Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Georg Bernd Pelster | -- | 2434 | 2023-03-23 13:20:22 | | | |
| 2 | Camila Xu | Meta information modification | 2434 | 2023-03-24 03:52:25 | | | | |
| 3 | Camila Xu | Meta information modification | 2434 | 2023-03-24 03:53:23 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Pelster, B. Swimbladder Function in Yellow Eels. Encyclopedia. Available online: https://encyclopedia.pub/entry/42480 (accessed on 10 May 2026).
Pelster B. Swimbladder Function in Yellow Eels. Encyclopedia. Available at: https://encyclopedia.pub/entry/42480. Accessed May 10, 2026.
Pelster, Bernd. "Swimbladder Function in Yellow Eels" Encyclopedia, https://encyclopedia.pub/entry/42480 (accessed May 10, 2026).
Pelster, B. (2023, March 23). Swimbladder Function in Yellow Eels. In Encyclopedia. https://encyclopedia.pub/entry/42480
Pelster, Bernd. "Swimbladder Function in Yellow Eels." Encyclopedia. Web. 23 March, 2023.
Copy Citation
Eels use the swimbladder for buoyancy control. The ductus pneumaticus connecting the esophagus with the swimbladder is closed soon after initial opening of the swimbladder in the glass eel stage, so that eels are functionally physoclist. Subsequent filling of the swimbladder is achieved by activity of gas gland cells in the swimbladder epithelium and countercurrent concentration in the rete mirabile.
gas gland
countercurrent exchange
metabolism
Root effect
pentose phosphate shunt
1. Introduction
Eels are usually considered catadromous fish spawning in the marine habitat, and spending most of their life cycle in freshwater systems. It has been shown, however, that some eels may skip the freshwater phase completely and stay in coastal water, or move between brackish water and freshwater (semi-catadromous behavior) [1]. The spawning area of the European eel Anguilla anguilla (L. 1758) is the Sargasso Sea. In a recent tracking experiment, it was shown for the first time that adult eels released near the Azores swim to the Sargasso Sea [2]. Tagged eels released near the European coast so far could not be tracked all the way down to the Sargasso Sea [3][4][5][6]. Fertilized eggs develop into a larvae named Leptocephalus [7], and later it was realized that the Leptocephali are the larvae of the European eel [8]. The Leptocephali drift with the Gulf stream [9][10] and reach the European or North African continental slope after a journey of about 7 months to 2 years [10]. Before entering the European freshwater system, Leptocephali metamorphose into glass eels. The translucent Leptocephali do not have a swimbladder and appear to be positively buoyant with overall densities of 1.028–1.043 g·mL−1 [11]. This low density value appears to be due to a high concentration of glycosaminoglycans in the translucent extracellular matrix. The swimbladder develops and is first inflated in glass eels. The glass eels then develop into so-called yellow eels, which typically spend 5–25 years in the European freshwater system [9][12]. In yellow eels, the ductus pneumaticus, i.e. the connection between esophagus and swimbladder is functionally closed, eels cannot take an air breath at the water surface. Filling of the swimbladder is achieved by diffusion of gas from the blood and from gas gland cells in the swimbladder epithelium. The necessary gas partial pressure gradients are generated by activity of swimbladder gas gland cells and countercurrent multiplication in a rete mirabile.
2. Opening of the Swimbladder in Glass Eel
The swimbladder develops as a dorsal outgrowth of the esophagus in early metamorphic stages to the glass eel, Leptocephali do not have a swimbladder [13]. In many glass eels caught on arrival along the European Coast, the swimbladder does not yet contain any gas, but is filled with surfactant, which is required to reduce surface tension [13]. Its presence has also been shown in yellow eel swimbladder, and it is produced and secreted by gas gland cells [14]. In early glass eels, the epithelial gas gland cells do not show the extensive basolateral labyrinth, characteristic for gas gland cells of yellow and silver eels (Figure 1). Moreover, the intimate connection between gas gland cells and blood capillaries is not yet visible [13]. Based on the histological appearance of the gas gland cells it therefore was concluded that the initial filling of the swimbladder with gas is not achieved by secretory activity of these cells, but by gulping air or by taking up small gas bubbles from the water [13]. Within 3 to 4 months of development after the initial inflation, the connective tissue of the swimbladder and the gas gland cells differentiate to the adult state present in yellow eels with an extensive basolateral labyrinth, which is in close contact with swimbladder capillaries [13]. The ductus pneumaticus, i.e. the connection between esophagus and the swimbladder, is functionally closed so that eels cannot gulp air at the water surface. The ductus pneumaticus is used as the resorbing section of the swimbladder with separate blood supply bypassing the rete mirabile. The resorbing section of the swimbladder is separated from the secretory section via a sphincter muscle, located between the two retia mirabilia [15].

