
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Eduardo Rocca | -- | 1792 | 2023-05-29 11:48:20 | | | |
| 2 | Catherine Yang | Meta information modification | 1792 | 2023-05-30 02:55:29 | | |
Video Upload Options
Lung ultrasound (LUS) has become a part of the daily examination of physicians working in intensive, sub-intensive, and general medical wards. The easy access to hand-held ultrasound machines in wards where they were not available in the past facilitated the widespread use of ultrasound, both for clinical examination and as a guide to procedures. The integration of bedside ultrasonography in the daily clinical activity of intensivists could reduce the risk of radiation exposure, need of patient transport, and hospital costs and may redirect patient’s management. A qualitative LUS approach is based on interpreting artifacts (A- and B-lines) and real images to distinguish between normal and pathological context. If a qualitative approach gives important information on the morphological assessment of the lung for the diagnosis, a quantitative approach allows us to extend the utility of the examination to lung monitoring.
1. Machine Setting and Probes

2. LUS Examination
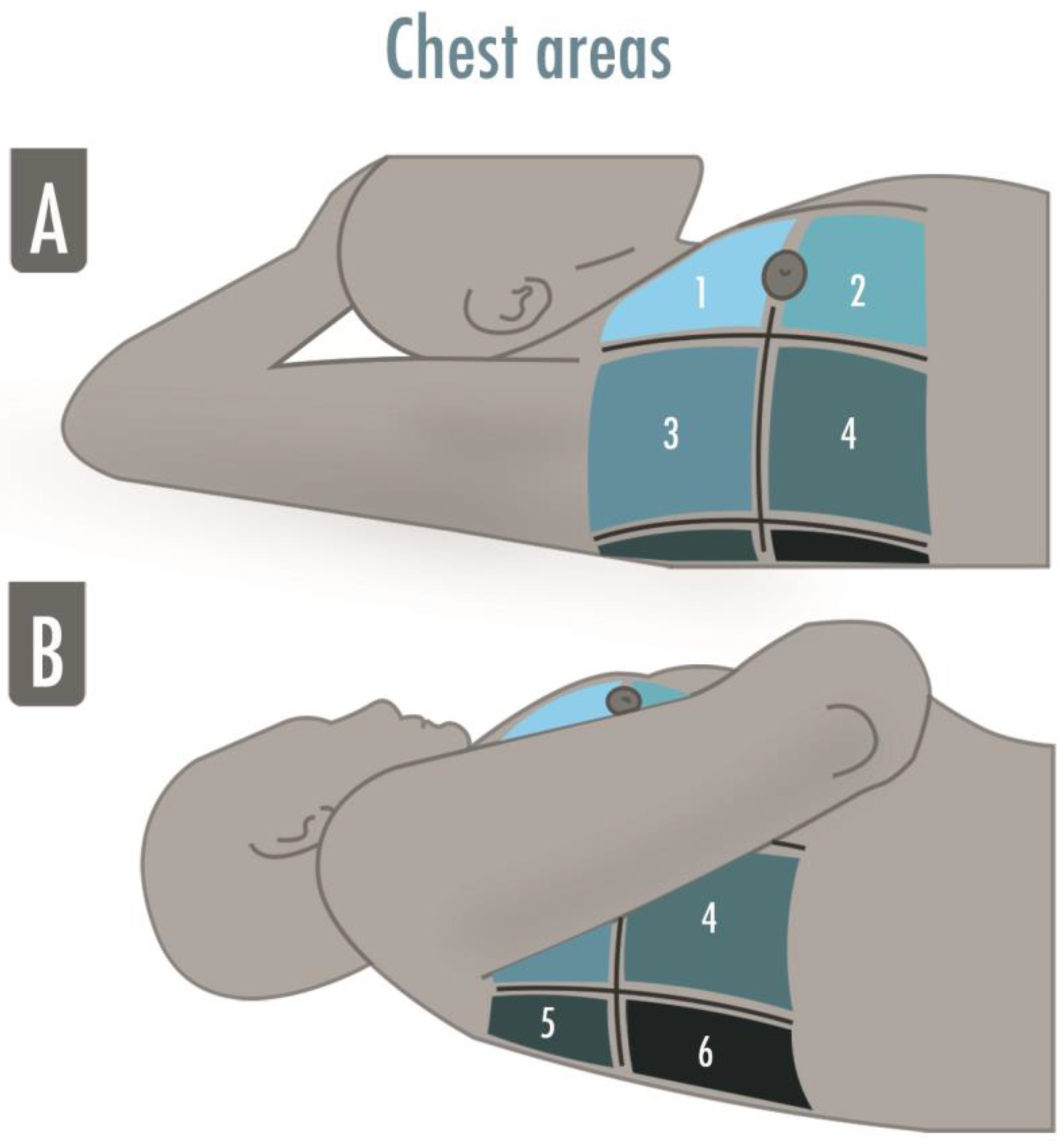
3. Obtaining and Optimizing LUS Images
4. Semeiotic
- -
-
Bat sign: the pleural line (bat’s body) is a horizontal hyperechoic line usually visualized in adult patients 0.5 cm below the ribs (bat’s wings) in a longitudinal approach; this is a basic landmark useful for proper identification of intercostal space and pleura. It is important, especially in those patients where it is difficult to identify the intercostal space as in case of subcutaneous emphysema or morbid obesity [3];
- -
-
A-lines: horizontal artifacts visualized as hyperechoic lines below the pleural line; repeated at a constant distance equal to the distance between the pleural line and the probe [4]; they are generated by the reverberation of the ultrasound beam between the pleura and the probe. The A-lines tell us there is air beneath the pleural line and correlate well with the high gas/volume ratio [2]; when associated with lung sliding, they correspond to normal lungs; otherwise, they can also be visualized in the case of hyperinflation and pneumothorax [1];
- -
-
B-lines: vertical artifacts originating from the pleural line, moving synchronously with it, erasing the A-lines and reaching the bottom of the screen. They are generated by increased density beneath the visceral pleura (altered air/tissue ratio) [5];
- -
-
Lung sliding: movement of the pleural line synchronous with tidal ventilation, it indicates that visceral and parietal pleura are in touch and regional ventilation is present [1];
- -
-
Seashore sign: straight lines above the pleural line and sandy pattern below the pleural line visualized in M-mode, confirm the lung sliding [1];
- -
-
Stratosphere sign: straight horizontal lines above and beneath the pleural line visualized in M-mode corresponding to an absence of pleural line movement, suggesting parietal and visceral pleura may not be in touch (i.e., pneumothorax), but also present in emphysematous bullae, pleural adherences, and severe hyperinflation;
- -
-
