Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Gundula Schulze-Tanzil | -- | 4043 | 2023-06-15 12:39:59 | | | |
| 2 | Peter Tang | Meta information modification | 4043 | 2023-06-16 03:48:56 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Gögele, C.; Hahn, J.; Schulze-Tanzil, G. Anatomical Tissue Engineering of Anterior Cruciate Ligament Entheses. Encyclopedia. Available online: https://encyclopedia.pub/entry/45645 (accessed on 06 June 2026).
Gögele C, Hahn J, Schulze-Tanzil G. Anatomical Tissue Engineering of Anterior Cruciate Ligament Entheses. Encyclopedia. Available at: https://encyclopedia.pub/entry/45645. Accessed June 06, 2026.
Gögele, Clemens, Judith Hahn, Gundula Schulze-Tanzil. "Anatomical Tissue Engineering of Anterior Cruciate Ligament Entheses" Encyclopedia, https://encyclopedia.pub/entry/45645 (accessed June 06, 2026).
Gögele, C., Hahn, J., & Schulze-Tanzil, G. (2023, June 15). Anatomical Tissue Engineering of Anterior Cruciate Ligament Entheses. In Encyclopedia. https://encyclopedia.pub/entry/45645
Gögele, Clemens, et al. "Anatomical Tissue Engineering of Anterior Cruciate Ligament Entheses." Encyclopedia. Web. 15 June, 2023.
Copy Citation
The firm integration of anterior cruciate ligament (ACL) grafts into bones remains the most demanding challenge in ACL reconstruction, since graft loosening means graft failure. For a functional-tissue-engineered ACL substitute to be realized in future, robust bone attachment sites (entheses) have to be re-established. The latter comprise four tissue compartments (ligament, non-calcified and calcified fibrocartilage, separated by the tidemark, bone) forming a histological and biomechanical gradient at the attachment interface between the ACL and bone. The ACL enthesis is surrounded by the synovium and exposed to the intra-articular micromilieu.
ACL
enthesis
ligament
synovioentheseal complex knee
tissue engineering
triphasic and graded scaffold
fibrocartilage
bone—ligament interface
zonality
tidemark
1. Introduction
Due to their high biomechanical loading, ACL entheses are prone to injury. As reported previously, many ruptures of ACL and ACL grafts can be observed at the area of the femoral enthesis; in particular, fibers of the posterolateral ACL bundle have a high risk of rupture during pivot landings [1] (Figure 1). Typically, autografts or allografts are used for reconstruction. Hence, the bone attachment site represents a major point of weakness after ACL reconstruction [2], since most of the techniques do not properly restore the entheses [3]. The reasons for this include a poor spontaneous healing ability and the fact that the regenerative tissue in the bone attachment site is mostly composed of poorly organized Sharpey-like fibers, leading to the insufficient stability of the graft [4]. Sharpey fibers represent connective tissue fibers mainly consisting of collagen type I which attach to the periosteum, but do not reflect a gradual and biomechanically competent fibrocartilaginous enthesis. In addition, several studies have shown that impaired healing can lead to bone tunnel widening and a consequent laxity of the graft as synovial fluid infiltrates into the tunnel [5][6][7], thereby impairing integration. The consequences can include knee instability after ACL graft loosening [2] and even osteoarthritis (OA) [8]. The onset of OA, which is associated with an inflammatory microenvironment, might further interfere with the integration process.
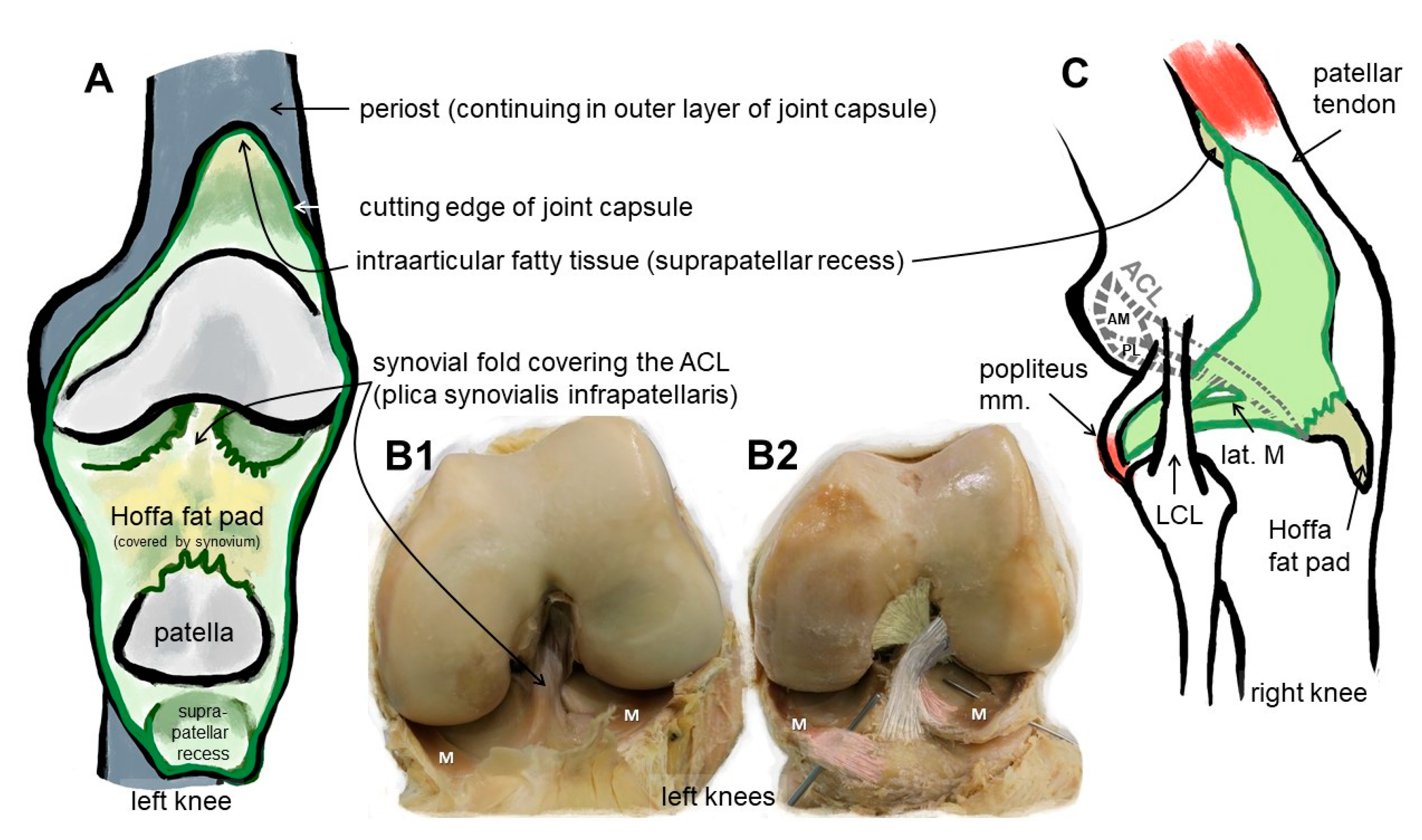
Figure 1. Macroscopical anatomy of the human ACL. Scheme of the insight into the knee joint cavity after the joint capsule of the knee is opened and its inner surface covered by the synovial membrane (except for the articulating surfaces) is shown (A). (B1) shows the same gross anatomical image and (B2) visualizes by coloration the relation of the ACL with anteromedial (beige) and posterolateral bundle (grey), menisci (M) and anterior meniscotibial ligaments (rose). (C) The extension of the synovial membrane is shown (lateral view, fibrous capsule layer removed). ACL: anterior cruciate ligament, AM: anteromedial bundle, lat.: lateral, LCL: lateral collateral ligament, M: meniscus, m: muscle, PL: posterolateral bundle. The image was created by G. Schulze-Tanzil using krita 4.1.7.
