Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Joana Castro | -- | 1775 | 2023-05-04 18:35:09 | | | |
| 2 | Conner Chen | Meta information modification | 1775 | 2023-05-06 04:41:12 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Barros, M.M.; Castro, J.; Araújo, D.; Campos, A.M.; Oliveira, R.; Silva, S.; Outor-Monteiro, D.; Almeida, C. Etiology of Swine Colibacillosis. Encyclopedia. Available online: https://encyclopedia.pub/entry/43794 (accessed on 24 May 2026).
Barros MM, Castro J, Araújo D, Campos AM, Oliveira R, Silva S, et al. Etiology of Swine Colibacillosis. Encyclopedia. Available at: https://encyclopedia.pub/entry/43794. Accessed May 24, 2026.
Barros, Maria Margarida, Joana Castro, Daniela Araújo, Ana Maria Campos, Ricardo Oliveira, Sónia Silva, Divanildo Outor-Monteiro, Carina Almeida. "Etiology of Swine Colibacillosis" Encyclopedia, https://encyclopedia.pub/entry/43794 (accessed May 24, 2026).
Barros, M.M., Castro, J., Araújo, D., Campos, A.M., Oliveira, R., Silva, S., Outor-Monteiro, D., & Almeida, C. (2023, May 04). Etiology of Swine Colibacillosis. In Encyclopedia. https://encyclopedia.pub/entry/43794
Barros, Maria Margarida, et al. "Etiology of Swine Colibacillosis." Encyclopedia. Web. 04 May, 2023.
Copy Citation
Swine pathogenic infection caused by Escherichia coli, known as swine colibacillosis, represents an epidemiological challenge not only for animal husbandry but also for health authorities. To note, virulent E. coli strains might be transmitted, and also cause disease, in humans.
swine colibacillosis
AMR bacteria
E. coli pathotypes
1. Introduction
Porcine infection caused by Escherichia coli (E. coli), so-called swine colibacillosis, is responsible for a wide range of problems, such as neonatal diarrhea, post-weaning diarrhea (PWD), edema disease (ED), septicemia, polyserositis, coliform mastitis, and urinary tract infection [1]. Among the huge diversity, certain strains of E. coli, named enterotoxigenic E. coli (ETEC), are able to cause intestinal disease, which results in neonatal diarrhea, PWD, and ED. These porcine infections are the most threatening for the swine industry worldwide due to significant economic losses associated with morbidity, mortality, decreased weight gain, the rising cost of treatments, vaccinations, and feed supplements [1][2][3].
PWD and ED may occur separately or together either in an individual outbreak or in the same pig [1]. Within 2–3 weeks after weaning, the piglets are more susceptible to microbial infections, owing to the existence of an immature immune system associated with sow milk removal and resulting from the interruption of the nutritive intake of immunoglobulin present in the milk [2][4]. Therefore, this period is crucial and usually associated with the most severe form of enteric E. coli infection, manifested by sudden death or severe diarrhea [1].
As an effort to promote health and growth performance, diverse approaches have been used to prevent and treat swine colibacillosis, with antibiotics being the most commonly used strategy [2][4]. Consequently, due to the growing selective pressure of antibiotic use to treat these E. coli infections, the emergence of the antimicrobial resistance (AMR) phenomenon has limited treatment options for pig producers and an increased public health concern because of the potential transfer of AMR genetic determinants directly by contact and indirectly into the food chain, water, and manure, among others [1][5]. It is important to note that E. coli has a great capacity to acquire resistance genes, mainly through horizontal gene transfer [6], in which the mobile genetic elements, such as plasmids, transposons, and gene cassettes in class 1 and class 2 integrons, seem to play a main role in the dissemination [5]. Furthermore, E. coli behaves as a donor and as a recipient of resistance genes and thus can exchange those genes with other bacteria and act as a reservoir of AMR genes [5]. Accordingly, extended-spectrum β-lactamases, carbapenemases, 16S rRNA methylases, plasmid-mediated quinolone resistance (PMQR) genes, and mcr genes constitute the most problematic genetic determinant classes of AMR in E. coli [5].