Figure 1. Histology of glass eel swimbladder. In Leptocephali developing into glass eels, the swimbladder initially is filled with surfactant; opposing gas gland cells appear to be separated by a thin layer of surfactant. Gas gland cells do not yet express an extensive basolateral labyrinth and the connection between gas gland cells and blood capillaries is not as tight as in yellow eels. gg, gas gland cell; s, surfactant; n, nucleus; pl, plasma membrane; lb, lamellar body; bm, basal membrane; lpm, lamina propria mucosae. With permission modified after [13].
3. Swimbladder Function in Yellow Eels
In the European freshwater system consisting of rivers and lakes, eels experience a limited depth range. Hydrostatic pressure increases by one atmosphere for every ten meters of water depth, so the range of hydrostatic pressures experienced in freshwater is limited. Nevertheless, the swimbladder wall is flexible, and therefore, according to Boyle’s law, in a fish descending from the water surface to a water depth of 10 m, it results in a 50% decrease in swimbladder volume due to the doubling of the hydrostatic pressure. The status of neutral buoyancy, a status in which a fish can stay at a certain water depth without any swimming movement, however, requires a constant swimbladder volume, exactly compensating for the compared to water density higher density of other fish tissues [16][17]. To avoid compression of the swimbladder when descending into deeper water therefore requires the secretion of gas in order to keep the swimbladder volume constant. Ascending closer to the water surface, in turn, requires the removal of gas to avoid an increase in swimbladder volume resulting in a status of positive buoyancy.
Although eels are anatomically physostome, the ductus pneumaticus is functionally closed soon after the original opening of the swimbladder [9][15]. The yellow eel therefore cannot gulp air at the surface and is functionally a physoclist fish. The thin-walled and almost translucent ductus pneumaticus is used as a resorbing part of the swimbladder. It is highly vascularized, draining into the main venous system. A sphincter muscle located between the two retia mirabilia allows for the transfer of gas from the secretory section to the resorbing part. Along partial pressure gradients, gases then diffuse from this resorbing section to the blood and the venous circulatory system.
The wall of the secretory swimbladder has a silvery appearance due to guanine incrustation in connective tissues, and the cell membranes are characterized by a high cholesterol content, resulting in low gas permeability of the swimbladder wall [18]. In the eel the whole swimblader epithelium consists of gas gland cells. Arterial blood supply to the secretory swimbladder passes the rete mirabile or red body, a remarkable countercurrent system. In the rete mirabile, the swimbladder artery gives rise to several tens of thousands of capillaries, running in parallel for a distance of several millimeters [19]. The capillaries reunify, forming two or three larger arterial vessels supplying the gas gland cells. Venous return to the rete again forms several tens of thousands of venous capillaries running in parallel and surrounding the arterial capillaries in the rete. The diffusion distance between arterial and venous capillaries is in the range of only one to two micrometer [19]. This arrangement of blood vessels allows taking blood samples in front of the countercurrent system, i.e., at the heart pole, but also between the rete and the gas gland cells, i.e., at the swimbladder pole. Thus, the concentrating function of the rete mirabile, and the function of gas gland cells can be analyzed separately. This explains why the eel became a model species for the analysis of swimbladder function in physoclist fish [20]. In many other species, this separation of rete mirabile and gas gland cell function is not possible, because the two structures are closely connected and rete capillaries do not give raise to larger blood vessels at the swimbladder pole. Blood samples collected at the swimbladder pole therefore may have been influenced by back-diffusion in the rete mirabile, but also by the secretory activity of the gas gland cells [21][22].
