Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Paweł Rasmus | -- | 3416 | 2023-03-16 12:14:05 | | | |
| 2 | Peter Tang | + 2 word(s) | 3418 | 2023-03-17 03:04:44 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Rasmus, P.; Kozłowska, E. Carotenoids Antioxidant and Anti-Inflammatory Effects in Mood Disorders. Encyclopedia. Available online: https://encyclopedia.pub/entry/42264 (accessed on 30 March 2026).
Rasmus P, Kozłowska E. Carotenoids Antioxidant and Anti-Inflammatory Effects in Mood Disorders. Encyclopedia. Available at: https://encyclopedia.pub/entry/42264. Accessed March 30, 2026.
Rasmus, Paweł, Elżbieta Kozłowska. "Carotenoids Antioxidant and Anti-Inflammatory Effects in Mood Disorders" Encyclopedia, https://encyclopedia.pub/entry/42264 (accessed March 30, 2026).
Rasmus, P., & Kozłowska, E. (2023, March 16). Carotenoids Antioxidant and Anti-Inflammatory Effects in Mood Disorders. In Encyclopedia. https://encyclopedia.pub/entry/42264
Rasmus, Paweł and Elżbieta Kozłowska. "Carotenoids Antioxidant and Anti-Inflammatory Effects in Mood Disorders." Encyclopedia. Web. 16 March, 2023.
Copy Citation
Depression has a multifactorial etiology comprising family history and unemployment. Antioxidant supplementation has been found to combat various stress-induced psychiatric disorders, including depression and anxiety. A growing body of evidence indicates that carotenoids have both antioxidant and anti-inflammatory. Studies also suggest that poor dietary intake, particularly low intakes of fruit and vegetables and high intakes of fast food and other convenience foods, may increase the risk of developing depression. Thus, dietary interventions have the potential to help mitigate the risk of mental health decline in both the general population and those with mood disorders.
depression
carotenoids
mental health
nutrition
1. Introduction
Some of the most common noncommunicable illnesses are psychiatric disorders such as depression; together, these are responsible for 14.3% of global fatalities. It is estimated that 322 million individuals live with depression globally and that depression is involved in 50–70% of suicides [1]. The World Health Organization predicts depression will become the second most prevalent disorder after ischemic heart disease [2]. Depression is most commonly manifested as hopelessness, disturbances in sleep or appetite, and avoiding social activity, and it also acts as an independent predictor of the beginning of somatic illness [3]. Studies have shown that people with depression have a higher risk of developing lifestyle disorders such as cardiovascular disease, obesity, and diabetes, as well as cancer; it has also been associated with cognitive impairment [4][5][6][7][8]. However, while it is impossible to avoid some of the most common risk factors, such as family history, poverty, and unemployment [9][10], it is possible to modify others, such as a sedentary lifestyle [11], cigarette smoking [12] and alcohol consumption [13].
Depression has a multifactorial etiology comprising functional deficits in monoamine, such as serotonin, and disturbances in the brain’s hypothalamic–pituitary–adrenal (HPA) axis [14]. In addition, emotional disorders such as anxiety and depression are often associated with a reduction in the activity of antioxidant enzymes in the HPA axis. HPA axis hyperactivity is often observed in cases of depression; this correlates with an increase in glucocorticoid concentration, resulting in the development of oxidative stress and glutamatergic excitotoxicity and thus the death of nerve cells, especially in the hippocampus, the key region related to the regulation of mood [15][16][17][18][19][20][21]. Also, chronic psychological stress, known to contribute to the development of depression, causes structural changes in the hippocampus associated with the regulation of cognition [22].
It should be emphasized that although there are many theories of depression development, its accurate pathomechanism is still not fully known. Depression is widely believed to be strongly influenced by immuno-inflammatory mechanisms [23][24][25]. Patients with depression tend to demonstrate significantly elevated levels of various chemokines (CCL2, CXCL10), and pro-inflammatory cytokines, such as interleukin (IL)−1β, IL-6, and tumor necrosis factor (TNF) [26][27][28][29][30]. Some studies found patients with depression to have increased levels of C reactive protein (CRP), an important inflammatory marker [31][32]. Current literature indicates a strong two-way association between the development of inflammation and psychiatric disorders, including depression. It should also be borne in mind that depression is related to an exacerbation of behaviors associated with the development of inflammation, such as nutritional deficiencies/poor eating habits, and addiction to psychoactive substances, such as alcohol, drugs, or smoking [33]. In addition, there is evidence that commensal gut microorganisms, which comprise the gut microbiota, may play an important role in the etiopathogenesis of depression via the gut-brain axis [34]. The gut-brain axis is a two-way communication pathway between the central nervous system and the gut. This communication occurs via hormonal, neurological, and immunological signaling systems, as well as gut microbe metabolites, which trigger changes in neurotransmission, neuroinflammation, and behavior [35][36]. Disturbances in the composition, quality, and functioning of the intestinal microbiota (intestinal dysbiosis) are correlated with some neuropsychiatric disorders, especially depression. Numerous studies have investigated the potential impact of gut microbiota on the onset of depression [37][38][39][40][41][42][43][44][45].
2. The Influence of Diet on the Development and Course of Mood Disorders
A number of studies indicate that an improperly-balanced diet is one of the elements associated with the development of depression and anxiety [46][47]. Recent years have seen a growth in interest in the relationship between nutrients and depression, particularly folic acid, vitamin D, and magnesium [48][49][50]. Mikkelsen et al. (2016) demonstrated a relationship between vitamin B deficiency, including B1, B3, B6, B9, B12, and depression [51][52]. Studies have also shown that a higher level of depression in adolescents is associated with irregular meals [53]. Research indicates that fat-soluble nutrients, such as vitamin E, protect against nerve damage, and low dietary intake is linked to mood changes and depression [54].
It is believed that carotenoid-cleaving enzymes, which take part in the metabolism of carotenoids, play a key role in depression. It should be pointed out that apocarotenoids are formed due to the oxidative breakdown of carotenoids catalyzed by carotenoid oxygenases. Apocarotenoids are, inter alia, retinal, retinol, retinoic acid, and abscisic acid. Some studies describe that retinoic acid, the active form of vitamin A, causes hyperactivity of the hypothalamic-pituitary-adrenal (HPA) axis and leads to the development of typical depressive behaviors. In addition, it has been shown that retinoic acid may cause suicide in some susceptible individuals [55][56].
A cross-sectional study of people of normal BMI by Nguyen et al. (2017) found significantly lower intake of β-carotene equivalent and vitamins C, E, B1, B3, B6, B9, and B5 in those with depressive symptoms [54]. It should be noted that vitamin C insufficiency has also been linked to an increased risk of depression symptoms [54]. Additionally, randomized, placebo-controlled clinical trials have found vitamin C to improve mood and lower the severity of depression in patients [57][58]. Interestingly, however, vitamin C supplementation appeared to have no effect on the depression score in these people [59]. A study conducted among elderly Japanese people showed that the consumption of carotene and vitamin C is associated with less severe depression symptoms [60]. Lower carotenoid concentrations may also reflect unhealthy eating patterns associated with overweight and obesity, which have been linked to an increased risk of depression by inflammation or dysregulation of the HPA axis [5][61][62].
Previous studies have shown that some dietary factors, such as fruit and vegetables, fish, dietary fiber, and some macro- and microelements, may play an important, protective role in the development of depression through their antioxidant and/or anti-inflammatory properties [63][64][65][66]. It is important to note that carotenoids are also known for their antioxidant activity and anti-inflammatory properties [67][68]. Additionally, research shows that depression leads to the development of diseases such as cardiovascular diseases, insulin resistance, metabolic syndrome, and obesity [69][70][71]. These data support the hypothesis that inflammation and oxidative stress may be involved in the pathophysiology of this disorder. Considering that carotenoids have both antioxidant and anti-inflammatory effects, it is expected that they may exert an antidepressant effect.
3. The Role of Stress in Unipolar Mood Disorder Pathology
The results of animal studies indicate that psychological stress can increase the level of lipid peroxidation, a significant source of the cell damage caused by reactive oxygen species (ROS) [72], and impair antioxidant protection in the plasma [73][74]. Due to its high oxygen consumption and relatively weak antioxidant defense, the brain is particularly susceptible to oxidative damage, which may increase the likelihood of developing depressive episodes. Therefore, oxidative stress, caused by an imbalance between antioxidants and prooxidants, may play a key role in the remission and chronic course of depressive disorder [75][76].
