
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Arup Indra | -- | 2987 | 2023-06-05 08:58:30 | | | |
| 2 | Lindsay Dong | Meta information modification | 2987 | 2023-06-05 10:18:35 | | |
Video Upload Options
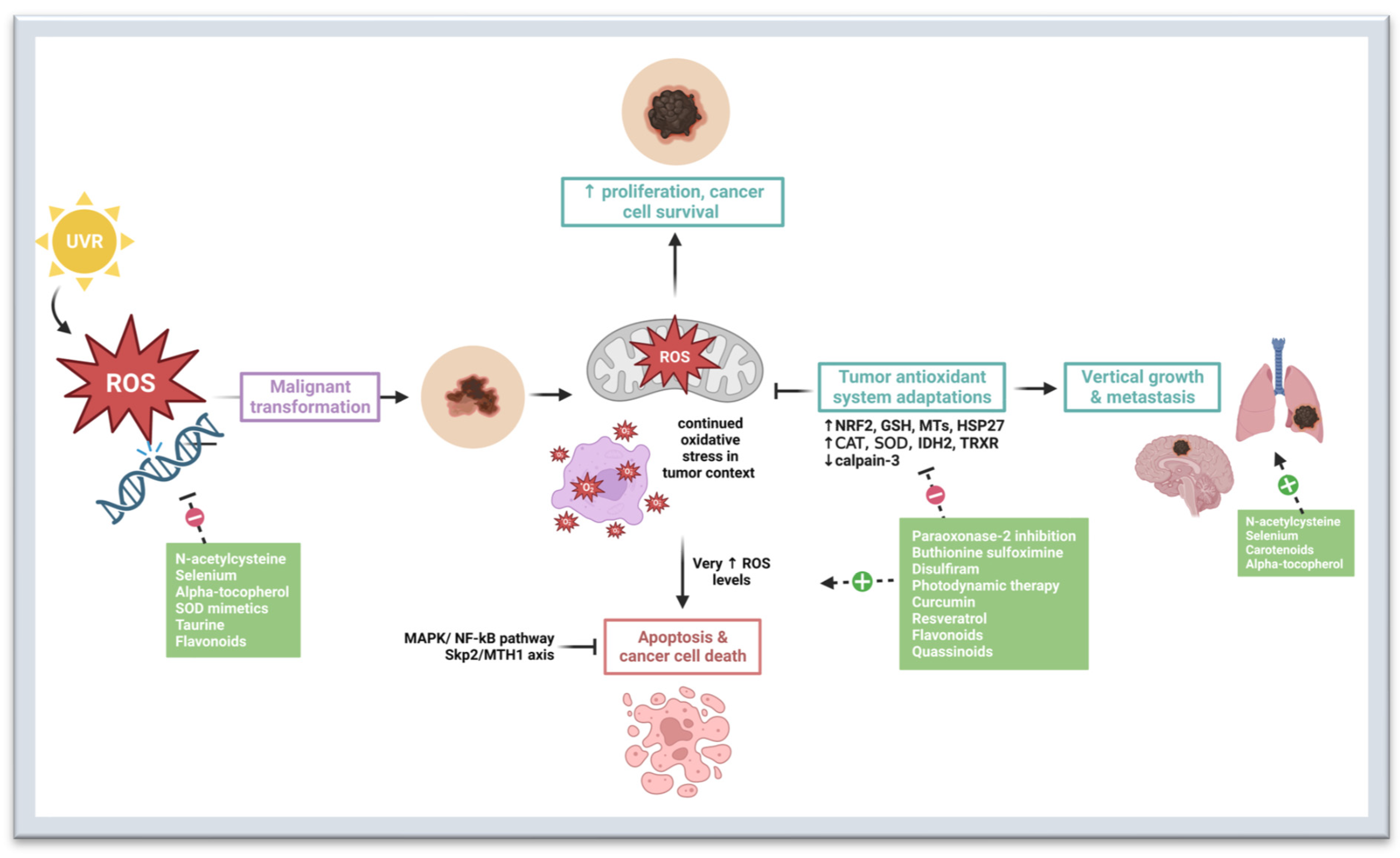
Cutaneous melanoma is one of the deadliest forms of skin cancer. While advancements in systemic targeted therapies and immunotherapies have greatly improved melanoma survival, tumor resistance can limit the efficacy of these therapies. Targeting redox homeostasis in melanoma progression is a promising therapeutic approach, especially in cases of melanoma drug resistance. The role of oxidative stress in melanoma is paradoxical in that it promotes tumor initiation but prevents vertical growth and metastasis. As the disease progresses, melanoma employs adaptive mechanisms to decrease oxidative stress in the tumor environment. Thus, agents with antioxidant properties may have the greatest utility in chemoprevention whereas those with pro-oxidant properties may be better suited for treatment.