2. Etiology
According to the taxonomy, the German pediatrician Theodor Escherich (1857–1911) gave the origin to the name of the genus Escherichia. This genus belongs to the family of Enterobacteriaceae, which contemplates the Gram-negative facultatively anaerobic rods, where the species Escherichia coli fits since they are Gram-negative, peritrichously flagellated rods of variable length and a diameter of about 1 μm [1].
Over the decades, the subdivision of species into types has been carried out by the development of several classification systems. Among these, serotyping (described in Table 1) is a recognized typing system to classify E. coli strains [1]. Nevertheless, since certain porcine pathogenic E. coli belong to a limited number of serotypes, this method is less used today for diagnostic purposes [1]. Thus, the serotyping technique has been substituted by the direct detection of genes coding for bacterial determinants involved in their pathogenesis, called virulence factors. Therefore, the term pathotype is applied to the classification of E. coli typologies according to the combinations of virulence factors [3].
Common E. coli pathotypes include Shiga toxin-producing E. coli (STEC) that contains two groups, edema disease E. coli (EDEC) and enterohemorrhagic E. coli (EHEC), enteropathogenic E. coli (EPEC), and extraintestinal pathogenic E. coli (ExPEC), with the most relevant E. coli pathotype in porcine, the enterotoxigenic E. coli (ETEC) [1]. It is important to highlight that EHEC and EPEC pathotypes are associated with the “attaching and effacing” (A/E) lesion development [1]. Nevertheless, EPEC is found in pigs with PWD, whereas EHEC is highly pathogenic in humans, and some zoonoses of this pathotype are sporadically recovered [1].
2.1. ETEC Virulence Factors and Their Impact on Trigger Colibacillosis Infection
E. coli is both a harmless commensal bacterium in the intestines of several mammals, as well as a dangerous pathogen [5]. Still, a small proportion of strains are pathogenic and can cause severe to life-threatening intestinal and extra-intestinal infections in humans and animals [5][7][8][9][10]. The pathogenicity of the strains is characterized by the presence of certain virulence factor combinations in particular adhesins and toxin secretions. As it has been described, the role of adhesins and surface proteins called fimbriae is to enable the adherence of ETEC to specific receptors on the brush borders of the small intestine’s enterocytes [8]. Regarding fimbriae, there are five common antigenically different types found in pigs: F4 (K88), F5 (K99), F41, F6 (987P), and F18 [11]. The first four fimbria types are responsible for mediating adhesion in neonates, while F18 is not associated with neonatal colibacillosis; however, it is common in postweaning colibacillosis as is F4. It is also important to highlight that hemolysis is a common trait for pathogenic F4 and F18 isolates [4]. The adhesion of ETEC through fimbriae conducts the release of toxins inside the epithelial cell that promote the secretion of water and electrolytes into the intestinal lumen [1][2][8]. ETEC constitutes the most relevant and pathogenic strains in porcine, where the following groups of toxins are produced, namely the two major classes of enterotoxins, heat-stable toxin (ST) and heat-labile toxin (LT), as well as the enteroaggregative heat-stable toxin 1 (EAST1), as described in Table 1. STs are divided into Sta (also nominated STI, ST1, or StaP) and STb (also nominated STII or ST2) [1][2]. Similarly, LT toxins are divided into two groups, LTI and LTII. On the other hand, EAST1 is widespread among porcine ETEC, but its role in this illness remains controversial [1]. It is important to note that EAST1 alone does not seem capable of developing the disease; however, together with LT, it takes effect [1][2]. Additionally, it is important to note that Shiga toxin (Stx or VT), namely the Shiga toxin type 2e (Stx2e), E. coli is also found in some ETEC strains, being commonly found in ETEC strains expressing LT and/or ST toxins [11]. This toxin is a causative factor of edema in swine, and it has been also associated with diarrhea commonly found in colibacillosis [1][11].