Patients with depression have been found to have a significantly lower mean intake of α-carotene compared to healthy subjects [77]. In addition, depression has been associated with lowered antioxidant levels, as evidenced by low levels of carotenoids and antioxidant enzymes [76][78][79][80]. Black et al. (2016) found reduced levels of carotenoids such as zeaxanthin/lutein, β-cryptoxanthin, lycopene, α-carotene, and β-carotene to be associated with an increase in depression symptoms. Most importantly, this relationship persisted after controlling for diet quality; as carotenoids are only acquired through the dietary route, diet could be considered a significant confounder [3]. Beydoun et al. (2013) report that among the studied carotenoids, β-carotene, lutein, and zeaxanthin levels were inversely related to the incidence of depressive symptoms among US adults [80]. Interestingly, these studies suggest that common genetic factors may influence the relationship between low carotenoid levels and depression: the presence of SNPs associated with low β-cryptoxanthin levels may also influence the occurrence of depression [81].
In turn, Tsubi et al. [72] and Nouri et al. (2020) [82] found no correlation between the serum level of lycopene and depressive symptoms. Zhang et al. (2016) report that seven days of pretreatment with 60 mg/kg lycopene could reverse LPS-induced depressive behavior in mice based on the tail suspension test and the forced swim test [83]. In turn, a mechanistic study by Lin et al. (2014) found that three-day treatment with 10 mg/kg lycopene reverses the LPS-induced increase in serum TNF and IL-6 concentrations and IL-1β levels in the hippocampus [84]; in addition, pretreatment with 5, 10, or 20 M lycopene inhibited LPS-induced production of cyclooxygenase-2, inducible nitric oxide synthase, and IL-6 in primary cultured microglia via the activation of heme oxygenase-1 [84]. There is a possibility that lycopene supplementation may help maintain cellular homeostasis by restoring normal cell cytokines levels turn. These results suggest that inhibiting neuroinflammation may be a key factor in the antidepressant effects of lycopene.
4. Oxidative Stress and Antioxidants in the Course of Mood Disorders
Oxidative stress occurs as a result of an imbalance between the build-up of reactive oxygen species (ROS) or reactive nitrogen species (RNS) and their removal. ROS levels are believed to increase due to various environmental features such as tobacco smoke, ionizing, UV radiation, and by the initiation of cell receptors [85]. At least 5% of inhaled oxygen is converted to ROS, which naturally occurs as a byproduct of aerobic metabolism. In metabolic processes, cytochrome oxidase completely reduces most of the molecular oxygen to water in the mitochondria. Only partially reduced oxygen can react with long-chain molecules such as proteins, carbohydrates, lipids, and DNA. In higher organisms, RNS are produced by the oxidation of one of the terminal guanidonitrogen atoms of L-arginine [86] by nitric oxide synthase. NO can then be converted to various other forms of RNS [87].
The human body has a range of antioxidant defense mechanisms in place to protect against the potentially damaging effects of such active species., for example, by removing free radicals from the body. It is now known that oxidative stress, as well as ROS and RNS, negatively affect a number of cellular processes. When ROS exposure (or generation) increases or antioxidant levels fall, lipids, proteins, and DNA can be damaged, resulting in cell malfunction and even cell death [88]. Importantly, ROS participate in a number of physiological reactions of the body, such as the phagocytosis process [89]. Most importantly, the brain is particularly susceptible to oxidative stress because the level of aerobic respiration is high in the brain tissue. Additionally, brain tissue is rich in polyunsaturated fatty acids (PUFAs) that are susceptible to ROS damage [90].
A growing body of data indicates that ROS may also play an essential role in the pathophysiology of various neurological and psychiatric disorders, including mood disorders. Numerous studies have shown that individuals with neuropsychiatric disorders have higher levels of free radicals, lipid peroxides, pro-apoptotic markers, and altered antioxidant defense mechanisms [91][92][93]. A meta-analysis by Black et al. (2014) found oxidative stress to be elevated in people with MDD and/or depressive symptoms [94]. Importantly, oxidative stress is linked to various socio-demographic, health, and lifestyle variables, including socioeconomic status and smoking, which are also linked to depression [5][88][95][96][97][98][99]. Cigarette smoke has been demonstrated to decrease the levels of carotenoids and other antioxidants in human plasma [100]; it has been proposed that smoking may reduce carotenoid concentration by increasing metabolic rate, resulting in greater oxidative stress [101]. Today it is well known that antioxidants defend against the harmful effects of oxidative stress, which is believed to be associated with depression [102][103].
The antioxidant system consists of enzymatic antioxidants such as inter alia glutathione reductase, SOD, and catalase, as well as non-enzymatic forms such as vitamin C and E, N-acetylcysteine, reduced glutathione, flavonoids, and carotenoids. Carotenoids are natural antioxidants that can effectively prevent oxidative damage [67]. There is evidence that antioxidants exert a neuroprotective effect through their ability to repair the central nervous system and prevent oxidative stress-induced neurodegeneration.
The total antioxidant capacity of a diet has been shown to have an inverse relationship with depression, anxiety, and stress [104][105]. Some data suggest that people with depression consume lower levels of antioxidants in the form of fruit and vegetables compared to those without [106]. In addition, data suggest that patients with depression have lower plasma vitamin E and C levels than those without [107][108]. Vitamin E has been found to exert an antidepressant-like effect in depressed animal models, and this has been attributed to it supporting the enzymatic glutathione-based antioxidant defense system in the hippocampus and prefrontal cortex [109]. However, growing evidence suggests that antioxidant treatment has proven unsatisfactory and even damaging in some oxidation-related diseases such as cancer [110][111][112]. While it is known that ROS plays a key role in defense against pathogens and intracellular signaling, the perception is that these compounds are harmful to cells. Likewise, antioxidants should not be regarded as purely beneficial agents. A clinical example of this is the finding that β-carotene supplementation in smokers leads to a significant increase in the incidence of lung cancer [113][114].
5. Carotenoids and Their Role in the Course of Depression
Carotenoids are fat-soluble color pigments that belong to the tetraterpene family, present in yellow-orange vegetables and fruits [115]. More than 700 carotenoids have been described, with the major forms being lycopene, β-carotene, ASTA, lutein, and zeaxanthin [116][117][118]. In nature, these pigments are found in many bacteria, fungi, and plants. The groups of carotenoids can also be divided into non-provitamin A and provitamin A (e.g., γ-carotene, β-carotene, α-carotene, and β-cryptoxanthin) [119]. The compounds can also be classified by the presence of specific functional groups: xanthophylls containing oxygen as a functional group (e.g., lutein, zeaxanthin), and carotenes containing only the parent hydrocarbon chain without any functional group (e.g., α-carotene, β-carotene, and lycopene) [119] (Figure 1).