1. Introduction
Melanoma develops from the malignant transformation of melanocytes, which are dendritic pigment-producing cells located in the basal layer of the epidermis, hair follicles, inner ear, and uvea of the eye [1][2][3]. Cutaneous melanoma is characterized by a high mutational burden and is frequently associated with mutations in genes that regulate cell proliferation and survival, including BRAF, NRAS, NF1, and tumor suppressor PTEN [4][5][6][7][8][9][10]. These mutations lead to overactivation of the mitogen-activated protein kinase (MAPK) pathway and phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT) signaling pathways. Currently, the most common genetic cause of melanoma is the BRAFV600E mutation, which is present in approximately 50% of melanomas, and contributes to increased cell proliferation and metabolic reprogramming [5][11][12]. Other genetic alterations have been identified in cutaneous melanoma, including mutations in cell cycle regulators like CDK4, and tumor suppressor genes TP53 and CDKN2A [6][7][8][13]. These mutations contribute to alterations in telomere maintenance, histone modification and methylation, which affect gene expression and contribute to the development and progression of the disease.
2. Oxidative Stress in Melanomagenesis
3. Preventive and Therapeutic Strategies That Target Oxidative Stress Pathways
3.1. N-Acetylcysteine
3.2. Selenium
3.3. Carotenoids
3.4. Alpha-Tocopherol
3.5. Superoxide Dismutase Mimetics
3.6. Taurine
3.7. Paraoxonase-2 Inhibitors
3.8. Buthionine Sulfoximine
3.9. Disulfiram
3.10. Photodynamic Therapy
3.11. Curcumin
3.12. Resveratrol
3.13. Flavonoids
References
- Lin, J.Y.; Fisher, D.E. Melanocyte Biology and Skin Pigmentation. Nature 2007, 445, 843–850.
- Slominski, R.M.; Zmijewski, M.A.; Slominski, A.T. The Role of Melanin Pigment in Melanoma. Exp. Dermatol. 2015, 24, 258–259.
- Davey, M.G.; Miller, N.; McInerney, N.M. A Review of Epidemiology and Cancer Biology of Malignant Melanoma. Cureus 2021, 13, e15087.
- Flaherty, K.T.; Puzanov, I.; Kim, K.B.; Ribas, A.; McArthur, G.A.; Sosman, J.A.; O’Dwyer, P.J.; Lee, R.J.; Grippo, J.F.; Nolop, K.; et al. Inhibition of Mutated, Activated BRAF in Metastatic Melanoma. N. Engl. J. Med. 2010, 363, 809–819.
- Arslanbaeva, L.R.; Santoro, M.M. Adaptive Redox Homeostasis in Cutaneous Melanoma. Redox Biol. 2020, 37, 101753.
- Mullenders, L.H.F. Solar UV Damage to Cellular DNA: From Mechanisms to Biological Effects. Photochem. Photobiol. Sci. 2018, 17, 1842–1852.
- Davis, E.J.; Johnson, D.B.; Sosman, J.A.; Chandra, S. Melanoma: What Do All the Mutations Mean? Cancer 2018, 124, 3490–3499.
- Schadendorf, D.; van Akkooi, A.C.J.; Berking, C.; Griewank, K.G.; Gutzmer, R.; Hauschild, A.; Stang, A.; Roesch, A.; Ugurel, S. Melanoma. Lancet 2018, 392, 971–984.
- Lim, S.Y.; Menzies, A.M.; Rizos, H. Mechanisms and Strategies to Overcome Resistance to Molecularly Targeted Therapy for Melanoma. Cancer 2017, 123, 2118–2129.
- Leonardi, G.C.; Falzone, L.; Salemi, R.; Zanghì, A.; Spandidos, D.A.; Mccubrey, J.A.; Candido, S.; Libra, M. Cutaneous Melanoma: From Pathogenesis to Therapy (Review). Int. J. Oncol. 2018, 52, 1071–1080.
- Ascierto, P.A.; Kirkwood, J.M.; Grob, J.-J.; Simeone, E.; Grimaldi, A.M.; Maio, M.; Palmieri, G.; Testori, A.; Marincola, F.M.; Mozzillo, N. The Role of BRAF V600 Mutation in Melanoma. J. Transl. Med. 2012, 10, 85.
- Forbes, S.A.; Bhamra, G.; Bamford, S.; Dawson, E.; Kok, C.; Clements, J.; Menzies, A.; Teague, J.W.; Futreal, P.A.; Stratton, M.R. The Catalogue of Somatic Mutations in Cancer (COSMIC). In Current Protocols in Human Genetics; John Wiley & Sons: Hoboken, NJ, USA, 2008; Chapter 10: Unit 10.11.
- Hayward, N.K.; Wilmott, J.S.; Waddell, N.; Johansson, P.A.; Field, M.A.; Nones, K.; Patch, A.-M.; Kakavand, H.; Alexandrov, L.B.; Burke, H.; et al. Whole-Genome Landscapes of Major Melanoma Subtypes. Nature 2017, 545, 175–180.
- Denat, L.; Kadekaro, A.L.; Marrot, L.; Leachman, S.A.; Abdel-Malek, Z.A. Melanocytes as Instigators and Victims of Oxidative Stress. J. Investig. Dermatol. 2014, 134, 1512–1518.
- Davalli, P.; Mitic, T.; Caporali, A.; Lauriola, A.; D’Arca, D. ROS, Cell Senescence, and Novel Molecular Mechanisms in Aging and Age-Related Diseases. Oxidative Med. Cell. Longev. 2016, 2016, 3565127.
