
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Meng Lin | -- | 5004 | 2023-05-18 15:58:57 | | | |
| 2 | Sirius Huang | Meta information modification | 5004 | 2023-05-19 03:13:45 | | |
Video Upload Options
Compared with traditional drugs, gene therapy can directly correct the disease-related genes at the genetic level, which guarantees a sustained effect. However, nucleic acids are unstable in circulation and have short half-lives. They cannot pass through biological membranes due to their high molecular weight and massive negative charges. To facilitate the delivery of nucleic acids, it is crucial to develop a suitable delivery strategy. The emergence of stimuli-responsive delivery systems has made it possible to control the release of nucleic acids in an intelligent manner and to precisely guide the therapeutic nucleic acids to the target site. Considering the unique properties of stimuli-responsive delivery systems, various stimuli-responsive nanocarriers have been developed.
1. Introduction
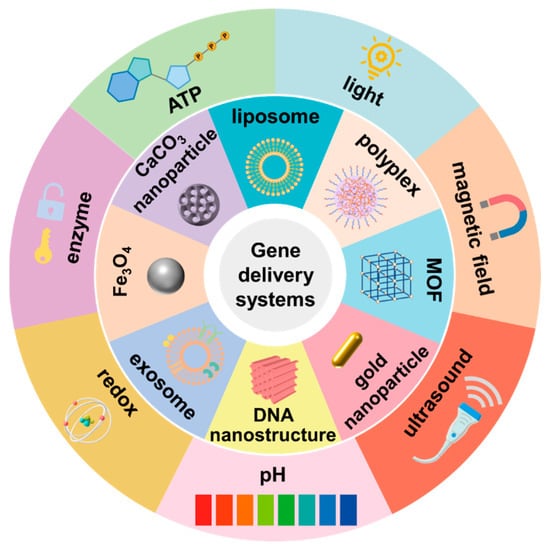
2. pH-Responsive Nanocarriers
2.1. pH-Responsive Detachment of Outlayer Coating
2.2. pH-Responsive Gene Release
2.3. pH-Responsive Charge Reversal
3. ROS-Responsive Nanocarriers
4. Enzyme-Responsive Nanocarriers
5. ATP-Responsive Nanocarriers
6. External Stimulus-Responsive Nanocarriers
7. Magnetic Field-Controlled Gene Delivery System
8. Light-Responsive Gene Delivery System
9. Ultrasound-Targeted Gene Delivery System
10. Multi-Stimulus Responsive Gene Delivery System
| Stimuli | Delivery Systems | Sensitive Linkers/Materials | Drugs | References |
|---|---|---|---|---|
| pH | PEI | Schiff base bond | Luciferase plasmid | [17] |
| Peptide nanoparticles (PNPs) | Peptide | Tissue factor (TF) siRNA | [33] | |
| Metal–organic framework (MOF) nanoparticle ZIF-8 | ZIF-8 | miR-34a mimic | [11] | |
| Solid lipid nanoparticles | Imine bond | miR-200 and irinotecan | [18] | |
| PEI−PBA/miR146a/PEI−DMA-C225, PMPC polyplexes | Ester bond | miR146a | [19] | |
| CaP-phospholipid complexes | CaP | As2O3 and HER2 siRNA | [21] | |
| Calcium carbonate nanohybrids | Calcium carbonate | p53 plasmid | [22] | |
| Dextran-quantum dot nanohybrids | Schiff base bond | p53 plasmid | [23] | |
| Black phosphorus nanosheets | Black phosphorus nanosheets | HTERT siRNA | [24] | |
| Lipid nanoparticles (LNP) | Ionizable lipids | CD47 siRNA and PD-L1 siRNA | [30] | |
| ROS | Heparin nanoparticles | Disulfide bond | miR-499 | [26] |
| Substance P (SP)-crosslinked BPEI | Boric acid ester bond | Plk1 siRNA | [35] | |
| DNA nanostructures | Disulfide bond | Doxorubicin, Bcl2 siRNA and P-gp siRNA | [39] | |
| Arginine-modified poly(disulfide amine)/siRNA nanocomplexes | Disulfide bond | KRAS siRNA | [41] | |
| Heparin nanoparticles | Disulfide bond | CRISPR/Cas 9 plasmid targeting survivin | [111] | |
| Fusogenic lipidic polyplexes | Boric acid ester bond | TRAIL plasmid | [42] | |
| Poly[(2-acryloyl)ethyl(p-boronic acid benzyl) diethylammonium Bromide] (B-PDEAEA) |
Boric acid ester bond | TRAIL plasmid | [43] | |
| Polyphotosensitizers (pPSs) | Thioketal | HIF-1α siRNA | [85] | |
| Enzyme | Cationic polymer PQDEA | Acetyloxybenzyl ester | TRAIL plasmid | [45] |
| Liposome-based layer-by-layer nanoparticles | MMP9-sensitive peptide | Luciferase plasmid | [50] | |
| Amphiphilic Dendrimer engineered nanocarrier system (ADENS) |
MMP2/9-sensitive peptide | Paclitaxel and VEGF siRNA | [57] | |
| PEI-based nanoparticles HPT-PF | MMP2/9-sensitive peptide | IL-12 plasmid and CRISPR/Cas 9 plasmid targeting CD47 | [58] | |
| Polyelectrolyte complexes | HA | Luciferase plasmid | [66] | |
| Hyaluronic acid (HA) coated nanoassembly | HA | Bcl-2 shRNA encoded plasmid and doxorubicin | [67] | |
| Polymetformin (pmet)-based nanosystem |
