
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Wissem mnif | -- | 2606 | 2023-05-04 09:57:48 | | | |
| 2 | Lindsay Dong | Meta information modification | 2606 | 2023-05-06 05:52:25 | | |
Video Upload Options
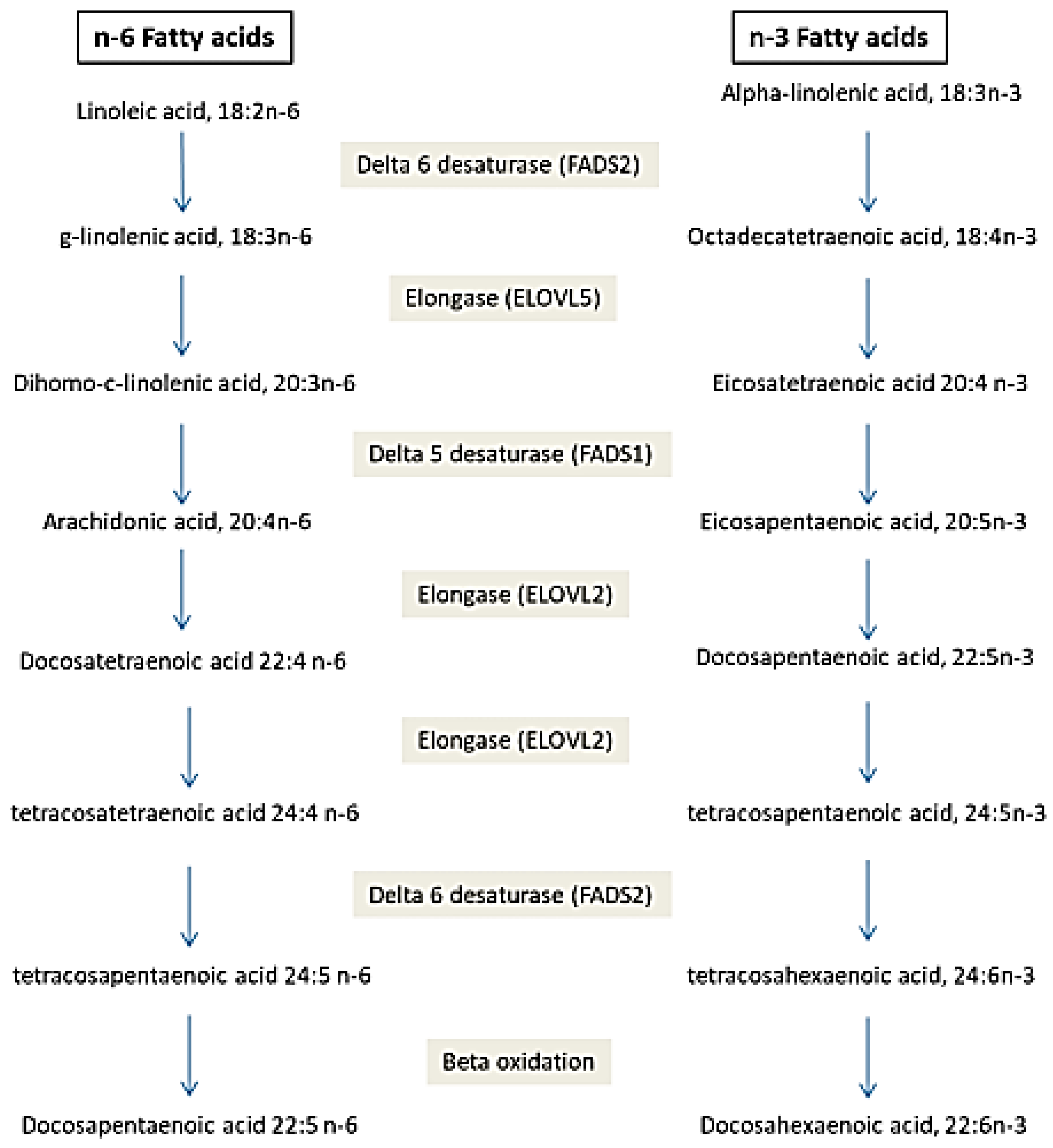
Circulating fatty acids (FA) have an endogenous or exogenous origin and are metabolized under the effect of many enzymes. They play crucial roles in many mechanisms: cell signaling, modulation of gene expression, etc., which leads to the hypothesis that their perturbation could be the cause of disease development. FA in erythrocytes and plasma rather than dietary FA could be used as a biomarker for many diseases. Cardiovascular disease was associated with elevated trans FA and decreased DHA and EPA. Increased arachidonic acid and decreased Docosahexaenoic Acids (DHA) were associated with Alzheimer’s disease. Low Arachidonic acid and DHA are associated with neonatal morbidities and mortality. Decreased saturated fatty acids (SFA), increased monounsaturated FA (MUFA) and polyunsaturated FA (PUFA) (C18:2 n-6 and C20:3 n-6) are associated with cancer. Additionally, genetic polymorphisms in genes coding for enzymes implicated in FA metabolism are associated with disease development. FA desaturase (FADS1 and FADS2) polymorphisms are associated with Alzheimer’s disease, Acute Coronary Syndrome, Autism spectrum disorder and obesity. Polymorphisms in FA elongase (ELOVL2) are associated with Alzheimer’s disease, Autism spectrum disorder and obesity. FA-binding protein polymorphism is associated with dyslipidemia, type 2 diabetes, metabolic syndrome, obesity, hypertension, non-alcoholic fatty liver disease, peripheral atherosclerosis combined with type 2 diabetes and polycystic ovary syndrome. Acetyl-coenzyme A carboxylase polymorphisms are associated with diabetes, obesity and diabetic nephropathy. FA profile and genetic variants of proteins implicated in FA metabolism could be considered as disease biomarkers and may help with the prevention and management of diseases.