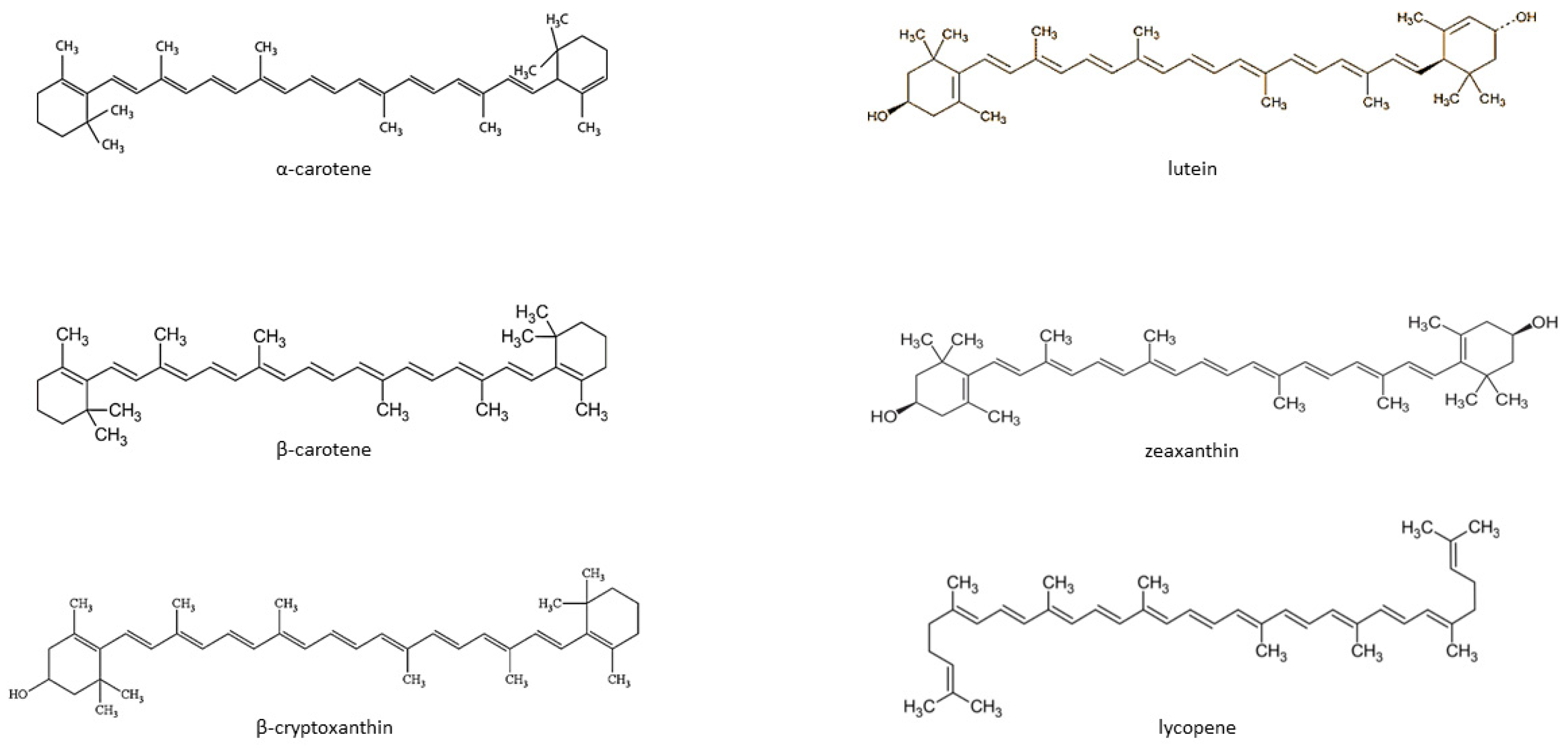
Carotenoids cannot be synthesized de novo by humans and can only be acquired through the dietary route. While around 700 carotenoids have been identified, only six are commonly found in the human diet and blood serum: α-carotene, β-carotene, lutein, zeaxanthin, lycopene, and β–cryptoxanthin. In addition, the typical human diet only includes about 40 carotenoids. Many studies show that providing the body with dietary carotenoids is associated with a reduced risk of developing lifestyle diseases, such as cancer, osteoporosis, diabetes, or cataracts, as well as certain infectious diseases, such as HIV infection [115][119][126]. The data also indicate that carotenoids may reduce the risk of developing CVD by lowering blood pressure and inflammatory markers and increasing insulin sensitivity in muscles, the liver, and adipose tissue. Interestingly, carotenoids could modulate the expression of specific genes involved in cell metabolism [127].
Carotenoids have many beneficial effects. They are mainly known for their antioxidant properties as major scavengers of ROS, including single molecular oxygen and peroxide radicals [119]. Recent epidemiological studies show that higher blood α-carotene and lycopene levels are linked to a lower risk of lung cancer, even among smokers [128]. Interestingly, an ever-increasing body of literature indicates that carotenoids may be effective in the treatment of a variety of cancers, e.g., neuroblastoma [129], cervical cancer [130], and prostate cancer [131]. A large number of existing in vitro and in vivo studies have revealed that carotenoids influence a variety of processes related to the body’s immune-inflammatory response. It has been demonstrated that these compounds influence both the cellular (lymphocyte proliferation, phagocytosis, and NK cell cytotoxicity) and humoral mechanisms of immunity (synthesis and secretion of cytokines) [132][133]. It was found that β-carotene can inhibit the upregulation of heme oxygenase 1 expression in human skin fibroblasts (FEK4) exposed to UV-A [115][134]. In turn, β-carotene has been shown to be less effective in preventing lipid peroxidation [134].
There is a growing body of evidence that the antioxidant and anti-inflammatory properties of carotenoids may promote efficient cognitive function [135][136][137][138][139] by increasing neuronal efficiency or stabilizing the lipid-protein bonds in neuronal membranes. Other neuroprotective mechanisms of carotenoids include enhancement of communication between clefts and modulation of the functional properties of synaptic membranes [138][139][140]. It should be stressed that some studies indicate that higher intake of β-carotene may be related to lower prevalence of depression, anxiety, and stress [90]. Epidemiological studies investigating the relationship between diet, carotenoids, and cognitive maintenance have reported that low levels of carotenoids may play a role in cognitive impairment [141][142]. Prohan et al. (2014) found depressed university male students to have a lower β-carotene intake compared to controls [143][144][145]. Increased β-carotene intake may also relieve depression and anxiety symptoms in cases of low blood antioxidant levels. Antioxidant supplementation has been found to resist stress-induced psychiatric disorders such as depression and anxiety [144].
One particularly potent antioxidant among the carotenoids is lycopene, which can trap singlet oxygen and reduce mutagenesis. Some authors have also suggested that lycopene effectively reduces smoke-generated ROS and modulates redox-sensitive target cells [135]. Research on neurodegenerative and psychiatric disorders also suggests that lycopene may have neuroprotective effects on the central nervous system (CNS) [145][146][147][148][149][150]. It has been shown that long-term intake of lycopene reduces the risk of stroke in men and reduces neuronal apoptosis in the case of cerebral ischemia [149][150][151].
Unlike other carotenoids, xanthophylls such as lutein, astaxanthin, and zeaxanthin are orientated within cell membranes by free hydroxyl groups at each end [152]. Lutein is present in high amounts in green plants and leaves such as spinach, kale, and broccoli. It is also the predominant carotenoid in the primate brain: it was present at almost 10–20 times higher levels in the occipital cortex, prefrontal cortex, and cerebellum than its isomer, zeaxanthin [153]. Zeaxanthin and lutein play a number of roles in both plants and humans, including photoprotection and the maintenance of the structural and functional integrity of biological membranes [154]. Numerous studies have shown that zeaxanthin and lutein exhibit antioxidant properties, thereby protecting cells from potential free radical damage [155]. Lutein, zeaxanthin, and other carotenoids can enter the brain from the blood and accumulate in the retinal macula [156][157]. Lutein is known to accumulate in all cortexes and membranes of the brain.
Serum concentrations of β-carotene, β -cryptoxanthin, and α-carotene can be used as biomarkers to predict the concentration of carotenoids in the brain [135][156][157][158]. It has been documented that a low serum lutein level was associated with depression in Alzheimer’s disease patients [78][159]. Previous research has shown that lutein reduces very low and medium-density lipoprotein levels, as well as inflammation and oxidative stress; it also inhibits the progression of atherosclerosis in humans by lowering serum IL-10 concentration [160][161]. Zeaxanthin can effectively scavenge water- and lipid-soluble peroxide radicals.
It should be stressed that carotenoids, as antioxidants, play an important role in counterbalancing the age-related rise in oxidative stress. It has also been shown that with age, the central nervous system becomes increasingly vulnerable to the impact of free radicals; indeed, seniors are much more vulnerable to protein and lipid damage caused by free radicals, resulting in impaired mitochondrial activity and increased free radical production [161]. The inability to counter oxidative stress may result from progressive neuronal deficits but also from neurodegenerative diseases [162][163][164]. Depression has also been found to cause structural and functional changes in some areas of the brain, especially around the hippocampus [165].
Taking the above into account, carotenoids have the potential to play a protective role in depression through various mechanisms. First, pro-inflammatory cytokines such as IL-6 and TNF impair the expression of BDNF, leading to the onset of depression [166]. Additionally, it should be noted that some studies have shown that patients with depression have lower serum levels of BDNF [167][168]. Interestingly, it has been shown that β-carotene and zeaxanthin may reduce the mRNA expression of IL-6 and TNF [169][170]. What is important, carotenoids have been tested for mechanisms in very important drug targets such as MAO or BDNF, known to be closely related to depression through molecular docking studies for possible inhibitory activity. Recent BDNF and carotenoid docking results conducted by Park et al. (2021) indicate the possibility of allosteric activation of BDNF by carotenoids [171]. On this basis, the authors suggested that dietary carotenoids may be used in the treatment of depressive symptoms. Secondly, this organ is prone to oxidative stress due to high oxygen consumption and high levels of lipids in the brain. At the same time, it is believed that the development of depression is closely related to oxidation and an imbalance between pro- and antioxidants. Studies have shown that people with depression have elevated levels of 8-hydroxy-2’-deoxyguanosine, which is considered a marker of oxidative DNA damage [172]. These results indicate that depression appears to be closely related to oxidative stress. Today it is known that carotenoids can effectively remove reactive oxygen species as well as other free radicals. Therefore, considering that carotenoids have both antioxidant and anti-inflammatory effects, it is expected that they may exert an antidepressant effect.