Regarding the route of contamination, ETEC is firstly ingested through the oral route and then passes through the stomach. When it reaches the intestine, in the presence of suitable environmental conditions, ETEC proliferates and causes disease [1][8]. At this point, they colonize the small intestine following the attachment of fimbria adhesins to specific receptors present on the epithelium as well as in the mucus coating the epithelium of the small intestine (see Figure 1). This promotes the production and release of enterotoxins, as mentioned above, inside the epithelial cell that stimulates the secretion of water and electrolytes into the intestinal lumen, which leads to diarrhea, weight loss, and possibly death [1][3][8]. To note, the combinations of different ETEC virulence factors (adhesins and toxins) are associated with the development of different diseases, namely neonatal diarrhea and PWD, as shown in Table 1.
Table 1. Virulence factors of enterotoxigenic Escherichia coli (ETEC) associated with swine enteric colibacillosis.
| Adhesins | Toxins | Serotypes | Disease |
|---|---|---|---|
| F5, F6, F41 | STa | O8, O9, O20, O64, O101 | Neonatal diarrhea |
| F4 | STa, STb, LT, EAST1, α-hemolysin b | O8, O138, O141, O145, O147, O149, O157 | Neonatal diarrhea Diarrhea in young pigs preweaning |
| F4, AIDA a, unknown | STa, STb, LT, EAST1, α-hemolysin b | O8, O138, O139, O141, O147, O149, O157 | PWD |
| F18, AIDA a | STa, STb, LT, Stx (or VT) c, EAST1, α-hemolysin b | O8, O138, O139, O141, O147, O149, O157 | PWD |
a AIDA is a non-fimbrial adhesin involved in diffuse adherence; nevertheless, the mechanism of AIDA in swine colibacillosis is not yet elucidated. However, this non-fimbrial adhesin has been associated with ETEC strains from weaned pigs with PWD [2]. b α-hemolysin is a pore-forming cytolysin associated with ETEC strains that cause diarrhea in animals [12]. c Shiga toxins (Stx or VT) are cytotoxins produced by Shiga-toxin-producing E. coli (STEC), and in swine, the most important STECs are those that cause edema disease (ED) [1].
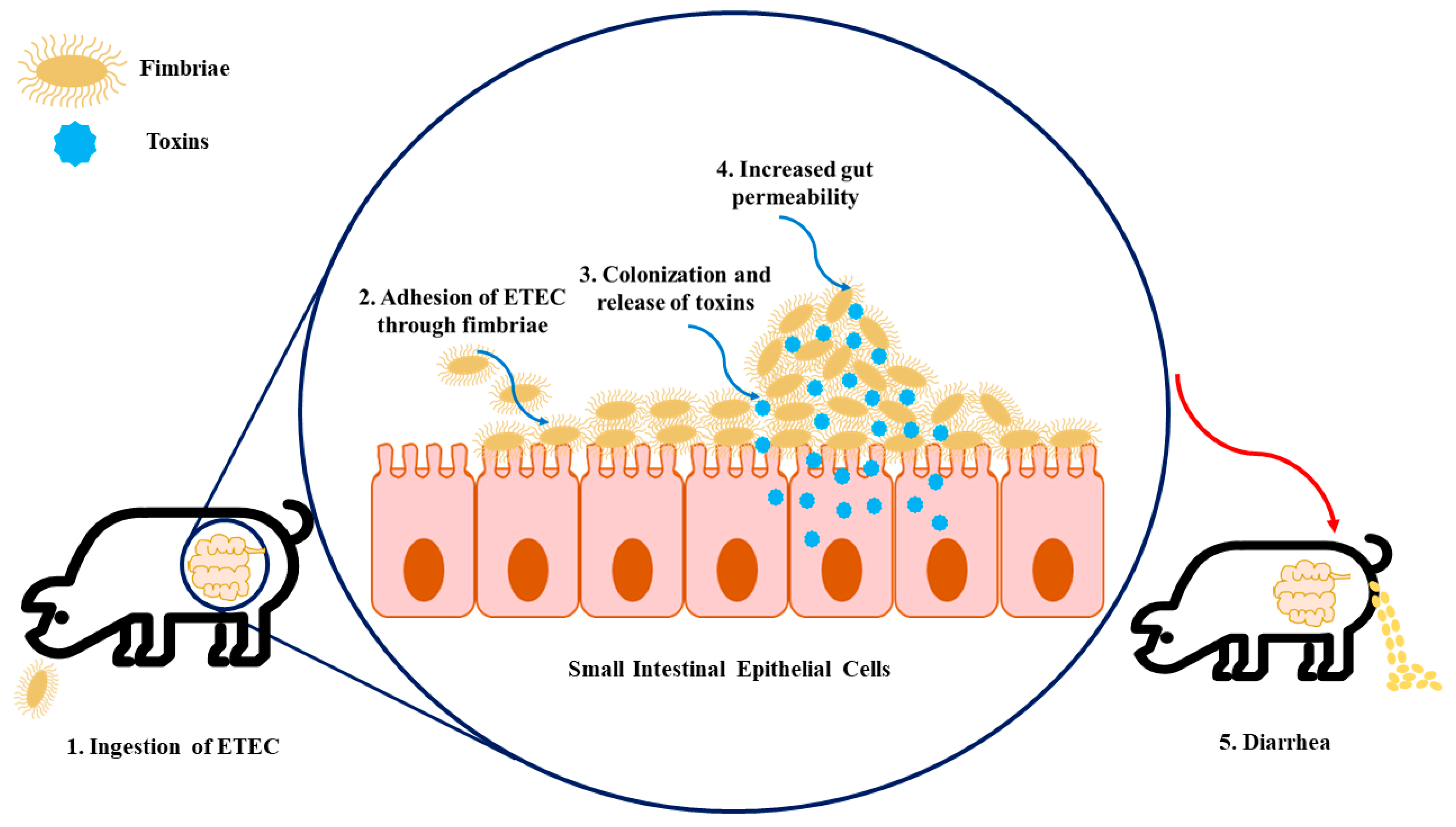
