
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Wang gang | -- | 504 | 2023-05-05 08:49:33 | | | |
| 2 | Wang gang | + 1331 word(s) | 1835 | 2023-05-06 08:26:42 | | | | |
| 3 | Beatrix Zheng | -75 word(s) | 1760 | 2023-05-08 11:20:01 | | |
Video Upload Options
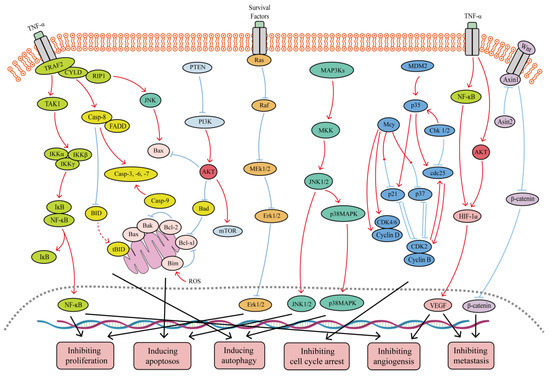
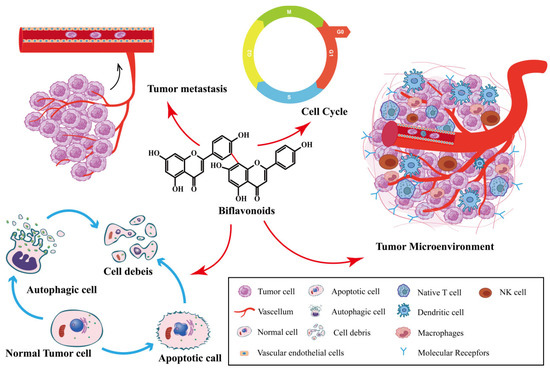
Despite the many strategies employed to slow the spread of cancer, the development of new anti-tumor drugs and the minimization of side effects have been major research hotspots in the anti-tumor field. Natural drugs are a huge treasure trove of drug development, and they have been widely used in the clinic as anti-tumor drugs. Selaginella species in the family Selaginellaceae are widely distributed worldwide, and they have been well-documented in clinical practice for the prevention and treatment of cancer. Biflavonoids are the main active ingredients in Selaginella, and they have good biological and anti-tumor activities, which warrant extensive research. The promise of biflavonoids from Selaginella (SFB) in the field of cancer therapy is being realized thanks to new research that offers insights into the multi-targeting therapeutic mechanisms and key signaling pathways.
1. Introduction
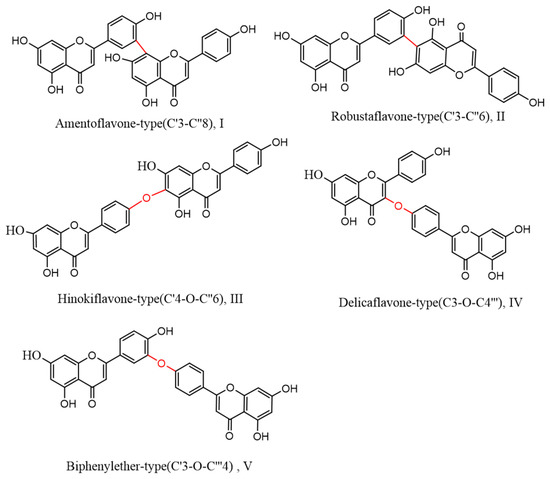
2. Structural Characteristics of Selaginella Biflavonoid
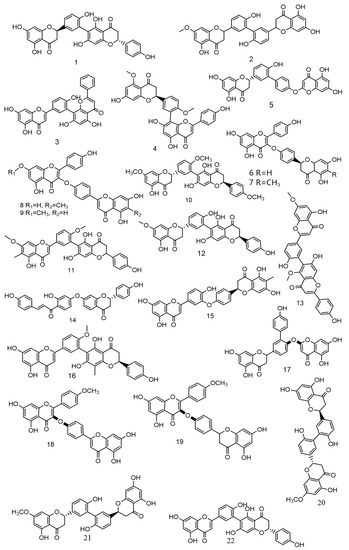
References
- Martincorena, I.; Raine, K.M.; Gerstung, M.; Dawson, K.J.; Haase, K.; Van Loo, P.; Davies, H.; Stratton, M.R.; Campbell, P.J. Universal Patterns of Selection in Cancer and Somatic Tissues. Cell 2017, 171, 1029–1041.e21.
- Williams, M.J.; Sottoriva, A.; Graham, T.A. Measuring Clonal Evolution in Cancer with Genomics. Annu. Rev. Genom. Hum. Genet. 2019, 20, 309–329.