HA | IL-12 plasmid | [68] | |
| Peptide | HA | LOX-1 siRNA | [69] | |
| ATP | Polyplex Micelles | Phenylboronic acid (PBA) group | Luciferase plasmid | [74] |
| 4-carboxyphenylboronic acid (PBA) and dopamine Grafted vitamin E (VEDA) |
Phenylboronic acid (PBA) group | Eg5 siRNA and EGFR siRNA | [75] | |
| PEI-PBA | Phenylboronic acid (PBA) group | Bcl2 siRNA | [76] | |
| DNA nanostructures | ATP aptamer | Plk1 siRNA | [77] | |
| Magnetic field | Magnetic mesoporous silica nanoparticles (M-MSNs) | Iron oxide nanoparticles | The herpes simplex virus thymidine kinase/ganciclovir (HSV-TK/GCV) plasmid | [81] |
| Exosome-based platforms | Iron oxide nanoparticles | DOX and molecular beacon targeting the miR-21 | [83] | |
| Lipoplexes functionalized artificial bacterial flagella | Artificial bacterial flagella | pDNA encoding yellow–green fluorescent Venus protein | [84] | |
| Light | Gold Nanorod | HSP70 promoter | CRISPR/Cas 9 plasmid targeting AAVS1 | [6] |
| Gold Nanorod | HSP70 promoter | CRISPR/Cas 9 plasmid targeting PD-L1 | [8] | |
| Coumarin modified PAMAM | Coumarin | TRAIL plasmid | [89] | |
| Prodrug-backboned polymeric nanoparticle system | Pt(IV) prodrug | Pt(IV) prodrug and c-fos siRNA | [91] | |
| TK-PEI/HAP/p53 NCs | Pheophytin a | p53 plasmid | [94] | |
| Spherical nucleic acid | Pheophorbide a | HIF-1α siRNA and Bcl2 siRNA | [95] | |
| Ultrasound | Mannose-modified bubble lipoplexes | Bubble lipoplexes | Plasmid co-expressing ubiquitylated gp100 and TRP-2 | [107] |
| Cationic biosynthetic Nanobubble (CBNB) |
Nanobubble | The pEGFP and pCMV-Luc reporter plasmids | [108] | |
| B-PDEAEA loaded liposome | IR780 | TRAIL plasmid | [110] |
References
- Zhang, L.; Wang, P.; Feng, Q.; Wang, N.; Chen, Z.; Huang, Y.; Zheng, W.; Jiang, X. Lipid nanoparticle-mediated efficient delivery of CRISPR/Cas9 for tumor therapy. NPG Asia Mater. 2017, 9, e441.
- Liu, Y.; Chen, J.; Tang, Y.; Li, S.; Dou, Y.; Zheng, J. Synthesis and Characterization of Quaternized Poly(β-amino ester) for Highly Efficient Delivery of Small Interfering RNA. Mol. Pharm. 2018, 15, 4558–4567.
- Rui, Y.; Wilson, D.R.; Sanders, K.; Green, J.J. Reducible Branched Ester-Amine Quadpolymers (rBEAQs) Codelivering Plasmid DNA and RNA Oligonucleotides Enable CRISPR/Cas9 Genome Editing. ACS Appl. Mater. Interfaces 2019, 11, 10472–10480.
- Wang, Y.; Wang, M.; Chen, H.; Liu, H.; Zhang, Q.; Cheng, Y. Fluorinated dendrimer for TRAIL gene therapy in cancer treatment. J. Mater. Chem. B 2016, 4, 1354–1360.
- Zhang, Z.; Wang, Q.; Liu, Q.; Zheng, Y.; Zheng, C.; Yi, K.; Zhao, Y.; Gu, Y.; Wang, Y.; Wang, C.; et al. Dual-Locking Nanoparticles Disrupt the PD-1/PD-L1 Pathway for Efficient Cancer Immunotherapy. Adv. Mater. 2019, 31, e1905751.
- Chen, X.; Chen, Y.; Xin, H.; Wan, T.; Ping, Y. Near-infrared optogenetic engineering of photothermal nanoCRISPR for programmable genome editing. Proc. Natl. Acad. Sci. USA 2020, 117, 2395–2405.
- Liu, Y.; Xu, M.; Zhao, Y.; Chen, X.; Zhu, X.; Wei, C.; Zhao, S.; Liu, J.; Qin, X. Flower-like gold nanoparticles for enhanced photothermal anticancer therapy by the delivery of pooled siRNA to inhibit heat shock stress response. J. Mater. Chem. B 2019, 7, 586–597.
- Tang, H.; Xu, X.; Chen, Y.; Xin, H.; Wan, T.; Li, B.; Pan, H.; Li, D.; Ping, Y. Reprogramming the tumor microenvironment through second-near-infrared-window photothermal genome editing of PD-L1 mediated by supramolecular gold nanorods for enhanced cancer immunotherapy. Adv. Mater. 2021, 33, e2006003.
- He, X.W.; Liu, T.; Chen, Y.X.; Cheng, D.J.; Li, X.R.; Xiao, Y.; Feng, Y.L. Calcium carbonate nanoparticle delivering vascular endothelial growth factor-C siRNA effectively inhibits lymphangiogenesis and growth of gastric cancer in vivo. Cancer Gene Ther. 2008, 15, 193–202.