1. Introduction
2. Fatty Acid Profile as Disease Predictor
2.1. Fatty Acids and Diseases
2.1.1. Fatty Acids and Cardiovascular Disease (CVD)
2.1.2. Fatty Acids and Diabetes
2.1.3. Fatty Acids and Cancer
2.1.4. Fatty Acids and Other Diseases
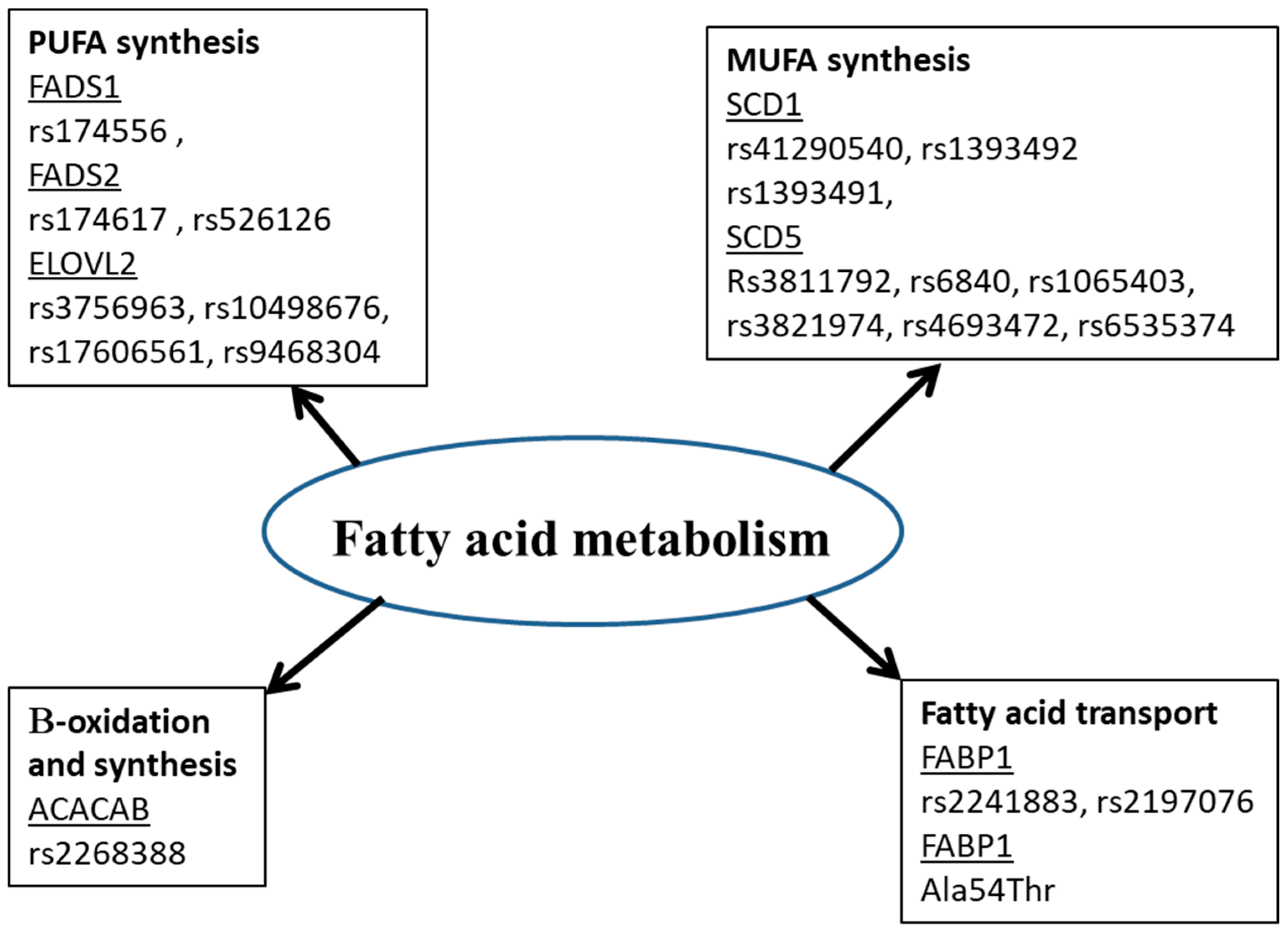
2.2. Polymorphisms of Gene Implicated in Fatty Acid Metabolism and Diseases
References
- Holman, R.T.; Johnson, S.B.; Hatch, T.F. A case of human linolenic acid deficiency involving neurological abnormalities. Am. J. Clin. Nutr. 1982, 35, 617–623.