2. Anatomy: The ACL Entheses
Since the enthesis acts as a functional unit allowing stress dissipation from the stretched ligament to its bone attachment sites, an organ concept arose: in the case of the enthesis of the Achilles tendon, it has been designated as an organ underlining the synergistic function of the different tissues (including the fat pad, fibrocartilage and synovial structures) forming and surrounding the enthesis [9]. This concept is transferable to the ACL entheses, where similar neighboring tissues (Hoffa fat pad, synovial fold and fibrocartilage) might contribute to their overall functions and homeostasis. An enthesis of the most common fibrocartilaginous type comprises four tissue compartments of increasing stiffness (ligament, non-calcified and calcified fibrocartilage, separated by the tidemark as a barrier between both and the underlying bone), generating a biomechanical gradient at the attachment to bone (Figure 2). The borders between the zones show undulating interdigitations [10], increasing the overall surface of contact areas and hence providing sufficient stability for repetitive loading. The tidemark is also a barrier between two nutrient diffusion fronts, from the subchondral bone and from the vascularized ACL midsubstance and outer synovial layer [11][12][13].
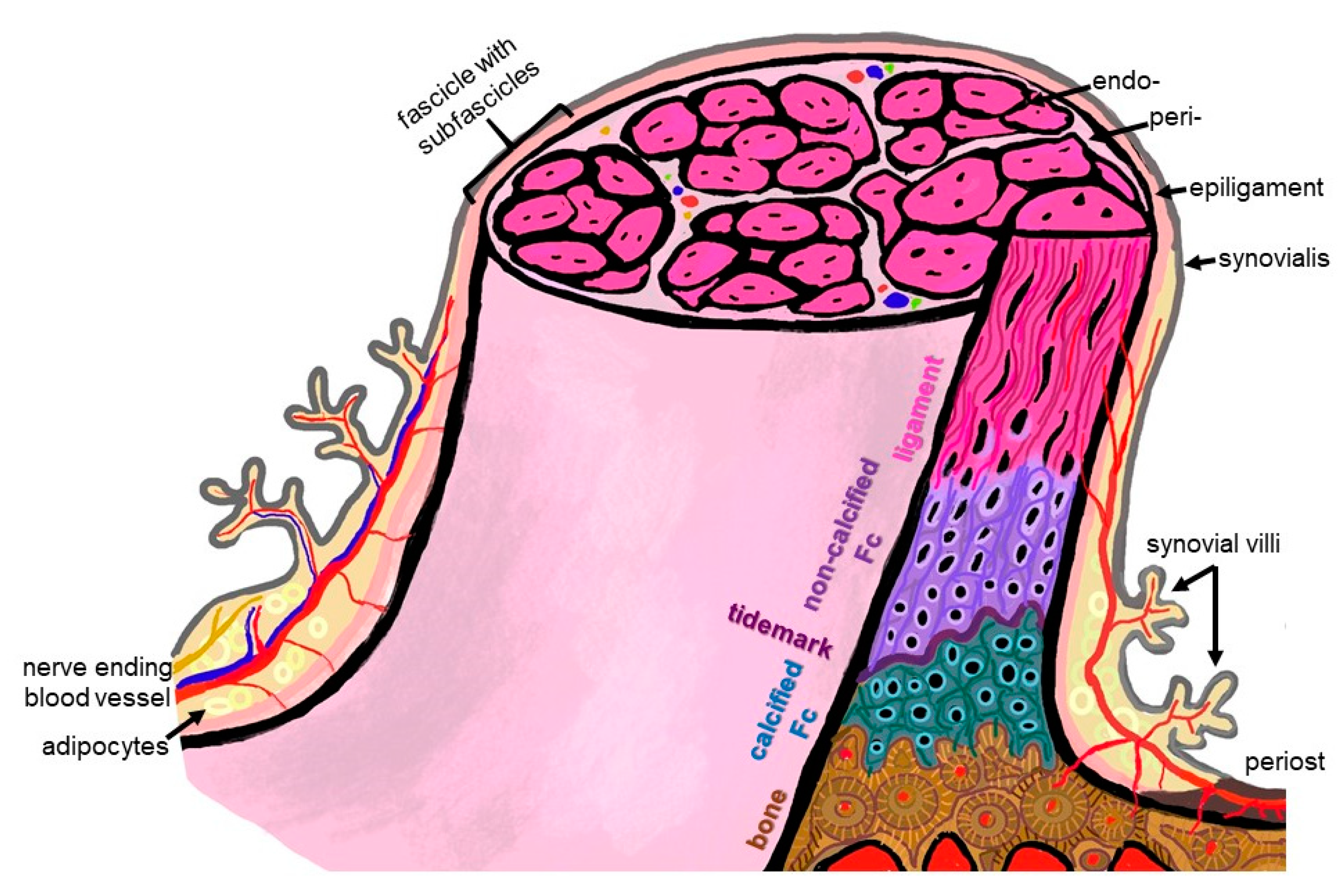
Figure 2. Scheme summarizing some histological features of the ACL enthesis. The ACL, covered by the synovial layer and embraced by the epiligament, is hierarchically organized in bundles (surrounded by the periligament) and sub bundles (encircled by the endoligament). Epi-, peri- and endoligament sheets represent flexible connective tissue layers, which also contain blood and lymphatic vessels as well as nerves. The enthesis comprises a graduated attachment into bone: starting with the ligament phase with increasing glycosaminoglycan/proteoglycan contents followed by non-vascularized and non-calcified fibrocartilage (FC), a tidemark forming the border to the calcification front of the calcified FC also lacking blood vessels down to the vascularized bone. The phases thoroughly interdigitate with each other. From ligament to bone, the ECM composition and collagen fiber quality and alignment change from anisotrop to isotrop. The image was created by G. Schulze-Tanzil using krita 4.1.7.
3. Differences between the Femoral and Tibial ACL Entheses
The femoral and tibial entheses of the ACL differ in regard to their overall and zone dimensions, fibrocartilage amount and fiber insertion angles [14]. The femoral enthesis had a 3.9-fold significantly sharper attachment angle of the fibers compared with that of the tibial enthesis. It also showed a significantly larger (43%) calcified and significantly greater (226%) non-calcified fibrocartilage zone, particularly in the central attachment area. The authors reported that these differences might provide an explanation of why it is prone to injury, since there possibly arises a stress concentration at the inferior border of the ACL’s femoral enthesis [14]. The femoral enthesis has a rather semilunar shape or represents a segment of a circle [15], whereas the tibial exhibits rather an elliptic attachment area [16]. The dimensions were reported as 11–24 mm for the long diameter for the femoral attachment zone and 17 × 11 mm as diameters for the tibial one [15].
Moreover, another study showed that the two bundles of the ACL (AM: anteromedial and PL: posterolateral) displayed different insertion modes, whereby the AM bundle formed more intimate interdigitations with the subligamentous cortical bone compared to the PL bundle attachment site, suggesting structure–function relationships and underlining the importance of sub-bundle anatomy [17], which could be incorporated in ACL reconstruction strategies in future.
A cadaver study revealed that two millimeters from its direct femoral attachment, the ACL fiber bundles formed a flat ribbon in all investigated donors and both, the AM and PL bundles, were barely separated in this area. This ribbon followed continuously the posterior femoral bone cortex. It had a width of between 11.43 and 16.18 mm and was only 2.54–3.38 mm thick [18].
4. ACL and Enthesis Development
Healing and reconstruction often recapitulate stages of tissue development. Hence, it is important to understand the key developmental processes of the ACL and its entheses. The ACL becomes visible before joint cavitation and starts its development at the ventral side of the future knee joint, invaginating gradually into the intercondylar space of the joint but remaining in an extrasynovial position over the entire time until adulthood. It migrates posteriorly. Initially, it derives from the same blastoma as the menisci, suggesting that both share functional aspects [15]. After birth, maturational changes (increase in collagen content and mineralization, but lesser collagen fiber alignment) can be observed in the ACL enthesis as a result of loading and remodeling as shown in bovine samples [19].