- Wang, C.Q.; Gong, M.Q.; Wu, J.L.; Zhuo, R.X.; Cheng, S.X. Dual-functionalized calcium carbonate based gene delivery system for efficient gene delivery. RSC Adv. 2014, 4, 38623–38629.
- Zhao, H.; Li, T.; Yao, C.; Gu, Z.; Liu, C.; Li, J.; Yang, D. Dual Roles of Metal-Organic Frameworks as Nanocarriers for miRNA Delivery and Adjuvants for Chemodynamic Therapy. ACS Appl. Mater. Interfaces 2021, 13, 6034–6042.
- Zhuang, J.; Gong, H.; Zhou, J.; Zhang, Q.; Gao, W.; Fang, R.H.; Zhang, L. Targeted gene silencing in vivo by platelet membrane-coated metal-organic framework nanoparticles. Sci. Adv. 2020, 6, eaaz6108.
- Lin, Y.; Wu, J.; Gu, W.; Huang, Y.; Tong, Z.; Huang, L.; Tan, J. Exosome-Liposome Hybrid Nanoparticles Deliver CRISPR/Cas9 System in MSCs. Adv. Sci. 2018, 5, 1700611.
- Munagala, R.; Aqil, F.; Jeyabalan, J.; Kandimalla, R.; Wallen, M.; Tyagi, N.; Wilcher, S.; Yan, J.; Schultz, D.J.; Spencer, W.; et al. Exosome-mediated delivery of RNA and DNA for gene therapy. Cancer Lett. 2021, 505, 58–72.
- Zhao, L.; Gu, C.; Gan, Y.; Shao, L.; Chen, H.; Zhu, H. Exosome-mediated siRNA delivery to suppress postoperative breast cancer metastasis. J. Control. Release 2020, 318, 1–15.
- Yang, J.; Zhang, Q.; Chang, H.; Cheng, Y. Surface-engineered dendrimers in gene delivery. Chem. Rev. 2015, 115, 5274–5300.
- Guan, X.; Guo, Z.; Wang, T.; Lin, L.; Chen, J.; Tian, H.; Chen, X. A pH-Responsive Detachable PEG Shielding Strategy for Gene Delivery System in Cancer Therapy. Biomacromolecules 2017, 18, 1342–1349.
- Lo, Y.L.; Chang, C.H.; Wang, C.S.; Yang, M.H.; Lin, A.M.; Hong, C.J.; Tseng, W.H. PEG-coated nanoparticles detachable in acidic microenvironments for the tumor-directed delivery of chemo- and gene therapies for head and neck cancer. Theranostics 2020, 10, 6695–6714.
- Ma, D.; Liu, H.; Zhao, P.; Ye, L.; Zou, H.; Zhao, X.; Dai, H.; Kong, X.; Liu, P. Programing Assembling/Releasing Multifunctional miRNA Nanomedicine to Treat Prostate Cancer. ACS Appl. Mater. Interfaces 2020, 12, 9032–9040.
- Wang, M.Z.; Niu, J.; Ma, H.J.; Dad, H.A.; Shao, H.T.; Yuan, T.J.; Peng, L.H. Transdermal siRNA delivery by pH-switchable micelles with targeting effect suppress skin melanoma progression. J. Control. Release 2020, 322, 95–107.
- Wang, Q.; Tian, Y.; Liu, L.; Chen, C.; Zhang, W.; Wang, L.; Guo, Q.; Ding, L.; Fu, H.; Song, H.; et al. Precise Targeting Therapy of Orthotopic Gastric Carcinoma by siRNA and Chemotherapeutic Drug Codelivered in pH-Sensitive Nano Platform. Adv. Healthc. Mater. 2021, 10, e2100966.
- Liu, Y.; Yu, B.; Dai, X.; Zhao, N.; Xu, F.J. Biomineralized calcium carbonate nanohybrids for mild photothermal heating-enhanced gene therapy. Biomaterials 2021, 274, 120885.
- Liu, Y.; Zhao, N.; Xu, F.J. pH-Responsive Degradable Dextran-Quantum Dot Nanohybrids for Enhanced Gene Delivery. ACS Appl. Mater. Interfaces 2019, 11, 34707–34716.
- Chen, L.; Chen, C.; Chen, W.; Li, K.; Chen, X.; Tang, X.; Xie, G.; Luo, X.; Wang, X.; Liang, H.; et al. Biodegradable Black Phosphorus Nanosheets Mediate Specific Delivery of hTERT siRNA for Synergistic Cancer Therapy. ACS Appl. Mater. Interfaces 2018, 10, 21137–21148.
- Saw, P.E.; Yao, H.; Lin, C.; Tao, W.; Farokhzad, O.C.; Xu, X. Stimuli-Responsive Polymer-Prodrug Hybrid Nanoplatform for Multistage siRNA Delivery and Combination Cancer Therapy. Nano Lett. 2019, 19, 5967–5974.
- Nie, J.J.; Qiao, B.K.; Duan, S.; Xu, C.; Chen, B.Y.; Hao, W.J.; Yu, B.R.; Li, Y.L.; Du, J.; Xu, F.J. Unlockable Nanocomplexes with Self-Accelerating Nucleic Acid Release for Effective Staged Gene Therapy of Cardiovascular Diseases. Adv. Mater. 2018, 30, 1801570.
