
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Colton Ladbury | -- | 1587 | 2023-06-08 16:09:59 | | | |
| 2 | Fanny Huang | Meta information modification | 1587 | 2023-06-12 03:04:06 | | | | |
| 3 | Fanny Huang | -3 word(s) | 1584 | 2023-06-12 10:14:22 | | |
Video Upload Options
Magnetic resonance-guided radiotherapy (MRgRT) is an emerging radiotherapy technology combining real-time magnetic resonance imaging and radiation delivery. By administering radiation with a linear accelerator with built in low-field or high-field MRI, practitioners have a greater ability to align to the target for daily set-up, precisely track the motion of and thereby target or avoid tissues and adapt to inter-treatment daily changes. This decreased uncertainty has implications for facilitating smaller, less-toxic treatment margins, potentially allowing delivery of higher dose radiotherapy that will lead to better control of tumors. The technology has already found success in treating breast, prostate, pancreatic, liver, lung, and limited metastatic cancers, in addition to non-oncologic indications such as cardiac ablation.
1. Introduction
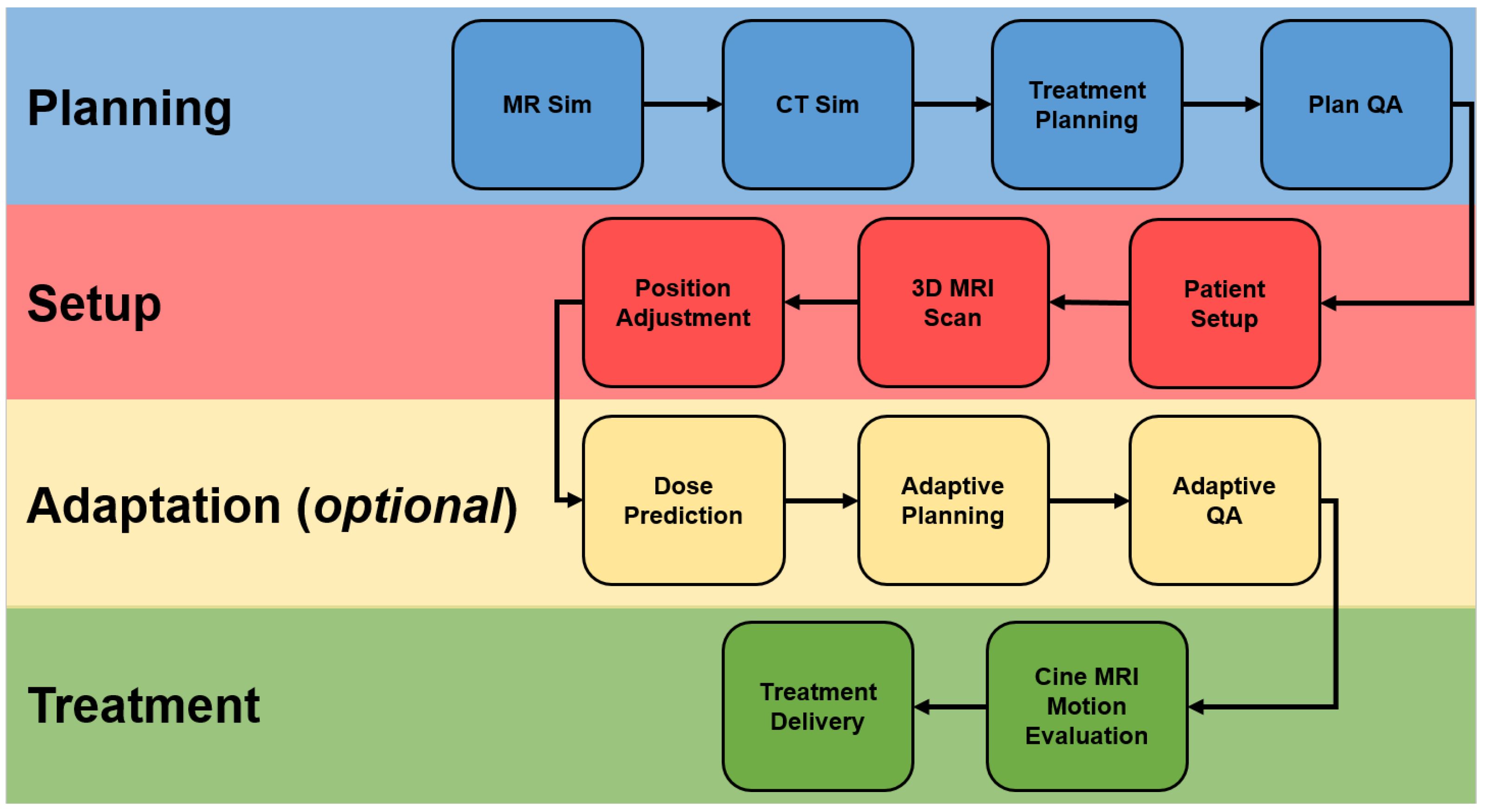
2. Types of MRgRT
2.1. Low-Field
| Characteristic | Low-Field | High-Field |
|---|---|---|
| Magnet Strength | 0.2–1.0 Tesla | 1.5–3.0 Tesla |
| Cost | Less | More |
| Space Requirement | Less | More |
| Compatibility with Metal Implants | More | Less |
| Commercially Available Models | Viewray MRIdian MagnetTx Aurora RT |
Elekta Unity |
| Signal-to-Noise Ratio | Reduced | Increased |
| Resolution | Decreased | Increased |
2.2. High Field
2.3. MagnetTx Aurora RT
3. Indications Treated by MRgRT
| Disease Site | Enhanced Soft Tissue Visualization | Motion Management | Inter-Fraction Adaptive Re-Planning | Margin Reduction | Facilitate Dose Escalation | Relevant Studies |
|---|---|---|---|---|---|---|