The clonal expansion of Growth and Differentiation Factor (GDF)5-positive progenitors triggers enthesis formation [20]. As crucial developmental actors, Indian hedgehog (Ihh), ParaThyroid Hormone related protein (PTHrP) and Patched 1 (Ptc1) were localized in the developing enthesis. The latter are known to regulate chondrocyte proliferation and differentiation, being in a negative feedback interrelation. The transcription factors SOX9 and scleraxis regulating several key ECM gene expressions also seem to contribute to enthesis formation. The zone-specific expression of these factors might be responsible for graded tissue formation at the enthesis [21]. Before mineralization, collagen-type-I-producing cells become associated with collagen-type-II-synthesizing cells. As a starting point of mineralization, cells at the base of the enthesis start to express Ihh, alkaline phosphatase and collagen type X. Ihh-responsive glioma associated oncogene homologue (Gli)1-positive cells differentiate from non-mineralized ECM producing fibrochondrocytes in those synthesizing a calcifying ECM [20]. One could estimate that the final ECM expression pattern in the maturating enthesis is strongly regulated by mechanobiology [22].
5. Histoarchitecture, Cellular and Biochemical Pecularities
The ECM of the different enthesis zones shows a unique expression profile. It comprises ECM components more typical of the dense regular connective tissue of the ACL (collagen types I and III) or more related to fibrocartilage (collagen type II, aggrecan), the latter to compensate pressure as well as components associated with calcification, e.g., collagen type X and hydroxyapatite. The orientation of the collagen fibers differs, e.g., more or less following one main direction (anisotrop, in response to unidirectional stress) or interwoven (isotrop) to compensate stress from different directions. The transition between zones is not abrupt, but forms a gradient [21]. The fibrocartilage of the enthesis generates a natural barrier separating the blood supply of the bone from that of the tendon/ligament [9].
6. Sex- and Training-Dependent Individual Aspects
It is well known that the anatomical dimensions of the knee joint differ substantially between females and males [23][24]. Sex-dependent differences in knee kinematics and neuromuscular control could also be observed [25]. It has been recommended to analyze sex-dimorphic landing mechanics data to understand differences in female and male ACL anatomy in the future in regard to estimating rupture risks [26]. In a recent study [27], the risk of rupture was 8.3 times higher in females than in males. Moreover, at the molecular level, the proteome of the ACL differed between female and male individuals: Comparing female with male ACL donors, alcohol dehydrogenase 1B and complement component 9 were more highly represented in female than in male. On the contrary, myocilin was the major protein detectable to higher degrees in males than in females [28]. The influence of estrogen on collagen in females might also be of interest in explaining sex-dependent higher risks of ACL injuries [29][30]. The menstrual cycle goes along with time-dependent differences in the biomechanical properties of the female ACL [31]. Not only this, in response to biomechanical challenges the ACL entheses comprise individual (influenced by personal loading history) architectures [1]. In addition to this, single-nucleotide polymorphisms of genes encoding for collagen correlated with the risk of non-contact ACL rupture in females [27]. The ACL can hypertrophy, showing increased cross-sectional areas in response to training in athletes [32]. However, the effect of extensive loading on the hypertrophy/remodeling of its enthesis is unclear. Individual aspects of the ACL were investigated to identify anatomical risk factors for rupture in males and females [33].
In regard to individual and sex-dependent risks, anatomical criteria such as the femoral notch diameter have been thoroughly investigated, with the hypothesis of a narrow notch, e.g., in females, being associated with higher risks of ACL failure, but not all published studies support this relation [15]. The posterior tibial slope was higher in females with ACL rupture compared to males [34].
7. ACL Animal Models
Animal models have been used extensively for experimental ACL reconstruction studies, including rabbit, pig, goat and sheep models. Since rodents (mice and rats) are too small, and in view of the costs of large animals, many of the ACL reconstruction studies are performed in rabbits [35][36]. Dogs, which are, in veterinary praxis, often patients with ACL rupture, are also sometimes used for experimental studies [37][38]. In view on these models, one should be aware of substantial species differences in the ACL anatomy, e.g., two anteriorly completely separately attaching anteromedial and posterolateral bundles in the porcine, caprine and sheep ACL models and larger dimensions of the pig ACL compared to that of humans [38][39][40]. Bascunan et al. concluded, based on their helpful review of comparative ACL anatomy and comparative biomechanical aspects, that the caprine and porcine models might show the closest similarity with human conditions [38]. However, the human knee is absolutely unique, e.g., in regard to tibial slope and plateau shape [38] and also concerning the ACL responding to loading during upright gait. Therefore, research approaches for anatomical ACL reconstruction using human ACL modeling/simulation data, e.g., derived from cadaver dissection, imaging or artificial human ACL models yet to be developed, would be very helpful in future.
8. In Silico ACL Modeling
In silico models based on human anatomical data sets from imaging, e.g., MRT or computer tomography, will become more important in future for research and clinical applications [41]. With an appropriate model of the knee for finite element (FE) analysis, it is feasible to simulate joint kinematics and kinetics. Furthermore, this enables statements to be made on failure behavior [42], reconstruction/fixation strategies [43][44] and implant design [45][46]. The development of adequate models is a complex and resource-intensive process. It requires a comprehensive workflow with data collection based on images and mechanical testing, an image segmentation step, surface geometry generation and meshing for finite element analysis as well as the definition of simulation settings [47]. Before the simulation step occurs, the model has to be customized by defining the material, loading and boundary conditions [48]. Since biological tissues exhibit highly anisotropic material behavior, the definition of material parameters and boundary conditions is a major challenge, especially when modeling the ACL enthesis as a connection between dissimilar tissue types over a small region. For comparison, bone is described as rigid and has a tensile strength of 120–180 MPa, a Young’s modulus of 15,000–25,000 MPa and a strain at failure between 2 and 3% [49]. In contrast, ligament tissue such as the ACL exhibits a tensile strength of 22–27 MPa, a Young’s modulus of 100–130 MPa and strain at failure between 25 and 30% [50].
There have been many mathematical and computational models, such as classical continuum structural mechanics approaches [51] or discrete fiber network models [52], developed focusing on different attachment modes with specific compositional and microstructural adaptations [53]. Hence, these modeling strategies address the mechanical role of fiber architecture [19][51][54], mineral reinforcements [55][56], surface roughness and interlocking [57].
Besides the different modeling approaches, the definition of the material properties is an essential point [55]. For this purpose, methods such as Fourier transform infrared spectroscopy imaging (FTIRI) are used to determine the collagen or proteoglycan content and the mineral distribution or extracellular matrix (ECM) composition [19][54]. Biomechanical investigations of the knee in terms of laxity tests in various degrees of freedom (DOF) [46] or traditional mechanical testing methods, such as uniaxial tensile tests, are required to determine characteristic properties, e.g., elastic modulus, tensile strength or displacement, and to ensure the correct prediction of knee motion and loading. Moreover, the anisotropic material properties of the ACL should be re-evaluated by full-field displacement measurements, which are independent of specimen geometry [47][58][59].
The body of literature focusing on the modeling and simulation of the ligament–bone complex with detailed information of the enthesis geometry and structural design is still lacking. Luetkemeyer et al. investigated the influence of ACL enthesis shape and attachment angle on the strain distribution in an FE model and demonstrated the possibility of making predictions in regard to injury risks [60]. Additionally, the constitutive model was further developed using full-field methods for ACL bundle modeling accounting for strain and principal material direction (collagen fiber splay) heterogeneity as well as enthesis shape [61]. Hence, it remains challenging to bridge the gap between clinical requirements and ACL modeling strategies, but this is essential for developing new substitutes for ACL TE approaches comprising the bone attachment. Machine learning could also be applied to predict cell behavior on scaffolds to reduce time-consuming and expensive experiments. This was demonstrated for cardiac tissue engineering [62]. Nevertheless, this approach needs sufficient data from the literature.
