
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Mohammad Al-Sayah | -- | 2146 | 2023-05-27 09:42:53 | | | |
| 2 | Catherine Yang | Meta information modification | 2146 | 2023-05-29 05:05:34 | | |
Video Upload Options
All currently approved antibiotics are being met by some degree of resistance by the bacteria they target. Biofilm formation is one of the crucial enablers of bacterial resistance, making it an important bacterial process to target for overcoming antibiotic resistance. Accordingly, several drug delivery systems that target biofilm formation have been developed. One of these systems is based on lipid-based nanocarriers (liposomes), which have shown strong efficacy against biofilms of bacterial pathogens. Liposomes come in various types, namely conventional (charged or neutral), stimuli-responsive, deformable, targeted, and stealth. This research reviews studies employing liposomal formulations against biofilms of medically salient gram-negative and gram-positive bacterial species reported recently.
1. Liposomes: Versatile Drug Delivery Systems
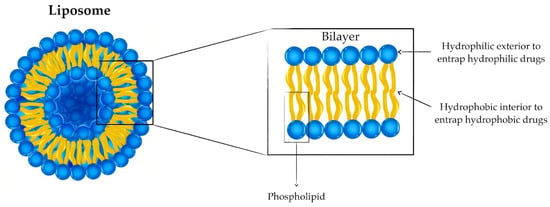
| Liposome Type | Description | Advantages | Limitations | Refs. |
|---|---|---|---|---|
| Conventional (cationic, anionic, neutral) | Bilayer composed of neutral, positively or negatively charged phospholipids | Reduced drug-associated toxicity | Rapid removal from circulation |
[11][12] |
| Deformable (elastic) | Elastic lipid bilayer | Deeper skin penetration for topical applications | Cargo release during prolonged storage times |
[17] |
| Stealth | PEG-coated liposomes | Prolonged circulation time | Non-biodegradability of high molar mass PEG and toxicity of low molar mass PEG |
[12][18] |
| Targeted | Liposomes surface modified with targeting moieties such as antibodies | Specificity | Rapid elimination from the bloodstream |
[24] |
| Stimuli-responsive | Liposomes change conformation and release encapsulated cargo in response to external and internal triggers | Reduced off-target toxicity | * | [13][33][34] |
2. Liposomal Formulations for the Treatment of Gram-Negative Biofilms
3. Liposomal Formulations for the Treatment of Gram-Positive Biofilms
References
- Wang, Y. Liposome as a Delivery System for the Treatment of Biofilm-mediated Infections. J. Appl. Microbiol. 2021, 131, 2626–2639.
- Wang, D.-Y.; van der Mei, H.C.; Ren, Y.; Busscher, H.J.; Shi, L. Lipid-Based Antimicrobial Delivery-Systems for the Treatment of Bacterial Infections. Front. Chem. 2020, 7, 872.
- Nsairat, H.; Khater, D.; Sayed, U.; Odeh, F.; Al Bawab, A.; Alshaer, W. Liposomes: Structure, Composition, Types, and Clinical Applications. Heliyon 2022, 8, e09394.
- Rukavina, Z.; Šegvić Klarić, M.; Filipović-Grčić, J.; Lovrić, J.; Vanić, Ž. Azithromycin-Loaded Liposomes for Enhanced Topical Treatment of Methicillin-Resistant Staphyloccocus Aureus (MRSA) Infections. Int. J. Pharm. 2018, 553, 109–119.
- Aiello, S.; Pagano, L.; Ceccacci, F.; Simonis, B.; Sennato, S.; Bugli, F.; Martini, C.; Torelli, R.; Sanguinetti, M.; Ciogli, A.; et al. Mannosyl, Glucosyl or Galactosyl Liposomes to Improve Resveratrol Efficacy against Methicillin Resistant Staphylococcus Aureus Biofilm. Colloids Surf. A Physicochem. Eng. Asp. 2021, 617, 126321.
- Alzahrani, N.M.; Booq, R.Y.; Aldossary, A.M.; Bakr, A.A.; Almughem, F.A.; Alfahad, A.J.; Alsharif, W.K.; Jarallah, S.J.; Alharbi, W.S.; Alsudir, S.A.; et al. Liposome-Encapsulated Tobramycin and IDR-1018 Peptide Mediated Biofilm Disruption and Enhanced Antimicrobial Activity against Pseudomonas Aeruginosa. Pharmaceutics 2022, 14, 960.
- Kluzek, M.; Oppenheimer-Shaanan, Y.; Dadosh, T.; Morandi, M.I.; Avinoam, O.; Raanan, C.; Goldsmith, M.; Goldberg, R.; Klein, J. Designer Liposomic Nanocarriers Are Effective Biofilm Eradicators. ACS Nano 2022, 16, 15792–15804.