| Prostate | ✓ | ✓ | Kishan et al. [15] Bruynzeel et al. [16] Ma et al. [17] |
|||
| Pancreas | ✓ | ✓ | ✓ | ✓ | Rudra et al. [18] Parikh et al. [19] |
|
| Liver | ✓ | ✓ | ✓ | ✓ | Rosenberg et al. [20] Luterstein et al. [21] van Dams et al. [22] Ugurluer et al. [23] Henke et al. [24] |
|
| Breast | ✓ | ✓ | ✓ | Kennedy et al. [25] Vasmel et al. [26] |
||
| Lung | ✓ | ✓ | Henke et al. [27] Finazzi et al. [28] Regnery et al. [29] Finazzi et al. [30] Finazzi et al. [31] |
|||
| Oligometastases | ✓ | ✓ | ✓ | ✓ | ✓ | Cuccia et al. [32] Yoon et al. [33] |
| Cardiac Ablation | ✓ | ✓ | Mayinger et al. [34] |
4. Future Directions
4.1. Imaging Improvements
4.2. Adaptive Workflows
5. Conclusions
References
- Raaijmakers, A.J.E.; Raaymakers, B.W.; Lagendijk, J.J.W. Magnetic-field-induced dose effects in MR-guided radiotherapy systems: Dependence on the magnetic field strength. Phys. Med. Biol. 2008, 53, 909–923.
- Keall, P.J.; Glide-Hurst, C.K.; Cao, M.; Lee, P.; Murray, B.; Raaymakers, B.W.; Tree, A.; van der Heide, U.A. ICRU REPORT 97: MRI-Guided Radiation Therapy Using MRI-Linear Accelerators. J. ICRU 2022, 22, 1–100.
- Thorwarth, D.; Low, D.A. Technical challenges of real-time adaptive MR-guided radiotherapy. Front. Oncol. 2021, 11, 634507.
- Marques, J.P.; Simonis, F.F.; Webb, A.G. Low-field MRI: An MR physics perspective. J. Magn. Reson. Imaging 2019, 49, 1528–1542.
- Fallone, B.G. The rotating biplanar linac–magnetic resonance imaging system. In Seminars in Radiation Oncology; Elsevier: Amsterdam, The Netherlands, 2014; pp. 200–202.
- Keyvanloo, A.; Burke, B.; Warkentin, B.; Tadic, T.; Rathee, S.; Kirkby, C.; Santos, D.M.; Fallone, B.G. Skin dose in longitudinal and transverse linac-MRIs using Monte Carlo and realistic 3D MRI field models. Med. Phys. 2012, 39, 6509–6521.
- Kirkby, C.; Murray, B.; Rathee, S.; Fallone, B. Lung dosimetry in a linac-MRI radiotherapy unit with a longitudinal magnetic field. Med. Phys. 2010, 37, 4722–4732.