- Venza, M.; Visalli, M.; Beninati, C.; De Gaetano, G.V.; Teti, D.; Venza, I. Cellular Mechanisms of Oxidative Stress and Action in Melanoma. Oxidative Med. Cell. Longev. 2015, 2015, 481782.
- Cannavò, S.P.; Tonacci, A.; Bertino, L.; Casciaro, M.; Borgia, F.; Gangemi, S. The Role of Oxidative Stress in the Biology of Melanoma: A Systematic Review. Pathol.-Res. Pract. 2019, 215, 21–28.
- Carvalho, L.A.C.; Queijo, R.G.; Baccaro, A.L.B.; Siena, Á.D.D.; Silva, W.A.; Rodrigues, T.; Maria-Engler, S.S. Redox-Related Proteins in Melanoma Progression. Antioxidants 2022, 11, 438.
- Emanuelli, M.; Sartini, D.; Molinelli, E.; Campagna, R.; Pozzi, V.; Salvolini, E.; Simonetti, O.; Campanati, A.; Offidani, A. The Double-Edged Sword of Oxidative Stress in Skin Damage and Melanoma: From Physiopathology to Therapeutical Approaches. Antioxidants 2022, 11, 612.
- Wheeler, M.D.; Smutney, O.M.; Samulski, R.J. Secretion of Extracellular Superoxide Dismutase from Muscle Transduced with Recombinant Adenovirus Inhibits the Growth of B16 Melanomas in Mice. Mol. Cancer Res. 2003, 1, 871–881.
- Aggarwal, V.; Tuli, H.S.; Varol, A.; Thakral, F.; Yerer, M.B.; Sak, K.; Varol, M.; Jain, A.; Khan, M.d.A.; Sethi, G. Role of Reactive Oxygen Species in Cancer Progression: Molecular Mechanisms and Recent Advancements. Biomolecules 2019, 9, 735.
- Liu-Smith, F.; Dellinger, R.; Meyskens, F.L. Updates of Reactive Oxygen Species in Melanoma Etiology and Progression. Arch. Biochem. Biophys. 2014, 563, 51–55.
- Moloney, J.N.; Cotter, T.G. ROS Signalling in the Biology of Cancer. Semin. Cell Dev. Biol. 2018, 80, 50–64.
- Sander, C.S.; Hamm, F.; Elsner, P.; Thiele, J.J. Oxidative Stress in Malignant Melanoma and Non-Melanoma Skin Cancer. Br. J. Dermatol. 2003, 148, 913–922.
- Ortega, A.L.; Carretero, J.; Obrador, E.; Gambini, J.; Asensi, M.; Rodilla, V.; Estrela, J.M. Tumor Cytotoxicity by Endothelial Cells. Impairment of the Mitochondrial System for Glutathione Uptake in Mouse B16 Melanoma Cells That Survive after in Vitro Interaction with the Hepatic Sinusoidal Endothelium. J. Biol. Chem. 2003, 278, 13888–13897.
- Woźniak, A.; Drewa, G.; Woźniak, B.; Schachtschabel, D.O. Activity of Antioxidant Enzymes and Concentration of Lipid Peroxidation Products in Selected Tissues of Mice of Different Ages, Both Healthy and Melanoma-Bearing. Z. Gerontol. Geriatr. 2004, 37, 184–189.
- Li, X.; Wu, J.; Zhang, X.; Chen, W. Glutathione Reductase-Mediated Thiol Oxidative Stress Suppresses Metastasis of Murine Melanoma Cells. Free Radic. Biol. Med. 2018, 129, 256–267.
- Schott, M.; de Jel, M.M.; Engelmann, J.C.; Renner, P.; Geissler, E.K.; Bosserhoff, A.K.; Kuphal, S. Selenium-Binding Protein 1 Is down-Regulated in Malignant Melanoma. Oncotarget 2018, 9, 10445–10456.
- Chen, H.; Zheng, Z.; Kim, K.-Y.; Jin, X.; Roh, M.R.; Jin, Z. Hypermethylation and Downregulation of Glutathione Peroxidase 3 Are Related to Pathogenesis of Melanoma. Oncol. Rep. 2016, 36, 2737–2744.
- Yi, Z.; Jiang, L.; Zhao, L.; Zhou, M.; Ni, Y.; Yang, Y.; Yang, H.; Yang, L.; Zhang, Q.; Kuang, Y.; et al. Glutathione Peroxidase 3 (GPX3) Suppresses the Growth of Melanoma Cells through Reactive Oxygen Species (ROS)-Dependent Stabilization of Hypoxia-Inducible Factor 1-α and 2-α. J. Cell. Biochem. 2019, 120, 19124–19136.
- Nogués, M.R.; Giralt, M.; Cervelló, I.; Del Castillo, D.; Espeso, O.; Argany, N.; Aliaga, A.; Mallol, J. Parameters Related to Oxygen Free Radicals in Human Skin: A Study Comparing Healthy Epidermis and Skin Cancer Tissue. J. Investig. Dermatol. 2002, 119, 645–652.