Several in situ studies and also experiments with primary cultured gas gland cells revealed that gas gland cells are specialized for the production of acidic metabolites in the anaerobic metabolism. Oxidation of metabolites in the aerobic metabolism is of minor importance, although gas gland cells are chronically exposed to high oxygen partial pressures. A large fraction of glucose taken up from the blood is converted into lactic acid [23][24][25][26]. Some of the glucose taken up from the blood, however, is also shifted to the pentose phosphate shunt, where the activity of 6-phosphogluconate dehydrogenase results in the production of CO2. In fact, CO2 production in the pentose phosphate shunt by far exceeds the amount of CO2 produced in the aerobic metabolism [25][27][28] (Figure 2). The activity of the pentose phosphate shunt also results in the generation of NADPH, an important reduction equivalent involved in the degradation of reactive oxygen species.
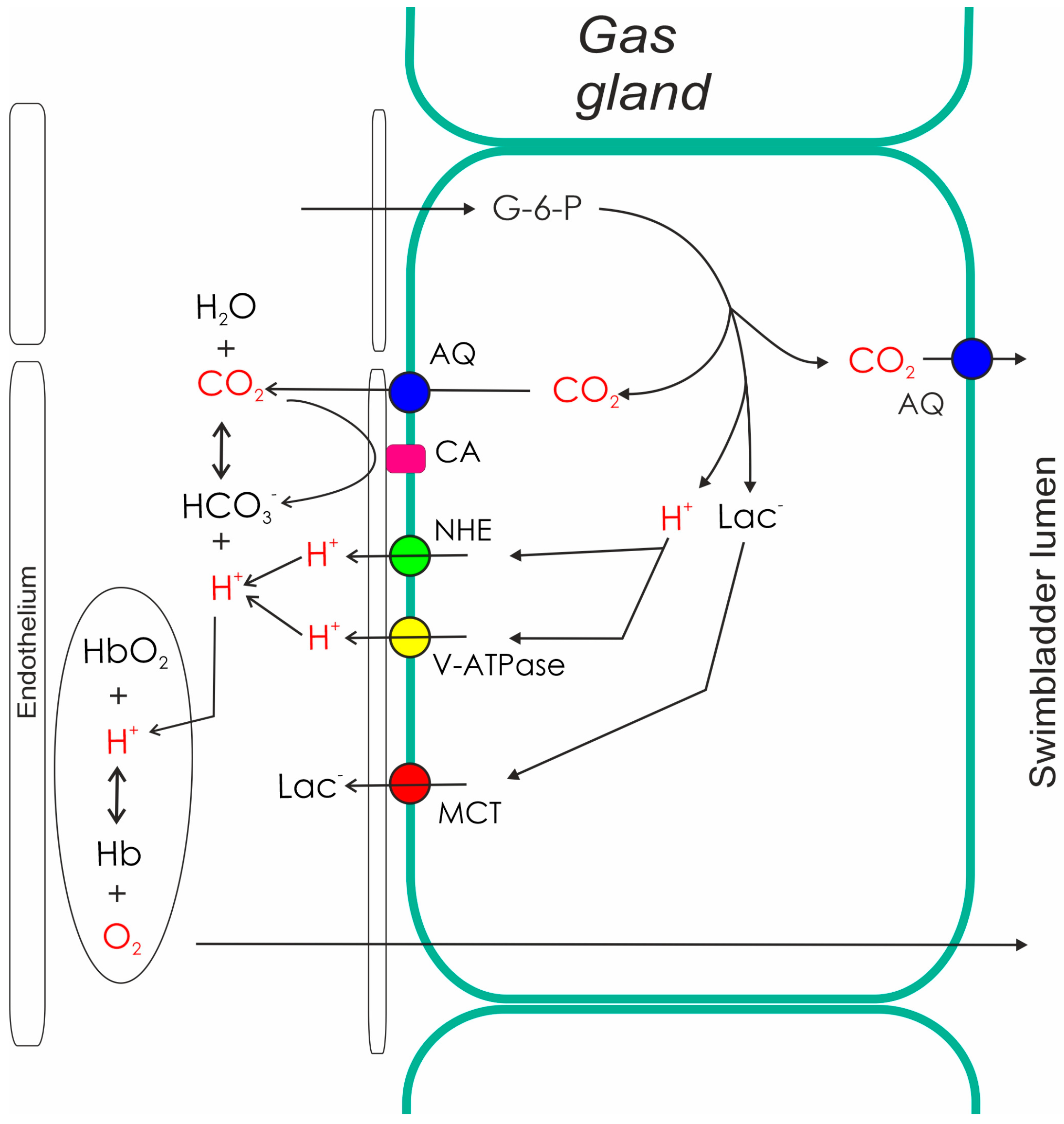
Figure 2. Metabolism and secretory activity of gas gland cells. The metabolism of gas gland cells of the European eel depends on blood glucose. Glucose is converted to lactic acid (lactate anion and a proton), and used for the production of CO2. CO2 production in the pentose phosphate shunt by far exceeds the amount of CO2 produced in the aerobic metabolism. Protons, CO2, and lactate are released into the blood, where the acidification switches on the Root effect. The resulting increase in PO2 provides the partial pressure gradient for the diffusion of oxygen into the swimbladder. The production of CO2 in gas gland cells also generates a partial pressure gradient towards the swimbladder lumen, allowing for the secretion of CO2 into the swimbladder. AQ, aquaporin; CA, carbonic anhydrase; G-6-P, glucose-6-phosphate; Hb, hemoglobin; Lac, lactate; NHE, sodium proton exchanger; MCT, monocarboxylate carrier.
Pharmacological studies indicated that gas gland cells produce and release lactate into the blood stream by a monocarboxylate carrier [29], and analysis of the transcriptome as well as of the proteome of these cells confirmed the presence of monocarboxylate carriers [30][31]. Proton secretion is achieved via sodium proton exchange proteins (NHE-proteins) and a proton ATPase (V-ATPase). V-ATPase subunits and several sodium proton exchange proteins, including NHE1 and NHE2, have been identified in the proteome and in the transcriptome [30][31][32][33]. Pharmacological studies confirmed that inhibition of NHE proteins and also inhibition of V-ATPase, both resulted in a significant decrease in acid secretion in isolated gas gland cells [29][34]. Figure 2 summarizes the activity of gas gland cells based on data obtained from the European eel.