These models could be used to select optimized scaffold/cell compositions and develop a stable fixation variant for the tissue engineered graded scaffolds to rebuild the enthesis before starting with in vivo tests.
9. Strategies for Multiphasic Enthesis Scaffolds for ACL Tissue Engineering
To address the challenge of firm attachment of the ACL substitute onto the subligamentous bone, enthesis TE is required. To rebuild the graded structure of the ACL enthesis means creating a bi-, or better, triphasic topology of the cell carrier. Furthermore, a significant impact on enthesis formation can be achieved by using functionalized or bioactive materials. The latter should be a transient template with a degradation profile adapted to neotissue formation, but resilient enough as a tissue-engineered ACL substitute to withstand loading. So far, developed gradient scaffolds and helpful techniques for creating gradients in vitro have been reviewed previously [63][64]. To realize the concept of multiphasic scaffolds, several material and technical approaches were designed. Nevertheless, considering the radial hierarchical structure of the ACL (Figure 2), in addition to the longitudinal triphasic composition, a radial gradient could also be addressed in future as realized by the electrospinning of synthetic polymers for the reconstruction of other tissues [65].
10. Biochemical Instruction of Cells Used for ACL Tissue Engineering
Different factors have been applied to enforce the enthesis lineage differentiation of different cell types. Factors were either added separately or integrated into the scaffold. As an early in vitro approach to address enthesis formation, a biphasic scaffold was generated and seeded with fibroblasts consisting of a fibrin gel with TGFβ (subtype not specified) and brushite supplementation to form the enthesis part. An enthesis-like tissue formation with a border between both parts resembling a tidemark could be detected [66]. A more recent study also based on a hydrogel described a tendency of multilineage differentiation of MSCs through the spatial presentation of growth factors (BMP-4 and TGFβ3) in a multiphasic gelatin-methacrylate hydrogel for enthesis cell type differentiation [67].
For tendon/ligament reconstruction TGFβ2 and GDF5 were zone-specifically tethered to a biphasic silk fibroin scaffold. In addition, anisotropic (ligament part) and isotropic (bone/cartilage part) pore alignment was arranged. TGFβ2 alone triggered in adipose-tissue-derived stem cells (ASC) the expression of ligament-related factors, whereas in combination with GDF5 and an isotropic scaffold topology, it favored cartilage marker expression [68]. In another study, lentiviral vectors encoding TGFβ3 and BMP-2 were selected and integrated using phosphatidylserine as an anchor for silk scaffold fibrocartilage and bone zones in ACL constructs to support enthesis cell differentiation [69]. TGFβ2 and TGFβ3 have time-dependent effects [70]. Some lineage-related differentiation of MSCs in response to TGFβ3 and BMP-2 could be shown by Fan et al. [69]. A poly-L-lactic acid scaffold with a bone and ligament compartment was braided and supplemented with BMP-2 before being tested in the rabbit ACL reconstruction model. The BMP-2 supplementation led to a trend of smaller bone tunnel cross-sectional areas [35].
Acidic fibroblast growth factor (FGF)2, combined with collagen, was added to the bone tunnel in which autografts in a rabbit ACL reconstruction model were fixed to support bone integration. This treatment induced well-organized Sharpey fiber and a fibrocartilage transition zone formation as a sign of firm integration [36].
In a comparable approach (rabbit ACL reconstruction model), osteoprotegerin (OPG) combined with deproteinized xenograft bone were installed into the bone tunnel. It indeed improved the bone integration of the tendon grafts compared to the groups with no OPG/bone treatment [71].
Despite another study not representing a true ACL implantation approach, it was interesting to see that hepatocyte growth factor (HGF) led to enthesis-like tissue formation in a tendon bone healing model in rabbits [72]. Looking at the promising results of the above-listed studies, one has to bear in mind that the rabbit model is well known to exert a high healing potential, which is possibly not fully transferable to human conditions.
Cellular and biochemical gradients can be applied using a novel bioreactor [73]. There are other promising factors which have so far not been investigated in much detail for ACL enthesis TE: Periostin, which seems to contribute to fibrocartilage layer growth, and is involved in ACL enthesis fibrocartilage layer formation [74][75], could be of major importance; moreover, distinct splice variants of periostin seem to be indicative either for ACL precursor cells or chondrocytes [76]. Additionally, cell stimulation, which has not been used for enthesis TE but for ACL, with mechanogrowth factor (MGF), which is an IGF1 splicing variant, is another candidate [77]. It stimulated ACL fibroblast proliferation and collagen synthesis [78][79].
11. Ligamentogenic/Entheseal Differentiation of Cells for ACL Enthesis Tissue Engineering
Ligament fibroblasts (ligamentocytes) have a slow growth rate [80] and are difficult to access. Theoretically, they could be harvested from the stumps of a ruptured ACL, isolated and after sufficient cell expansion be used for the TE construct. A more accessible cell source with full ligamentogenic differentiation potential might be iliotibial-band-derived fibroblasts [81].
The plasticity of tendon cells is known; when the tendon goes around a pulley, fibrocartilage develops in response to compressive loading [82]. In tendons, different stem cell niches have been shown [83] which could also be hypothesized for ligaments. It remains a point of discussion whether resident stem cell populations are responsible for aberrant lineage differentiation mediating tissue metaplasia in pathological ACLs [84][85], and whether enthesis neoformation in implanted grafts or even differentiated ligament fibroblasts can transdifferentiate. By single-cell sequencing, Fang et al. showed a stem cell pool (Gli+ progenitor population) important for tendon enthesis formation and healing [86]. This Gli1+ progenitor cell population can differentiate into fibrocartilage-producing cells [87]. This cell population remains to be shown for the ACL.
ASCs were easily isolated from lipoaspirates [80][88], and hence represent an attractive source for musculoskeletal TE. However, the in vitro supplementation of human ASCs with anabolic growth factors (TGFβ1, insulin-like growth factor (IGF), epidermal growth factor (EGF) and FGF were tested) was not sufficient for commitment into the ligamentogenic lineage [80]. On the contrary, ASC cell sheets promoted ligament bone healing in a rabbit model [89]. ASCs applied to tendon enthesis scaffolds supplemented with HA on the bone and chondroitinsulphate on the ligament part expressed the typical ECM components of tendon in the tendon zone with the aligned fibers, or bone tissue in the random mineralized part [90]. In another study, ASCs changed their expression profile depending on the pore alignment in multiphasic enthesis scaffolds [91]. The autologous stromal vascular fraction of ASCs, an adipose-derived, stem cell-rich isolate from knee fat pads, was also used to seed silk fibroin scaffolds which were subsequently successfully tested in an ovine ACL reconstruction model. However, the implantation of the cell-seeded scaffolds did not lead to histologically different results compared to cell-free implantation in regard to bone integration [92][93].
Many of the aforementioned ACL TE studies based on BMSCs were tested on a multiphasic scaffold dotted with GFs which promoted their lineage differentiation [69].
Interesting is the observation that even skin fibroblasts seeded on collagen scaffolds and implanted in a goat model led to formation of enthesis-like tissue in vivo with Sharpey fibers and fibrocartilage [94]. Nevertheless, it remains unclear whether fibroblast plasticity or the trophic factors released by them triggered the observed tissue formation.