References
- Bachmann, S. Epidemiology of suicide and the psychiatric perspective. Int. J. Environ. Res. Public Health 2018, 6, 1425.
- Reddy, M.S. Depression: The disorder and the burden. Indian J. Psychol. Med. 2010, 32, 1–2.
- Black, C.N.; Penninx, B.W.; Bot, M.; Odegaard, A.O.; Gross, M.D.; Matthews, K.A.; Jacobs, D.R., Jr. Oxidative stress, anti-oxidants and the cross-sectional and longitudinal association with depressive symptoms: Results from the CARDIA study. Transl. Psychiatry 2016, 23, 6, e743.
- Nicholson, A.; Kuper, H.; Hemingway, H. Depression as an aetiologic and prognostic factor in coronary heart disease: A meta-analysis of 6362 events among 146,538 participants in 54 observational studies. Eur. Heart J. 2006, 27, 2763–2774.
- Luppino, F.S.; de Wit, L.M.; Bouvy, P.F.; Stijnen, T.; Cuijpers, P.; Penninx, B.W.J.H.; Zitman, F.G. Overweight, obesity, and depression: A systematic review and meta-analysis of longitudinal studies. Arch. Gen. Psychiatry 2010, 67, 220–229.
- Mezuk, B.; Eaton, W.W.; Albrecht, S.; Golden, S.H. Depression and type 2 diabetes over the lifespan: A meta-analysis. Diabetes Care 2008, 31, 2383–2390.
- Chida, Y.; Hamer, M.; Wardle, J.; Steptoe, A. Do stress-related psychosocial factors contribute to cancer incidence and survival? Nat. Clin. Pract. Oncol. 2008, 5, 466–475.
- Barnes, D.E.; Alexopoulos, G.S.; Lopez, O.L.; Williamson, J.D.; Yaffe, K. Depressive 5-3 the cardiovascular health study. Arch. Gen. Psychiatry. 2006, 63, 273–279.
- Sullivan, P.F.; Neale, M.C.; Kendler, K.S. Genetic epidemiology of major depression: Review and meta-analysis. Am. J. Psychiatry 2000, 157, 1552–1562.
- Tozzi, F.; Prokopenko, I.; Perry, J.D.; Kennedy, J.L.; McCarthy, A.D.; Holsboer, F.; Berrettini, W.; Middleton, L.T.; Chilcoat, H.D.; Muglia, P. Family history of depression is associated with younger age of onset in patients with recurrent depression. Psychol. Med. 2008, 38, 641–649.
- Zhai, L.; Zhang, Y.; Zhang, D. Sedentary behaviour and the risk of depression: A meta-analysis. Br. J. Sports Med. 2015, 49, 705–709.
- Boden, J.M.; Fergusson, D.M.; Horwood, L.J. Cigarette smoking and depression: Tests of causal linkages using a longitudinal birth cohort. Br. J. Psychiatry. 2010, 196, 440–446.
- Paljarvi, T.; Koskenvuo, M.; Poikolainen, K.; Kauhanen, J.; Sillanmaki, L.; Makela, P. Binge drinking and depressive symptoms: A 5-year population-based cohort study. Addiction 2009, 104, 1168–1178.
- Kumar, P.V.N.; Elango, P.; Asmathulla, S.; Kavimani, S. Lycopene treatment transposed antidepressant-like action in rats provoked to chronic mild stress. Biomed. Pharmacol. J. 2019, 12, 981–988.
- Lee, A.L.; Ogle, W.O.; Sapolsky, R.M. Stress and depression: Possible links to neuron death in the hippocampus. Bipolar Disord. 2002, 4, 117–128.
- Freitas, A.E.; Egea, J.; Buendía, I.; Navarro, E.; Rada, P.; Cuadrado, A.; Rodrigues, A.L.S.; López, M.G. Agmatine induces Nrf2 and protects against corticosterone effects in hippocampal neuronal cell line. Mol. Neurobiol. 2015, 51, 1504–1519.
- Myers, B.; McKlveen, J.M.; Herman, J.P. Glucocorticoid actions on synapses, circuits, and behavior: Implications for the energetics of stress. Front. Neuroendocrinol. 2014, 35, 180–196.
- Spiers, J.G.; Chen, H.J.C.; Sernia, C.; Lavidis, N.A. Activation of the hypothalamicpituitary- adrenal stress axis induces cellular oxidative stress. Front. Neurosci. 2015, 9, 456.
- Stanić, D.; Plećaš-Solarović, B.; Petrović, J.; Bogavac-Stanojević, N.; Sopić, M.; Kotur-Stevuljević, J.; Ignjatović, S.; Pešić, V. Hydrogen peroxide-induced oxidative damage in peripheral blood lymphocytes from rats chronically treated with corticosterone: The protective effect of oxytocin treatment. Chem. Biol. Interact. 2016, 256, 134–141.
- Lau, A.; Tymianski, M. Glutamate receptors, neurotoxicity and neurodegeneration. Pflügers Arch. Eur. J. Physiol. 2010, 460, 525–542.
- Freitas, A.E.; Egea, J.; Buendia, I.; Gómez-Rangel, V.; Parada, E.; Navarro, E.; Casas, A.I.; Wojnicz, A.; Ortiz, J.A.; Cuadrado, A.; et al. Agmatine, by improving neuroplasticity markers and inducing Nrf2, prevents corticosterone-induced depressive-like behavior in mice. Mol. Neurobiol. 2016, 53, 3030–3045.
- Czéh, B.; Lucassen, P.J. What causes the hippocampal volume decrease in depression? Are neurogenesis, glial changes and apoptosis implicated? Eur. Arch. Psychiatry Clin. Neurosci. 2007, 257, 250–260.
- Hong, H.; Kim, B.S.; Im, H.-I. Pathophysiological role of neuroinflammation in neurodegenerative diseases and psychiatric disorders. Int. Neurourol. J. 2016, 20, S2.
- Müller, N. The role of anti-inflammatory treatment in psychiatric disorders. Psychiatr. Danub. 2013, 25, 292–298.
- Najjar, S.; Pearlman, D.M.; Alper, K.; Najjar, A.; Devinsky, O. Neuroinflammation and psychiatric illness. J. Neuroinflammation 2013, 10, 43.
- Eyre, H.A.; Air, T.; Pradhan, A. A meta-analysis of chemokines in major depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 68, 1–8.
- Dowlati, Y.; Herrmann, N.; Swardfager, W.; Liu, H.; Sham, L.; Reim, E.K. A metaanalysis of cytokines in major depression. Biol. Psychiatry 2010, 67, 446–457.
- Raison, C.L.; Capuron, L.; Miller, A.H. Cytokines sing the blues: Inflammation and the pathogenesis of depression. Trends Immunol. 2006, 27, 24–31.
- Verduijn, J.; Milaneschi, Y.; Schoevers, R.A.; van Hemert, A.M.; Beekman, A.T. Pathophysiology of major depressive disorder: Mechanisms involved in etiology are not associated with clinical progression. Transl. Psychiatry 2015, 5, e649.
- Wu, W.; Zheng, Y.L.; Tian, L.P.; Lai, J.B.; Hu, C.C.; Zhang, P.; Chen, J.K.; Hu, J.B.; Huang, M.L.; Wei, N. Circulating T lymphocyte subsets, cytokines, and immune checkpoint inhibitors in patients with bipolar II or major depression: A preliminary study. Sci. Rep. 2017, 7, 40530.
- Wium-Andersen, M.K.; Ørsted, D.D.; Nordestgaard, B.G. Elevated C-reactive protein, depression, somatic diseases, and all-cause mortality: A mendelian randomization study. Biol. Psychiatry 2014, 76, 249–257.