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer Statistics, 2023. CA Cancer J. Clin. 2023, 73, 17–48.
- Nie, Z.; Chen, M.; Gao, Y.; Huang, D.; Cao, H.; Peng, Y.; Guo, N.; Wang, F.; Zhang, S. Ferropto sis and Tumor Drug Resistance: Current Status and Major Challenges. Front. Pharmacol. 2022, 13, 879317.
- Bray, F.; Laversanne, M.; Weiderpass, E.; Soerjomataram, I. The Ever-increasing Importance of Cancer as a Leading Cause of Premature Death Worldwide. Cancer 2021, 127, 3029–3030.
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249.
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674.
- Zhang, Y.; Liu, Q.; Zhang, X.; Huang, H.; Tang, S.; Chai, Y.; Xu, Z.; Li, M.; Chen, X.; Liu, J.; et al. Recent Advances in Exosome-Mediated Nucleic Acid Delivery for Cancer Therapy. J. Nanobiotechnol. 2022, 20, 279.
- Zaimy, M.A.; Saffarzadeh, N.; Mohammadi, A.; Pourghadamyari, H.; Izadi, P.; Sarli, A.; Moghaddam, L.K.; Paschepari, S.R.; Azizi, H.; Torkamandi, S.; et al. New Methods in the Diag nosis of Cancer and Gene Therapy of Cancer Based on Nanoparticles. Cancer Gene Ther. 2017, 24, 233–243.
- Gu, Y.; Zheng, Q.; Fan, G.; Liu, R. Advances in Anti-Cancer Activities of Flavonoids in Scute llariae Radix: Perspectives on Mechanism. Int. J. Mol. Sci. 2022, 23, 11042.
- Cheng, C.-S.; Chen, J.; Tan, H.-Y.; Wang, N.; Chen, Z.; Feng, Y. Scutellaria Baicalensis and Can cer Treatment: Recent Progress and Perspectives in Biomedical and Clinical Studies. Am. J. Chin. Med. 2018, 46, 25–54.
- Zhang, W.; Li, S.; Li, C.; Li, T.; Huang, Y. Remodeling Tumor Microenvironment with Natural Products to Overcome Drug Resistance. Front. Immunol. 2022, 13, 1051998.
- Guo, Q.; Cao, H.; Qi, X.; Li, H.; Ye, P.; Wang, Z.; Wang, D.; Sun, M. Research Progress in Reversal of Tumor Multi-Drug Resistance via Natural Products. Anticancer Agents Med. Chem. 2017, 17, 1466–1476.
- Deng, L.-J.; Qi, M.; Li, N.; Lei, Y.-H.; Zhang, D.-M.; Chen, J.-X. Natural Products and Their Derivatives: Promising Modulators of Tumor Immunotherapy. J. Leukoc. Biol. 2020, 108, 493–508.
- Agarwal, G.; Carcache, P.J.B.; Addo, E.M.; Kinghorn, A.D. Current Status and Contemporary Approaches to the Discovery of Antitumor Agents from Higher Plants. Biotechnol. Adv. 2020, 38, 107337.
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803.
- Liu, Y.; Wang, X.; Zeng, S.; Zhang, X.; Zhao, J.; Zhang, X.; Chen, X.; Yang, W.; Yang, Y.; Dong, Z.; et al. The Natural Polyphenol Curcumin Induces Apoptosis by Suppressing STAT3 Signaling in Esophageal Squamous Cell Carcinoma. J. Exp. Clin. Cancer Res. 2018, 37, 303.
- Luo, H.; Vong, C.T.; Chen, H.; Gao, Y.; Lyu, P.; Qiu, L.; Zhao, M.; Liu, Q.; Cheng, Z.; Zou, J.; et al. Naturally Occurring Anti-Cancer Compounds: Shining from Chinese Herbal Medic ine. Chin. Med. 2019, 14, 48.
- Abu Samaan, T.M.; Samec, M.; Liskova, A.; Kubatka, P.; Büsselberg, D. Paclitaxel’s Mechanistic and Clinical Effects on Breast Cancer. Biomolecules 2019, 9, 789.
- Zhu, S.; Yu, Q.; Huo, C.; Li, Y.; He, L.; Ran, B.; Chen, J.; Li, Y.; Liu, W. Ferroptosis: A Novel Mechanism of Artemisinin and Its Derivatives in Cancer Therapy. Curr. Med. Chem. 2020, 28, 329–345.