Figure 1. The infection model of enterotoxigenic Escherichia coli (ETEC) on intestinal epithelial cells. (1) Firstly, the swine ingest ETEC, enabling its transition to the gastrointestinal tract. (2) The fimbriae expressed by ETEC allow the adhesion of bacteria to specific receptors present in the intestinal epithelial cells. (3) Colonization arises in the small intestinal mucosa, which leads to the production of toxins. (4) These enterotoxins promote water and electrolyte loss into the intestinal lumen, resulting in increased gut permeability. (5) As a consequence of increased gut permeability and massive water loss, diarrhea, weight loss, and mortality can happen.
2.1.1. Neonatal ETEC
Neonatal diarrhea caused by ETEC is only associated with STa and might have one or more of the fimbriae associated, including F4 (K88), F5 (K99), F6 (987P), and F41 [1]. Of note, F4 is usually related to the colonization of the length of the jejunum and ileum, whereas F5, F6, and F41 mostly colonize the posterior jejunum and ileum [2]. Recently, Dubreuil and colleagues reviewed the prevalence of F4-, F5-, and F6-fimbriated ETEC from neonatal diarrhea and concluded that they exhibited both temporal and geographic variations [13]. This illness usually occurs in the first four days after the piglet is born and is characterized by whitish-yellow diarrhea, with a watery or creamy consistency, in large quantities [2]. Once ETEC has adhered to the epithelium, it binds to specific receptors in the apical region of the epithelial cells in the jejunal region and begins to produce the enterotoxin Sta, culminating in the hypersecretion of electrolytes and fluids in the small intestine. Eventually, if the large intestine is unable to reabsorb these excess fluids, the piglet enters a state of dehydration (and in more severe cases, metabolic acidosis) and eventually dies [1].
2.1.2. Postweaning ETEC
The enterotoxins STa, STb, LT, and EAST-1 are typically produced individually or together in the ETECs that cause diarrhea in postweaning or older suckling pigs [1]. The fimbriae involved in this disease are mainly F4 and F18, and both possess several variant subtypes based on antigenic differences. In this respect, F4 and F18 evidenced the following subtypes, ab, ac, and ad and ab and ac, respectively [2]. Post-weaning diarrhea usually appears 2 to 3 weeks after weaning and is characterized by the appearance of watery diarrhea that can vary from yellowish-grey or a slightly pinkish color, which lasts for up to a week [1]. The ETEC responsible for this condition generally colonizes the duodenal and jejunal portions of the small intestine, thus inducing the hypersecretion of fluids, very similar to that described in neonatal diarrhea [1].