Lung Pulse: movement of the pleural line synchronous to the cardiac rhythm caused by the transmission of the heart beats; heart beats are always visible between breaths, but the sign is defined as a lung pulse only in the absence of lung sliding. It indicates that the pleurae are in touch, but regional ventilation is impaired (e.g., selective intubation, initial phase of atelectasis, pulmonary contusion, hyperinflation) [6][7];
- -
-
Lung Point: contact point between collapsed lung and pneumothorax air collection; a normal LUS pattern is visualized close to a motionless pleura; in M-mode, it could be visualized as an alternation between seashore and stratosphere sign, it represents the lateral edge of the intrapleural air layer [8].
- -
-
Shred sign: subpleural echo-poor images delimitated by irregular borders, indicating juxtapleural small consolidation [1];
- -
-
Tissue-like pattern: homogeneous texture of a lobe, similar to abdominal parenchyma, corresponds to a complete loss of aeration [9];
- -
-
Air bronchogram: hyperechoic intraparenchymal images visualized within a tissue-like pattern that corresponds to air trapped within the consolidation and that could be classified in absent, static (not patent airway), and dynamic (patent airways); the latter is then subclassified in linear/arborescent (specific for ventilator associated pneumonia) or punctiform (low specificity);
- -
-
Pleural effusions: hypo or anechoic space between the pleurae usually visualized in the most dependent areas of the chest. Its position could change in accordance with the patient’s posture, and the lung could be floating in it or compressed by it in a tissue-like pattern. Its echogenicity could help in differentiating the effusion’s type: transudative (i.e., homogeneously anechoic) or exudative (i.e., anechoic or homogeneously echogenic with internal echoes, fibrin strands, or septation).
5. Score for Lung Aeration Quantification

References
- Mojoli, F.; Bouhemad, B.; Mongodi, S.; Lichtenstein, D.A. Lung Ultrasound for Critically Ill Patients. Am. J. Respir. Crit. Care Med. 2019, 199, 701–714.
- Mongodi, S.; De Luca, D.; Colombo, A.; Stella, A.; Santangelo, E.; Corradi, F.; Gargani, L.; Rovida, S.; Volpicelli, S.; Bouhemad, B. Quantitative Lung Ultrasound: Technical Aspects and Clinical Applications. Anesthesiology 2021, 134, 949–965.
- Volpicelli, G.; Gargani, L. How I do it: Lung ultrasound. Cardiovasc. Ultrasound 2014, 12, 25.
- Bhoil, R.; Ahluwalia, A.; Chopra, R.; Surya, M.; Bhoil, S. Signs and lines in lung ultrasound. J. Ultrason. 2021, 21, e225–e233.
- Lichtenstein, D.A.; Meziere, G.A. Relevance of lung ultrasound in the diagnosis of acute respiratory failure: The BLUE protocol. Chest 2008, 134, 117–125.
- Lichtenstein, D.A.; Lascols, N.; Prin, S.; Meziere, G. The lung pulse: An early ultrasound sign of complete atelectasis. Intensive Care Med. 2003, 29, 2187–2192.
- Volpicelli, G. Sonographic diagnosis of pneumothorax. Intensive Care Med. 2011, 37, 224–232.
- Blaivas, M.; Lyon, M.; Duggal, S. A prospective comparison of supine chest radiography and bedside ultrasound for the diagnosis of traumatic pneumothorax. Acad. Emerg. Med. 2005, 12, 844–849.
- Mongodi, S.; Bouhemad, B.; Iotti, G.A.; Mojoli, F. An ultrasonographic sign of intrapulmonary shunt. Intensive Care Med. 2016, 42, 912–913.
- Chiumello, D.; Mongodi, S.; Algieri, I.; Vergani, G.L.; Orlando, A.; Via, G.; Crimella, F.; Cressoni, M.; Mojoli, F. Assessment of Lung Aeration and Recruitment by CT Scan and Ultrasound in Acute Respiratory Distress Syndrome Patients. Crit. Care Med. 2018, 46, 1761–1768.