- Chu, V.W.; Kan, M.W.; Lee, L.K.; Wong, K.C.; Tong, M.; Chan, A.T. The effect of the magnetic fields from three different configurations of the MRIgRT systems on the dose deposition from lateral opposing photon beams in a laryngeal geometry—A Monte Carlo study. Radiat. Med. Prot. 2021, 2, 103–111.
- Boeke, S.; Mönnich, D.; van Timmeren, J.E.; Balermpas, P. MR-Guided Radiotherapy for Head and Neck Cancer: Current Developments, Perspectives, and Challenges. Front. Oncol. 2021, 11, 616156.
- van Dams, R.; Raldow, A.C.; Lee, P. Role of mr-guided radiotherapy (mrgrt) in colorectal cancer. Curr. Color. Cancer Rep. 2021, 17, 69–76.
- Maziero, D.; Straza, M.W.; Ford, J.C.; Bovi, J.A.; Diwanji, T.; Stoyanova, R.; Paulson, E.S.; Mellon, E.A. MR-Guided Radiotherapy for Brain and Spine Tumors. Front. Oncol. 2021, 11, 626100.
- Boldrini, L.; Piras, A.; Chiloiro, G.; Autorino, R.; Cellini, F.; Cusumano, D.; Fionda, B.; D’aviero, A.; Campitelli, M.; Marazzi, F.; et al. Low Tesla magnetic resonance guided radiotherapy for locally advanced cervical cancer: First clinical experience. Tumori J. 2020, 106, 497–505.
- Yavas, G.; Kuscu, U.E.; Ayhan, A.; Yavas, C.; Onal, C. The utility of 1.5 tesla MR-guided adaptive stereotactic body radiotherapy for recurrent ovarian tumor—Case reports and review of the literature. Int. J. Surg. Case Rep. 2022, 99, 107696.
- Tetar, S.; Bruynzeel, A.; Bohoudi, O.; Nieboer, T.; Lagerwaard, F. Pitfalls of Ovarian Ablative Magnetic Resonance-guided Radiation Therapy for Refractory Endometriosis. Cureus 2018, 10, e2294.
- Kishan, A.U.; Ma, T.M.; Lamb, J.M.; Casado, M.; Wilhalme, H.; Low, D.A.; Sheng, K.; Sharma, S.; Nickols, N.G.; Pham, J.; et al. Magnetic Resonance Imaging–Guided vs Computed Tomography–Guided Stereotactic Body Radiotherapy for Prostate Cancer: The MIRAGE Randomized Clinical Trial. JAMA Oncol. 2023, 9, 365–373.
- Bruynzeel, A.M.; Tetar, S.U.; Oei, S.S.; Senan, S.; Haasbeek, C.J.; Spoelstra, F.O.; Piet, A.H.; Meijnen, P.; van der Jagt, M.A.B.; Fraikin, T.; et al. A prospective single-arm phase 2 study of stereotactic magnetic resonance guided adaptive radiation therapy for prostate cancer: Early toxicity results. Int. J. Radiat. Oncol. Biol. Phys. 2019, 105, 1086–1094.
- Ma, T.M.; Neylon, J.; Casado, M.; Sharma, S.; Sheng, K.; Low, D.; Yang, Y.; Steinberg, M.L.; Lamb, J.; Cao, M.; et al. Dosimetric impact of interfraction prostate and seminal vesicle volume changes and rotation: A post-hoc analysis of a phase III randomized trial of MRI-guided versus CT-guided stereotactic body radiotherapy. Radiother. Oncol. 2022, 167, 203–210.
- Rudra, S.; Jiang, N.; Rosenberg, S.A.; Olsen, J.R.; Roach, M.C.; Wan, L.; Portelance, L.; Mellon, E.A.; Bruynzeel, A.; Lagerwaard, F.; et al. Using adaptive magnetic resonance image-guided radiation therapy for treatment of inoperable pancreatic cancer. Cancer Med. 2019, 8, 2123–2132.
- Parikh, P.J.; Lee, P.; Low, D.; Kim, J.; Mittauer, K.E.; Bassetti, M.F.; Glide-Hurst, C.; Raldow, A.; Yang, Y.; Portelance, L.; et al. Stereotactic MR-Guided On-Table Adaptive Radiation Therapy (SMART) for Patients with Borderline or Locally Advanced Pancreatic Cancer: Primary Endpoint Outcomes of a Prospective Phase II Multi-Center International Trial. Int. J. Radiat. Oncol. Biol. Phys. 2022, 114, 1062–1063.