- Nicolosi, D.; Scalia, M.; Nicolosi, V.M.; Pignatello, R. Encapsulation in Fusogenic Liposomes Broadens the Spectrum of Action of Vancomycin against Gram-Negative Bacteria. Int. J. Antimicrob. Agents 2010, 35, 553–558.
- Scriboni, A.B.; Couto, V.M.; de Morais Ribeiro, L.N.; Freires, I.A.; Groppo, F.C.; de Paula, E.; Franz-Montan, M.; Cogo-Müller, K. Fusogenic Liposomes Increase the Antimicrobial Activity of Vancomycin Against Staphylococcus Aureus Biofilm. Front. Pharmacol. 2019, 10, 1401.
- Zahra, M.-J.; Hamed, H.; Mohammad, R.-Y.; Nosratollah, Z.; Akbarzadeh, A.; Morteza, M. Evaluation and Study of Antimicrobial Activity of Nanoliposomal Meropenem against Pseudomonas Aeruginosa Isolates. Artif. Cells Nanomed. Biotechnol. 2017, 45, 975–980.
- Sercombe, L.; Veerati, T.; Moheimani, F.; Wu, S.Y.; Sood, A.K.; Hua, S. Advances and Challenges of Liposome Assisted Drug Delivery. Front. Pharmacol. 2015, 6, 286.
- Vassallo, A.; Silletti, M.F.; Faraone, I.; Milella, L. Nanoparticulate Antibiotic Systems as Antibacterial Agents and Antibiotic Delivery Platforms to Fight Infections. J. Nanomater. 2020, 2020, 6905631.
- Antoniou, A.I.; Giofrè, S.; Seneci, P.; Passarella, D.; Pellegrino, S. Stimulus-Responsive Liposomes for Biomedical Applications. Drug Discov. Today 2021, 26, 1794–1824.
- Kannan, S.; Solomon, A.; Krishnamoorthy, G.; Marudhamuthu, M. Liposome Encapsulated Surfactant Abetted Copper Nanoparticles Alleviates Biofilm Mediated Virulence in Pathogenic Pseudomonas Aeruginosa and MRSA. Sci. Rep. 2021, 11, 1102.
- Harper, R.A.; Carpenter, G.H.; Proctor, G.B.; Harvey, R.D.; Gambogi, R.J.; Geonnotti, A.R.; Hider, R.; Jones, S.A. Diminishing Biofilm Resistance to Antimicrobial Nanomaterials through Electrolyte Screening of Electrostatic Interactions. Colloids Surf. B Biointerfaces 2019, 173, 392–399.
- Elsayed, M.M.A.; Abdallah, O.Y.; Naggar, V.F.; Khalafallah, N.M. Deformable Liposomes and Ethosomes: Mechanism of Enhanced Skin Delivery. Int. J. Pharm. 2006, 322, 60–66.
- Nayak, D.; Tippavajhala, V.K. A Comprehensive Review on Preparation, Evaluation and Applications of Deformable Liposomes. Iran. J. Pharm. Res. 2021, 20, 186.
- Knop, K.; Hoogenboom, R.; Fischer, D.; Schubert, U.S. Poly(Ethylene Glycol) in Drug Delivery: Pros and Cons as Well as Potential Alternatives. Angew. Chem. Int. Ed. 2010, 49, 6288–6308.
- Gabizon, A.; Catane, R.; Uziely, B.; Kaufman, B.; Safra, T.; Cohen, R.; Martin, F.; Huang, A.; Barenholz, Y. Prolonged Circulation Time and Enhanced Accumulation in Malignant Exudates of Doxorubicin Encapsulated in Polyethylene-Glycol Coated Liposomes. Cancer Res. 1994, 54, 987–992.
- Ghaferi, M.; Raza, A.; Koohi, M.; Zahra, W.; Akbarzadeh, A.; Ebrahimi Shahmabadi, H.; Alavi, S.E. Impact of PEGylated Liposomal Doxorubicin and Carboplatin Combination on Glioblastoma. Pharmaceutics 2022, 14, 2183.
- Maruyama, K.; Yuda, T.; Okamoto, A.; Kojima, S.; Suginaka, A.; Iwatsuru, M. Prolonged Circulation Time in Vivo of Large Unilamellar Liposomes Composed of Distearoyl Phosphatidylcholine and Cholesterol Containing Amphipathic Poly(Ethylene Glycol). Biochim. Biophys. Acta (BBA) Lipids Lipid Metab. 1992, 1128, 44–49.