- Zhang, Q.; Shen, C.N.; Zhao, N.; Xu, F.J. Redox-Responsive and Drug-Embedded Silica Nanoparticles with Unique Self-Destruction Features for Efficient Gene/Drug Codelivery. Adv. Funct. Mater. 2017, 27, 1606229.
- Samaridou, E.; Heyes, J.; Lutwyche, P. Lipid nanoparticles for nucleic acid delivery: Current perspectives. Adv. Drug Deliv. Rev. 2020, 154–155, 37–63.
- Qiu, M.; Li, Y.; Bloomer, H.; Xu, Q. Developing Biodegradable Lipid Nanoparticles for Intracellular mRNA Delivery and Genome Editing. Acc. Chem. Res. 2021, 54, 4001–4011.
- Liu, S.; Liu, J.; Li, H.; Mao, K.; Wang, H.; Meng, X.; Wang, J.; Wu, C.; Chen, H.; Wang, X.; et al. An optimized ionizable cationic lipid for brain tumor-targeted siRNA delivery and glioblastoma immunotherapy. Biomaterials 2022, 287, 121645.
- Hafez, I.M.; Maurer, N.; Cullis, P.R. On the mechanism whereby cationic lipids promote intracellular delivery of polynucleic acids. Gene Ther. 2001, 8, 1188–1196.
- Wittrup, A.; Ai, A.; Liu, X.; Hamar, P.; Trifonova, R.; Charisse, K.; Manoharan, M.; Kirchhausen, T.; Lieberman, J. Visualizing lipid-formulated siRNA release from endosomes and target gene knockdown. Nat. Biotechnol. 2015, 33, 870–876.
- Liu, S.; Zhang, Y.; Zhao, X.; Wang, J.; Di, C.; Zhao, Y.; Ji, T.; Cheng, K.; Wang, Y.; Chen, L.; et al. Tumor-Specific Silencing of Tissue Factor Suppresses Metastasis and Prevents Cancer-Associated Hypercoagulability. Nano Lett. 2019, 19, 4721–4730.
- Wang, Y.; Qi, H.; Liu, Y.; Duan, C.; Liu, X.; Xia, T.; Chen, D.; Piao, H.L.; Liu, H.X. The double-edged roles of ROS in cancer prevention and therapy. Theranostics 2021, 11, 4839–4857.
- Ruan, C.; Liu, L.; Wang, Q.; Chen, X.; Chen, Q.; Lu, Y.; Zhang, Y.; He, X.; Zhang, Y.; Guo, Q.; et al. Reactive Oxygen Species-Biodegradable Gene Carrier for the Targeting Therapy of Breast Cancer. ACS Appl. Mater. Interfaces 2018, 10, 10398–10408.
- Trachootham, D.; Alexandre, J.; Huang, P. Targeting cancer cells by ROS-mediated mechanisms: A radical therapeutic approach? Nat. Rev. Drug Discov. 2009, 8, 579–591.
- Zhou, Z.; Li, H.; Wang, K.; Guo, Q.; Li, C.; Jiang, H.; Hu, Y.; Oupicky, D.; Sun, M. Bioreducible Cross-Linked Hyaluronic Acid/Calcium Phosphate Hybrid Nanoparticles for Specific Delivery of siRNA in Melanoma Tumor Therapy. ACS Appl. Mater. Interfaces 2017, 9, 14576–14589.
- Wang, G.; Zhu, D.; Zhou, Z.; Piao, Y.; Tang, J.; Shen, Y. Glutathione-Specific and Intracellularly Labile Polymeric Nanocarrier for Efficient and Safe Cancer Gene Delivery. ACS Appl. Mater. Interfaces 2020, 12, 14825–14838.
- Wang, Z.; Song, L.; Liu, Q.; Tian, R.; Shang, Y.; Liu, F.; Liu, S.; Zhao, S.; Han, Z.; Sun, J.; et al. A Tubular DNA Nanodevice as a siRNA/Chemo-Drug Co-delivery Vehicle for Combined Cancer Therapy. Angew. Chem. Int. Ed. Engl. 2021, 60, 2594–2598.
- Wu, Y.; Zhong, D.; Li, Y.; Wu, H.; Zhang, H.; Mao, H.; Yang, J.; Luo, K.; Gong, Q.; Gu, Z. A tumor-activatable peptide supramolecular nanoplatform for the delivery of dual-gene targeted siRNAs for drug-resistant cancer treatment. Nanoscale 2021, 13, 4887–4898.
- Zhang, X.; Hong, K.; Sun, Q.; Zhu, Y.; Du, J. Bioreducible, arginine-rich polydisulfide-based siRNA nanocomplexes with excellent tumor penetration for efficient gene silencing. Biomater. Sci. 2021, 9, 5275–5292.
- Liu, X.; Xiang, J.; Zhu, D.; Jiang, L.; Zhou, Z.; Tang, J.; Liu, X.; Huang, Y.; Shen, Y. Fusogenic Reactive Oxygen Species Triggered Charge-Reversal Vector for Effective Gene Delivery. Adv. Mater. 2016, 28, 1743–1752.
- Xiang, J.; Liu, X.; Zhou, Z.; Zhu, D.; Zhou, Q.; Piao, Y.; Jiang, L.; Tang, J.; Liu, X.; Shen, Y. Reactive Oxygen Species (ROS)-Responsive Charge-Switchable Nanocarriers for Gene Therapy of Metastatic Cancer. ACS Appl. Mater. Interfaces 2018, 10, 43352–43362.