12. Co-Cultures for Zonal ACL Tissue Engineering and Mechanical Training of Cells
Several co-culture approaches have been reported for ACL tissue-engineered constructs. A trilineage co-culture system (osteoblasts–BMSCs–ligament-derived fibroblasts) was established on a hybrid silk scaffold. Particularly, the influence of TGFβ3 was tested, but seemed rather to inhibit the mineralization [95]. A dual-chamber system was used with blended fibrin–alginate hydrogel scaffolds for ligament-interface TE. The endochondral, fibrocartilaginous or ligamentous attachments were triggered by the spatial exposition of the same cell type (MSCs) to growth factors such as TGFβ3 [96]. Mesenchymal stem cells (BMSCs), chondrocytes and osteoblasts were co-cultured on triphasic silk enthesis scaffolds. The construct showed a successful integration into bone in the rabbit ACL reconstruction model [97]. Fibroblasts and osteoblasts were co-cultured on a triphasic scaffold (melt-electrospun/written) with different grid crimp microtopologies, which supported lineage differentiation [98].
In the cases of co-culturing zone-related cell types of the enthesis on the same area, directed seeding strategies are required. Different approaches to achieve an exact position of the cells on the constructs have been applied. The usage of spheroids is a method to bring cells into porous scaffolds. Cells stored in the spheroids emigrate on the carrier. The size of the spheroids could be adapted to the pore size. It is known that ACL ligamentocytes maintain their expression profile in spheroids [99][100][101][102][103]. Using cell sheets is another tool to place cells on a defined position on the scaffolds, leading to cell emigration on them [104]. Capturing cells within a hydrogel and fixing them in the scaffold presents another tool of directed seeding. This could be combined with bioprinting, which allows cells or cell aggregates to be placed exactly in the area wanted. Cells can be mixed into a hydrogel; however, the contact to the scaffold material is not given in the beginning, and it could happen that cells fall out and are lost when the hydrogel starts degradation. Another challenge in co-culturing cells with different proliferative activity might be the overgrowth of zones by one cell type. Hence, the concept of a transient barrier integrated into scaffolds to inhibit cell overgrowth was proposed [105]. This barrier should allow nutrition to pass through and disappear at the time point that it is not required anymore.
References
- Beaulieu, M.L.; Carey, G.E.; Schlecht, S.H.; Wojtys, E.M.; Ashton-Miller, J.A. On the heterogeneity of the femoral enthesis of the human ACL: Microscopic anatomy and clinical implications. J. Exp. Orthop. 2016, 3, 14.
- Chen, C.H. Strategies to enhance tendon graft–bone healing in anterior cruciate ligament reconstruction. Chang. Gung Med. J. 2009, 32, 483–493.
- Lu, H.; Chen, C.; Xie, S.; Tang, Y.; Qu, J. Tendon Healing in Bone Tunnel after Human Anterior Cruciate Ligament Reconstruction: A Systematic Review of Histological Results. J. Knee Surg. 2019, 32, 454–462.
- Tabuchi, K.; Soejima, T.; Kanazawa, T.; Noguchi, K.; Nagata, K. Chronological changes in the collagen-type composition at tendon-bone interface in rabbits. Bone Joint Res. 2012, 1, 218–224.
- Lin, K.M.; Boyle, C.; Marom, N.; Marx, R.G. Graft Selection in Anterior Cruciate Ligament Reconstruction. Sports Med. Arthrosc. Rev. 2020, 28, 41–48.
- Hoher, J.; Moller, H.D.; Fu, F.H. Bone tunnel enlargement after anterior cruciate ligament reconstruction: Fact or fiction? Knee Surg. Sports Traumatol. Arthrosc. 1998, 6, 231–240.
- Fahey, M.; Indelicato, P.A. Bone tunnel enlargement after anterior cruciate ligament replacement. Am. J. Sports Med. 1994, 22, 410–414.
- Brandt, K.D. Insights into the natural history of osteoarthritis provided by the cruciate-deficient dog. An animal model of osteoarthritis. Ann. N. Y. Acad. Sci. 1994, 732, 199–205.
- Benjamin, M.; McGonagle, D. Entheses: Tendon and ligament attachment sites. Scand. J. Med. Sci. Sports 2009, 19, 520–527.
- Lyons, T.J.; McClure, S.F.; Stoddart, R.W.; McClure, J. The normal human chondro-osseous junctional region: Evidence for contact of uncalcified cartilage with subchondral bone and marrow spaces. BMC Musculoskelet. Disord. 2006, 7, 52.
- Arkill, K.P.; Winlove, C.P. Fatty acid transport in articular cartilage. Arch. Biochem. Biophys. 2006, 456, 71–78.
- Pan, J.; Zhou, X.; Li, W.; Novotny, J.E.; Doty, S.B.; Wang, L. In situ measurement of transport between subchondral bone and articular cartilage. J. Orthop. Res. 2009, 27, 1347–1352.
- Gongadze, L.R. Basophilic line of the articular cartilage in normal and various pathological states. Arkh. Anat. Gistol. Embriol. 1987, 92, 52–57.
- Beaulieu, M.L.; Carey, G.E.; Schlecht, S.H.; Wojtys, E.M.; Ashton-Miller, J.A. Quantitative comparison of the microscopic anatomy of the human ACL femoral and tibial entheses. J. Orthop. Res. 2015, 33, 1811–1817.
- Dienst, M.; Burks, R.T.; Greis, P.E. Anatomy and biomechanics of the anterior cruciate ligament. Orthop. Clin. N. Am. 2002, 33, 605–620.
- Kohn, L.; Rembeck, E.; Rauch, A. Anterior cruciate ligament injury in adults: Diagnostics and treatment. Orthopade 2020, 49, 1013–1028.
- Zhao, L.; Lee, P.V.S.; Ackland, D.C.; Broom, N.D.; Thambyah, A. Microstructure Variations in the Soft-Hard Tissue Junction of the Human Anterior Cruciate Ligament. Anat. Rec. 2017, 300, 1547–1559.
- Smigielski, R.; Zdanowicz, U.; Drwiega, M.; Ciszek, B.; Ciszkowska-Lyson, B.; Siebold, R. Ribbon like appearance of the midsubstance fibres of the anterior cruciate ligament close to its femoral insertion site: A cadaveric study including 111 knees. Knee Surg. Sports Traumatol. Arthrosc. 2015, 23, 3143–3150.
- Qu, D.; Subramony, S.D.; Boskey, A.L.; Pleshko, N.; Doty, S.B.; Lu, H.H. Compositional mapping of the mature anterior cruciate ligament-to-bone insertion. J. Orthop. Res. 2017, 35, 2513–2523.
- Dyment, N.A.; Breidenbach, A.P.; Schwartz, A.G.; Russell, R.P.; Aschbacher-Smith, L.; Liu, H.; Hagiwara, Y.; Jiang, R.; Thomopoulos, S.; Butler, D.L.; et al. Gdf5 progenitors give rise to fibrocartilage cells that mineralize via hedgehog signaling to form the zonal enthesis. Dev. Biol. 2015, 405, 96–107.
- Thomopoulos, S.; Genin, G.M.; Galatz, L.M. The development and morphogenesis of the tendon-to-bone insertion—What development can teach us about healing. J. Musculoskelet. Neuronal. Interact. 2010, 10, 35–45.
- Milz, S.; Boszczyk, B.M.; Boszczyk, A.A.; Putz, R.; Benjamin, M. The enthesis. Physiological morphology, molecular composition and pathoanatomical alterations. Orthopade 2005, 34, 526–532.
- Bellemans, J.; Carpentier, K.; Vandenneucker, H.; Vanlauwe, J.; Victor, J. The John Insall Award: Both morphotype and gender influence the shape of the knee in patients undergoing TKA. Clin. Orthop. Relat. Res. 2010, 468, 29–36.
- Guy, S.P.; Farndon, M.A.; Sidhom, S.; Al-Lami, M.; Bennett, C.; London, N.J. Gender differences in distal femoral morphology and the role of gender specific implants in total knee replacement: A prospective clinical study. Knee 2012, 19, 28–31.
- Beaulieu, M.L.; Lamontagne, M.; Xu, L. Gender differences in time-frequency EMG analysis of unanticipated cutting maneuvers. Med. Sci. Sports Exerc. 2008, 40, 1795–1804.