- Wium-Andersen, M.K.; Ørsted, D.D.; Nordestgaard, B.G. Elevated C-reactive protein associated with late- and very-late-onset schizophrenia in the general population: A prospective study. Schizophr. Bull. 2014, 40, 1117–1127.
- Mac Giollabhui, N.; Ng, T.H.; Ellman, L.M.; Alloy, L.B. The longitudinal associations of inflammatory biomarkers and depression revisited: Systematic review, meta-analysis, and meta-regression. Mol. Psychiatry 2020, 26, 3302–3314.
- Radjabzadeh, D.; Bosch, J.A.; Uitterlinden, A.G.; Zwinderman, A.H.; Ikram, M.A.; van Meurs, J.B.J.; Luik, A.I.; Nieuwdorp, M.; Lok, A.; van Duijn, C.M.; et al. Gut microbiome-wide association study of depressive symptoms. Nat. Commun. 2022, 13, 7128.
- Chen, L.; Liu, B.; Ren, L.; Du, H.; Fei, C.; Qian, C.; Li, B.; Zhang, R.; Liu, H.; Li, Z.; et al. High-fiber diet ameliorates gut microbiota, serum metabolism and emotional mood in type 2 diabetes patients. Front. Cell. Infect. Microbiol. 2023, 13, 1069954.
- Carlessi, A.S.; Borba, L.A.; Zugno, A.I.; Quevedo, J.; Réus, G.Z. Gut microbiota-brain axis in depression: The role of neuroinflammation. Eur. J. Neurosci. 2021, 53, 222–235.
- Cruz-Pereira, J.S.; Rea, K.; Nolan, Y.M.; O’Leary, O.F.; Dinan, T.G.; Cryan, J.F. Depression’s unholy trinity: Dysregulated stress, immunity, and the microbiome. Annu. Rev. Psychol. 2020, 71, 49–78.
- Yang, Z.; Li, J.; Gui, X.; Shi, X.; Bao, Z.; Han, H.; Li, M.D. Updated review of research on the gut microbiota and their relation to depression in animals and human beings. Mol. Psychiatry 2020, 25, 2759–2772.
- Leclercq, S.; Le Roy, T.; Furgiuele, S.; Coste, V.; Bindels, L.B.; Leyrolle, Q.; Neyrinck, A.M.; Quoilin, C.; Amadieu, C.; Petit, G.; et al. Gut microbiota-induced changes in β-hydroxybutyrate metabolism are linked to altered sociability and depression in alcohol use disorder. Cell. Rep. 2020, 33, 108238.
- Bharwani, A.; West, C.; Champagne-Jorgensen, K.; McVey Neufeld, K.A.; Ruberto, J.; Kunze, W.A.; Bienenstock, J.; Forsythe, P. The vagus nerve is necessary for the rapid and widespread neuronal activation in the brain following oral administration of psychoactive bacteria. Neuropharmacology 2020, 170, 108067.
- Abildgaard, A.; Kern, T.; Pedersen, O.; Hansen, T.; Lund, S.; Wegener, G. A diet-induced gut microbiota component and related plasma metabolites are associated with depressive-like behaviour in rats. Eur. Neuropsychopharmacol. 2021, 43, 10–21.
- Zhuang, Z.; Yang, R.; Wang, W.; Qi, L.; Huang, T. Associations between gut microbiota and Alzheimer’s disease, major depressive disorder, and schizophrenia. J. Neuroinflamm. 2020, 17, 88.
- Chen, M.; Xie, C.R.; Shi, Y.Z.; Tang, T.C.; Zheng, H. Gut microbiota and major depressive disorder: A bidirectional Mendelian randomization. J. Affect. Disord. 2022, 316, 187–193.
- McGuinness, A.J.; Davis, J.A.; Dawson, S.L.; Loughman, A.; Collier, F.; O’Hely, M.; Simpson, C.A.; Green, J.; Marx, W.; Hair, C.; et al. A systematic review of gut microbiota composition in observational studies of major depressive disorder, bipolar disorder and schizophrenia. Mol. Psychiatry 2022, 27, 1920–1935.
- Winter, G.; Hart, R.A.; Charlesworth, R.P.G.; Sharpley, C.F. Gut microbiome and depression: What we know and what we need to know. Rev. Neurosci. 2018, 29, 629–643.
- Bodnar, L.; Wisner, K. Nutrition and depression: Implications for improving mental health among childbearing-aged women. Biol. Psychiatry 2005, 58, 679–685.
- Murphy, J.; Wehler, C.; Pagano, M.; Little, M.; Kleinman, R.; Jellinek, M. Relationship between hunger and psychosocial functioning in lowincome American children. J. Am. Acad. Child Adolesc. Psychiatry 1998, 37, 163–170.
- Petridou, E.T.; Kousoulis, A.A.; Michelakos, T.; Papathoma, P.; Dessypris, N.; Papadopoulos, F.C.; Stefanadis, C. Folate and B12 serum levels in association with depression in the aged: A systematic review and meta-analysis. Aging Ment. 2016, 20, 965–973.
- Li, B.; Lv, J.; Wang, W.; Zhang, D. Dietary magnesium and calcium intake and risk of depression in the general population: A meta-analysis. Aust. N. Z. J. Psychiatry. 2017, 51, 219–229.
- Anglin, R.E.; Samaan, Z.; Walter, S.D.; McDonald, S.D. Vitamin D deficiency and depression in adults: Systematic review and meta-analysis. Br. J. Psychiatry 2013, 202, 100–107.
- Mikkelsen, K.; Stojanovska, L.; Apostolopoulos, V. The effects of vitamin B in depression. Curr. Med. Chem. 2016, 23, 4317–4337.
- Mikkelsen, K.; Stojanovska, L.; Prakash, M.; Apostolopoulos, V. The effects of vitamin B on the immune/cytokine network and their involvement in depression. Maturitas 2017, 96, 58–71.
- Fulkerson, J.A.; Sherwood, N.; Perry, C.; Neumark-Sztainer, D.; Story, M. Depressive symptoms and adolescent eating and health behaviors: A multifaceted view in a population-based sample. Prev. Med. 2004, 38, 865–875.
- Nguyen, T.T.T.; Tsujiguchi, H.; Kambayashi, Y.; Hara, A.; Miyagi, S.; Yamada, Y.; Nakamura, H.; Shimizu, Y.; Hori, D.; Suzuki, F.; et al. Relationship between vitamin intake and depressive symptoms in elderly Japanese individuals: Differences with gender and body mass index. Nutrients 2017, 3, 1319.
- Bremner, J.D.; Shearer, K.D.; McCaffery, P.J. Retinoic acid and affective disorders: The evidence for an association. J. Clin. Psychiatry 2012, 73, 37–50.
- Ludot, M.; Mouchabac, S.; Ferreri, F. Inter-relationships between isotretinoin treatment and psychiatric disorders: Depression, bipolar disorder, anxiety, psychosis and suicide risks. World J. Psychiatry 2015, 5, 222–227.
- Brody, S. High-dose ascorbic acid increases intercourse frequency and improves mood: A randomized controlled clinical trial. Biol. Psychiatry 2002, 52, 371–374.
- Khajehnasiri, F.; Mortazavi, S.B.; Allameh, A.; Akhondzadeh, S. Effect of omega-3 and ascorbic acid on inflammation markers in depressed shift workers in Shahid Tondgoyan Oil Refinery, Iran: A randomized double-blind placebo-controlled study. J. Clin. Biochem. Nutr. 2013, 53, 36–40.
- Mazloom, Z.; Ekramzadeh, M.; Hejazi, N. Efficacy of supplementary vitamins c and e on anxiety, depression and stress in type 2 diabetic patients: A randomized, single-blind, placebo-controlled trial. Pak. J. Biol. Sci. 2013, 16, 1597–1600.
- Oishi, J.; Doi, H.; Kawakami, N. Nutrition and depressive symptoms in community-dwelling elderly persons in Japan. Acta Med. Okayama 2009, 63, 9–17.
- Vogelzangs, N.; Kritchevsky, S.B.; Beekman, A.T.; Newman, A.B.; Satterfield, S.; Simonsick, E.M.; Yaffe, K.; Harris, T.B.; Penninx, B.W. Depressive symptoms and change in abdominal obesity in older persons. Arch. Gen. Psychiatry 2008, 65, 1386–1389.
- Miller, G.E.; Freedland, K.E.; Carney, R.M.; Stetler, C.A.; Banks, W.A. Pathways linking depression, adiposity, and inflammatory markers in healthy young adults. Brain Behav. Immun. 2003, 17, 276–285.