Due to the production of CO2 in gas gland cells, the highest PCO2 is expected in these cells, providing a driving force for the diffusion of CO2 into the swimbladder lumen as well as into the blood stream (Figure 2). A membrane-bound carbonic anhydrase has been detected in gas gland cells, establishing a rapid equilibrium between CO2 and bicarbonate in swimbladder blood, and the inhibition of carbonic anhydrase activity reduced the rate of acid secretion [29][35]. In a recent study, the presence of aquaporin 1 was demonstrated in apical and basolateral membranes of gas gland cells, and also in endothelial cells of the swimbladder [18]. In a physoclist swimbladder, water movements between the gas cavity and the surrounding tissue do not make sense. Aquaporin 1 is known to be a water channel, but studies in mammalian and in fish cells revealed that it is also permeable to CO2 [36][37][38]. Therefore, it was suggested that these aquaporins act as a CO2 channel [18], facilitating the diffusion of CO2 into the swimbladder as well as into the blood.
Acidification of the blood during the passage of the gas gland cells causes a decrease in the oxygen carrying capacity of the hemoglobin, switching on the Root effect [39][40][41][42]. Root effect hemoglobins are partially deoxygenated on acidification. The acidification of swimbladder blood during passage of the gas gland cells therefore results in a significant increase in blood PO2, establishing a partial pressure gradient for the diffusion of oxygen into the swimbladder. It also assures that the PO2 in venous blood returning to the rete mirabile exceeds the PO2 of arterial blood leaving the rete.
The activity of the gas gland cells thus induces an initial increase in gas partial pressures in swimbladder capillaries and assures that the gas partial pressure in blood returning to the rete mirabile exceeds the partial pressures recorded in blood leaving the rete in arterial vessels at the swimbladder pole. The gas partial pressure gradient between venous and arterial rete vessels allows for back-diffusion of gas molecules from the venous to the arterial side. This initial increase in gas partial pressure thus in second step is multiplied by back-diffusion and countercurrent concentration in the rete mirabile [39][43]. Experiments in the European eel reveald that back-diffusion of oxygen and CO2 from venous to arterial capillaries caused a 7-fold increase in PO2 and an 8-fold increase in PCO2 during arterial passage of the rete mirabile [44].
