; Wisløff, U. Temporal changes in cardiorespiratory fitness and risk of dementia incidence and mortality: A population-based prospective cohort study. Lancet Public Health 2019, 4, e565–e574.
- Birkenhäger, W.H.; Forette, F.; Seux, M.-L.; Wang, J.-G.; Staessen, J.A. Blood Pressure, Cognitive Functions, and Prevention of Dementias in Older Patients with Hypertension. Arch. Intern. Med. 2001, 161, 152–156.
- Donaghy, P.C.; Ciafone, J.; Durcan, R.; Hamilton, C.A.; Barker, S.; Lloyd, J.; Firbank, M.; Allan, L.M.; O’Brien, J.T.; Taylor, J.-P.; et al. Mild cognitive impairment with Lewy bodies: Neuropsychiatric supportive symptoms and cognitive profile. Psychol. Med. 2020, 52, 1147–1155.
- Burns, A.; Iliffe, S. Alzheimer’s disease. BMJ Br. Med. J. (Int. Ed.) 2009, 338, 467–471.
- Roberts, R.; Knopman, D.S. Classification and Epidemiology of MCI. Clin. Geriatr. Med. 2013, 29, 753–772.
- Petersen, R.C.; Lopez, O.; Armstrong, M.J.; Getchius, T.S.; Ganguli, M.; Gloss, D.; Gronseth, G.S.; Marson, D.; Pringsheim, T.; Day, G.S.; et al. Author response: Practice guideline update summary: Mild cognitive impairment: Report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology. Neurology 2018, 91, 373–374.
- Ward, A.; Tardiff, S.; Dye, C.; Arrighi, H.M. Rate of Conversion from Prodromal Alzheimer’s Disease to Alzheimer’s Dementia: A Systematic Review of the Literature. Dement. Geriatr. Cogn. Disord. Extra 2013, 3, 320–332.
- Mitchell, A.J.; Shiri-Feshki, M. Rate of progression of mild cognitive impairment to dementia—Meta-analysis of 41 robust inception cohort studies. Acta Psychiatr. Scand. 2009, 119, 252–265.
- Sherman, D.S.; Mauser, J.; Nuno, M.; Sherzai, D. The Efficacy of Cognitive Intervention in Mild Cognitive Impairment (MCI): A Meta-Analysis of Outcomes on Neuropsychological Measures. Neuropsychol. Rev. 2017, 27, 440–484.
- Ala, T.; Bakir, D.; Goel, S.; Feller, N.; Botchway, A.; Womack, C. A Mini-Mental State Examination Formula May Help to Distinguish Alzheimer’s Disease from Dementia with Lewy Bodies. J. Alzheimer’s Dis. 2022, 89, 1119–1129.
- McGurn, M.; Dworkin, J.D.; Chapman, S.; Huey, E.D.; Cosentino, S.; Louis, E.D. Can the Montreal Cognitive Assessment and Mini-Mental State Examination detect cognitive decline in elderly patients with essential tremor? Clin. Neuropsychol. 2022, 1–18.
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-Mental State”. A Practical Method for Grading the Cognitive State of Patients for the Clinician. J. Psychiatr. Res. 1975, 12, 189–198.
- Tzeng, R.-C.; Yang, Y.-W.; Hsu, K.-C.; Chang, H.-T.; Chiu, P.-Y. Sum of boxes of the clinical dementia rating scale highly predicts conversion or reversion in predementia stages. Front. Aging Neurosci. 2022, 14, 1021792.
- Hughes, C.P.; Berg, L.; Danziger, W.L.; Coben, L.A.; Martin, R.L. A New Clinical Scale for the Staging of Dementia. Br. J. Psychiatry 1982, 140, 566–572.
- Titheradge, D.; Isaac, M.; Bremner, S.; Tabet, N. Cambridge Cognitive Examination and Hachinski Ischemic Score as predictors of MRI confirmed pathology in dementia: A cross-sectional study. Int. J. Clin. Pract. 2019, 74, e13446.