- Hansen, A.E.; Haggard, M.E.; Boelsche, A.N.; Adam, D.J.; Wiese, H.F. Essential fatty acids in infant nutrition: III. Clinical manifestations of linoleic acid deficiency. J. Nutr. 1958, 66, 565–576.
- Guesnet, P.; Alessandri, J.-M.; Astorg, P.; Pifferi, F.; Lavialle, M. Les rôles physiologiques majeurs exercés par les acides gras polyinsaturés (AGPI). Oléagineux Corps Gras Lipides 2005, 12, 333–343.
- Maulucci, G.; Cohen, O.; Daniel, B.; Sansone, A.; Petropoulou, P.I.; Filou, S.; Spyridonidis, A.; Pani, G.; De Spirito, M.; Chatgilialoglu, C.; et al. Fatty acid-related modulations of membrane fluidity in cells: Detection and implications. Free. Radic. Res. 2016, 50, S40–S50.
- Stillwell, W.; Shaikh, S.R.; Zerouga, M.; Siddiqui, R.; Wassall, S.R. Docosahexaenoic acid affects cell signaling by altering lipid rafts. Reprod. Nutr. Dev. 2005, 45, 559–579.
- Ghosh, A.; Gao, L.; Thakur, A.; Siu, P.M.; Lai, C.W.K. Role of free fatty acids in endothelial dysfunction. J. Biomed. Sci. 2017, 24, 50.
- Scott, J.S.; Nassar, Z.D.; Swinnen, J.V.; Butler, L.M. Monounsaturated Fatty Acids: Key Regulators of Cell Viability and Intracellular Signaling in Cancer. Mol. Cancer Res. 2022, 20, 1354–1364.
- Wahle, K.W.J.; Rotondo, D.; Heys, S.D. Polyunsaturated fatty acids and gene expression in mammalian systems. Proc. Nutr. Soc. 2003, 62, 349–360.
- Sun, Q.; Ma, J.; Campos, H.; Hankinson, S.E.; Manson, J.E.; Stampfer, M.J.; Rexrode, K.M.; Willett, W.C.; Hu, F.B. A Prospective Study of Trans Fatty Acids in Erythrocytes and Risk of Coronary Heart Disease. Circulation 2007, 115, 1858–1865.
- Mallick, R.; Duttaroy, A.K. Modulation of endothelium function by fatty acids. Mol. Cell. Biochem. 2021, 477, 15–38.
- Fan, W.X.; Parker, R.; Parpia, B.; Qu, Y.S.; Cassano, P.; Crawford, M.; Leyton, J.; Tian, J.; Li, J.Y.; Chen, J.S. Erythrocyte fatty acids, plasma lipids, and cardiovascular disease in rural China. Am. J. Clin. Nutr. 1990, 52, 1027–1036.
- Lemaitre, R.N.; McKnight, B.; Sotoodehnia, N.; Fretts, A.M.; Qureshi, W.T.; Song, X.; King, I.B.; Sitlani, C.M.; Siscovick, D.S.; Psaty, B.M.; et al. Circulating Very Long-Chain Saturated Fatty Acids and Heart Failure: The Cardiovascular Health Study. J. Am. Heart Assoc. 2018, 7, e010019.
- Ahmed, S.H.; Koubaa, N.; Kharroubi, W.; Zarrouk, A.; Mnari, A.; Batbout, F.; Gamra, H.; Hammami, S.; Lizard, G.; Hammami, M. Identification of long and very long chain fatty acids, plasmalogen-C16: 0 and phytanic acid as new lipid biomarkers in Tunisian coronary artery disease patients. Prostaglandins Other Lipid Mediat. 2017, 131, 49–58.
- Ahmed, S.H.; Kharroubi, W.; Kaoubaa, N.; Zarrouk, A.; Batbout, F.; Gamra, H.; Najjar, M.F.; Lizard, G.; Hininger-Favier, I.; Hammami, M. Correlation of trans fatty acids with the severity of coronary artery disease lesions. Lipids Health Dis. 2018, 17, 52.
- Guo, M.; Fan, X.; Tuerhongjiang, G.; Wang, C.; Wu, H.; Lou, B.; Wu, Y.; Yuan, Z.; She, J. Targeted metabolomic analysis of plasma fatty acids in acute myocardial infarction in young adults. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 3131–3141.
- Nishizaki, Y.; Shimada, K.; Tani, S.; Ogawa, T.; Ando, J.; Takahashi, M.; Yamamoto, M.; Shinozaki, T.; Miyazaki, T.; Miyauchi, K.; et al. Association between the ratio of serum n-3 to n-6 polyunsaturated fatty acids and acute coronary syndrome in non-obese patients with coronary risk factor: A multicenter cross-sectional study. BMC Cardiovasc. Disord. 2020, 20, 160–167.
- Bittner, D.O.; Goeller, M.; Zopf, Y.; Achenbach, S.; Marwan, M. Early-onset coronary atherosclerosis in patients with low levels of omega-3 fatty acids. Eur. J. Clin. Nutr. 2020, 74, 651–656.