- Rosenberg, S.A.; Henke, L.E.; Shaverdian, N.; Mittauer, K.; Wojcieszynski, A.P.; Hullett, C.R.; Kamrava, M.; Lamb, J.; Cao, M.; Green, O.L.; et al. A multi-institutional experience of MR-guided liver stereotactic body radiation therapy. Adv. Radiat. Oncol. 2019, 4, 142–149.
- Luterstein, E.; Cao, M.; Lamb, J.M.; Raldow, A.; Low, D.; Steinberg, M.L.; Lee, P. Clinical outcomes using magnetic resonance–guided stereotactic body radiation therapy in patients with locally advanced cholangiocarcinoma. Adv. Radiat. Oncol. 2020, 5, 189–195.
- van Dams, R.; Wu, T.C.; Kishan, A.U.; Raldow, A.C.; Chu, F.I.; Hernandez, J.; Cao, M.; Lamb, J.M.; Mikaeilian, A.; Low, D.A.; et al. Ablative radiotherapy for liver tumors using stereotactic MRI-guidance: A prospective phase I trial. Radiother. Oncol. 2022, 170, 14–20.
- Ugurluer, G.; Mustafayev, T.Z.; Gungor, G.; Atalar, B.; Abacioglu, U.; Sengoz, M.; Agaoglu, F.; Demir, G.; Ozyar, E. Stereotactic MR-guided online adaptive radiation therapy (SMART) for the treatment of liver metastases in oligometastatic patients: Initial clinical experience. Radiat. Oncol. J. 2021, 39, 33.
- Henke, L.; Kashani, R.; Robinson, C.; Curcuru, A.; DeWees, T.; Bradley, J.; Green, O.; Michalski, J.; Mutic, S.; Parikh, P.; et al. Phase I trial of stereotactic MR-guided online adaptive radiation therapy (SMART) for the treatment of oligometastatic or unresectable primary malignancies of the abdomen. Radiother. Oncol. 2018, 126, 519–526.
- Kennedy, W.R.; Thomas, M.A.; Stanley, J.A.; Luo, J.; Ochoa, L.L.; Clifton, K.K.; Cyr, A.E.; Margenthaler, J.A.; DeWees, T.A.; Price, A.; et al. Single-institution phase 1/2 prospective clinical trial of single-fraction, high-gradient adjuvant partial-breast irradiation for hormone sensitive stage 0-I breast cancer. Int. J. Radiat. Oncol. Biol. Phys. 2020, 107, 344–352.
- Vasmel, J.E.; Charaghvandi, R.K.; Houweling, A.C.; Philippens, M.E.; van Asselen, B.; Vreuls, C.P.; van Diest, P.J.; van Leeuwen, A.G.; van Gorp, J.; Witkamp, A.J.; et al. Tumor response after neoadjuvant magnetic resonance guided single ablative dose partial breast irradiation. Int. J. Radiat. Oncol. Biol. Phys. 2020, 106, 821–829.
- Henke, L.E.; Olsen, J.R.; Contreras, J.A.; Curcuru, A.; DeWees, T.A.; Green, O.L.; Michalski, J.; Mutic, S.; Roach, M.C.; Bradley, J.D.; et al. Stereotactic MR-guided online adaptive radiation therapy (SMART) for ultracentral thorax malignancies: Results of a phase 1 trial. Adv. Radiat. Oncol. 2019, 4, 201–209.
- Finazzi, T.; Haasbeek, C.J.; Spoelstra, F.O.; Palacios, M.A.; Admiraal, M.A.; Bruynzeel, A.M.; Slotman, B.J.; Lagerwaard, F.J.; Senan, S. Clinical outcomes of stereotactic MR-guided adaptive radiation therapy for high-risk lung tumors. Int. J. Radiat. Oncol. Biol. Phys. 2020, 107, 270–278.
- Regnery, S.; Buchele, C.; Weykamp, F.; Pohl, M.; Hoegen, P.; Eichkorn, T.; Held, T.; Ristau, J.; Rippke, C.; König, L.; et al. Adaptive MR-guided stereotactic radiotherapy is beneficial for ablative treatment of lung tumors in high-risk locations. Front. Oncol. 2022, 11, 5431.