- Schmand, B.; Walstra, G.; Lindeboom, J.; Teunisse, S.; Jonker, C. Early detection of Alzheimer’s disease using the Cambridge Cognitive Examination (CAMCOG). Psychol. Med. 2000, 30, 619–627.
- López-Cuenca, I.; Marcos-Dolado, A.; Yus-Fuertes, M.; Salobrar-García, E.; Elvira-Hurtado, L.; Fernández-Albarral, J.A.; Salazar, J.J.; Ramírez, A.I.; Sánchez-Puebla, L.; Fuentes-Ferrer, M.E.; et al. The relationship between retinal layers and brain areas in asymptomatic first-degree relatives of sporadic forms of Alzheimer’s disease: An exploratory analysis. Alzheimer’s Res. Ther. 2022, 14, 79.
- Rocha, A.; Bellaver, B.; Souza, D.G.; Schu, G.; Fontana, I.C.; Venturin, G.T.; Greggio, S.; Fontella, F.U.; Schiavenin, M.L.; Machado, L.S.; et al. Clozapine induces astrocyte-dependent FDG-PET hypometabolism. Eur. J. Nucl. Med. 2022, 49, 2251–2264.
- Oe, K.; Zeng, F.; Niikura, T.; Fukui, T.; Sawauchi, K.; Matsumoto, T.; Nogami, M.; Murakami, T.; Kuroda, R. Influence of Metal Implants on Quantitative Evaluation of Bone Single-Photon Emission Computed Tomography/Computed Tomography. J. Clin. Med. 2022, 11, 6732.
- Madetko-Alster, N.; Alster, P.; Migda, B.; Nieciecki, M.; Koziorowski, D.; Królicki, L. The Use of Cerebellar Hypoperfusion Assessment in the Differential Diagnosis of Multiple System Atrophy with Parkinsonism and Progressive Supranuclear Palsy-Parkinsonism Predominant. Diagnostics 2022, 12, 3022.
- Charpentier, P.; Lavenu, I.; Defebvre, L.; Duhamel, A.; Lecouffe, P.; Pasquier, F.; Steinling, M. Alzheimer’s disease and frontotemporal dementia are differentiated by discriminant analysis applied to 99mTc HmPAO SPECT data. J. Neurol. Neurosurg. Psychiatry 2000, 69, 661–663.
- Garriga, M.; Emila, M.; Mir, M.; Eal-Baradie, R.; Ehuertas, S.; Ecastejon, C.; Ecasas, L.; Badenes, D.; Gimenez, N.; Font, M.A.; et al. 123I-FP-CIT SPECT imaging in early diagnosis of dementia in patients with and without a vascular component. Front. Syst. Neurosci. 2015, 9, 99.
- Fortea, J.; Carmona-Iragui, M.; Benejam, B.; Fernández, S.; Videla, L.; Barroeta, I.; Alcolea, D.; Pegueroles, J.; Muñoz, L.; Belbin, O.; et al. Plasma and CSF biomarkers for the diagnosis of Alzheimer’s disease in adults with Down syndrome: A cross-sectional study. Lancet Neurol. 2018, 17, 860–869.
- Olsson, B.; Lautner, R.; Andreasson, U.; Öhrfelt, A.; Portelius, E.; Bjerke, M.; Hölttä, M.; Rosén, C.; Olsson, C.; Strobel, G.; et al. CSF and blood biomarkers for the diagnosis of Alzheimer’s disease: A systematic review and meta-analysis. Lancet Neurol. 2016, 15, 673–684.
- Chen, C.L.; Lu, Q.; Moorakonda, R.B.; Kandiah, N.; Tan, B.Y.; Villaraza, S.G.; Cano, J.; Venketasubramanian, N. Alzheimer’s Disease THErapy with NEuroaid (ATHENE): A Randomized Double-Blind Delayed-Start Trial. J. Am. Med. Dir. Assoc. 2021, 23, 379–386.e3.