Gas molecules diffuse through membranes, and the rete mirabile was considered to be a passive exchange system [43]. However, analysis of water and lactate movements in the rete provided the first evidence that not only oxygen and CO2, but also lactate diffuses back to the arterial side in the eel rete [45]. A detailed study of the transcriptome and the proteome of rete capillaries revealed the expression of a large number of membrane transport proteins and several membrane ATPases, including Ca2+-ATPase, V-ATPase, and Na+/K+-ATPase [46]. This suggests that ion transport and proton transport mechanisms indeed contribute to the countercurrent exchange and support the back-diffusion of lactate and protons to the arterial side of the rete in order to switch on the Root effect during the arterial passage of the rete. The data also revealed aquaporin expression in the rete. Because no water shift was detected in the rete, this suggests that aquaporin facilitates the diffusion of CO2 not only in gas gland cells but also in the rete, supporting acidification of the arterial blood in order to switch on the Root effect [46].
The main gases secreted into the swimbladder based on these mechanisms are oxygen and CO2 [44]. Due to the small increase in inert gas partial pressures (mainly nitrogen) inert gases contribute only small fraction to total gas secretion. Although the swimbladder wall has a low gas permeability due to guanine incrustation and a high content in cholesterol, it is not totally impermeable [18], and gases with a high gas solubility (like CO2) preferentially leave the swimbladder by diffusion, leaving behind the less soluble gases like nitrogen. The amount of gas secreted based on these mechanisms typically is in range of several ml per hour. In the European eel a gas secretion rate of 1-2 mL*h−1 has been recorded [44][47]. In the American eel Anguilla rostrata secretion rates of up to 3 mL*h−1 have been measured [48].
References
- Tzeng, W.N.; Wang, C.H.; Wickström, H.; Reizenstein, M. Occurrence of the semi-catadromous European eel Anguilla anguilla in the Baltic Sea. Marine Biol. 2000, 137, 93–98.
- Wright, R.; Piper, A.; Aarestrup, K.; Azevedo, J.; Cowan, G.; Don, A.; Gollock, M.; Rodriguez Ramallo, S.; Velterop, R.; Walker, A.; et al. First direct evidence of adult European eels migrating to their breeding place in the Sargasso Sea. Sci. Rep. 2022, 12, 15362.
- Righton, D.; Aarestrup, K.; Jellyman, D.; Sebert, P.; Van den Thillart, G.; Tsukamoto, K. The Anguilla spp. migration problem: 40 million years of evolution and two millennia of speculation. J. Fish Biol. 2012, 81, 365–386.
- Righton, D.; Westerberg, H.; Feunteun, E.; Okland, F.; Gargan, P.; Amilhart, E.; Metcalfe, J.; Lobon-Cervia, J.; Sjöberg, J.; Acou, A.; et al. Empirical observations of the spawning migration of European eels: The long and dangerous road to the Sargasso Sea. Sci. Adv. 2016, 2, e1501694.
- Wysujack, K.; Westerberg, H.; Aarestrup, K.; Trautner, J.; Kurwie, T.; Nagel, F.; Hanel, R. The migration behaviour of European silver eels (Anguilla anguilla) released in open ocean conditions. Mar. Freshw. Res. 2015, 66, 145–157.
- Aarestrup, K.; Okland, F.; Hansen, M.; Righton, D.; Gargan, P.; Castonguay, M.; Bernatchez, L.; Howey, P.; Sparholt, H.; Pedersen, M.; et al. Oceanic spawning migration of the european eel (Anguilla anguilla). Science 2009, 325, 1660.
- Kaup, J.J. Catalogue of Apodal Fish, in the Collection of the British Museum; British Museum: London, UK, 1856.
- Grassi, G.B.; Calandruccio, S. Fortpflanzung und Metamorphose des Aales. Allg. Fisch Zeitung. 1897, 22, 402–408.
- Tesch, F.-W. Der Aal; Paul Parey Verlag: Singhofen, Germany, 1999.
- Bonhommeau, S.; Castonguay, M.; Rivot, E.; Sabatié, R.; Le Pape, O. The duration of migration of Atlantic Anguilla larvae. Fish Fish. 2010, 11, 289–306.