- Schadendorf, D.; Jurgovsky, K.; Kohlmus, C.M.; Czarnetzki, B.M. Glutathione and Related Enzymes in Tumor Progression and Metastases of Human Melanoma. J. Investig. Dermatol. 1995, 105, 109–112.
- Kanetsky, P.A.; Holmes, R.; Walker, A.; Najarian, D.; Swoyer, J.; Guerry, D.; Halpern, A.; Rebbeck, T.R. Interaction of Glutathione S-Transferase M1 and T1 Genotypes and Malignant Melanoma. Cancer Epidemiol. Biomark. Prev. 2001, 10, 509–513.
- Kwee, J.K.; Mitidieri, E.; Affonso, O.R. Lowered Superoxide Dismutase in Highly Metastatic B16 Melanoma Cells. Cancer Lett. 1991, 57, 199–202.
- Schadendorf, D.; Zuberbier, T.; Diehl, S.; Schadendorf, C.; Czarnetzki, B.M. Serum Manganese Superoxide Dismutase Is a New Tumour Marker for Malignant Melanoma. Melanoma Res. 1995, 5, 351–353.
- Radojičić, R.M.; Spasić, S.; Saičić, Z.S.; Jovanović, T.B.; Simić-Krstić, J.B. Superoxide Dismutase Activity as a Function of Culture Aging of B-16 Mouse Melanoma Cells. J. Serb. Chem. Soc. 2004, 69, 1005–1011.
- Grammatico, P.; Maresca, V.; Roccella, F.; Roccella, M.; Biondo, L.; Catricalà, C.; Picardo, M. Increased Sensitivity to Peroxidizing Agents Is Correlated with an Imbalance of Antioxidants in Normal Melanocytes from Melanoma Patients. Exp. Dermatol. 1998, 7, 205–212.
- Bisevac, J.P.; Djukic, M.; Stanojevic, I.; Stevanovic, I.; Mijuskovic, Z.; Djuric, A.; Gobeljic, B.; Banovic, T.; Vojvodic, D. Association Between Oxidative Stress and Melanoma Progression. J. Med. Biochem. 2018, 37, 12–20.
- Meyskens, F.L.; McNulty, S.E.; Buckmeier, J.A.; Tohidian, N.B.; Spillane, T.J.; Kahlon, R.S.; Gonzalez, R.I. Aberrant Redox Regulation in Human Metastatic Melanoma Cells Compared to Normal Melanocytes. Free Radic. Biol. Med. 2001, 31, 799–808.
- Wang, J.Y.; Liu, G.Z.; Wilmott, J.S.; La, T.; Feng, Y.C.; Yari, H.; Yan, X.G.; Thorne, R.F.; Scolyer, R.A.; Zhang, X.D.; et al. Skp2-Mediated Stabilization of MTH1 Promotes Survival of Melanoma Cells upon Oxidative Stress. Cancer Res. 2017, 77, 6226–6239.
- Pieri, L.; Dominici, S.; Del Bello, B.; Maellaro, E.; Comporti, M.; Paolicchi, A.; Pompella, A. Redox Modulation of Protein Kinase/Phosphatase Balance in Melanoma Cells: The Role of Endogenous and Gamma-Glutamyltransferase-Dependent H2O2 Production. Biochim. Biophys. Acta 2003, 1621, 76–83.
- Giommarelli, C.; Corti, A.; Supino, R.; Favini, E.; Paolicchi, A.; Pompella, A.; Zunino, F. Cellular Response to Oxidative Stress and Ascorbic Acid in Melanoma Cells Overexpressing Gamma-Glutamyltransferase. Eur. J. Cancer 2008, 44, 750–759.
- Li, J.; Peng, L.; Wu, L.; Kuang, Y.; Su, J.; Yi, M.; Hu, X.; Li, D.; Xie, H.; Kanekura, T.; et al. Depletion of CD147 Sensitizes Human Malignant Melanoma Cells to Hydrogen Peroxide-Induced Oxidative Stress. J. Dermatol. Sci. 2010, 58, 204–210.
- Luo, Y.; Dallaglio, K.; Chen, Y.; Robinson, W.A.; Robinson, S.E.; McCarter, M.D.; Wang, J.; Gonzalez, R.; Thompson, D.C.; Norris, D.A.; et al. ALDH1A Isozymes Are Markers of Human Melanoma Stem Cells and Potential Therapeutic Targets. Stem Cells 2012, 30, 2100–2113.
- Feng, Z.; Hom, M.E.; Bearrood, T.E.; Rosenthal, Z.C.; Fernández, D.; Ondrus, A.E.; Gu, Y.; McCormick, A.K.; Tomaske, M.G.; Marshall, C.R.; et al. Targeting Colorectal Cancer with Small-Molecule Inhibitors of ALDH1B1. Nat. Chem. Biol. 2022, 18, 1065–1075.
- Mameishvili, E.; Serafimidis, I.; Iwaszkiewicz, S.; Lesche, M.; Reinhardt, S.; Bölicke, N.; Büttner, M.; Stellas, D.; Papadimitropoulou, A.; Szabolcs, M.; et al. Aldh1b1 Expression Defines Progenitor Cells in the Adult Pancreas and Is Required for Kras-Induced Pancreatic Cancer. Proc. Natl. Acad. Sci. USA 2019, 116, 20679–20688.