- Bazan, H.A.; Lu, Y.; Jun, B.; Fang, Z.; Woods, T.C.; Hong, S. Circulating inflammation-resolving lipid mediators RvD1 and DHA are decreased in patients with acutely symptomatic carotid disease. Prostaglandins Leukot. Essent. Fat. Acids 2017, 125, 43–47.
- Yang, W.-S.; Chen, Y.-Y.; Chen, P.-C.; Hsu, H.-C.; Su, T.-C.; Lin, H.-J.; Chen, M.-F.; Lee, Y.-T.; Chien, K.-L. Association between Plasma N-6 Polyunsaturated Fatty Acids Levels and the Risk of Cardiovascular Disease in a Community-based Cohort Study. Sci. Rep. 2019, 9, 19298.
- Chien, K.-L.; Lin, H.-J.; Hsu, H.-C.; Chen, P.-C.; Su, T.-C.; Chen, M.-F.; Lee, Y.-T. Comparison of predictive performance of various fatty acids for the risk of cardiovascular disease events and all-cause deaths in a community-based cohort. Atherosclerosis 2013, 230, 140–147.
- Würtz, P.; Havulinna, A.S.; Soininen, P.; Tynkkynen, T.; Prieto-Merino, D.; Tillin, T.; Ghorbani, A.; Artati, A.; Wang, Q.; Tiainen, M.; et al. Metabolite profiling and cardiovascular event risk: A prospective study of 3 population-based cohorts. Circulation 2015, 131, 774–785.
- Dozio, E.; Vianello, E.; Grossi, E.; Menicanti, L.; Schmitz, G.; Romanelli, M.M.C. Plasma fatty acid profile as biomarker of coronary artery disease: A pilot study using fourth generation artificial neural networks. J. Biol. Regul. Homeost. Agents 2018, 32, 1007–1013.
- Muldoon, M.F.; Erickson, K.I.; Goodpaster, B.H.; Jakicic, J.M.; Conklin, S.M.; Sekikawa, A.; Yao, J.K.; Manuck, S.B. Concurrent Physical Activity Modifies the Association between n3 Long-Chain Fatty Acids and Cardiometabolic Risk in Midlife Adults. J. Nutr. 2013, 143, 1414–1420.
- Harris, K.; Oshima, M.; Sattar, N.; Würtz, P.; Jun, M.; Welsh, P.; Hamet, P.; Harrap, S.; Poulter, N.; Chalmers, J.; et al. Plasma fatty acids and the risk of vascular disease and mortality outcomes in individuals with type 2 diabetes: Results from the ADVANCE study. Diabetologia 2020, 63, 1637–1647.
- Shoji, T.; Kakiya, R.; Hayashi, T.; Tsujimoto, Y.; Sonoda, M.; Shima, H.; Mori, K.; Fukumoto, S.; Tahara, H.; Shioi, A.; et al. Serum n-3 and n-6 Polyunsaturated Fatty Acid Profile as an Independent Predictor of Cardiovascular Events in Hemodialysis Patients. Am. J. Kidney Dis. 2013, 62, 568–576.
- Noguchi, H.; Okubo, R.; Hamazaki, K.; Yamashita, A.; Narisawa, T.; Matsuoka, Y.J. Serum polyunsaturated fatty acids and risk of psychiatric disorder at 6 months after acute coronary syndrome: A prospective cohort study. Prostaglandins Leukot. Essent. Fat. Acids 2019, 149, 18–23.
- Ouchi, S.; Miyazaki, T.; Shimada, K.; Sugita, Y.; Shimizu, M.; Murata, A.; Kato, T.; Aikawa, T.; Suda, S.; Shiozawa, T.; et al. Decreased circulating dihomo-gamma-linolenic acid levels are associated with total mortality in patients with acute cardiovascular disease and acute decompensated heart failure. Lipids Health Dis. 2017, 16, 150.
- Li, Z.; Zhang, Y.; Su, D.; Lv, X.; Wang, M.; Ding, D.; Ma, J.; Xia, M.; Wang, D.; Yang, Y.; et al. The opposite associations of long-chain versus very long-chain monounsaturated fatty acids with mortality among patients with coronary artery disease. Heart 2014, 100, 1597–1605.
- Harris, W.S.; Kennedy, K.F.; O’Keefe, J.H.; Spertus, J.A. Red blood cell fatty acid levels improve GRACE score prediction of 2-yr mortality in patients with myocardial infarction. Int. J. Cardiol. 2013, 168, 53–59.
- Salgin, B.; Ong, K.K.; Thankamony, A.; Emmett, P.; Wareham, N.J.; Dunger, D.B. Higher Fasting Plasma Free Fatty Acid Levels Are Associated with Lower Insulin Secretion in Children and Adults and a Higher Incidence of Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2012, 97, 3302–3309.
- Clore, J.N.; Allred, J.; White, D.; Li, J.; Stillman, J. The role of plasma fatty acid composition in endogenous glucose production in patients with type 2 diabetes mellitus. Metabolism 2002, 51, 1471–1477.
- Liu, L.; Li, Y.; Guan, C.; Li, K.; Wang, C.; Feng, R.; Sun, C. Free fatty acid metabolic profile and biomarkers of isolated post-challenge diabetes and type 2 diabetes mellitus based on GC–MS and multivariate statistical analysis. J. Chromatogr. B 2010, 878, 2817–2825.