- Li, F.; Liu, X.; Zhang, D. Fish consumption and risk of depression: A meta-analysis. J. Epidemiol. Community Health 2016, 70, 299–304.
- Liu, X.; Yan, Y.; Li, F.; Zhang, D. Fruit and vegetable consumption and the risk of depression: A meta-analysis. Nutrition 2016, 32, 296–302.
- Xu, H.; Li, S.; Song, X.; Li, Z.; Zhang, D. Exploration of the association between dietary fiber intake and depressive symptoms in adults. Nutrition 2018, 54, 48–53.
- Sun, C.; Wang, R.; Li, Z.; Zhang, D. Dietary magnesium intake and risk of depression. J. Affect. Disord. 2019, 246, 627–632.
- Fiedor, J.; Burda, K. Potential role of carotenoids as antioxidants in human health and disease. Nutrients 2014, 6, 466–488.
- Rubin, L.P.; Ross, A.C.; Stephensen, C.B.; Bohn, T.; Tanumihardjo, S.A. Metabolic effects of inflammation on vitamin A and carotenoids in humans and animal models. Adv. Nutr. 2017, 8, 197–212.
- Ali, S.; Stone, M.A.; Peters, J.L.; Davies, M.J.; Khunti, K. The prevalence of comorbid depression in adults with Type 2 diabetes: A systematic review and meta-analysis. Diabet. Med. 2006, 23, 1165–1173.
- González, H.M.; Tarraf, W. Comorbid cardiovascular disease and major depression among ethnic and racial groups in the United States. Int. Psychogeriatr. 2013, 25, 833–841.
- Euesden, J.; Danese, A.; Lewis, C.M.; Maughan, B. A bidirectional relationship between depression and the autoimmune disorders—New perspectives from the National Child Development Study. PLoS ONE 2017, 12, 0173015.
- Tsuboi, H.; Shimoi, K.; Kinae, N.; Oguni, I.; Hori, R.; Kobayashi, F. Depressive symptoms are independently correlated with lipid peroxidation in a female population: Comparison with vitamins and carotenoids. J. Psychosom. Res. 2004, 56, 53–58.
- Moller, P.; Wallin, H.; Knudsen, L.E. Oxidative stress associated with exercise, psychological stress and life-style factors. Chem. Biol. Interact. 1996, 102, 17–36.
- Cernak, I.; Savic, V.; Kotur, J.; Prokic, V.; Kuljic, B.; Grbovic, D.; Veljovic, M. Alterations in magnesium and oxidative status during chronic emotional stress. Magnes. Res. 2000, 13, 29–36.
- Moylan, S.; Maes, M.; Wray, N.R.; Berk, M. The neuroprogressive nature of major depressive disorder: Pathways to disease evolution and resistance, and therapeutic implications. Mol. Psychiatry 2013, 18, 595–606.
- Sarandol, A.; Sarandol, E.; Eker, S.S.; Erdinc, S.; Vatansever, E.; Kirli, S. Major depressive disorder is accompanied with oxidative stress: Short-term antidepressant treatment does not alter oxidative-antioxidative systems. Hum. Psychopharmacol. 2007, 22, 67–73.
- Park, J.Y.; You, J.S.; Chang, K.J. Dietary taurine intake, nutrients intake, dietary habits and life stress by depression in Korean female college students: A case-control study. J. Biomed. Sci. 2010, 17, 40.
- Milaneschi, Y.; Bandinelli, S.; Penninx, B.W.; Corsi, A.M.; Lauretani, F.; Vazzana, R.; Semba, R.D.; Guralnik, J.M.; Ferrucci, L. The relationship between plasma carotenoids and depressive symptoms in older persons. World J. Biol. Psychiatr. 2012, 13, 588–598.
- Palta, P.; Samuel, L.J.; Miller, E.R.; Szanton, S.L. Depression and oxidative stress: Results from a meta-analysis of observational studies. Psychosom. Med. 2014, 76, 12–19.
- Beydoun, M.A.; Beydoun, H.A.; Boueiz, A.; Shroff, M.R.; Zonderman, A.B. Antioxidant status and its association with elevated depressive symptoms among US adults: National Health and Nutrition Examination Surveys 2005–6. Br. J. Nutr. 2013, 109, 1714–1729.
- Beydoun, M.A.; Nalls, M.A.; Canas, J.A.; Evans, M.K.; Zonderman, A.B. Gene polymorphisms and gene scores linked to low serum carotenoid status and their associations with metabolic disturbance and depressive symptoms in African–American adults. Br. J. Nutr. 2014, 112, 992–1003.
- Nouri, M.; Nasr-Esfahani, M.H.; Tarrahi, M.J.; Amani, R. The effect of lycopene supplementation on mood status and quality of life in infertile men: A randomized, double-blind, placebo-controlled clinical trial. Int. J. Fertil. Steril. 2020, 14, 17–22.
- Zhang, F.; Fu, Y.; Zhou, X.; Pan, W.; Shi, Y.; Wang, M.; Zhang, X.; Qi, D.; Li, L.; Ma, K. Depression-like behaviors and heme oxygenase-1 are regulated by Lycopene in lipopolysaccharide-induced neuroinflammation. J. Neuroimmunol. 2016, 15, 298.
- Lin, H.Y.; Huang, B.R.; Yeh, W.L.; Lee, C.H.; Huang, S.S.; Lai, C.H.; Lin, H.; Lu, D.Y. Antineuroinflammatory effects of lycopene via activation of adenosine monophosphate-activated protein kinase-α1/heme oxygenase-1 pathways. Neurobiol. Aging 2014, 35, 191–202.
- Felicity, N.; Michael, B.; Olivia, D.; Ashley, I.B. Oxidative stress in psychiatric disorders: Evidence base and therapeutic implications. Int. J. Neuropsychopharmacol. 2008, 11, 851–876.
- Ferret, P.J.; Soum, E.; Negre, O.; Wollman, E.E.; Fradelizi, D. Protective effect of thioredoxin upon NO- mediated cell injury in THP1 monocytic human cells. Biochem. J. 2000, 346, 759–765.
- Hughes, M.N. Relationships between nitric oxide, nitroxyl ion, nitrosonium cation and peroxynitrite. Biochim. Biophys. Acta 1999, 1411, 263–272.
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.D.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84.
- Uribe-Querol, E.; Rosales, C. Phagocytosis: Our current understanding of a universal biological process. Front. Immunol. 2020, 11, 1066.
- Farhadnejad, H.; Neshatbini Tehrani, A.; Salehpour, A.; Hekmatdoost, A. Antioxidant vitamin intakes and risk of depression, anxiety and stress among female adolescents. Clin. Nutr. ESPEN 2020, 40, 257–262.
- Boskovic, M.; Vovk, T.; Kores Plesnicar, B.; Grabnar, I. Oxidative stress in schizophrenia. Curr. Neuropharmacol. 2011, 9, 301–312.
- Casademont, J.; Garrabou, G.; Miro, O.; Lopez, S.; Pons, A.; Bernardo, M.; Cardellach, F. Neuroleptic treatment effect on mitochondrial electron transport chain: Peripheral blood mononuclear cells analysis in psychotic patients. J. Clin. Psychopharmacol. 2007, 27, 284–288.
- Rezin, G.T.; Amboni, G.; Zugno, A.I.; Quevedo, J.; Streck, E.L. Mitochondrial dysfunction and psychiatric disorders. Neurochem. Res. 2009, 34, 1021–1029.
- Black, C.N.; Bot, M.; Scheffer, P.G.; Cuijpers, P.; Penninx, B.W.J.H. Is depression associated with increased oxidative stress? A systematic review and meta-analysis. Psychoneuroendocrinology 2014, 51C, 164–175.
- Dalle-Donne, I.; Rossi, R.; Colombo, R.; Giustarini, D.; Milzani, A. Biomarkers of oxidative damage in human disease. Clin. Chem. 2006, 52, 601–623.
- Janicki-Deverts, D.; Cohen, S.; Matthews, K.A.; Gross, M.D.; Jacobs, D.R.J. Socioeconomic status, antioxidant micronutrients, and correlates of oxidative damage: The Coronary Artery Risk Development in Young Adults (CARDIA) study. Psychosom. Med. 2009, 71, 541–548.