- Wu, L.; Dong, B.; Zhang, F.; Li, Y.; Liu, L. Prediction of the Engendering Mechanism and Specific Genes of Primary Melanoma by Bioinformatics Analysis. Dermatol. Sin. 2016, 34, 14–19.
- Yan, B.Y.; Garcet, S.; Gulati, N.; Kiecker, F.; Fuentes-Duculan, J.; Gilleaudeau, P.; Sullivan-Whalen, M.; Shemer, A.; Mitsui, H.; Krueger, J.G. Novel Immune Signatures Associated with Dysplastic Naevi and Primary Cutaneous Melanoma in Human Skin. Exp. Dermatol. 2019, 28, 35–44.
- Rózanowska, M.; Sarna, T.; Land, E.J.; Truscott, T.G. Free Radical Scavenging Properties of Melanin Interaction of Eu- and Pheo-Melanin Models with Reducing and Oxidising Radicals. Free Radic. Biol. Med. 1999, 26, 518–525.
- El-Naggar, N.E.-A.; El-Ewasy, S.M. Bioproduction, Characterization, Anticancer and Antioxidant Activities of Extracellular Melanin Pigment Produced by Newly Isolated Microbial Cell Factories Streptomyces Glaucescens NEAE-H. Sci. Rep. 2017, 7, 42129.
- Slominski, A.; Kim, T.K.; Brozyna, A.A.; Janjetovic, Z.; Brooks, D.L.; Schwab, L.P.; Skobowiat, C.; Jozwicki, W.; Seagroves, T.N. The Role of Melanogenesis in Regulation of Melanoma Behavior: Melanogenesis Leads to Stimulation of HIF-1alpha Expression and HIF-Dependent Attendant Pathways. Arch. Biochem. Biophys. 2014, 563, 79–93.
- Trachootham, D.; Alexandre, J.; Huang, P. Targeting Cancer Cells by ROS-Mediated Mechanisms: A Radical Therapeutic Approach? Nat. Rev. Drug Discov. 2009, 8, 579–591.
- Goodson, A.G.; Cotter, M.A.; Cassidy, P.; Wade, M.; Florell, S.R.; Liu, T.; Boucher, K.M.; Grossman, D. Use of Oral N-Acetylcysteine for Protection of Melanocytic Nevi against UV-Induced Oxidative Stress: Towards a Novel Paradigm for Melanoma Chemoprevention. Clin. Cancer Res. 2009, 15, 7434–7440.
- Cotter, M.A.; Thomas, J.; Cassidy, P.; Robinette, K.; Jenkins, N.; Florell, S.R.; Leachman, S.; Samlowski, W.E.; Grossman, D. N-Acetylcysteine Protects Melanocytes against Oxidative Stress/Damage and Delays Onset of Ultraviolet-Induced Melanoma in Mice. Clin. Cancer Res. 2007, 13, 5952–5958.
- Cassidy, P.B.; Fain, H.D.; Cassidy, J.P.; Tran, S.M.; Moos, P.J.; Boucher, K.M.; Gerads, R.; Florell, S.R.; Grossman, D.; Leachman, S.A. Selenium for the Prevention of Cutaneous Melanoma. Nutrients 2013, 5, 725–749.
- Gębka, N.; Gębka-Kępińska, B.; Adamczyk, J.; Mizgała-Izworska, E. The Use of Flavonoids in Skin Cancer Prevention and Treatment. J. Pre. Clin. Clin. Res. 2022, 16, 108–113.
- Chung, J.H.; Seo, J.Y.; Lee, M.K.; Eun, H.C.; Lee, J.H.; Kang, S.; Fisher, G.J.; Voorhees, J.J. Ultraviolet Modulation of Human Macrophage Metalloelastase in Human Skin in Vivo. J. Investig. Dermatol. 2002, 119, 507–512.
- Sfaxi, I.H.; Ferraro, D.; Fasano, E.; Pani, G.; Limam, F.; Marzouki, M.N. Inhibitory Effects of a Manganese Superoxide Dismutase Isolated from Garlic (Allium sativum L.) on in Vitro Tumoral Cell Growth. Biotechnol. Prog. 2009, 25, 257–264.
- Yu, J.; Kim, A.K. Effect of Taurine on Antioxidant Enzyme System in B16F10 Melanoma Cells. Adv. Exp. Med. Biol. 2009, 643, 491–499.
- Obrador, E.; Salvador-Palmer, R.; López-Blanch, R.; Oriol-Caballo, M.; Moreno-Murciano, P.; Estrela, J.M. N-Acetylcysteine Promotes Metastatic Spread of Melanoma in Mice. Cancers 2022, 14, 3614.
- Kashif, M.; Yao, H.; Schmidt, S.; Chen, X.; Truong, M.; Tüksammel, E.; Liu, Y.; Bergo, M.O. ROS-Lowering Doses of Vitamins C and A Accelerate Malignant Melanoma Metastasis. Redox Biol. 2023, 60, 102619.