- Jo, S.; An, W.-S.; Park, Y. Erythrocyte n-3 Polyunsaturated Fatty Acids and the Risk of Type 2 Diabetes in Koreans: A Case-Control Study. Ann. Nutr. Metab. 2013, 63, 283–290.
- Ma, Y.; Xiong, J.; Zhang, X.; Qiu, T.; Pang, H.; Li, X.; Zhu, J.; Wang, J.; Pan, C.; Yang, X.; et al. Potential biomarker in serum for predicting susceptibility to type 2 diabetes mellitus: Free fatty acid 22:6. J. Diabetes Investig. 2020, 12, 950–962.
- Shetty, S.S.; N., S.K.; Shetty, P.K. ω-6/ω-3 fatty acid ratio as an essential predictive biomarker in the management of type 2 diabetes mellitus. Nutrition 2020, 79-80, 110968.
- Lu, Y.; Wang, Y.; Ong, C.-N.; Subramaniam, T.; Choi, H.W.; Yuan, J.-M.; Koh, W.-P.; Pan, A. Metabolic signatures and risk of type 2 diabetes in a Chinese population: An untargeted metabolomics study using both LC-MS and GC-MS. Diabetologia 2016, 59, 2349–2359.
- Grapov, D.; Adams, S.H.; Pedersen, T.L.; Garvey, W.T.; Newman, J.W. Type 2 Diabetes Associated Changes in the Plasma Non-Esterified Fatty Acids, Oxylipins and Endocannabinoids. PLoS ONE 2012, 7, e48852.
- Yi, L.; He, J.; Liang, Y.; Yuan, D.; Gao, H.; Zhou, H. Simultaneously quantitative measurement of comprehensive profiles of esterified and non-esterified fatty acid in plasma of type 2 diabetic patients. Chem. Phys. Lipids 2007, 150, 204–216.
- Sobczak, A.I.S.; Blindauer, C.A.; Stewart, A.J. Changes in Plasma Free Fatty Acids Associated with Type-2 Diabetes. Nutrients 2019, 11, 2022.
- Ruddock, M.W.; Stein, A.; Landaker, E.; Park, J.; Cooksey, R.C.; McClain, D.; Patti, M.-E. Saturated Fatty Acids Inhibit Hepatic Insulin Action by Modulating Insulin Receptor Expression and Post-receptor Signalling. J. Biochem. 2008, 144, 599–607.
- Miao, Z.; Lin, J.-S.; Mao, Y.; Chen, G.-D.; Zeng, F.-F.; Dong, H.-L.; Jiang, Z.; Wang, J.; Xiao, C.; Shuai, M.; et al. Erythrocyte n-6 Polyunsaturated Fatty Acids, Gut Microbiota, and Incident Type 2 Diabetes: A Prospective Cohort Study. Diabetes Care 2020, 43, 2435–2443.
- Amézaga, J.; Arranz, S.; Urruticoechea, A.; Ugartemendia, G.; Larraioz, A.; Louka, M.; Uriarte, M.; Ferreri, C.; Tueros, I. Altered Red Blood Cell Membrane Fatty Acid Profile in Cancer Patients. Nutrients 2018, 10, 1853.
- Jiao, J.; Kwan, S.-Y.; Sabotta, C.M.; Tanaka, H.; Veillon, L.; Warmoes, M.O.; Lorenzi, P.L.; Wang, Y.; Wei, P.; Hawk, E.T.; et al. Circulating Fatty Acids Associated with Advanced Liver Fibrosis and Hepatocellular Carcinoma in South Texas Hispanics. Cancer Epidemiol. Biomark. Prev. 2021, 30, 1643–1651.
- Chen, Q.; Wang, J.; Lin, J.; Chen, L.; Lin, L.-S.; Pan, L.-Z.; Shi, B.; Qiu, Y.; Zheng, X.-Y.; Chen, F.; et al. Erythrocyte ω-3 polyunsaturated fatty acids are inversely associated with the risk of oral cancer: A case-control study. Nutr. Diabetes 2020, 10, 35.
- Shishavan, N.G.; Mohamadkhani, A.; Sepanlou, S.G.; Masoudi, S.; Sharafkhah, M.; Poustchi, H.; Hekmatdoost, A.; Pourshams, A. Circulating plasma fatty acids and risk of pancreatic cancer: Results from the Golestan Cohort Study. Clin. Nutr. 2020, 40, 1897–1904.
- Zaridze, D.G.; Chevchenko, V.E.; Levtshuk, A.A.; Lifanova, Y.E.; Maximovitch, D.M. Fatty acid composition of phospholipids in erythrocyte membranes and risk of breast cancer. Int. J. Cancer 1990, 45, 807–810.
- Hirko, K.A.; Chai, B.; Spiegelman, D.; Campos, H.; Farvid, M.S.; Hankinson, S.E.; Willett, W.C.; Eliassen, A.H. Erythrocyte membrane fatty acids and breast cancer risk: A prospective analysis in the nurses’ health study II. Int. J. Cancer 2017, 142, 1116–1129.