- McNeeley, K.M.; Annapragada, A.; Bellamkonda, R.V. Decreased Circulation Time Offsets Increased Efficacy of PEGylated Nanocarriers Targeting Folate Receptors of Glioma. Nanotechnology 2007, 18, 385101.
- Zhang, L.; Han, L.; Sun, X.; Gao, D.; Qin, J.; Wang, J. The Use of PEGylated Liposomes to Prolong the Circulation Lifetime of Salvianolic Acid B. Fitoterapia 2012, 83, 678–689.
- Bendas, G. Immunoliposomes: A Promising Approach to Targeting Cancer Therapy. BioDrugs 2001, 15, 215–224.
- Gholizadeh, S.; Dolman, E.M.; Wieriks, R.; Sparidans, R.W.; Hennink, W.E.; Kok, R.J. Anti-GD2 Immunoliposomes for Targeted Delivery of the Survivin Inhibitor Sepantronium Bromide (YM155) to Neuroblastoma Tumor Cells. Pharm. Res. 2018, 35, 85.
- Huwyler, J.; Wu, D.; Pardridge, W.M. Brain Drug Delivery of Small Molecules Using Immunoliposomes. Proc. Natl. Acad. Sci. USA 1996, 93, 14164–14169.
- Loureiro, J.A.; Gomes, B.; Fricker, G.; Cardoso, I.; Ribeiro, C.A.; Gaiteiro, C.; Coelho, M.A.N.; do Carmo Pereira, M.; Rocha, S. Dual Ligand Immunoliposomes for Drug Delivery to the Brain. Colloids Surf. B Biointerfaces 2015, 134, 213–219.
- Robinson, A.M.; Creeth, J.E.; Jones, M.N. The Use of Immunoliposomes for Specific Delivery of Antimicrobial Agents to Oral Bacteria Immobilized on Polystyrene. J. Biomater. Sci. Polym. Ed. 2000, 11, 1381–1393.
- Zhang, Y.; Zhao, Y.; Dong, D.; Li, X.; Li, Z.; Li, S.; Wang, J. Effects of Isosorbide Mononitrate Loaded Nanoparticles Conjugated with Anti-Staphylococcus Aureus A-toxin on Staphylococcus Aureus Biofilms. Exp. Med. 2019, 19, 1267–1274.
- Ommen, P.; Hansen, L.; Hansen, B.K.; Vu-Quang, H.; Kjems, J.; Meyer, R.L. Aptamer-Targeted Drug Delivery for Staphylococcus Aureus Biofilm. Front. Cell. Infect. Microbiol. 2022, 12, 814340.
- Wei, Z.; Zhou, Y.; Wang, R.; Wang, J.; Chen, Z. Aptamers as Smart Ligands for Targeted Drug Delivery in Cancer Therapy. Pharmaceutics 2022, 14, 2561.
- Yang, K.; Gitter, B.; Rüger, R.; Wieland, G.D.; Chen, M.; Liu, X.; Albrecht, V.; Fahr, A. Antimicrobial Peptide-Modified Liposomes for Bacteria Targeted Delivery of Temoporfin in Photodynamic Antimicrobial Chemotherapy. Photochem. Photobiol. Sci. 2011, 10, 1593–1601.
- Ali, A.A.; Al-Othman, A.; Al-Sayah, M.H. Multifunctional Stimuli-Responsive Hybrid Nanogels for Cancer Therapy: Current Status and Challenges. J. Control. Release 2022, 351, 476–503.
- Zhao, X.; Bai, J.; Yang, W. Stimuli-Responsive Nanocarriers for Therapeutic Applications in Cancer. Cancer Biol. Med. 2021, 18, 319–335.
- Ding, M.; Zhao, W.; Song, L.-J.; Luan, S.-F. Stimuli-Responsive Nanocarriers for Bacterial Biofilm Treatment. Rare Met. 2022, 41, 482–498.
- Koga, K.; Tagami, T.; Ozeki, T. Gold Nanoparticle-Coated Thermosensitive Liposomes for the Triggered Release of Doxorubicin, and Photothermal Therapy Using a near-Infrared Laser. Colloids Surf. A Physicochem. Eng. Asp. 2021, 626, 127038.
- Cressey, P.; Bronstein, L.-G.; Benmahmoudi, R.; Rosilio, V.; Regeard, C.; Makky, A. Novel Liposome-like Assemblies Composed of Phospholipid-Porphyrin Conjugates with Photothermal and Photodynamic Activities against Bacterial Biofilms. Int. J. Pharm. 2022, 623, 121915.
- Xu, M.; Hu, Y.; Xiao, Y.; Zhang, Y.; Sun, K.; Wu, T.; Lv, N.; Wang, W.; Ding, W.; Li, F.; et al. Near-Infrared-Controlled Nanoplatform Exploiting Photothermal Promotion of Peroxidase-like and OXD-like Activities for Potent Antibacterial and Anti-Biofilm Therapies. ACS Appl. Mater. Interfaces 2020, 12, 50260–50274.