- Fruman, D.A.; Chiu, H.; Hopkins, B.D.; Bagrodia, S.; Cantley, L.C.; Abraham, R.T. The PI3K Pathway in Human Disease. Cell 2017, 170, 605–635.
- Yan, W.; Ma, X.; Zhao, X.; Zhang, S. Baicalein Induces Apoptosis and Autophagy of Breast Cancer Cells via Inhibiting PI3K/AKT Pathway in Vivo and Vitro. Drug Des. Dev. Ther. 2018, 12, 3961–3972.
- Tolomeo, M.; Cascio, A. The Multifaced Role of STAT3 in Cancer and Its Implication for Anticancer Therapy. Int. J. Mol. Sci. 2021, 22, 603.
- Luo, Y.; Gao, X.; Zou, L.; Lei, M.; Feng, J.; Hu, Z. Bavachin Induces Ferroptosis through the STAT3/P53/SLC7A11 Axis in Osteosarcoma Cells. Oxid. Med. Cell. Longev. 2021, 2021, e1783485.
- Hepworth, E.M.W.; Hinton, S.D. Pseudophosphatases as Regulators of MAPK Signaling. Int. J. Mol. Sci. 2021, 22, 12595.
- Jiang, Y.-M.; Liang, L.-Z.; Gan, T.-Q. Phytochemistry and Bioactivities of Biflavonoids: A Review. Assoc. Comput. Mach. 2017, 2017, 105–108.
- Křížkovská, B.; Kumar, R.; Řehořová, K.; Sýkora, D.; Dobiasová, S.; Kučerová, D.; Tan, M.C.; Linis, V.; Oyong, G.; Ruml, T.; et al. Comparison of Chemical Composition and Biological Activities of Eight Selaginella Species. Pharmaceuticals 2020, 14, 16.
- Gang, W.; Hua, L.S.; Lian, Z.H.; Mei, J.Y.; Mei, S.M.; Jiang, Y.L.; Mei, Z.X. Phytochemical Screening, Antioxidant, Antibacterial and Cytotoxic Activities of Different Extracts of Selaginella Doederleinii. Bangladesh J. Bot. 2017, 46, 1193–1201.
- Li, S.; Wang, X.; Wang, G.; Shi, P.; Lin, S.; Xu, D.; Chen, B.; Liu, A.; Huang, L.; Lin, X.; et al. Ethyl Acetate Extract of Selaginella Doederleinii Hieron Induces Cell Autophagic Death and Apoptosis in Colorectal Cancer via PI3K-Akt-MTOR and AMPKα-Signaling Pathways. Front. Pharmacol. 2020, 11, 565090.
- Lei, J.; Wang, Y.; Li, W.; Fu, S.; Zhou, J.; Lu, D.; Wang, C.; Sheng, X.; Zhang, M.; Xiao, S.; et al. Natural Green Deep Eutectic Solvents-Based Eco-Friendly and Efficient Extraction of Flavonoids from Selaginella Moellendorffii: Process Optimization, Composition Identification and Biological Activity. Sep. Purif. Technol. 2022, 283, 120203.
- Qin, G.; Lei, J.; Li, S.; Jiang, Y.; Qiao, L.; Ren, M.; Gao, Q.; Song, C.; Fu, S.; Zhou, J.; et al. Efficient, Green Extraction of Two Biflavonoids from Selaginella Uncinata with Deep Eutectic Solvents. Microchem. J. 2022, 183, 108085.
- Zhang, G.; Jing, Y.; Zhang, H.; Ma, E.; Guan, J.; Xue, F.; Liu, H.; Sun, X. Isolation and Cytotoxic Activity of Selaginellin Derivatives and Biflavonoids from Selaginella Tamariscina. Planta Med. 2012, 78, 390–392.
- Thamnarak, W.; Eurtivong, C.; Pollawatn, R.; Ruchirawat, S.; Thasana, N. Two New Nor-Lignans, Siamensinols A and B, from Selaginella Siamensis Hieron. and Their Biological Activities. Nat. Prod. Res. 2022, 36, 5591–5599.
- He, X.; Yang, F.; Huang, X. Proceedings of Chemistry, Pharmacology, Pharmacokinetics and Synthesis of Biflavonoids. Molecules 2021, 26, 6088.
- Xu, J.; Yang, L.; Wang, R.; Zeng, K.; Fan, B.; Zhao, Z. The Biflavonoids as Protein Tyrosine Phosphatase 1B Inhibitors from Selaginella Uncinata and Their Antihyperglycemic Action. Fitoterapia 2019, 137, 104255.