- McGee, E.E.; Kim, C.H.; Wang, M.; Spiegelman, D.; Stover, D.G.; Heng, Y.J.; Collins, L.C.; Baker, G.M.; Farvid, M.S.; Schedin, P.; et al. Erythrocyte membrane fatty acids and breast cancer risk by tumor tissue expression of immuno-inflammatory markers and fatty acid synthase: A nested case-control study. Breast Cancer Res. 2020, 22, 78.
- Ruan, X.; Wang, Y.; Zhou, L.; Zheng, Q.; Hao, H.; He, D. Evaluation of Untargeted Metabolomic Strategy for the Discovery of Biomarker of Breast Cancer. Front. Pharmacol. 2022, 13, 894099.
- Kaikkonen, J.E.; Jula, A.; Viikari, J.S.A.; Juonala, M.; Hutri-Kähönen, N.; Kähönen, M.; Lehtimäki, T.; Raitakari, O.T. Associations of Serum Fatty Acid Proportions with Obesity, Insulin Resistance, Blood Pressure, and Fatty Liver: The Cardiovascular Risk in Young Finns Study. J. Nutr. 2021, 151, 970–978.
- Ma, Y.; Qiu, T.; Zhu, J.; Wang, J.; Li, X.; Deng, Y.; Zhang, X.; Feng, J.; Chen, K.; Wang, C.; et al. Serum FFAs profile analysis of Normal weight and obesity individuals of Han and Uygur nationalities in China. Lipids Health Dis. 2020, 19, 13.
- Ni, Y.; Zhao, L.; Yu, H.; Ma, X.; Bao, Y.; Rajani, C.; Loo, L.W.; Shvetsov, Y.B.; Yu, H.; Chen, T.; et al. Circulating Unsaturated Fatty Acids Delineate the Metabolic Status of Obese Individuals. eBioMedicine 2015, 2, 1513–1522.
- Zarrouk, A.; Riedinger, J.-M.; Ahmed, S.H.; Hammami, S.; Chaabane, W.; Debbabi, M.; Ben Ammou, S.; Rouaud, O.; Frih, M.; Lizard, G.; et al. Fatty Acid Profiles in Demented Patients: Identification of Hexacosanoic Acid (C26:0) as a Blood Lipid Biomarker of Dementia. J. Alzheimer’s Dis. 2015, 44, 1349–1359.
- Yamagishi, K.; Ikeda, A.; Chei, C.-L.; Noda, H.; Umesawa, M.; Cui, R.; Muraki, I.; Ohira, T.; Imano, H.; Sankai, T.; et al. Serum α-linolenic and other ω-3 fatty acids, and risk of disabling dementia: Community-based nested case–control study. Clin. Nutr. 2016, 36, 793–797.
- Hammouda, S.; Ghzaiel, I.; Khamlaoui, W.; Hammami, S.; Mhenni, S.Y.; Samet, S.; Hammami, M.; Zarrouk, A. Genetic variants in FADS1 and ELOVL2 increase level of arachidonic acid and the risk of Alzheimer’s disease in the Tunisian population. Prostaglandins Leukot. Essent. Fat. Acids 2020, 160, 102159.
- Sala-Vila, A.; Satizabal, C.L.; Tintle, N.; van Lent, D.M.; Vasan, R.S.; Beiser, A.S.; Seshadri, S.; Harris, W.S. Red Blood Cell DHA Is Inversely Associated with Risk of Incident Alzheimer’s Disease and All-Cause Dementia: Framingham Offspring Study. Nutrients 2022, 14, 2408.
- Tomata, Y.; Larsson, S.C.; Hägg, S. Polyunsaturated fatty acids and risk of Alzheimer’s disease: A Mendelian randomization study. Eur. J. Nutr. 2019, 59, 1763–1766.
- Jaček, M.; Hrnčířová, D.; Rambousková, J.; Dlouhý, P.; Tůma, P. Effect of Food with Low Enrichment of N-3 Fatty Acids in a Two-Month Diet on the Fatty Acid Content in the Plasma and Erythrocytes and on Cardiovascular Risk Markers in Healthy Young Men. Nutrients 2020, 12, 2207.
- Seethaler, B.; Basrai, M.; Vetter, W.; Lehnert, K.; Engel, C.; Siniatchkin, M.; Halle, M.; Kiechle, M.; Bischoff, S.C. Fatty acid profiles in erythrocyte membranes following the Mediterranean diet—Data from a multicenter lifestyle intervention study in women with hereditary breast cancer (LIBRE). Clin. Nutr. 2019, 39, 2389–2398.
- Lankinen, M.; Uusitupa, M.; Schwab, U. Genes and Dietary Fatty Acids in Regulation of Fatty Acid Composition of Plasma and Erythrocyte Membranes. Nutrients 2018, 10, 1785.
- Raeisi, M.; Hassanbeigi, L.; Khalili, F.; Kharrati-Shishavan, H.; Yousefi, M.; Mehdizadeh, A. Stearoyl-CoA desaturase 1 as a therapeutic target for cancer: A focus on hepatocellular carcinoma. Mol. Biol. Rep. 2022, 49, 8871–8882.
- Stearoyl-CoA desaturase 1: A potential target for non-alcoholic fatty liver disease?-perspective on emerging experimental evidence. World J. Hepatol. 2022, 14, 168–179.