- Tsukamoto, K.; Yamada, Y.; Okamura, A.; Kaneko, T.; Tanaka, H.; Miller, M.; Horie, N.; Mikawa, N.; Utoh, T.; Tanaka, S. Positive buoyancy in eel leptocephali: An adaptation for life in the ocean surface layer. Mar. Biol. 2009, 156, 835–846.
- Durif, C.M.F.; van Ginneken, V.; Dufour, S.; Müller, T.; Elie, P. Seasonal Evolution and Individual Differences in Silvering Eels from Different Locations. In Spawning Migration of the European Eel; Springer: Dordrecht, The Netherlands, 2009; pp. 13–38.
- Zwerger, P.; Nimeth, K.; Würtz, J.; Salvenmoser, W.; Pelster, B. Development of the swimbladder in the European eel (Anguilla anguilla). Cell Tissue Res. 2002, 307, 155–164.
- Prem, C.; Salvenmoser, W.; Würtz, J.; Pelster, B. Swimbladder gas gland cells produce surfactant: In vivo and in culture. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2000, 279, 2336–2343.
- Dorn, E. Über den Feinbau der Schwimmblase von Anguilla vulgaris L. Licht- und Elektronenmikroskopische Untersuchungen. Zeitschrift für Zellforsch. 1961, 55, 849–912.
- Alexander, R.M. Physical aspects of swimbladder function. Biol. Rev. 1966, 41, 141–176.
- Pelster, B. Buoyancy control in aquatic vertebrates. In Cardio-Respiratory Control in Vertebrates; Glass, M.L., Wood, S.C., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 65–98.
- Drechsel, V.; Schneebauer, G.; Fiechtner, B.; Cutler, C.P.; Pelster, B. Aquaporin expression and cholesterol content in eel swimbladder tissue. J. Fish Biol. 2022, 100, 609–618.
- Wagner, R.C.; Froehlich, R.; Hossler, F.E.; Andrews, S.B. Ultrastructure of capillaries in the red body (rete mirabile) of the eel swim bladder. Microvasc. Res. 1987, 34, 349–362.
- Pelster, B. The swimbladder. In Eel Physiology; Trischitta, F., Takei, Y., Sebert, P., Eds.; CRC Press: Enfield, FL, USA, 2013; pp. 44–67.
- Fänge, R. The mechanisms of gas transport in the euphysoclist swimbladder. Acta Physiol. Scand. Suppl. 1953, 30, 1–133.
- Jones, F.R.H.; Marshall, N.B. The structure and functions of the teleostean swimbladder. Biol. Bull. 1953, 28, 16–83.
- Steen, J.B. The physiology of the swimbladder in the eel Anguilla vulgaris. III. The mechanism of gas secretion. Acta Physiol. Scand. 1963, 59, 221–241.
- Pelster, B.; Scheid, P. Glucose Metabolism of the Swimbladder Tissue of the European Eel Anguilla anguilla. J. Exp. Biol. 1993, 185, 169–178.
- Pelster, B. Metabolism of the swimbladder tissue. Biochem. Mol. Biol. Fishes 1995, 4, 101–118.
- Drechsel, V.; Schneebauer, G.; Sandbichler, A.M.; Fiechtner, B.; Pelster, B. Oxygen consumption and acid secretion in isolated gas gland cells of the European eel Anguilla anguilla. J. Comp. Physiol. B 2022, 192, 447–457.
- Walsh, P.J.; Milligan, C.L. Roles of buffering capacity and pentose phosphate pathway activity in the gas gland of the gulf toadfish Opsanus beta. J. Exp. Biol. 1993, 176, 311–316.
- Pelster, B.; Hicks, J.; Driedzic, W.R. Contribution of the Pentose Phosphate Shunt to the formation of CO2 in swimbladder tissue of the eel. J. Exp. Biol. 1994, 197, 119–128.
- Pelster, B. Mechanisms of acid release in isolated gas gland cells of the European eel Anguilla anguilla. Am. J. Physiol. 1995, 269, R793–R799.
- Sialana, F.J.; Schneebauer, G.; Paunkov, A.; Pelster, B.; Lubec, G. Proteomic Studies on the Swim Bladder of the European Eel (Anguilla anguilla). Proteomics 2018, 18, 1700445.