- Beaulieu, M.L.; McLean, S.G. Sex-dimorphic landing mechanics and their role within the noncontact ACL injury mechanism: Evidence, limitations and directions. Sports Med. Arthrosc. Rehabil. Ther. Technol. 2012, 4, 10.
- Rodas, G.; Caceres, A.; Ferrer, E.; Balague-Dobon, L.; Osaba, L.; Lucia, A.; Gonzalez, J.R. Sex Differences in the Association between Risk of Anterior Cruciate Ligament Rupture and COL5A1 Polymorphisms in Elite Footballers. Genes 2022, 14, 33.
- Little, D.; Thompson, J.W.; Dubois, L.G.; Ruch, D.S.; Moseley, M.A.; Guilak, F. Proteomic differences between male and female anterior cruciate ligament and patellar tendon. PLoS ONE 2014, 9, e96526.
- Liu, S.H.; Al-Shaikh, R.A.; Panossian, V.; Finerman, G.A.; Lane, J.M. Estrogen affects the cellular metabolism of the anterior cruciate ligament. A potential explanation for female athletic injury. Am. J. Sports Med. 1997, 25, 704–709.
- Liu, X.; Luo, Z.P. Combined effects of estrogen and mechanical loading on anterior cruciate ligament fibroblast biosynthesis. ScientificWorldJournal 2005, 5, 5–8.
- Lee, H.; Petrofsky, J.S.; Daher, N.; Berk, L.; Laymon, M.; Khowailed, I.A. Anterior cruciate ligament elasticity and force for flexion during the menstrual cycle. Med. Sci. Monit. 2013, 19, 1080–1088.
- Beaulieu, M.L.; DeClercq, M.G.; Rietberg, N.T.; Li, S.H.; Harker, E.C.; Weber, A.E.; Ashton-Miller, J.A.; Wojtys, E.M. The Anterior Cruciate Ligament Can Become Hypertrophied in Response to Mechanical Loading: A Magnetic Resonance Imaging Study in Elite Athletes. Am. J. Sports Med. 2021, 49, 2371–2378.
- Beaulieu, M.L.; Nowak, E.K.; Beynnon, B.D.; Ashton-Miller, J.A.; Sturnick, D.R.; Wojtys, E.M. Clinical-Grade MRI-Based Methods to Identify Combined Anatomic Factors That Predict ACL Injury Risk in Male and Female Athletes. Am. J. Sports Med. 2021, 49, 2615–2623.
- Hohmann, E.; Bryant, A.; Reaburn, P.; Tetsworth, K. Is there a correlation between posterior tibial slope and non-contact anterior cruciate ligament injuries? Knee Surg. Sports Traumatol. Arthrosc. 2011, 19 (Suppl. S1), S109–S114.
- Mengsteab, P.Y.; Conroy, P.; Badon, M.; Otsuka, T.; Kan, H.M.; Vella, A.T.; Nair, L.S.; Laurencin, C.T. Evaluation of a bioengineered ACL matrix’s osteointegration with BMP-2 supplementation. PLoS ONE 2020, 15, e0227181.
- Lu, D.; Yang, C.; Zhang, Z.; Xiao, M. Enhanced tendon-bone healing with acidic fibroblast growth factor delivered in collagen in a rabbit anterior cruciate ligament reconstruction model. J. Orthop. Surg. Res. 2018, 13, 301.
- Jiang, Q.; Wang, L.; Liu, Z.; Su, J.; Tang, Y.; Tan, P.; Zhu, X.; Zhang, K.; Ma, X.; Jiang, J.; et al. Canine ACL reconstruction with an injectable hydroxyapatite/collagen paste for accelerated healing of tendon-bone interface. Bioact. Mater. 2023, 20, 1–15.
- Bascunan, A.L.; Biedrzycki, A.; Banks, S.A.; Lewis, D.D.; Kim, S.E. Large Animal Models for Anterior Cruciate Ligament Research. Front. Vet. Sci. 2019, 6, 292.
- Schulze-Tanzil, G.; Silawal, S.; Hoyer, M. Anatomical feature of knee joint in Aachen minipig as a novel miniature pig line for experimental research in orthopaedics. Ann. Anat. 2020, 227, 151411.
- Shi, Q.; Wang, H.; He, K.; Tao, M.; Cheng, C.K. Comparison of the morphology of the anterior cruciate ligament and related bony structures between pigs and humans. Front. Vet. Sci. 2022, 9, 1045785.
- Erdemir, A.; Guess, T.M.; Halloran, J.; Tadepalli, S.C.; Morrison, T.M. Considerations for reporting finite element analysis studies in biomechanics. J. Biomech. 2012, 45, 625–633.
- Kiapour, A.; Kiapour, A.M.; Kaul, V.; Quatman, C.E.; Wordeman, S.C.; Hewett, T.E.; Demetropoulos, C.K.; Goel, V.K. Finite element model of the knee for investigation of injury mechanisms: Development and validation. J. Biomech. Eng. 2014, 136, 011002.
- Pena, E.; Calvo, B.; Martinez, M.A.; Palanca, D.; Doblare, M. Finite element analysis of the effect of meniscal tears and meniscectomies on human knee biomechanics. Clin. Biomech. 2005, 20, 498–507.
- Benos, L.; Stanev, D.; Spyrou, L.; Moustakas, K.; Tsaopoulos, D.E. A Review on Finite Element Modeling and Simulation of the Anterior Cruciate Ligament Reconstruction. Front. Bioeng. Biotechnol. 2020, 8, 967.
- Catani, F.; Innocenti, B.; Belvedere, C.; Labey, L.; Ensini, A.; Leardini, A. The Mark Coventry Award: Articular contact estimation in TKA using in vivo kinematics and finite element analysis. Clin. Orthop. Relat. Res. 2010, 468, 19–28.
- Harris, M.D.; Cyr, A.J.; Ali, A.A.; Fitzpatrick, C.K.; Rullkoetter, P.J.; Maletsky, L.P.; Shelburne, K.B. A Combined Experimental and Computational Approach to Subject-Specific Analysis of Knee Joint Laxity. J. Biomech. Eng. 2016, 138, 0810041–0810048.
- Readioff, R.; Geraghty, B.; Comerford, E.; Elsheikh, A. A full-field 3D digital image correlation and modelling technique to characterise anterior cruciate ligament mechanics ex vivo. Acta Biomater. 2020, 113, 417–428.
- Chokhandre, S.; Schwartz, A.; Klonowski, E.; Landis, B.; Erdemir, A. Open Knee(s): A Free and Open Source Library of Specimen-Specific Models and Related Digital Assets for Finite Element Analysis of the Knee Joint. Ann. Biomed. Eng. 2023, 51, 10–23.
- Currey, J.D. Bones: Structure and Mechanics; Princeton University Press: Princeton, NJ, USA, 2002.
- Chandrashekar, N.; Mansouri, H.; Slauterbeck, J.; Hashemi, J. Sex-based differences in the tensile properties of the human anterior cruciate ligament. J. Biomech. 2006, 39, 2943–2950.
- Thomopoulos, S.; Marquez, J.P.; Weinberger, B.; Birman, V.; Genin, G.M. Collagen fiber orientation at the tendon to bone insertion and its influence on stress concentrations. J. Biomech. 2006, 39, 1842–1851.
- Ristaniemi, A.; Tanska, P.; Stenroth, L.; Finnila, M.A.J.; Korhonen, R.K. Comparison of material models for anterior cruciate ligament in tension: From poroelastic to a novel fibril-reinforced nonlinear composite model. J. Biomech. 2021, 114, 110141.
- Tits, A.; Ruffoni, D. Joining soft tissues to bone: Insights from modeling and simulations. Bone Rep. 2021, 14, 100742.