- Iida, T.; Ubukata, M.; Mitani, I.; Nakagawa, Y.; Maeda, K.; Imai, H.; Ogoshi, Y.; Hotta, T.; Sakata, S.; Sano, R.; et al. Discovery of potent liver-selective stearoyl-CoA desaturase-1 (SCD1) inhibitors, thiazole-4-acetic acid derivatives, for the treatment of diabetes, hepatic steatosis, and obesity. Eur. J. Med. Chem. 2018, 158, 832–852.
- Liu, Z.; Yin, X.; Mai, H.; Li, G.; Lin, Z.; Jie, W.; Li, K.; Zhou, H.; Wei, S.; Hu, L.; et al. SCD rs41290540 single-nucleotide polymorphism modifies miR-498 binding and is associated with a decreased risk of coronary artery disease. Mol. Genet. Genom. Med. 2020, 8, e1136.
- Martín-Núñez, G.M.; Cabrera-Mulero, R.; Rojo-Martínez, G.; Gómez-Zumaquero, J.M.; Chaves, F.J.; de Marco, G.; Soriguer, F.; Castaño, L.; Morcillo, S. Polymorphisms in the SCD1 gene are associated with indices of stearoyl CoA desaturase activity and obesity: A prospective study. Mol. Nutr. Food Res. 2013, 57, 2177–2184.
- Byberg, L.; Kilander, L.; Lemming, E.W.; Michaëlsson, K.; Vessby, B. Cancer death is related to high palmitoleic acid in serum and to polymorphisms in the SCD-1 gene in healthy Swedish men. Am. J. Clin. Nutr. 2014, 99, 551–558.
- Tibori, K.; Orosz, G.; Zámbó, V.; Szelényi, P.; Sarnyai, F.; Tamási, V.; Rónai, Z.; Mátyási, J.; Tóth, B.; Csala, M.; et al. Molecular Mechanisms Underlying the Elevated Expression of a Potentially Type 2 Diabetes Mellitus Associated SCD1 Variant. Int. J. Mol. Sci. 2022, 23, 6221.
- Zámbó, V.; Orosz, G.; Szabó, L.; Tibori, K.; Sipeki, S.; Molnár, K.; Csala, M.; Kereszturi, É. A Single Nucleotide Polymorphism (rs3811792) Affecting Human SCD5 Promoter Activity Is Associated with Diabetes Mellitus. Genes 2022, 13, 1784.
- Yu, G.I.; Mun, K.H.; Yang, S.H.; Shin, D.H.; Hwang, J.S. Polymorphisms in the 3′-UTR of SCD5 gene are associated with hepatocellular carcinoma in Korean population. Mol. Biol. Rep. 2018, 45, 1705–1714.
- Song, Z.; Cao, H.; Qin, L.; Jiang, Y. A Case-Control Study between Gene Polymorphisms of Polyunsaturated Fatty Acid Metabolic Rate-Limiting Enzymes and Acute Coronary Syndrome in Chinese Han Population. BioMed Res. Int. 2013, 2013, 928178.
- Khamlaoui, W.; Mehri, S.; Hammami, S.; Hammouda, S.; Chraeif, I.; Elosua, R.; Hammami, M. Association Between Genetic Variants in FADS1-FADS2 and ELOVL2 and Obesity, Lipid Traits, and Fatty Acids in Tunisian Population. Clin. Appl. Thromb. 2020, 26.
- Sun, C.; Zou, M.; Wang, X.; Xia, W.; Ma, Y.; Liang, S.; Hao, Y.; Wu, L.; Fu, S. FADS1-FADS2 and ELOVL2 gene polymorphisms in susceptibility to autism spectrum disorders in Chinese children. BMC Psychiatry 2018, 18, 283.
- Valizadeh, M.; Aghasizadeh, M.; Nemati, M.; Hashemi, M.; Aghaee-Bakhtiari, S.H.; Zare-Feyzabadi, R.; Esmaily, H.; Ghazizdaeh, H.; Sahebi, R.; Ahangari, N.; et al. The association between a Fatty Acid Binding Protein 1 (FABP1) gene polymorphism and serum lipid abnormalities in the MASHAD cohort study. Prostaglandins Leukot. Essent. Fat. Acids 2021, 172, 102324.
- Xue, H.; Zhao, H.; Liu, X.; Zhao, Y.-R.; Chen, Z.-J.; Ma, J. Association of single-nucleotide polymorphisms rs2197076 and rs2241883 of FABP1 gene with polycystic ovary syndrome. J. Assist. Reprod. Genet. 2015, 33, 75–83.
- Mansego, M.L.; Martínez, F.; Martínez-Larrad, M.T.; Zabena, C.; Rojo, G.; Morcillo, S.; Soriguer, F.; Martín-Escudero, J.C.; Serrano-Ríos, M.; Redon, J.; et al. Common Variants of the Liver Fatty Acid Binding Protein Gene Influence the Risk of Type 2 Diabetes and Insulin Resistance in Spanish Population. PLoS ONE 2012, 7, e31853.
- Mozaffari, M.; Ghayour-Mobarhan, M.; Zare-Feyzabadi, R.; Valizadeh, M. FABP1 gene variant associated with risk of metabolic syndrome. Comb. Chem. High Throughput Screen. 2022, 25, 1355–1360.