- Campagna, R.; Bacchetti, T.; Salvolini, E.; Pozzi, V.; Molinelli, E.; Brisigotti, V.; Sartini, D.; Campanati, A.; Ferretti, G.; Offidani, A.; et al. Paraoxonase-2 Silencing Enhances Sensitivity of A375 Melanoma Cells to Treatment with Cisplatin. Antioxidants 2020, 9, 1238.
- Ongaro, A.; Pellati, A.; De Mattei, M.; De Terlizzi, F.; Rossi, C.R.; Campana, L.G. Enhancement of Melphalan Activity by Buthionine Sulfoximine and Electroporation in Melanoma Cells. Anticancer Drugs 2015, 26, 284–292.
- Calderon-Aparicio, A.; Cornejo, A.; Orue, A.; Rieber, M. Anticancer Response to Disulfiram May Be Enhanced by Co-Treatment with MEK Inhibitor or Oxaliplatin: Modulation by Tetrathiomolybdate, KRAS/BRAF Mutations and c-MYC/P53 Status. Ecancermedicalscience 2019, 13, 890.
- Cen, D.; Brayton, D.; Shahandeh, B.; Meyskens, F.L.; Farmer, P.J. Disulfiram Facilitates Intracellular Cu Uptake and Induces Apoptosis in Human Melanoma Cells. J. Med. Chem. 2004, 47, 6914–6920.
- Morrison, B.W.; Doudican, N.A.; Patel, K.R.; Orlow, S.J. Disulfiram Induces Copper-Dependent Stimulation of Reactive Oxygen Species and Activation of the Extrinsic Apoptotic Pathway in Melanoma. Melanoma Res. 2010, 20, 11–20.
- Fontes, S.S.; Nogueira, M.L.; Dias, R.B.; Rocha, C.A.G.; Soares, M.B.P.; Vannier-Santos, M.A.; Bezerra, D.P. Combination Therapy of Curcumin and Disulfiram Synergistically Inhibits the Growth of B16-F10 Melanoma Cells by Inducing Oxidative Stress. Biomolecules 2022, 12, 1600.
- Bush, J.A.; Cheung, K.J.; Li, G. Curcumin Induces Apoptosis in Human Melanoma Cells through a Fas Receptor/Caspase-8 Pathway Independent of P53. Exp. Cell Res. 2001, 271, 305–314.
- Kocyigit, A.; Guler, E.M. Curcumin Induce DNA Damage and Apoptosis through Generation of Reactive Oxygen Species and Reducing Mitochondrial Membrane Potential in Melanoma Cancer Cells. Cell. Mol. Biol. 2017, 63, 97–105.
- Bhattacharya, S.; Darjatmoko, S.R.; Polans, A.S. Resveratrol Modulates the Malignant Properties of Cutaneous Melanoma through Changes in the Activation and Attenuation of the Antiapoptotic Protooncogenic Protein Akt/PKB. Melanoma Res. 2011, 21, 180–187.
- Chen, Y.-J.; Chen, Y.-Y.; Lin, Y.-F.; Hu, H.-Y.; Liao, H.-F. Resveratrol Inhibits Alpha-Melanocyte-Stimulating Hormone Signaling, Viability, and Invasiveness in Melanoma Cells. Evid.-Based Complement. Alternat. Med. 2013, 2013, 632121.
- Heo, J.-R.; Kim, S.-M.; Hwang, K.-A.; Kang, J.-H.; Choi, K.-C. Resveratrol Induced Reactive Oxygen Species and Endoplasmic Reticulum Stress-mediated Apoptosis, and Cell Cycle Arrest in the A375SM Malignant Melanoma Cell Line. Int. J. Mol. Med. 2018, 42, 1427–1435.
- Lu, J.; Papp, L.V.; Fang, J.; Rodriguez-Nieto, S.; Zhivotovsky, B.; Holmgren, A. Inhibition of Mammalian Thioredoxin Reductase by Some Flavonoids: Implications for Myricetin and Quercetin Anticancer Activity. Cancer Res. 2006, 66, 4410–4418.
- Netcharoensirisuk, P.; Abrahamian, C.; Tang, R.; Chen, C.-C.; Rosato, A.S.; Beyers, W.; Chao, Y.-K.; Filippini, A.; Di Pietro, S.; Bartel, K.; et al. Flavonoids Increase Melanin Production and Reduce Proliferation, Migration and Invasion of Melanoma Cells by Blocking Endolysosomal/Melanosomal TPC2. Sci. Rep. 2021, 11, 8515.
- Liu-Smith, F.; Meyskens, F.L. Molecular Mechanisms of Flavonoids in Melanin Synthesis and the Potential for the Prevention and Treatment of Melanoma. Mol. Nutr. Food Res. 2016, 60, 1264–1274.
- Balyan, R.; Kudugunti, S.K.; Hamad, H.A.; Yousef, M.S.; Moridani, M.Y. Bioactivation of Luteolin by Tyrosinase Selectively Inhibits Glutathione S-Transferase. Chem. Biol. Interact. 2015, 240, 208–218.