- Wang, Q.Y.; Xu, Y.S.; Zhang, N.X.; Dong, Z.P.; Zhao, B.N.; Liu, L.C.; Lu, T.; Wang, Y. Phenylboronic ester-modified anionic micelles for ROS-stimuli response in HeLa cell. Drug Deliv. 2020, 27, 681–690.
- Qiu, N.; Gao, J.; Liu, Q.; Wang, J.; Shen, Y. Enzyme-Responsive Charge-Reversal Polymer-Mediated Effective Gene Therapy for Intraperitoneal Tumors. Biomacromolecules 2018, 19, 2308–2319.
- Ren, F.; Tang, R.; Zhang, X.; Madushi, W.M.; Luo, D.; Dang, Y.; Li, Z.; Wei, K.; Chen, G. Overexpression of MMP Family Members Functions as Prognostic Biomarker for Breast Cancer Patients: A Systematic Review and Meta-Analysis. PLoS ONE 2015, 10, e0135544.
- Klassen, L.M.B.; Chequin, A.; Manica, G.C.M.; Biembengut, I.V.; Toledo, M.B.; Baura, V.A.; de Pedrosa, F.O.; Ramos, E.A.S.; Costa, F.F.; de Souza, E.M.; et al. MMP9 gene expression regulation by intragenic epigenetic modifications in breast cancer. Gene 2018, 642, 461–466.
- Malik, R.; Lelkes, P.I.; Cukierman, E. Biomechanical and biochemical remodeling of stromal extracellular matrix in cancer. Trends Biotechnol. 2015, 33, 230–236.
- Paduch, R.; Kandefer-Szerszeń, M.; Szuster-Ciesielska, A.; Plewka, K. Transforming growth factor-beta1 modulates metalloproteinase-2 and -9, nitric oxide, RhoA and alpha-smooth muscle actin expression in colon adenocarcinoma cells. Cell Biol. Int. 2010, 34, 213–223.
- Boehnke, N.; Correa, S.; Hao, L.; Wang, W.; Straehla, J.P.; Bhatia, S.N.; Hammond, P.T. Theranostic Layer-by-Layer Nanoparticles for Simultaneous Tumor Detection and Gene Silencing. Angew. Chem. Int. Ed. Engl. 2020, 59, 2776–2783.
- Komin, A.; Russell, L.M.; Hristova, K.A.; Searson, P.C. Peptide-based strategies for enhanced cell uptake, transcellular transport, and circulation: Mechanisms and challenges. Adv. Drug. Deliv. Rev. 2017, 110–111, 52–64.
- Shi, N.Q.; Qi, X.R.; Xiang, B.; Zhang, Y. A survey on “Trojan Horse” peptides: Opportunities, issues and controlled entry to “Troy”. J. Control. Release 2014, 194, 53–70.
- Ma, D.X.; Shi, N.Q.; Qi, X.R. Distinct transduction modes of arginine-rich cell-penetrating peptides for cargo delivery into tumor cells. Int. J. Pharm. 2011, 419, 200–208.
- Gao, W.; Meng, T.; Shi, N.; Zhuang, H.; Yang, Z.; Qi, X. Targeting and microenvironment-responsive lipid nanocarrier for the enhancement of tumor cell recognition and therapeutic efficiency. Adv. Healthc. Mater. 2015, 4, 748–759.
- Gao, W.; Xiang, B.; Meng, T.T.; Liu, F.; Qi, X.R. Chemotherapeutic drug delivery to cancer cells using a combination of folate targeting and tumor microenvironment-sensitive polypeptides. Biomaterials 2013, 34, 4137–4149.
- Han, Q.J.; Lan, X.T.; Wen, Y.; Zhang, C.Z.; Cleary, M.; Sayyed, Y.; Huang, G.; Tuo, X.; Yi, L.; Xi, Z.; et al. Matrix Metalloproteinase-9-Responsive Surface Charge-Reversible Nanocarrier to Enhance Endocytosis as Efficient Targeted Delivery System for Cancer Diagnosis and Therapy. Adv. Healthc. Mater. 2021, 10, e2002143.
- Li, X.; Sun, A.; Liu, Y.; Zhang, W.; Pang, N.; Cheng, S.; Qi, X. Amphiphilic dendrimer engineered nanocarrier systems for co-delivery of siRNA and paclitaxel to matrix metalloproteinase-rich tumors for synergistic therapy. NPG Asia Mater. 2018, 10, 238–254.
- Lin, M.; Yang, Z.; Yang, Y.; Peng, Y.; Li, J.; Du, Y.; Sun, Q.; Gao, D.; Yuan, Q.; Zhou, Y.; et al. CRISPR-based in situ engineering tumor cells to reprogram macrophages for effective cancer immunotherapy. Nano Today 2022, 42, 101359.
- Lv, J.; Chang, H.; Wang, Y.; Wang, M.; Xiao, J.; Zhang, Q.; Cheng, Y. Fluorination on polyethylenimine allows efficient 2D and 3D cell culture gene delivery. J. Mater. Chem. B 2015, 3, 642–650.
- Wang, H.; Wang, Y.; Wang, Y.; Hu, J.; Li, T.; Liu, H.; Zhang, Q.; Cheng, Y. Self-Assembled Fluorodendrimers Combine the Features of Lipid and Polymeric Vectors in Gene Delivery. Angew. Chem. Int. Ed. Engl. 2015, 54, 11647–11651.