- Peng, X.-E.; Wu, Y.-L.; Lu, Q.-Q.; Hu, Z.-J.; Lin, X. Two genetic variants in FABP1 and susceptibility to non-alcohol fatty liver disease in a Chinese population. Gene 2012, 500, 54–58.
- Fisher, E.; Li, Y.; Burwinkel, B.; Kühr, V.; Hoffmann, K.; Möhlig, M.; Spranger, J.; Pfeiffer, A.; Boeing, H.; Schrezenmeir, J.; et al. Preliminary Evidence of FABP2 A54T Polymorphism Associated with Reduced Risk of Type 2 Diabetes and Obesity in Women from a German Cohort. Horm. Metab. Res. 2006, 38, 341–345.
- Shabana; Hasnain, S. The fatty acid binding protein 2 (FABP2) polymorphism Ala54Thr and obesity in Pakistan: A population based study and a systematic meta-analysis. Gene 2015, 574, 106–111.
- Liu, Y.; Wu, G.; Han, L.; Zhao, K.; Qu, Y.; Xu, A.; Huang, Q. Association of the FABP2 Ala54Thr polymorphism with type 2 diabetes, obesity, and metabolic syndrome: A population-based case-control study and a systematic meta-analysis. Genet. Mol. Res. 2015, 14, 1155–1168.
- Peng, X.; Zhang, L.; Wang, Q.; Cui, X. Study on the relationship between FABP2 Ala54Thr polymorphism and the risk of non-alcoholic fatty liver diseases. Wei Sheng Yan Jiu 2009, 38, 401–404.
- Khattab, S.A.; Abo-Elmatty, D.; Ghattas, M.H.; Mesbah, N.; Mehanna, E.T. Intestinal fatty acid binding protein Ala54Thr polymorphism is associated with peripheral atherosclerosis combined with type 2 diabetes mellitus. J. Diabetes 2016, 9, 821–826.
- Li, Z.; Ni, C.-L.; Niu, W.-Y.; Chang, B.-C.; Chen, L.-M. The intestinal fatty acid binding protein-2 Ala54Thr polymorphism is associated with diabetic retinopathy in Chinese population. Diabetol. Metab. Syndr. 2015, 7, 23.
- Canani, L.H.; Capp, C.; Ng, D.P.; Choo, S.G.; Maia, A.L.; Nabinger, G.B.; Santos, K.; Crispim, D.; Roisemberg, I.; Krolewski, A.S.; et al. The Fatty Acid–Binding Protein-2 A54T Polymorphism Is Associated With Renal Disease in Patients With Type 2 Diabetes. Diabetes 2005, 54, 3326–3330.
- Abbas, S.; Raza, S.T.; Chandra, A.; Rizvi, S.; Ahmed, F.; Eba, A.; Mahdi, F. Association of ACE, FABP2 and GST genes polymorphism with essential hypertension risk among a North Indian population. Ann. Hum. Biol. 2014, 42, 461–469.
- Sikhayeva, N.; Iskakova, A.; Saigi-Morgui, N.; Zholdybaeva, E.; Eap, C.-B.; Ramanculov, E. Association between 28 single nucleotide polymorphisms and type 2 diabetes mellitus in the Kazakh population: A case-control study. BMC Med. Genet. 2017, 18, 76.
- An, L.; Jiang, H.; Tang, R.-N. The ACACB gene rs2268388 polymorphism is associated with nephropathy in Caucasian patients with diabetes: A meta-analysis. Ren. Fail. 2015, 37, 925–928.
- Tang, S.C.W.; Leung, V.T.M.; Chan, L.Y.Y.; Wong, S.S.H.; Chu, D.W.S.; Leung, J.C.K.; Ho, Y.W.; Lai, K.N.; Ma, L.; Elbein, S.C.; et al. The acetyl-coenzyme A carboxylase beta (ACACB) gene is associated with nephropathy in Chinese patients with type 2 diabetes. Nephrol. Dial. Transplant. 2010, 25, 3931–3934.
- Shah, V.N.; Cheema, B.S.; Sharma, R.; Khullar, M.; Kohli, H.S.; Ahluwalia, T.S.; Mohan, V.; Bhansali, A. ACACβ gene (rs2268388) and AGTR1 gene (rs5186) polymorphism and the risk of nephropathy in Asian Indian patients with type 2 diabetes. Mol. Cell. Biochem. 2012, 372, 191–198.
- Chan, G.C.W.; Zhi, H.; Hicks, P.J.; Freedman, B.I.; Tang, S.C.W. Acetyl-coenzyme A carboxylase beta gene polymorphism does not predict cardiovascular risk susceptibility in Chinese type 2 diabetic individuals. Nephrology 2022, 27, 404–409.
- Riancho, J.; Vázquez, L.; García-Pérez, M.; Sainz, J.; Olmos, J.; Hernández, J.; Pérez-López, J.; Amado, J.; Zarrabeitia, M.; Cano, A.; et al. Association of ACACB polymorphisms with obesity and diabetes. Mol. Genet. Metab. 2011, 104, 670–676.