- Cucci, M.A.; Grattarola, M.; Dianzani, C.; Damia, G.; Ricci, F.; Roetto, A.; Trotta, F.; Barrera, G.; Pizzimenti, S. Ailanthone Increases Oxidative Stress in CDDP-Resistant Ovarian and Bladder Cancer Cells by Inhibiting of Nrf2 and YAP Expression through a Post-Translational Mechanism. Free Radic. Biol. Med. 2020, 150, 125–135.
- Daga, M.; Pizzimenti, S.; Dianzani, C.; Cucci, M.A.; Cavalli, R.; Grattarola, M.; Ferrara, B.; Scariot, V.; Trotta, F.; Barrera, G. Ailanthone Inhibits Cell Growth and Migration of Cisplatin Resistant Bladder Cancer Cells through Down-Regulation of Nrf2, YAP, and c-Myc Expression. Phytomedicine 2019, 56, 156–164.
- Ding, H.; Yu, X.; Hang, C.; Gao, K.; Lao, X.; Jia, Y.; Yan, Z. Ailanthone: A Novel Potential Drug for Treating Human Cancer. Oncol. Lett. 2020, 20, 1489–1503.
- Lara-Vega, I.; Vega-López, A. Combinational Photodynamic and Photothermal—Based Therapies for Melanoma in Mouse Models. Photodiagn. Photodyn. Ther. 2023, in press.
- Piskounova, E.; Agathocleous, M.; Murphy, M.M.; Hu, Z.; Huddlestun, S.E.; Zhao, Z.; Leitch, A.M.; Johnson, T.M.; DeBerardinis, R.J.; Morrison, S.J. Oxidative Stress Inhibits Distant Metastasis by Human Melanoma Cells. Nature 2015, 527, 186–191.
- Le Gal, K.; Ibrahim, M.X.; Wiel, C.; Sayin, V.I.; Akula, M.K.; Karlsson, C.; Dalin, M.G.; Akyürek, L.M.; Lindahl, P.; Nilsson, J.; et al. Antioxidants Can Increase Melanoma Metastasis in Mice. Sci. Transl. Med. 2015, 7, 308re8.
- Schrauzer, G.N.; Surai, P.F. Selenium in Human and Animal Nutrition: Resolved and Unresolved Issues. A Partly Historical Treatise in Commemoration of the Fiftieth Anniversary of the Discovery of the Biological Essentiality of Selenium, Dedicated to the Memory of Klaus Schwarz (1914–1978) on the Occasion of the Thirtieth Anniversary of His Death. Crit. Rev. Biotechnol. 2009, 29, 2–9.
- Blot, W.J.; Li, J.Y.; Taylor, P.R.; Guo, W.; Dawsey, S.; Wang, G.Q.; Yang, C.S.; Zheng, S.F.; Gail, M.; Li, G.Y. Nutrition Intervention Trials in Linxian, China: Supplementation with Specific Vitamin/Mineral Combinations, Cancer Incidence, and Disease-Specific Mortality in the General Population. J. Natl. Cancer Inst. 1993, 85, 1483–1492.
- Maoka, T. Carotenoids as Natural Functional Pigments. J. Nat. Med. 2020, 74, 1–16.
- Asgari, M.M.; Brasky, T.M.; White, E. Association of Vitamin A and Carotenoid Intake with Melanoma Risk in a Large Prospective Cohort. J. Investig. Dermatol. 2012, 132, 1573–1582.
- Chhabra, G.; Ndiaye, M.A.; Garcia-Peterson, L.M.; Ahmad, N. Melanoma Chemoprevention: Current Status and Future Prospects. Photochem. Photobiol. 2017, 93, 975–989.
- Miura, K.; Green, A.C. Dietary Antioxidants and Melanoma: Evidence from Cohort and Intervention Studies. Nutr. Cancer 2015, 67, 867–876.
- Hercberg, S.; Galan, P.; Preziosi, P.; Bertrais, S.; Mennen, L.; Malvy, D.; Roussel, A.-M.; Favier, A.; Briançon, S. The SU.VI.MAX Study: A Randomized, Placebo-Controlled Trial of the Health Effects of Antioxidant Vitamins and Minerals. Arch. Intern. Med. 2004, 164, 2335–2342.
- Robbins, D.; Zhao, Y. Manganese Superoxide Dismutase in Cancer Prevention. Antioxid. Redox Signal. 2014, 20, 1628–1645.
- Tu, S.; Zhang, X.-L.; Wan, H.-F.; Xia, Y.-Q.; Liu, Z.-Q.; Yang, X.-H.; Wan, F.-S. Effect of Taurine on Cell Proliferation and Apoptosis Human Lung Cancer A549 Cells. Oncol Lett 2018, 15, 5473–5480.
- Mates, J.M.; Segura, J.A.; Alonso, F.J.; Marquez, J. Sulphur-Containing Non Enzymatic Antioxidants: Therapeutic Tools against Cancer. Front Biosci (Schol Ed) 2012, 4, 722–748.
- Higuchi, K.; Sugiyama, K.; Tomabechi, R.; Kishimoto, H.; Inoue, K. Mammalian Monocarboxylate Transporter 7 (MCT7/Slc16a6) Is a Novel Facilitative Taurine Transporter. J. Biol. Chem. 2022, 298, 101800.