- Pelster, B.; Schneebauer, G.; Dirks, R.P. Anguillicola crassus infection significantly affects the silvering related modifications in steady state mRNA levels in gas gland tissue of the European eel. Front. Physiol. 2016, 7, 175.
- Schneebauer, G.; Dirks, R.P.; Pelster, B. Anguillicola crassus infection affects mRNA expression levels in gas gland tissue of European yellow and silver eel. PLoS ONE 2017, 12, e0183128.
- Niederstätter, H.; Pelster, B. Expression of two vacuolar-type ATPase B subunit isoforms in swimbladder gas gland cells of the European eel: Nucleotide sequences and deduced amino acid sequences. Bba. Gene Struct. Express 2000, 1491, 133–142.
- Pelster, B. pH regulation and swimbladder function in fish. Respir. Physiol. Neurobiol. 2004, 144, 179–790.
- Würtz, J.; Taraschewski, H.; Pelster, B. Changes in gas composition in the swimbladder of the European eel (Anguilla anguilla) infected with Anguillicola crassus (Nematoda). Parasitology 1996, 112, 233–238.
- Chen, L.M.; Zhao, J.; Musa-Aziz, R.; Pelletier, M.F.; Drummond, I.A.; Boron, W.F. Cloning and characterization of a zebrafish homologue of human AQP1: A bifunctional water and gas channel. AJP-Regul. Integr. Comp. Physiol. 2010, 299, R1163–R1174.
- Perry, S.F.; Braun, M.H.; Noland, M.; Dawdy, J.; Walsh, P.J. Do zebrafish Rh proteins act as dual ammonia-CO2 channels? J. Exp. Zool. Part A Ecol. Genet. Physiol. 2010, 313, 618–621.
- Talbot, K.; Kwong, R.W.M.; Gilmour, K.M.; Perry, S.F. The water channel aquaporin-1a1 facilitates movement of CO2 and ammonia in zebrafish (Danio rerio) larvae. J. Exp. Biol. 2015, 218, 3931–3940.
- Pelster, B. The Generation of Hyperbaric Oxygen Tensions in Fish. Physiology 2001, 16, 287–291.
- Root, R.W. The respiratory function of the blood of marine fishes. Biol. Bull. 1931, 61, 427–456.
- Berenbrink, M.; Koldkjaer, P.; Kepp, O.; Cossins, A.R. Evolution of Oxygen Secretion in Fishes and the Emergence of a Complex Physiological System. Science 2005, 307, 1752–1757.
- Berenbrink, M.; Koldkaer, P.; Hannah Wright, E.; Kepp, O.; Jose da Silva, A. Magnitude of the Root effect in red blood cells and haemoglobin solutions of fishes: A tribute to August Krogh. Acta Physiol. 2011, 202, 583–592.
- Kuhn, W.; Ramel, A.; Kuhn, H.J.; Marti, E. The filling mechanism of the swimbladder. Generation of high gas pressures through hairpin countercurrent multiplication. Experientia 1963, 19, 497–511.
- Kobayashi, H.; Pelster, B.; Scheid, P. CO2 back-diffusion in the rete aids O2 secretion in the swimbladder of the eel. Respir. Physiol. 1990, 79, 231–242.
- Kobayashi, H.; Pelster, B.; Scheid, P. Water and lactate movement in the swimbladder of the eel, Anguilla anguilla. Respir. Physiol. 1989, 78, 45–57.
- Schneebauer, G.; Drechsel, V.; Dirks, R.; Faserl, K.; Sarg, B.; Pelster, B. Expression of transport proteins in the rete mirabile of european silver and yellow eel. BMC Genom. 2021, 22, 866.
- Pelster, B.; Scheid, P. The influence of gas gland metabolism and blood flow on gas deposition into the swimbladder of the European eel Anguilla anguilla. J. Exp. Biol. 1992, 173, 205–216.
- Kleckner, R.C. Swim bladder volume maintenance related to migratory depth in silver phase Anguilla rostrata. Science 1980, 208, 1481–1482.
More
Information
Subjects:
Fisheries
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
747
Revisions:
3 times
(View History)
Update Date:
27 Mar 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