- Glassman, A.H.; Helzer, J.E.; Covey, L.S.; Cottler, L.B.; Stetner, F.; Tipp, J.E.; Johnson, J. Smoking, smoking cessation, and major depression. JAMA 1990, 264, 1546–1549.
- Abu-Omar, K.; Rutten, A.; Lehtinen, V. Mental health and physical activity in the European Union. Soz. Praventivmed. 2004, 49, 301–309.
- Sullivan, L.E.; Fiellin, D.A.; O’Connor, P.G. The prevalence and impact of alcohol problems in major depression: A systematic review. Am. J. Med. 2005, 118, 330–341.
- Handelman, G.J.; Packer, L.; Cross, C.E. Destruction of tocopherols, carotenoids, and retinol in human plasma by cigarette smoke. Am. J. Clin. Nutr. 1996, 63, 559–565.
- Alberg, A. The influence of cigarette smoking on circulating concentrations of antioxidant micronutrients. Toxicology 2002, 180, 121–137.
- Kodydková, J.; Vávrová, L.; Zeman, M.; Jirák, R.; Macásek, J.; Stanková, B.; Tvrzická, E.; Zák, A. Antioxidative enzymes and increased oxidative stress in depressive women. Clin. Biochem. 2009, 42, 1368–1374.
- Maes, M.; Mihaylova, I.; Kubera, M.; Uytterhoeven, M.; Vrydags, N.; Bosmans, E. Increased 8-hydroxy-deoxyguanosine, a marker of oxidative damage to DNA, in major depression and myalgic encephalomyelitis / chronic fatigue syndrome. Neuro. Endocrinol. Lett. 2009, 30, 15–22.
- Abshirini, M.; Siassi, F.; Koohdani, F.; Qorbani, M.; Mozaffari, H.; Aslani, Z.; Soleymani, M.; Entezarian, M.; Sotoudeh, G. Dietary total antioxidant capacity is inversely associated with depression, anxiety and some oxidative stress biomarkers in postmenopausal women: A cross-sectional study. Ann. Gen. Psychiatr. 2019, 18, 3.
- Daneshzad, E.; Keshavarz, S.-A.; Qorbani, M.; Larijani, B.; Azadbakht, L. Dietary total antioxidant capacity and its association with sleep, stress, anxiety, and depression score: A cross-sectional study among diabetic women. Clin. Nutr. ESPEN 2020, 37, 187–194.
- Payne, M.E.; Steck, S.E.; George, R.R.; Steffens, D.C. Fruit, vegetable, and antioxidant intakes are lower in older adults with depression. J. Acad. Nutr. Diet. 2012, 112, 2022–2027.
- Khanzode, S.D.; Dakhale, G.N.; Khanzode, S.S.; Saoji, A.; Palasodkar, R. Oxidative damage and major depression: The potential antioxidant action of selective serotonin reuptake inhibitors. Redox Rep. 2003, 8, 365–370.
- Owen, A.; Batterham, M.; Probst, Y.; Grenyer, B.; Tapsell, L.C. Low plasma vitamin E levels in major depression: Diet or disease? Eur. J. Clin. Nutr. 2005, 59, 304.
- Lobato, K.R.; Cardoso, C.C.; Binfare, R.W.; Budni, J.; Wagner, C.L.; Brocardo, P.S.; de Souza, L.F.; Brocardo, C.; Flesch, S.; Freitas, A.E.; et al. a-Tocopherol administration produces an antidepressant-like effect in predictive animal models of depression. Behav. Brain Res. 2010, 209, 249–259.
- Bjelakovic, G.; Nikolova, D.; Gluud, L.L.; Simonetti, R.G.; Gluud, C. Antioxidant supplements for prevention of mortality in healthy participants and patients with various diseases. Cochrane Database Syst. Rev. 2012, 3, CD007176.
- Bjelakovic, G.; Nikolova, D.; Simonetti, R.G.; Gluud, C. Antioxidant supplements for preventing gastrointestinal cancers. Cochrane Database Syst. Rev. 2008, 3, CD004183.
- Cortés-Jofré, M.; Rueda, J.-R.; Corsini-Muñoz, G.; Fonseca-Cortés, C.; Caraballoso, M.; Bonfill Cosp, X. Drugs for preventing lung cancer in healthy people. Cochrane Database Syst Rev. 2012, 10, CD002141.
- Satia, J.A.; Littman, A.; Slatore, C.G.; Galanko, J.A.; White, E. Long-term use of betacarotene, retinol, lycopene, and lutein supplements and lung cancer risk: Results from the VITamins and Lifestyle (VITAL) study. Am. J. Epidemiol. 2009, 169, 815–828.
- Rao, A.V.; Rao, L.G. Carotenoids and human health. Pharmacol. Res. 2007, 55, 207–216.
- Jomova, K.; Valko, M. Health protective effects of carotenoids and their interactions with other biological antioxidants. Eur. J. Med. Chem. 2013, 70, 102–110.
- Rutz, J.K.; Borges, C.D.; Zambiazi, R.C.; da Rosa, C.G.; da Silva, M.M. Elaboration of microparticles of carotenoids from natural and syn- thetic sources for applications in food. Food Chem. 2016, 202, 324–333.
- Tapiero, H.; Townsend, D.M.; Tew, K.D. The role of carotenoids in the prevention of human pathologies. Biomed. Pharmacother. 2004, 58, 100–110.
- Stahl, W.; Sies, H. Bioactivity and protective effects of natural carotenoids. Biochim. Biophys. Acta 2005, 1740, 101–107.
- Saini, R.K.; Nile, S.H.; Park, W. Carotenoids from fruits and vegetables: Chemistry, analysis, occurrence, bioavailability and biological activities. Food Res Int. 2015, 76, 735–750.
- National Center for Biotechnology Information. “PubChem Compound Summary for CID 5281235, Beta-Cryptoxanthin” PubChem. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/beta-Cryptoxanthin (accessed on 26 January 2022).
- National Center for Biotechnology Information. “PubChem Compound Summary for CID 5281243, Lutein” PubChem. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Lutein (accessed on 26 January 2022).
- National Center for Biotechnology Information. “PubChem Compound Summary for CID 5280899, Zeaxanthin” PubChem. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Zeaxanthin (accessed on 26 January 2022).
- National Center for Biotechnology Information. “PubChem Compound Summary for CID 5280489, Beta-Carotene” PubChem. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/beta-Carotene (accessed on 26 January 2022).
- National Center for Biotechnology Information. “PubChem Compound Summary for CID 6419725, Alpha-Carotene” PubChem. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/alpha-Carotene (accessed on 26 January 2022).
- National Center for Biotechnology Information. “PubChem Compound Summary for CID 446925, Lycopene” PubChem. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Lycopene (accessed on 26 January 2022).
- Pechinskii, S.V.; Kuregyan, A.G. Molecular-biological problems of drug design and mechanism of drug action. Pharmaceut. Chem. J. 2014, 47, 509–513.
- Gammone, M.A.; Riccioni, G.; D’Orazio, N. Carotenoids: Potential allies of cardiovascular health? Food Nutr. Res. 2015, 59, 26762.
- Heber, D.; Lu, Q.Y. Overview of mechanisms of action of lycopene. Exp. Biol. Med. 2002, 227, 920–923.
- Kim, Y.S.; Lee, H.A.; Lim, J.Y.; Kim, Y.; Jung, C.H.; Yoo, S.H.; Kim, Y. β-Carotene inhibits neuroblastoma cell invasion and metastasis in vitro and in vivo by decreasing level of hypoxia-inducible factor-1α. J. Nutr. Biochem. 2014, 25, 655–664.
- Guo, L.; Zhu, H.; Lin, C.; Che, J.; Tian, X.; Han, S.; Zhao, H.; Zhu, Y.; Mao, D. Associations between antioxidant vitamins and the risk of invasive cervical cancer in chinese women: A case–control study. Sci. Rep. 2015, 5, 13607.
- Karppi, J.; Kurl, S.; Laukkanen, J.A.; Kauhanen, J. Serum betacarotene in relation to risk of prostate cancer: The kuopio ischaemic heart disease risk factor study. Nutr. Cancer 2012, 64, 361–367.
- Park, J.S.; Chyun, J.H.; Kim, Y.K.; Line, L.L.; Chew, B.P. Astaxanthin decreased oxidative stress and inflammation and enhanced immune response in humans. Nutr. Metab. 2010, 7, 18.