- Finazzi, T.; Palacios, M.A.; Haasbeek, C.J.; Admiraal, M.A.; Spoelstra, F.O.; Bruynzeel, A.M.; Slotman, B.J.; Lagerwaard, F.J.; Senan, S. Stereotactic MR-guided adaptive radiation therapy for peripheral lung tumors. Radiother. Oncol. 2020, 144, 46–52.
- Finazzi, T.; de Koste, J.R.v.S.; Palacios, M.A.; Spoelstra, F.O.; Slotman, B.J.; Haasbeek, C.J.; Senan, S. Delivery of magnetic resonance-guided single-fraction stereotactic lung radiotherapy. Phys. Imaging Radiat. Oncol. 2020, 14, 17–23.
- Cuccia, F.; Rigo, M.; Gurrera, D.; Nicosia, L.; Mazzola, R.; Figlia, V.; Giaj-Levra, N.; Ricchetti, F.; Attinà, G.; Pastorello, E.; et al. Mitigation on bowel loops daily variations by 1.5-T MR-guided daily-adaptive SBRT for abdomino-pelvic lymph-nodal oligometastases. J. Cancer Res. Clin. Oncol. 2021, 147, 3269–3277.
- Yoon, S.M.; Luterstein, E.; Chu, F.; Cao, M.; Lamb, J.; Agazaryan, N.; Low, D.; Raldow, A.; Steinberg, M.L.; Lee, P. Clinical outcomes of stereotactic magnetic resonance image-guided adaptive radiotherapy for primary and metastatic tumors in the abdomen and pelvis. Cancer Med. 2021, 10, 5897–5906.
- Mayinger, M.; Kovacs, B.; Tanadini-Lang, S.; Ehrbar, S.; Wilke, L.; Chamberlain, M.; Moreira, A.; Weitkamp, N.; Brunckhorst, C.; Duru, F.; et al. First magnetic resonance imaging-guided cardiac radioablation of sustained ventricular tachycardia. Radiother. Oncol. 2020, 152, 203–207.
- Wahl, R.L.; Herman, J.M.; Ford, E. The promise and pitfalls of positron emission tomography and single-photon emission computed tomography molecular imaging–guided radiation therapy. In Seminars in Radiation Oncology; Elsevier: Amsterdam, The Netherlands, 2011; pp. 88–100.
- Hamstra, D.A.; Rehemtulla, A.; Ross, B.D. Diffusion magnetic resonance imaging: A biomarker for treatment response in oncology. J. Clin. Oncol. 2007, 25, 4104–4109.
- Shaverdian, N.; Yang, Y.; Hu, P.; Hart, S.; Sheng, K.; Lamb, J.; Cao, M.; Agazaryan, N.; Thomas, D.; Steinberg, M.; et al. Feasibility evaluation of diffusion-weighted imaging using an integrated MRI-radiotherapy system for response assessment to neoadjuvant therapy in rectal cancer. Br. J. Radiol. 2017, 90, 20160739.
- Matsuo, M.; Matsumoto, S.; Mitchell, J.B.; Krishna, M.C.; Camphausen, K. Magnetic resonance imaging of the tumor microenvironment in radiotherapy: Perfusion, hypoxia, and metabolism. In Seminars in Radiation Oncology; Elsevier: Amsterdam, The Netherlands, 2014; pp. 210–217.
- Lambin, P.; Leijenaar, R.T.; Deist, T.M.; Peerlings, J.; De Jong, E.E.; Van Timmeren, J.; Sanduleanu, S.; Larue, R.T.H.M.; Even, A.J.G.; Jochems, A.; et al. Radiomics: The bridge between medical imaging and personalized medicine. Nat. Rev. Clin. Oncol. 2017, 14, 749–762.
- Boldrini, L.; Cusumano, D.; Chiloiro, G.; Casà, C.; Masciocchi, C.; Lenkowicz, J.; Cellini, F.; Dinapoli, N.; Azario, L.; Teodoli, S.; et al. Delta radiomics for rectal cancer response prediction with hybrid 0.35 T magnetic resonance-guided radiotherapy (MRgRT): A hypothesis-generating study for an innovative personalized medicine approach. Radiol. Med. 2019, 124, 145–153.