- Altenhöfer, S.; Witte, I.; Teiber, J.F.; Wilgenbus, P.; Pautz, A.; Li, H.; Daiber, A.; Witan, H.; Clement, A.M.; Förstermann, U.; et al. One Enzyme, Two Functions: PON2 Prevents Mitochondrial Superoxide Formation and Apoptosis Independent from Its Lactonase Activity. J. Biol. Chem. 2010, 285, 24398–24403.
- Devarajan, A.; Bourquard, N.; Hama, S.; Navab, M.; Grijalva, V.R.; Morvardi, S.; Clarke, C.F.; Vergnes, L.; Reue, K.; Teiber, J.F.; et al. Paraoxonase 2 Deficiency Alters Mitochondrial Function and Exacerbates the Development of Atherosclerosis. Antioxid. Redox Signal. 2011, 14, 341–351.
- Bacchetti, T.; Salvolini, E.; Pompei, V.; Campagna, R.; Molinelli, E.; Brisigotti, V.; Togni, L.; Lucarini, G.; Sartini, D.; Campanati, A.; et al. Paraoxonase-2: A Potential Biomarker for Skin Cancer Aggressiveness. Eur. J. Clin. Investig. 2021, 51, e13452.
- Meraz-Torres, F.; Plöger, S.; Garbe, C.; Niessner, H.; Sinnberg, T. Disulfiram as a Therapeutic Agent for Metastatic Malignant Melanoma-Old Myth or New Logos? Cancers 2020, 12, 3538.
- Cvek, B.; Dvorak, Z. The Value of Proteasome Inhibition in Cancer. Can the Old Drug, Disulfiram, Have a Bright New Future as a Novel Proteasome Inhibitor? Drug Discov. Today 2008, 13, 716–722.
- Li, C.; Zhou, S.; Chen, C.; Zhu, L.; Li, S.; Song, Z.; Liang, J.; Tang, C.; Xu, N.; Liu, T.; et al. DDTC-Cu(I) Based Metal-Organic Framework (MOF) for Targeted Melanoma Therapy by Inducing SLC7A11/GPX4-Mediated Ferroptosis. Colloids Surf. B Biointerfaces 2023, 225, 113253.
- Tang, S.-J.; Wang, M.-F.; Yang, R.; Liu, M.; Li, Q.-F.; Gao, F. More-Is-Better Strategy for Constructing Homoligand Polypyridyl Ruthenium Complexes as Photosensitizers for Infrared Two-Photon Photodynamic Therapy. Inorg. Chem. 2023, 62, 8210–8218.
- Balas, M.; Nistorescu, S.; Badea, M.A.; Dinischiotu, A.; Boni, M.; Dinache, A.; Smarandache, A.; Udrea, A.-M.; Prepelita, P.; Staicu, A. Photodynamic Activity of TMPyP4/TiO2 Complex under Blue Light in Human Melanoma Cells: Potential for Cancer-Selective Therapy. Pharmaceutics 2023, 15, 1194.
- Xin, J.; Wang, J.; Yao, Y.; Wang, S.; Zhang, Z.; Yao, C. Improved Simulated-Daylight Photodynamic Therapy and Possible Mechanism of Ag-Modified TiO2 on Melanoma. Int. J. Mol. Sci. 2023, 24, 7061.
- Salehi, B.; Mishra, A.P.; Nigam, M.; Sener, B.; Kilic, M.; Sharifi-Rad, M.; Fokou, P.V.T.; Martins, N.; Sharifi-Rad, J. Resveratrol: A Double-Edged Sword in Health Benefits. Biomedicines 2018, 6, 91.
- Ravikumar, P.; Katariya, M.; Patil, S.; Tatke, P.; Pillai, R. Skin Delivery of Resveratrol Encapsulated Lipidic Formulation for Melanoma Chemoprevention. J. Microencapsul. 2019, 36, 535–551.
- Davoodvandi, A.; Darvish, M.; Borran, S.; Nejati, M.; Mazaheri, S.; Reza Tamtaji, O.; Hamblin, M.R.; Masoudian, N.; Mirzaei, H. The Therapeutic Potential of Resveratrol in a Mouse Model of Melanoma Lung Metastasis. Int. Immunopharmacol. 2020, 88, 106905.
- Júnior, R.G.d.O.; Ferraz, C.A.A.; Silva, M.G.e.; Lavor, É.M.d.; Rolim, L.A.; Lima, J.T.; Fleury, A.; Picot, L.; Siqueira Quintans, J.; Júnior, L.; et al. Flavonoids: Promising Natural Products for Treatment of Skin Cancer (Melanoma). In Natural Products and Cancer Drug Discovery; Intech: London, UK, 2017.
- Carpenter, E.L.; Wyant, M.B.; Indra, A.; Ito, S.; Wakamatsu, K.; Merrill, G.F.; Moos, P.J.; Cassidy, P.B.; Leachman, S.A.; Ganguli-Indra, G.; et al. Thioredoxin Reductase 1 Modulates Pigmentation and Photobiology of Murine Melanocytes in Vivo. J. Investig. Dermatol. 2022, 142, 1903–1911.e5.