References
- Fairbrother, J.M.; Nadeau, É. Colibacillosis. In Diseases of Swine; Zimmerman, J.J., Karriker, L.A., Ramirez, A., Schwartz, K.J., Stevenson, G.W., Zhang, J., Eds.; John Wiley & Son: Hoboken, NJ, USA, 2019; pp. 807–834.
- Luppi, A. Swine Enteric Colibacillosis: Diagnosis, Therapy and Antimicrobial Resistance. Porcine Health Manag. 2017, 3, 16.
- Fairbrother, J.M.; Nadeau, É.; Gyles, C.L. Escherichia coli in Postweaning Diarrhea in Pigs: An Update on Bacterial Types, Pathogenesis, and Prevention Strategies. Anim. Health Res. Rev. 2005, 6, 17–39.
- Castro, J.; Barros, M.M.; Araújo, D.; Campos, A.M.; Oliveira, R.; Silva, S.; Almeida, C. Swine Enteric Colibacillosis: Current Treatment Avenues and Future Directions. Front. Vet. Sci. 2022, 9, 981207.
- Poirel, L.; Madec, J.-Y.; Lupo, A.; Schink, A.-K.; Kieffer, N.; Nordmann, P.; Schwarz, S. Antimicrobial Resistance in Escherichia coli. Microbiol. Spectr. 2018, 6, 4.
- Javadi, M.; Bouzari, S.; Oloomi, M. Horizontal Gene Transfer and the Diversity of Escherichia coli. In Escherichia coli—Recent Advances on Physiology, Pathogenesis and Biotechnological Applications; InTech: London, UK, 2017; pp. 318–331.
- Nielsen, S.S.; Bicout, D.J.; Calistri, P.; Canali, E.; Drewe, J.A.; Garin-Bastuji, B.; Gonzales Rojas, J.L.; Gortázar, C.; Herskin, M.; Michel, V.; et al. Assessment of Listing and Categorisation of Animal Diseases within the Framework of the Animal Health Law (Regulation (EU) No 2016/429): Antimicrobial-Resistant Escherichia coli in Dogs and Cats, Horses, Swine, Poultry, Cattle, Sheep and Goats. EFSA J. 2022, 20, e07311.
- Gyles, C.L.; Fairbrother, J.M. Escherichia coli. In Pathogenesis of Bacterial Infections in Animals; Gyles, C.L., Prescott, J.F., Songer, J.G., Thoen, C.O., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2010; pp. 267–279. ISBN 9780813812373.
- Loayza, F.; Graham, J.P.; Trueba, G. Factors Obscuring the Role of E. coli from Domestic Animals in the Global Antimicrobial Resistance Crisis: An Evidence-Based Review. Int. J. Environ. Res. Public Health 2020, 17, 3061.
- Valat, C.; Drapeau, A.; Beurlet, S.; Bachy, V.; Boulouis, H.-J.; Pin, R.; Cazeau, G.; Madec, J.-Y.; Haenni, M. Pathogenic Escherichia coli in Dogs Reveals the Predominance of ST372 and the Human-Associated ST73 Extra-Intestinal Lineages. Front. Microbiol. 2020, 11, 580.
- Kim, K.; Song, M.; Liu, Y.; Ji, P. Enterotoxigenic Escherichia coli Infection of Weaned Pigs: Intestinal Challenges and Nutritional Intervention to Enhance Disease Resistance. Front. Immunol. 2022, 13, 885253.
- Burgos, Y.; Beutin, L. Common Origin of Plasmid Encoded Alpha-Hemolysin Genes in Escherichia coli. BMC Microbiol. 2010, 10, 193.
- Dubreuil, J.D.; Isaacson, R.E.; Schifferli, D.M. Animal Enterotoxigenic Escherichia coli. EcoSal Plus 2016, 7, 1–47.
More
Information
Subjects:
Veterinary Sciences
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
912
Revisions:
2 times
(View History)
Update Date:
06 May 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