- Spalazzi, J.P.; Boskey, A.L.; Pleshko, N.; Lu, H.H. Quantitative mapping of matrix content and distribution across the ligament-to-bone insertion. PLoS ONE 2013, 8, e74349.
- Armitage, O.E.; Oyen, M.L. Indentation across interfaces between stiff and compliant tissues. Acta Biomater. 2017, 56, 36–43.
- Ruffoni, D.; Fratzl, P.; Roschger, P.; Phipps, R.; Klaushofer, K.; Weinkamer, R. Effect of temporal changes in bone turnover on the bone mineralization density distribution: A computer simulation study. J. Bone Miner. Res. 2008, 23, 1905–1914.
- Dai, C.; Guo, L.; Yang, L.; Wu, Y.; Gou, J.; Li, B. Regional fibrocartilage variations in human anterior cruciate ligament tibial insertion: A histological three-dimensional reconstruction. Connect. Tissue Res. 2015, 56, 18–24.
- Mallett, K.F.; Arruda, E.M. Digital image correlation-aided mechanical characterization of the anteromedial and posterolateral bundles of the anterior cruciate ligament. Acta Biomater. 2017, 56, 44–57.
- Luetkemeyer, C.M.; Cai, L.; Neu, C.P.; Arruda, E.M. Full-volume displacement mapping of anterior cruciate ligament bundles with dual MRI. Extrem. Mech. Lett. 2018, 19, 7–14.
- Luetkemeyer, C.M.; Marchi, B.C.; Ashton-Miller, J.A.; Arruda, E.M. Femoral entheseal shape and attachment angle as potential risk factors for anterior cruciate ligament injury. J. Mech. Behav. Biomed. Mater. 2018, 88, 313–321.
- Luetkemeyer, C.M.; Scheven, U.; Estrada, J.B.; Arruda, E.M. Constitutive modeling of the anterior cruciate ligament bundles and patellar tendon with full-field methods. J. Mech. Phys. Solids 2021, 156, 104577.
- Rafieyan, S.; Vasheghani-Farahani, E.; Baheiraei, N.; Keshavarz, H. MLATE: Machine learning for predicting cell behavior on cardiac tissue engineering scaffolds. Comput. Biol. Med. 2023, 158, 106804.
- Seidi, A.; Ramalingam, M.; Elloumi-Hannachi, I.; Ostrovidov, S.; Khademhosseini, A. Gradient biomaterials for soft-to-hard interface tissue engineering. Acta Biomater. 2011, 7, 1441–1451.
- Cai, J.; Wang, J.; Sun, C.; Dai, J.; Zhang, C. Biomaterials with stiffness gradient for interface tissue engineering. Biomed. Mater. 2022, 17, 064103.
- Klabukov, I.; Tenchurin, T.; Shepelev, A.; Baranovskii, D.; Mamagulashvili, V.; Dyuzheva, T.; Krasilnikova, O.; Balyasin, M.; Lyundup, A.; Krasheninnikov, M.; et al. Biomechanical Behaviors and Degradation Properties of Multilayered Polymer Scaffolds: The Phase Space Method for Bile Duct Design and Bioengineering. Biomedicines 2023, 11, 745.
- Paxton, J.Z.; Grover, L.M.; Baar, K. Engineering an in vitro model of a functional ligament from bone to bone. Tissue Eng. Part A 2010, 16, 3515–3525.
- Hurley-Novatny, A.; Arumugasaamy, N.; Kimicata, M.; Baker, H.; Mikos, A.G.; Fisher, J.P. Concurrent multi-lineage differentiation of mesenchymal stem cells through spatial presentation of growth factors. Biomed. Mater. 2020, 15, 055035.
- Font Tellado, S.; Chiera, S.; Bonani, W.; Poh, P.S.P.; Migliaresi, C.; Motta, A.; Balmayor, E.R.; van Griensven, M. Heparin functionalization increases retention of TGF-beta2 and GDF5 on biphasic silk fibroin scaffolds for tendon/ligament-to-bone tissue engineering. Acta Biomater. 2018, 72, 150–166.
- Fan, J.; Sun, L.; Chen, X.; Qu, L.; Li, H.; Liu, X.; Zhang, Y.; Cheng, P.; Fan, H. Implementation of a stratified approach and gene immobilization to enhance the osseointegration of a silk-based ligament graft. J. Mater. Chem. B 2017, 5, 7035–7050.
- Ferguson, M.W.; O’Kane, S. Scar-free healing: From embryonic mechanisms to adult therapeutic intervention. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2004, 359, 839–850.
- Zou, G.; Song, E.; Wei, B. Effects of tendon-bone healing of anterior cruciate ligament reconstruction by osteoprotegerin combined with deproteinized bovine bone. Muscles Ligaments Tendons J. 2017, 7, 256–262.
- Nakase, J.; Kitaoka, K.; Matsumoto, K.; Tomita, K. Facilitated tendon-bone healing by local delivery of recombinant hepatocyte growth factor in rabbits. Arthroscopy 2010, 26, 84–90.
- Iannucci, L.E.; Boys, A.J.; McCorry, M.C.; Estroff, L.A.; Bonassar, L.J. Cellular and Chemical Gradients to Engineer the Meniscus-to-Bone Insertion. Adv. Healthc. Mater. 2019, 8, e1800806.
- Mutsuzaki, H.; Kuwahara, K.; Nakajima, H. Influence of periostin on the development of fibrocartilage layers of anterior cruciate ligament insertion. Orthop. Traumatol. Surg. Res. 2022, 103215.
- Mutsuzaki, H.; Yoshida, Y.; Nakajima, H. Periostin Contributes to Fibrocartilage Layer Growth of the Patella Tendon Tibial Insertion in Mice. Medicina 2022, 58, 957.
- Cai, L.; Brophy, R.H.; Tycksen, E.D.; Duan, X.; Nunley, R.M.; Rai, M.F. Distinct expression pattern of periostin splice variants in chondrocytes and ligament progenitor cells. FASEB J. 2019, 33, 8386–8405.
- Miescher, I.; Rieber, J.; Calcagni, M.; Buschmann, J. In Vitro and In Vivo Effects of IGF-1 Delivery Strategies on Tendon Healing: A Review. Int. J. Mol. Sci. 2023, 24, 2370.
- Sha, Y.; Afandi, R.; Zhang, B.; Yang, L.; Lv, Y. MGF E peptide pretreatment improves collagen synthesis and cell proliferation of injured human ACL fibroblasts via MEK-ERK1/2 signaling pathway. Growth Factors 2017, 35, 29–38.
- Sha, Y.; Yang, L.; Lv, Y. MGF E peptide improves anterior cruciate ligament repair by inhibiting hypoxia-induced cell apoptosis and accelerating angiogenesis. J. Cell. Physiol. 2019, 234, 8846–8861.
- Eagan, M.J.; Zuk, P.A.; Zhao, K.W.; Bluth, B.E.; Brinkmann, E.J.; Wu, B.M.; McAllister, D.R. The suitability of human adipose-derived stem cells for the engineering of ligament tissue. J. Tissue Eng. Regen. Med. 2012, 6, 702–709.
- Schwarz, S.; Goegele, C.; Ondruschka, B.; Hammer, N.; Kohl, B.; Schulze-Tanzil, G. Migrating Myofibroblastic Iliotibial Band-Derived Fibroblasts Represent a Promising Cell Source for Ligament Reconstruction. Int. J. Mol. Sci. 2019, 20, 1972.
- Benjamin, M.; Ralphs, J.R. Fibrocartilage in tendons and ligaments—An adaptation to compressive load. J. Anat. 1998, 193 Pt 4, 481–494.
- Schulze-Tanzil, G.G.; Delgado-Calcares, M.; Stange, R.; Wildemann, B.; Docheva, D. Tendon healing: A concise review on cellular and molecular mechanisms with a particular focus on the Achilles tendon. Bone Joint Res. 2022, 11, 561–574.