- Zou, L.; Hu, D.; Wang, F.; Jin, Q.; Ji, J. The Relief of Hypoxic Microenvironment Using an O2 Self-Sufficient Fluorinated Nanoplatform for Enhanced Photodynamic Eradication of Bacterial Biofilms. Nano Res. 2022, 15, 1636–1644.
- Alamoudi, K.; Martins, P.; Croissant, J.G.; Patil, S.; Omar, H.; Khashab, N.M. Thermoresponsive Pegylated Bubble Liposome Nanovectors for Efficient SiRNA Delivery via Endosomal Escape. Nanomedicine 2017, 12, 1421–1433.
- Fu, Y.-Y.; Zhang, L.; Yang, Y.; Liu, C.; He, Y.-N.; Li, P.; Yu, X. Synergistic Antibacterial Effect of Ultrasound Microbubbles Combined with Chitosan-Modified Polymyxin B-Loaded Liposomes on Biofilm-Producing Acinetobacter Baumannii. Int. J. Nanomed. 2019, 14, 1805–1815.
- Zhou, H.; Fang, S.; Kong, R.; Zhang, W.; Wu, K.; Xia, R.; Shang, X.; Zhu, C. Effect of Low Frequency Ultrasound plus Fluorescent Composite Carrier in the Diagnosis and Treatment of Methicillin-Resistant Staphylococcus Aureus Biofilm Infection of Bone Joint Implant. Int. J. Clin. Exp. Med. 2018, 11, 799–805.
- Raza, A.; Hayat, U.; Rasheed, T.; Bilal, M.; Iqbal, H.M.N. “Smart” Materials-Based near-Infrared Light-Responsive Drug Delivery Systems for Cancer Treatment: A Review. J. Mater. Res. Technol. 2019, 8, 1497–1509.
- Andra, V.V.S.N.L.; Pammi, S.V.N.; Bhatraju, L.V.K.P.; Ruddaraju, L.K. A Comprehensive Review on Novel Liposomal Methodologies, Commercial Formulations, Clinical Trials and Patents. Bionanoscience 2022, 12, 274–291.
- Ferreira, M.; Ogren, M.; Dias, J.N.R.; Silva, M.; Gil, S.; Tavares, L.; Aires-da-Silva, F.; Gaspar, M.M.; Aguiar, S.I. Liposomes as Antibiotic Delivery Systems: A Promising Nanotechnological Strategy against Antimicrobial Resistance. Molecules 2021, 26, 2047.
- Breijyeh, Z.; Jubeh, B.; Karaman, R. Resistance of Gram-Negative Bacteria to Current Antibacterial Agents and Approaches to Resolve It. Molecules 2020, 25, 1340.
- Zhang, G.; Meredith, T.C.; Kahne, D. On the Essentiality of Lipopolysaccharide to Gram-Negative Bacteria. Curr. Opin. Microbiol. 2013, 16, 779–785.
- Bertani, B.; Ruiz, N. Function and Biogenesis of Lipopolysaccharides. EcoSal Plus 2018, 8, ecosalplus.ESP-0001-2018.
- Jubeh, B.; Breijyeh, Z.; Karaman, R. Resistance of Gram-Positive Bacteria to Current Antibacterial Agents and Overcoming Approaches. Molecules 2020, 25, 2888.
- Khatoon, Z.; McTiernan, C.D.; Suuronen, E.J.; Mah, T.-F.; Alarcon, E.I. Bacterial Biofilm Formation on Implantable Devices and Approaches to Its Treatment and Prevention. Heliyon 2018, 4, e01067.
- Oliva, A.; Stefani, S.; Venditti, M.; Di Domenico, E.G. Biofilm-Related Infections in Gram-Positive Bacteria and the Potential Role of the Long-Acting Agent Dalbavancin. Front. Microbiol. 2021, 12, 749685.
- Vazquez-Guillamet, C.; Kollef, M.H. Treatment of Gram-Positive Infections in Critically Ill Patients. BMC Infect. Dis. 2014, 14, 92.
- Lebeaux, D.; Ghigo, J.-M.; Beloin, C. Biofilm-Related Infections: Bridging the Gap between Clinical Management and Fundamental Aspects of Recalcitrance toward Antibiotics. Microbiol Mol. Biol. Rev. 2014, 78, 510–543.
- Römling, U.; Balsalobre, C. Biofilm Infections, Their Resilience to Therapy and Innovative Treatment Strategies. J. Intern. Med. 2012, 272, 541–561.