- Feng, G.; Zha, Z.; Huang, Y.; Li, J.; Wang, Y.; Ke, W.; Chen, H.; Liu, L.; Song, Y.; Ge, Z. Sustained and Bioresponsive Two-Stage Delivery of Therapeutic miRNA via Polyplex Micelle-Loaded Injectable Hydrogels for Inhibition of Intervertebral Disc Fibrosis. Adv. Healthc. Mater. 2018, 7, e1800623.
- Girish, K.S.; Kemparaju, K. The magic glue hyaluronan and its eraser hyaluronidase: A biological overview. Life Sci. 2007, 80, 1921–1943.
- McAtee, C.O.; Barycki, J.J.; Simpson, M.A. Emerging roles for hyaluronidase in cancer metastasis and therapy. Adv. Cancer Res. 2014, 123, 1–34.
- Bertrand, P.; Girard, N.; Duval, C.; d’Anjou, J.; Chauzy, C.; Ménard, J.F.; Delpech, B. Increased hyaluronidase levels in breast tumor metastases. Int. J. Cancer 1997, 73, 327–331.
- Lokeshwar, V.B.; Rubinowicz, D.; Schroeder, G.L.; Forgacs, E.; Minna, J.D.; Block, N.L.; Nadji, M.; Lokeshwar, B.L. Stromal and epithelial expression of tumor markers hyaluronic acid and HYAL1 hyaluronidase in prostate cancer. J. Biol. Chem. 2001, 276, 11922–11932.
- He, Y.; Zhou, J.; Ma, S.; Nie, Y.; Yue, D.; Jiang, Q.; Wali, A.R.; Tang, J.Z.; Gu, Z. Multi-Responsive “Turn-On” Nanocarriers for Efficient Site-Specific Gene Delivery In Vitro and In Vivo. Adv. Healthc. Mater. 2016, 5, 2799–2812.
- Rajendrakumar, S.K.; Venu, A.; Revuri, V.; George Thomas, R.; Thirunavukkarasu, G.K.; Zhang, J.; Vijayan, V.; Choi, S.Y.; Lee, J.Y.; Lee, Y.K.; et al. Hyaluronan-Stabilized Redox-Sensitive Nanoassembly for Chemo-Gene Therapy and Dual T1/T2 MR Imaging in Drug-Resistant Breast Cancer Cells. Mol. Pharm. 2019, 16, 2226–2234.
- Sun, Y.; Liu, L.; Zhou, L.; Yu, S.; Lan, Y.; Liang, Q.; Liu, J.; Cao, A.; Liu, Y. Tumor Microenvironment-Triggered Charge Reversal Polymetformin-Based Nanosystem Co-Delivered Doxorubicin and IL-12 Cytokine Gene for Chemo-Gene Combination Therapy on Metastatic Breast Cancer. ACS Appl. Mater. Interfaces 2020, 12, 45873–45890.
- Zhao, Y.; He, Z.; Gao, H.; Tang, H.; He, J.; Guo, Q.; Zhang, W.; Liu, J. Fine Tuning of Core-Shell Structure of Hyaluronic Acid/Cell-Penetrating Peptides/siRNA Nanoparticles for Enhanced Gene Delivery to Macrophages in Antiatherosclerotic Therapy. Biomacromolecules 2018, 19, 2944–2956.
- Choi, K.Y.; Silvestre, O.F.; Huang, X.; Min, K.H.; Howard, G.P.; Hida, N.; Jin, A.J.; Carvajal, N.; Lee, S.W.; Hong, J.I.; et al. Versatile RNA interference nanoplatform for systemic delivery of RNAs. ACS Nano 2014, 8, 4559–4570.
- Jiang, C.; Chen, J.; Li, Z.; Wang, Z.; Zhang, W.; Liu, J. Recent advances in the development of polyethylenimine-based gene vectors for safe and efficient gene delivery. Expert Opin. Drug Deliv. 2019, 16, 363–376.
- Naito, M.; Ishii, T.; Matsumoto, A.; Miyata, K.; Miyahara, Y.; Kataoka, K. A phenylboronate-functionalized polyion complex micelle for ATP-triggered release of siRNA. Angew. Chem. Int. Ed. Engl. 2012, 51, 10751–10755.
- Zhang, J.; Wang, Y.; Chen, J.; Liang, X.; Han, H.; Yang, Y.; Li, Q.; Wang, Y. Inhibition of cell proliferation through an ATP-responsive co-delivery system of doxorubicin and Bcl-2 siRNA. Int. J. Nanomed. 2017, 12, 4721–4732.
- Yoshinaga, N.; Ishii, T.; Naito, M.; Endo, T.; Uchida, S.; Cabral, H.; Osada, K.; Kataoka, K. Polyplex Micelles with Phenylboronate/Gluconamide Cross-Linking in the Core Exerting Promoted Gene Transfection through Spatiotemporal Responsivity to Intracellular pH and ATP Concentration. J. Am. Chem. Soc. 2017, 139, 18567–18575.
- Fan, X.; Zhao, X.; Su, W.; Tang, X. Triton X-100-Modified Adenosine Triphosphate-Responsive siRNA Delivery Agent for Antitumor Therapy. Mol. Pharm. 2020, 17, 3696–3708.