- McKhann, G.; Drachman, D.; Folstein, M.; Katzman, R.; Price, D.; Stadlan, E.M. Clinical diagnosis of Alzheimer’s disease: Report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 1984, 34, 939–944.
- Dubois, B.; Feldman, H.H.; Jacova, C.; DeKosky, S.T.; Barberger-Gateau, P.; Cummings, J.L.; Delacourte, A.; Galasko, D.; Gauthier, S.; Jicha, G.A.; et al. Research Criteria for the Diagnosis of Alzheimer’s Disease: Revising the NINCDS–ADRDA Criteria. Lancet Neurol. 2007, 6, 734–746.
- Dubois, B.; Feldman, H.H.; Jacova, C.; Cummings, J.L.; DeKosky, S.T.; Barberger-Gateau, P.; Delacourte, A.; Frisoni, G.; Fox, N.C.; Galasko, D.; et al. Revising the definition of Alzheimer’s disease: A new lexicon. Lancet Neurol. 2010, 9, 1118–1127.
- Dubois, B.; Feldman, H.H.; Jacova, C.; Hampel, H.; Molinuevo, J.L.; Blennow, K.; DeKosky, S.T.; Gauthier, S.; Selkoe, D.; Bateman, R.; et al. Advancing research diagnostic criteria for Alzheimer’s disease: The IWG-2 criteria. Lancet Neurol. 2014, 13, 614–629.
- Jack, C.R., Jr.; Albert, M.; Knopman, D.S.; McKhann, G.M.; Sperling, R.A.; Carillo, M.; Thies, W.; Phelps, C.H. Introduction to revised criteria for the diagnosis of Alzheimer’s disease: National Institute on Aging and the Alzheimer Association Workgroups. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2011, 7, 257.
- Zhou, J.; Benoit, M.; Sharoar, G. Recent advances in pre-clinical diagnosis of Alzheimer’s disease. Metab. Brain Dis. 2021, 37, 1703–1725.
- Sperling, R.A.; Aisen, P.S.; Beckett, L.A.; Bennett, D.A.; Craft, S.; Fagan, A.M.; Iwatsubo, T.; Jack, C.R., Jr.; Kaye, J.; Montine, T.J.; et al. Toward Defining the Preclinical Stages of Alzheimer’s Disease: Recommendations from the National Institute on Aging-Alzheimer’s Association Workgroups on Diagnostic Guidelines for Alzheimer’s Disease. Alzheimer’s Dement. 2011, 7, 280–292.
- Chao, L.; Mueller, S.; Buckley, S.; Peek, K.; Raptentsetseng, S.; Elman, J.; Yaffe, K.; Miller, B.; Kramer, J.; Madison, C.; et al. Evidence of neurodegeneration in brains of older adults who do not yet fulfill MCI criteria. Neurobiol. Aging 2010, 31, 368–377.
- Albert, M.S.; DeKosky, S.T.; Dickson, D.; Dubois, B.; Feldman, H.H.; Fox, N.C.; Gamst, A.; Holtzman, D.M.; Jagust, W.J.; Petersen, R.C.; et al. The Diagnosis of Mild Cognitive Impairment due to Alzheimer’s Disease: Recommendations from the National Institute on Aging-Alzheimer’s Association Workgroups on Diagnostic Guidelines for Alzheimer’s Disease. Alzheimer’s Dement. 2011, 7, 270–279.
- McGrattan, A.M.; Pakpahan, E.; Siervo, M.; Mohan, D.; Reidpath, D.D.; Prina, M.; Allotey, P.; Zhu, Y.; Shulin, C.; Yates, J. Risk of conversion from mild cognitive impairment to dementia in low-and middle-income countries: A systematic review and meta-analysis. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2022, 8, e12267.
- McKhann, G.M.; Knopman, D.S.; Chertkow, H.; Hyman, B.T.; Jack, C.R., Jr.; Kawas, C.H.; Klunk, W.E.; Koroshetz, W.J.; Manly, J.J.; Mayeux, R. The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s Dement. 2011, 7, 263–269.