- Schulze-Tanzil, G. Intraarticular Ligament Degeneration Is Interrelated with Cartilage and Bone Destruction in Osteoarthritis. Cells 2019, 8, 99.
- Ruschke, K.; Meier, C.; Ullah, M.; Krebs, A.C.; Silberreis, K.; Kohl, B.; Knaus, P.; Jagielski, M.; Arens, S.; Schulze-Tanzil, G. Bone morphogenetic protein 2/SMAD signalling in human ligamentocytes of degenerated and aged anterior cruciate ligaments. Osteoarthr. Cartil. 2016, 24, 1816–1825.
- Fang, F.; Xiao, Y.; Zelzer, E.; Leong, K.W.; Thomopoulos, S. A mineralizing pool of Gli1-expressing progenitors builds the tendon enthesis and demonstrates therapeutic potential. Cell Stem Cell 2022, 29, 1669–1684.e6.
- Schwartz, A.G.; Galatz, L.M.; Thomopoulos, S. Enthesis regeneration: A role for Gli1+ progenitor cells. Development 2017, 144, 1159–1164.
- Buschmann, J.; Gao, S.; Harter, L.; Hemmi, S.; Welti, M.; Werner, C.M.; Calcagni, M.; Cinelli, P.; Wanner, G.A. Yield and proliferation rate of adipose-derived stromal cells as a function of age, body mass index and harvest site-increasing the yield by use of adherent and supernatant fractions? Cytotherapy 2013, 15, 1098–1105.
- Matsumoto, T.; Sato, Y.; Kobayashi, T.; Suzuki, K.; Kimura, A.; Soma, T.; Ito, E.; Kikuchi, T.; Kobayashi, S.; Harato, K.; et al. Adipose-Derived Stem Cell Sheets Improve Early Biomechanical Graft Strength in Rabbits After Anterior Cruciate Ligament Reconstruction. Am. J. Sports Med. 2021, 49, 3508–3518.
- Bianchi, E.; Faccendini, A.; Del Favero, E.; Ricci, C.; Caliogna, L.; Vigani, B.; Pavesi, F.C.; Perotti, C.; Domingues, R.M.A.; Gomes, M.E.; et al. Topographical and Compositional Gradient Tubular Scaffold for Bone to Tendon Interface Regeneration. Pharmaceutics 2022, 14, 2153.
- Font Tellado, S.; Bonani, W.; Balmayor, E.R.; Foehr, P.; Motta, A.; Migliaresi, C.; van Griensven, M. (*) Fabrication and Characterization of Biphasic Silk Fibroin Scaffolds for Tendon/Ligament-to-Bone Tissue Engineering. Tissue Eng. Part A 2017, 23, 859–872.
- Teuschl, A.; Heimel, P.; Nuernberger, S.; van Griensven, M.; Redl, H.; Nau, T. A Novel Silk Fiber-Based Scaffold for Regeneration of the Anterior Cruciate Ligament: Histological Results From a Study in Sheep. Am. J. Sports Med. 2016, 44, 1547–1557.
- Teuschl, A.H.; Tangl, S.; Heimel, P.; Schwarze, U.Y.; Monforte, X.; Redl, H.; Nau, T. Osteointegration of a Novel Silk Fiber-Based ACL Scaffold by Formation of a Ligament-Bone Interface. Am. J. Sports Med. 2019, 47, 620–627.
- Tremblay, P.; Cloutier, R.; Lamontagne, J.; Belzil, A.M.; Larkin, A.M.; Chouinard, L.; Chabaud, S.; Laverty, S.; Lussier, B.; Goulet, F. Potential of skin fibroblasts for application to anterior cruciate ligament tissue engineering. Cell Transplant. 2011, 20, 535–542.
- He, P.; Ng, K.S.; Toh, S.L.; Goh, J.C. In vitro ligament-bone interface regeneration using a trilineage coculture system on a hybrid silk scaffold. Biomacromolecules 2012, 13, 2692–2703.
- Harris, E.; Liu, Y.; Cunniffe, G.; Morrissey, D.; Carroll, S.; Mulhall, K.; Kelly, D.J. Biofabrication of soft tissue templates for engineering the bone-ligament interface. Biotechnol. Bioeng. 2017, 114, 2400–2411.
- Li, H.; Fan, J.; Sun, L.; Liu, X.; Cheng, P.; Fan, H. Functional regeneration of ligament-bone interface using a triphasic silk-based graft. Biomaterials 2016, 106, 180–192.
- Xiong, J.; Wang, H.; Lan, X.; Wang, Y.; Wang, Z.; Bai, J.; Ou, W.; Cai, N.; Wang, W.; Tang, Y. Fabrication of bioinspired grid-crimp micropatterns by melt electrospinning writing for bone-ligament interface study. Biofabrication 2022, 14, 025008.
- Schulze-Tanzil, G.; Arnold, P.; Goegele, C.; Hahn, J.; Breier, A.; Meyer, M.; Kohl, B.; Schropfer, M.; Schwarz, S. SV40 Transfected Human Anterior Cruciate Ligament Derived Ligamentocytes-Suitable as a Human in Vitro Model for Ligament Reconstruction? Int. J. Mol. Sci. 2020, 21, 593.
- Hoyer, M.; Meier, C.; Breier, A.; Hahner, J.; Heinrich, G.; Drechsel, N.; Meyer, M.; Rentsch, C.; Garbe, L.A.; Ertel, W.; et al. In vitro characterization of self-assembled anterior cruciate ligament cell spheroids for ligament tissue engineering. Histochem. Cell Biol. 2015, 143, 289–300.
- Goegele, C.; Hahn, J.; Elschner, C.; Breier, A.; Schroepfer, M.; Prade, I.; Meyer, M.; Schulze-Tanzil, G. Enhanced Growth of Lapine Anterior Cruciate Ligament-Derived Fibroblasts on Scaffolds Embroidered from Poly(l-lactide-co-epsilon-caprolactone) and Polylactic Acid Threads Functionalized by Fluorination and Hexamethylene Diisocyanate Cross-Linked Collagen Foams. Int. J. Mol. Sci. 2020, 21, 1132.
- Goegele, C.; Konrad, J.; Hahn, J.; Breier, A.; Schroepfer, M.; Meyer, M.; Merkel, R.; Hoffmann, B.; Schulze-Tanzil, G. Maintenance of Ligament Homeostasis of Spheroid-Colonized Embroidered and Functionalized Scaffolds after 3D Stretch. Int. J. Mol. Sci. 2021, 22, 8204.
- Zahn, I.; Braun, T.; Goegele, C.; Schulze-Tanzil, G. Minispheroids as a Tool for Ligament Tissue Engineering: Do the Self-Assembly Techniques and Spheroid Dimensions Influence the Cruciate Ligamentocyte Phenotype? Int. J. Mol. Sci. 2021, 22, 11011.
- Zahn, I.; Stoebener, D.D.; Weinhart, M.; Goegele, C.; Breier, A.; Hahn, J.; Schroepfer, M.; Meyer, M.; Schulze-Tanzil, G. Cruciate Ligament Cell Sheets Can Be Rapidly Produced on Thermoresponsive poly(glycidyl ether) Coating and Successfully Used for Colonization of Embroidered Scaffolds. Cells 2021, 10, 877.
- Hahner, J.; Hoyer, M.; Hillig, S.; Schulze-Tanzil, G.; Meyer, M.; Schroepfer, M.; Lohan, A.; Garbe, L.A.; Heinrich, G.; Breier, A. Diffusion chamber system for testing of collagen-based cell migration barriers for separation of ligament enthesis zones in tissue-engineered ACL constructs. J. Biomater. Sci. Polym. Ed. 2015, 26, 1085–1099.
More
Information
Subjects:
Anatomy & Morphology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.3K
Revisions:
2 times
(View History)
Update Date:
16 Jun 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