- Zhou, Z.; Zhang, Q.; Zhang, M.; Li, H.; Chen, G.; Qian, C.; Oupicky, D.; Sun, M. ATP-activated decrosslinking and charge-reversal vectors for siRNA delivery and cancer therapy. Theranostics 2018, 8, 4604–4619.
- Li, F.; Yu, W.; Zhang, J.; Dong, Y.; Ding, X.; Ruan, X.; Gu, Z.; Yang, D. Spatiotemporally programmable cascade hybridization of hairpin DNA in polymeric nanoframework for precise siRNA delivery. Nat. Commun. 2021, 12, 1138.
- Bauer, L.M.; Situ, S.F.; Griswold, M.A.; Samia, A.C. High-performance iron oxide nanoparticles for magnetic particle imaging-guided hyperthermia (hMPI). Nanoscale 2016, 8, 12162–12169.
- Cole, A.J.; David, A.E.; Wang, J.; Galbán, C.J.; Yang, V.C. Magnetic brain tumor targeting and biodistribution of long-circulating PEG-modified, cross-linked starch-coated iron oxide nanoparticles. Biomaterials 2011, 32, 6291–6301.
- Dwivedi, P.; Kiran, S.; Han, S.; Dwivedi, M.; Khatik, R.; Fan, R.; Mangrio, F.A.; Du, K.; Zhu, Z.; Yang, C.; et al. Magnetic Targeting and Ultrasound Activation of Liposome-Microbubble Conjugate for Enhanced Delivery of Anticancer Therapies. ACS Appl. Mater. Interfaces 2020, 12, 23737–23751.
- Wang, Z.; Chang, Z.; Lu, M.; Shao, D.; Yue, J.; Yang, D.; Zheng, X.; Li, M.; He, K.; Zhang, M.; et al. Shape-controlled magnetic mesoporous silica nanoparticles for magnetically-mediated suicide gene therapy of hepatocellular carcinoma. Biomaterials 2018, 154, 147–157.
- Zhou, Z.; Sun, Y.; Shen, J.; Wei, J.; Yu, C.; Kong, B.; Liu, W.; Yang, H.; Yang, S.; Wang, W. Iron/iron oxide core/shell nanoparticles for magnetic targeting MRI and near-infrared photothermal therapy. Biomaterials 2014, 35, 7470–7478.
- Wang, J.; Chen, P.; Dong, Y.; Xie, H.; Wang, Y.; Soto, F.; Ma, P.; Feng, X.; Du, W.; Liu, B.F. Designer exosomes enabling tumor targeted efficient chemo/gene/photothermal therapy. Biomaterials 2021, 276, 121056.
- Qiu, F.M.; Fujita, S.; Mhanna, R.; Zhang, L.; Simona, B.R.; Nelson, B.J. Magnetic Helical Microswimmers Functionalized with Lipoplexes for Targeted Gene Delivery. Adv. Funct. Mater. 2015, 25, 1666–1671.
- Zheng, N.; Luo, X.; Zhang, Z.; Wang, A.; Song, W. Cationic Polyporphyrins as siRNA Delivery Vectors for Photodynamic and Gene Synergistic Anticancer Therapy. ACS Appl. Mater. Interfaces 2021, 13, 27513–27521.
- Sun, Q.; Yang, Z.; Lin, M.; Peng, Y.; Wang, R.; Du, Y.; Zhou, Y.; Li, J.; Qi, X. Phototherapy and anti-GITR antibody-based therapy synergistically reinvigorate immunogenic cell death and reject established cancers. Biomaterials 2021, 269, 120648.
- Tang, H.; Wang, J.; Yu, L.; Zhang, S.; Yang, H.; Li, X.; Brash, J.L.; Wang, L.; Chen, H. Ultrahigh Efficiency and Minimalist Intracellular Delivery of Macromolecules Mediated by Latent-Photothermal Surfaces. ACS Appl. Mater. Interfaces 2021, 13, 12594–12602.
- Yang, Z.; Du, Y.; Sun, Q.; Peng, Y.; Wang, R.; Zhou, Y.; Wang, Y.; Zhang, C.; Qi, X. Albumin-Based Nanotheranostic Probe with Hypoxia Alleviating Potentiates Synchronous Multimodal Imaging and Phototherapy for Glioma. ACS Nano 2020, 14, 6191–6212.
- Wang, H.; Miao, W.; Wang, F.; Cheng, Y. A Self-Assembled Coumarin-Anchored Dendrimer for Efficient Gene Delivery and Light-Responsive Drug Delivery. Biomacromolecules 2018, 19, 2194–2201.
- Lin, X.; Wu, M.; Li, M.; Cai, Z.; Sun, H.; Tan, X.; Li, J.; Zeng, Y.; Liu, X.; Liu, J. Photo-responsive hollow silica nanoparticles for light-triggered genetic and photodynamic synergistic therapy. Acta Biomater. 2018, 76, 178–192.
- Zhang, Q.; Kuang, G.; He, S.; Lu, H.; Cheng, Y.; Zhou, D.; Huang, Y. Photoactivatable Prodrug-Backboned Polymeric Nanoparticles for Efficient Light-Controlled Gene Delivery and Synergistic Treatment of Platinum-Resistant Ovarian Cancer. Nano Lett. 2020, 20, 3039–3049.
- Kuang, G.; Lu, H.; He, S.; Xiong, H.; Yu, J.; Zhang, Q.; Huang, Y. Near-Infrared Light-Triggered Polyprodrug/siRNA Loaded Upconversion Nanoparticles for Multi-Modality Imaging and Synergistic Cancer Therapy. Adv. Healthc. Mater. 2021, 10, e2100938.