- Hyman, B.T.; Phelps, C.H.; Beach, T.G.; Bigio, E.H.; Cairns, N.J.; Carrillo, M.C.; Dickson, D.W.; Duyckaerts, C.; Frosch, M.P.; Masliah, E. National Institute on Aging–Alzheimer’s Association guidelines for the neuropathologic assessment of Alzheimer’s disease. Alzheimer’s Dement. 2012, 8, 1–13.
- Risacher, S.; Saykin, A.; Wes, J.; Shen, L.; Firpi, H.; McDonald, B. Baseline MRI Predictors of Conversion from MCI to Probable AD in the ADNI Cohort. Curr. Alzheimer Res. 2009, 6, 347–361.
- Qiu, A.; Fennema-Notestine, C.; Dale, A.M.; Miller, M.I. Regional shape abnormalities in mild cognitive impairment and Alzheimer’s disease. Neuroimage 2009, 45, 656–661.
- Guévremont, D.; Tsui, H.; Knight, R.; Fowler, C.J.; Masters, C.L.; Martins, R.N.; Abraham, W.C.; Tate, W.P.; Cutfield, N.; Williams, J.M. Plasma microRNA vary in association with the progression of Alzheimer’s disease. Alzheimer’s Dement. 2022, 14, e12251.
- Mesa-Herrera, F.; Marin, R.; Torrealba, E.; Santos, G.; Diaz, M. Neuronal ER-Signalosome Proteins as Early Biomarkers in Prodromal Alzheimer’s Disease Independent of Amyloid-beta Production and Tau Phosphorylation. Front. Mol. Neurosci. 2022, 15, 1–20.
- Shahid, S.S.; Wen, Q.; Risacher, S.L.; Farlow, M.R.; Unverzagt, F.W.; Apostolova, L.G.; Foroud, T.M.; Zetterberg, H.; Blennow, K.; Saykina, A.J.; et al. Hippocampal-subfield microstructures and their relation to plasma biomarkers in Alzheimer’s disease. Brain 2022, 145, 2149–2160.
- Vaghari, D.; Kabir, E.; Henson, R.N. Late combination shows that MEG adds to MRI in classifying MCI versus controls. Neuroimage 2022, 252, 119054.
- Klöppel, S.; Stonnington, C.M.; Chu, C.; Draganski, B.; Scahill, R.I.; Rohrer, J.D.; Fox, N.C.; Jack, C.R., Jr.; Ashburner, J.; Frackowiak, R.S. Automatic classification of MR scans in Alzheimer’s disease. Brain 2008, 131, 681–689.
- Janousova, E.; Vounou, M.; Wolz, R.; Gray, K.R.; Rueckert, D.; Montana, G. Biomarker discovery for sparse classification of brain images in Alzheimer’s disease. Ann. BMVA 2012, 2, 1–11.
- Zhang, D.; Wang, Y.; Zhou, L.; Yuan, H.; Shen, D. Multimodal classification of Alzheimer’s disease and mild cognitive impairment. Neuroimage 2011, 55, 856–867.
- Liu, S.; Song, Y.; Cai, W.; Pujol, S.; Kikinis, R.; Wang, X.; Feng, D. Multifold Bayesian kernelization in Alzheimer’s diagnosis. In Proceedings of the International Conference on Medical Image Computing and Computer-Assisted Intervention, Nagoya, Japan, 22–26 September 2013; Springer: Berlin/Heidelberg, Germany, 2013.
- Zhang, Y.; Dong, Z.; Phillips, P.; Wang, S.; Ji, G.; Yang, J.; Yuan, T.-F. Detection of subjects and brain regions related to Alzheimer’s disease using 3D MRI scans based on eigenbrain and machine learning. Front. Comput. Neurosci. 2015, 9, 66.
- Hong, S.; Coelho, C.A.; Park, J. An Exact and Near-Exact Distribution Approach to the Behrens–Fisher Problem. Mathematics 2022, 10, 2953.