- Zou, Z.-X.; Zhang, S.; Tan, J.-B.; Chen, D.-K.; Xu, Y.-R.; Xu, K.-P.; Tan, G.-S. Two New Biflavonoids from Selaginella Doederleinii. Phytochem. Lett. 2020, 40, 126–129.
- Liu, L.-F.; Sun, H.-H.; Tan, J.-B.; Huang, Q.; Cheng, F.; Xu, K.-P.; Zou, Z.-X.; Tan, G.-S. New Cytotoxic Biflavones from Selaginella Doederleinii. Nat. Prod. Res. 2021, 35, 930–936.
- Kang, F.; Zhang, S.; Chen, D.; Tan, J.; Kuang, M.; Zhang, J.; Zeng, G.; Xu, K.; Zou, Z.; Tan, G. Biflavonoids from Selaginella Doederleinii as Potential Antitumor Agents for Intervention of Non-Small Cell Lung Cancer. Molecules 2021, 26, 5401.
- Yang, P.; Tian, Y.-M.; Cheng, Y.-T.; Yang, Y. A New Biflavonoid from Selaginella Uncinata. Chem. Nat. Compd. 2021, 57, 23–25.
- Xie, Y.; Xu, P.-S.; Xu, K.-P.; Zou, Z.-X.; Zhou, G.; Li, D.; Li, D.; Li, X.-M.; Li, J.; Tan, G.-S. Two New Biflavanoids from Selaginella Trichoclada Alsto. Nat. Prod. Res. 2021, 35, 3410–3416.
- Xie, Y.; Zhou, X.; Li, J.; Yao, X.-C.; Liu, W.-L.; Kang, F.-H.; Zou, Z.-X.; Xu, K.-P.; Xu, P.-S.; Tan, G.-S. Identification of a New Natural Biflavonoids against Breast Cancer Cells Induced Ferroptosis via the Mitochondrial Pathway. Bioorgan. Chem. 2021, 109, 104744.
- Xie, Y.; Yao, X.-C.; Tan, L.-H.; Long, H.-P.; Xu, P.-S.; Li, J.; Tan, G.-S. Trichocladabiflavone A, a Chalcone-Flavonone Type Biflavonoid from Selaginella Trichoclada Alsto. Nat. Prod. Res. 2022, 36, 1797–1802.
- Xie, Y.; Zhou, X.; Li, J.; Yao, X.; Liu, W.; Xu, P.; Tan, G. Cytotoxic Effects of the Biflavonoids Isolated from Selaginella Trichoclada on MCF-7 Cells and Its Potential Mechanism. Bioorg. Med. Chem. Lett. 2022, 56, 128486.
- Liu, X.-F.; Fu, T.-F.; Wu, J.-S.; Lu, B.-C.; Zhang, P.; Liu, H.-J. Cytotoxic Biflavonoids from Selaginella Braunii. Nat. Prod. Commun. 2022, 17, 1934578X2211250.
- Demehin, A.A.; Thamnarak, W.; Lamtha, T.; Chatwichien, J.; Eurtivong, C.; Choowongkomon, K.; Chainok, K.; Ruchirawat, S.; Thasana, N. Siamenflavones A-C, Three Undescribed Biflavonoids from Selaginella Siamensis Hieron. and Biflavonoids from Spike Mosses as EGFR Inhibitor. Phytochemistry 2022, 203, 113374.
- Long, H.-P.; Liu, J.; Xu, P.-S.; Xu, K.-P.; Li, J.; Tan, G.-S. Hypoglycemic Flavonoids from Selaginella Tamariscina (P.Beauv.) Spring. Phytochemistry 2022, 195, 113073.
- Ogunwa, T.H.; Taii, K.; Sadakane, K.; Kawata, Y.; Maruta, S.; Miyanishi, T. Morelloflavone as a Novel Inhibitor of Mitotic Kinesin Eg5. J. Biochem. 2019, 166, 129–137.
- Zhang, C.; Zhou, S.; Feng, L.; Zhang, D.; Lin, N.; Zhang, L.; Pan, J.; Wang, J.; Li, J. In Vitro Anti-Cancer Activity of Chamaejasmenin B and Neochamaejasmin C Isolated from the Root of Stellera Chamaejasme L. Acta Pharmacol. Sin. 2013, 34, 262–270.
- Wan, H.; Ge, L.; Li, J.; Zhang, K.; Wu, W.; Peng, S.; Zou, X.; Zhou, H.; Zhou, B.; Zeng, X. Effects of a Novel Biflavonoid of Lonicera Japonica Flower Buds on Modulating Apoptosis under Different Oxidative Conditions in Hepatoma Cells. Phytomedicine 2019, 57, 282–291.