- Mallidi, S.; Anbil, S.; Bulin, A.L.; Obaid, G.; Ichikawa, M.; Hasan, T. Beyond the Barriers of Light Penetration: Strategies, Perspectives and Possibilities for Photodynamic Therapy. Theranostics 2016, 6, 2458–2487.
- Wang, J.; He, H.; Xu, X.; Wang, X.; Chen, Y.; Yin, L. Far-red light-mediated programmable anti-cancer gene delivery in cooperation with photodynamic therapy. Biomaterials 2018, 171, 72–82.
- Chen, L.; Li, G.; Wang, X.; Li, J.; Zhang, Y. Spherical Nucleic Acids for Near-Infrared Light-Responsive Self-Delivery of Small-Interfering RNA and Antisense Oligonucleotide. ACS Nano 2021, 15, 11929–11939.
- Høgset, A.; Prasmickaite, L.; Selbo, P.K.; Hellum, M.; Engesaeter, B.; Bonsted, A.; Berg, K. Photochemical internalisation in drug and gene delivery. Adv. Drug Deliv. Rev. 2004, 56, 95–115.
- Park, S.J.; Park, W.; Na, K. Tumor Intracellular-Environment Responsive Materials Shielded Nano-Complexes for Highly Efficient Light-Triggered Gene Delivery without Cargo Gene Damage. Adv. Funct. Mater. 2015, 25, 3472–3482.
- Selbo, P.K.; Weyergang, A.; Høgset, A.; Norum, O.J.; Berstad, M.B.; Vikdal, M.; Berg, K. Photochemical internalization provides time- and space-controlled endolysosomal escape of therapeutic molecules. J. Control. Release 2010, 148, 2–12.
- Zhao, L.; Luo, Y.; Huang, Q.; Cao, Z.; Yang, X. Photo-Enhanced CRISPR/Cas9 System Enables Robust PD-L1 Gene Disruption in Cancer Cells and Cancer Stem-Like Cells for Efficient Cancer Immunotherapy. Small 2020, 16, 2004879.
- Defelippis, M.R.; Faraggi, M.; Klapper, M.H. Redox Potentials Of The Azide And Dithiocyanate Radicals. J. Phys. Chem. 1990, 94, 2420–2424.
- Ma, Z.; Du, Z.; Chen, X.; Wang, X.; Yang, Y. Fine tuning the LightOn light-switchable transgene expression system. Biochem. Biophys. Res. Commun. 2013, 440, 419–423.
- Wang, X.; Chen, X.; Yang, Y. Spatiotemporal control of gene expression by a light-switchable transgene system. Nat. Methods 2012, 9, 266–269.
- He, M.; Wang, Y.; Chen, X.; Zhao, Y.; Lou, K.; Wang, Y.; Huang, L.; Hou, X.; Xu, J.; Cai, X.; et al. Spatiotemporally controllable diphtheria toxin expression using a light-switchable transgene system combining multifunctional nanoparticle delivery system for targeted melanoma therapy. J. Control. Release 2020, 319, 1–14.
- Sirsi, S.R.; Borden, M.A. Advances in ultrasound mediated gene therapy using microbubble contrast agents. Theranostics 2012, 2, 1208–1222.
- Wu, J.; Li, R.K. Ultrasound-targeted microbubble destruction in gene therapy: A new tool to cure human diseases. Genes Dis. 2017, 4, 64–74.
- Walsh, A.P.G.; Gordon, H.N.; Peter, K.; Wang, X. Ultrasonic particles: An approach for targeted gene delivery. Adv. Drug Deliv. Rev. 2021, 179, 113998.
- Un, K.; Kawakami, S.; Suzuki, R.; Maruyama, K.; Yamashita, F.; Hashida, M. Suppression of melanoma growth and metastasis by DNA vaccination using an ultrasound-responsive and mannose-modified gene carrier. Mol. Pharm. 2011, 8, 543–554.
- Tayier, B.; Deng, Z.; Wang, Y.; Wang, W.; Mu, Y.; Yan, F. Biosynthetic nanobubbles for targeted gene delivery by focused ultrasound. Nanoscale 2019, 11, 14757–14768.
- Shapiro, M.G.; Goodwill, P.W.; Neogy, A.; Yin, M.; Foster, F.S.; Schaffer, D.V.; Conolly, S.M. Biogenic gas nanostructures as ultrasonic molecular reporters. Nat. Nanotechnol. 2014, 9, 311–316.
- Zhang, C.; Chen, J.; Song, Y.; Luo, J.; Jin, P.; Wang, X.; Xin, L.; Qiu, F.; Yao, J.; Wang, G.; et al. Ultrasound-Enhanced Reactive Oxygen Species Responsive Charge-Reversal Polymeric Nanocarriers for Efficient Pancreatic Cancer Gene Delivery. ACS Appl. Mater. Interfaces 2022, 14, 2587–2596.
- Nie, J.J.; Liu, Y.; Qi, Y.; Zhang, N.; Yu, B.; Chen, D.F.; Yang, M.; Xu, F.J. Charge-reversal nanocomolexes-based CRISPR/Cas9 delivery system for loss-of-function oncogene editing in hepatocellular carcinoma. J. Control. Release 2021, 333, 362–373.