- Esteki, S.; Naghsh-Nilchi, A.R. Frequency component Kernel for SVM. Neural Comput. Appl. 2022, 34, 22449–22464.
- Nayak, J.; Swapnarekha, H.; Naik, B.; Dhiman, G.; Vimal, S. 25 Years of Particle Swarm Optimization: Flourishing Voyage of Two Decades. Arch. Comput. Methods Eng. 2022, 30, 1663–1725.
- Sonoda, S.; Murata, N. Neural network with unbounded activation functions is universal approximator. Appl. Comput. Harmon. Anal. 2017, 43, 233–268.
- McKinney, S.M.; Sieniek, M.; Godbole, V.; Godwin, J.; Antropova, N.; Ashrafian, H.; Back, T.; Chesus, M.; Corrado, G.S.; Darzi, A.; et al. International evaluation of an AI system for breast cancer screening. Nature 2020, 577, 89–94.
- Zaidi, S.M.A.; Habib, S.S.; Van Ginneken, B.; Ferrand, R.A.; Creswell, J.; Khowaja, S.; Khan, A. Evaluation of the diagnostic accuracy of Computer-Aided Detection of tuberculosis on Chest radiography among private sector patients in Pakistan. Sci. Rep. 2018, 8, 12339.
- Kang, D.; Park, J.E.; Kim, Y.-H.; Kim, J.H.; Oh, J.Y.; Kim, J.; Kim, Y.; Kim, S.T.; Kim, H.S. Diffusion radiomics as a diagnostic model for atypical manifestation of primary central nervous system lymphoma: Development and multicenter external validation. Neuro-Oncology 2018, 20, 1251–1261.
- Feng, X.; Provenzano, F.A.; Small, S.A.; Initiative, F.T.A.D.N. A deep learning MRI approach outperforms other biomarkers of prodromal Alzheimer’s disease. Alzheimer’s Res. Ther. 2022, 14, 1–11.
- Zheng, Q.; Zhang, Y.Y.; Li, H.L.; Tong, X.R.; Ouyang, M.H. How segmentation methods affect hippocampal radiomic feature accuracy in Alzheimer’s disease analysis? Eur. Radiol. 2022, 32, 6965–6976.
- Aly, M.F.A.; Kleijn, S.A.; van Lenthe, J.H.; Menken-Negroiu, R.F.; Robbers, L.F.; Beek, A.M.; Kamp, O. Prediction of prognosis in patientswith left ventricular dysfunction using three-dimensional strain echocardiography and cardiac magnetic resonance imaging. Neth. Heart J. 2022, 30, 572–579.
- Khojaste-Sarakhsi, M.; Haghighi, S.S.; Ghomi, S.; Marchiori, E. Deep learning for Alzheimer’s disease diagnosis: A survey. Artif. Intell. Med. 2022, 130, 102332.
- Reith, F.H.; Mormino, E.C.; Zaharchuk, G. Predicting future amyloid biomarkers in dementia patients with machine learning to improve clinical trial patient selection. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2021, 7, e12212.
- Kim, Y.; Jiang, X.; Giancardo, L.; Pena, D.; Bukhbinder, A.S.; Amran, A.Y.; Schulz, P.E.; Initiative, A.D.N. Multimodal Phenotyping of Alzheimer’s Disease with Longitudinal Magnetic Resonance Imaging and Cognitive Function Data. Sci. Rep. 2020, 10, 5527.
- Chen, K.T.; Gong, E.; Macruz, F.B.D.C.; Xu, J.; Boumis, A.; Khalighi, M.; Poston, K.L.; Sha, S.J.; Greicius, M.D.; Mormino, E.; et al. Ultra–Low-Dose18F-Florbetaben Amyloid PET Imaging Using Deep Learning with Multi-Contrast MRI Inputs. Radiology 2019, 290, 649–656.
More
Information
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
528
Revisions:
2 times
(View History)
Update Date:
25 Jun 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