- Seyedzadeh, M.H.; Safari, Z.; Zare, A.; Gholizadeh Navashenaq, J.; Razavi, S.A.; Kardar, G.A. Study of curcumin immunomodulatory effects on reactive astrocyte cell function. Int. Immunopharmacol. 2014, 22, 230–235.
- El-Agamey, A.; Lowe, G.M.; McGarvey, D.J.; Mortensen, A.; Phillip, D.M.; Truscott, T.G.; Young, A.J. Carotenoid radical chemistry and antioxidant/pro-oxidant properties. Arch. Biochem. Biophys. 2004, 430, 37–48.
- Kaulmann, A.; Bohn, T. Carotenoids, inflammation, and oxidative stress—Implications of cellular signaling pathways and relation to chronic disease prevention. Nutr. Res. 2014, 34, 907–929.
- Honarvar, N.M.; Saedisomeolia, A.; Abdolahi, M.; Shayeganrad, A.; Sangsari, G.T.; Rad, B.H.; Muench, G. Molecular anti-inflammatory mechanisms of retinoids and carotenoids in Alzheimer’s disease: A review of current evidence. J. Mol. Neurosci. 2017, 61, 289–304.
- Sorrenti, V.; Davinelli, S.; Scapagnini, G.; Willcox, B.J.; Allsopp, R.C.; Willcox, D.C. Astaxanthin as a putative geroprotector: Molecular basis and focus on brain aging. Mar. Drugs 2020, 18, 351.
- Sujak, A.; Gabrielska, J.; Grudzinski, W.; Borc, R.; Mazurek, P.; Gruszecki, W.I. Lutein and zeaxanthin as protectors of lipid membranes against oxidative damage: The structural aspects. Arch. Biochem. Biophys. 1999, 371, 301–307.
- Gruszecki, W. Carotenoid orientation: Role in membrane stabilization. In Carotenoids in Health and Disease, 1st ed.; Norman, I.K., Susan, T.M., Helmut, S., Eds.; CRC Press: Boca Raton, FL, USA, 2004; pp. 170–183.
- Stahl, W.; Sies, H. Effects of carotenoids and retinoids on gap junctional communication. BioFactors 2001, 15, 95–98.
- Akbaraly, N.T.; Faure, H.; Gourlet, V.; Favier, A.; Berr, C. Plasma carotenoid levels and cognitive performance in an elderly population: Results of the EVA study. J. Gerontol. Ser. A 2007, 62, 308–316.
- Christensen, K.; Gleason, C.E.; Mares, J.A. Dietary carotenoids and cognitive function among US adults, NHANES 2011–2014. Nutr. Neurosci. 2020, 23, 554–562.
- Prohan, M.; Amani, R.; Nematpour, S.; Jomehzadeh, N.; Haghighizadeh, M.H. Total antioxidant capacity of diet and serum, dietary antioxidant vitamins intake, and serum hs-CRP levels in relation to depression scales in university male students. Redox Rep. 2014, 19, 133–139.
- Gautam, M.; Agrawal, M.; Gautam, M.; Sharma, P.; Gautam, A.S.; Gautam, S. Role of antioxidants in generalised anxiety disorder and depression. Indian J. Psychiatr. 2012, 54, 244.
- Yu, L.; Wang, W.; Pang, W.; Xiao, Z.; Jiang, Y.; Hong, Y. Dietary lycopene supplementation improves cognitive performances in Tau transgenic mice expressing P301L mutation via inhibiting oxidative stress and Tau hyperphosphorylation. J. Alzheimers Dis. 2017, 57, 475–482.
- Qu, M.; Li, L.; Chen, C.; Li, M.; Pei, L.; Chu, F.; Yang, J.; Yu, Z.; Wang, D.; Zhou, Z. Protective effects of lycopene against amyloid β-induced neurotoxicity in cultured rat cortical neurons. Neurosci. Lett. 2011, 21, 286–290.
- Sandhir, R.; Mehrotra, A.; Kamboj, S.S. Lycopene prevents 3-nitropropionic acid-induced mitochondrial oxidative stress and dysfunctions in nervous system. Neurochem. Int. 2010, 57, 579–587.
- Prema, A.; Janakiraman, U.; Manivasagam, T.; Thenmozhi, A.J. Neuroprotective effect of lycopene against MPTP induced experimental Parkinson’s disease in mice. Neurosci. Lett. 2015, 10, 599.
- Lei, X.; Lei, L.; Zhang, Z.; Cheng, Y. Neuroprotective effects of lycopene pretreatment on transient global cerebral ischemia reperfusion in rats: The role of the Nrf2/HO-1 signaling pathway. Mol. Med. Rep. 2016, 13, 412–418.
- Fujita, K.; Yoshimoto, N.; Kato, T.; Imada, H.; Matsumoto, G.; Inakuma, T.; Nagata, Y.; Miyachi, E. Lycopene inhibits ischemia/reperfusion-induced neuronal apoptosis in gerbil hippocampal tissue. Neurochem. Res. 2013, 38, 461–469.
- Karppi, J.; Laukkanen, J.A.; Sivenius, J.; Ronkainen, K.; Kurl, S. Serum lycopene decreases the risk of stroke in men: A population-based follow-up study. Neurology 2012, 9, 1540–1547.
- Roberts, R.L.; Green, J.; Lewis, B. Lutein and zeaxanthin in eye and skin health. Clin. Dermatol. 2009, 27, 195–201.
- Vishwanathan, R.; Neuringer, M.; Snodderly, D.M.; Schalch, W.; Johnson, E.J. Macular lutein and zeaxanthin are related to brain lutein and zeaxanthin in primates. Nutr. Neurosci. 2013, 16, 21–29.
- Demmig-Adams, B.; López-Pozo, M.; Stewart, J.J.; Adams, W.W. Zeaxanthin and Lutein: Photoprotectors, anti-inflammatories, and brain food. Molecules 2020, 25, 3607.
- Krinsky, N.I.; Landrum, J.T.; Bone, R.A. Biologic mechanisms of the protective role of lutein and zeaxanthin in the eye. Annu. Rev. Nutr. 2003, 23, 171–201.
- Johnson, E.J.; Vishwanathan, R.; Scott, T.M.; Schalch, W.; Wittwer, J.; Hausman, D.B.; Davey, A.; Johnson, M.A.; Green, R.C.; Gearing, M.; et al. Serum carotenoids as a biomarker for carotenoid concentrations in the brain. FASEB J. 2011, 25, 3442.
- Johnson, E.J.; Vishwanathan, R.; Johnson, M.A.; Hausman, D.B.; Davey, A.; Scott, T.M.; Joffe, S.; Miller, L.S.; Gearing, M.; Woodard, J.; et al. Relationship between serum and brain carotenoids, -tocopherol, and retinol concentrations and cognitive performance in the oldest old from the Georgia Centenarian Study. J. Aging Res. 2013, 2013, 951786.
- Tanprasertsuk, M.J.; Mohn, P.E.S.; Matthan, P.N.R.; Lichtenstein, D.A.H.; Barger, P.K.; Vishwanathan, P.R.; Johnson, M.A.; Poon, P.L.W.; Johnson, E.J. Serum carotenoids, tocopherols, total n-3 polyunsaturated fatty acids, and n-6/n-3 polyunsaturated fatty acid ratio reflect brain concentrations in a cohort of centenarians. J. Gerontol. Ser. A 2019, 74, 306–314.
- Wang, M.; Jiao, J.; Li, Z.; Liu, R.; Shi, Q.; Ma, L. Lutein supplementation reduces plasma lipid peroxidation and C-reactive protein in healthy nonsmokers. Atherosclerosis 2013, 227, 380–385.
- Dwyer, J.H.; Navab, M.; Dwyer, K.M.; Hassan, K.; Sun, P.; Shircore, A.; Hama-Levy, S.; Hough, G.; Wang, X.; Drake, T.; et al. Oxygenated carotenoid lutein and progression of early atherosclerosis: The Los Angeles atherosclerosis study. Circulation 2001, 103, 2922–2927.
- Kim, H. Inhibitory mechanism of lycopene on cytokine expression in experimental pancreatitis. Ann. NY Acad. Sci. 2011, 1229, 99–102.
- Balaban, R.S.; Nemoto, S.; Finkel, T. Mitochondria, oxidants, and aging. Cell 2005, 120, 483–495.
- Beal, M.F. Mitochondria take center stage in aging and neurodegeneration. Ann. Neurol. 2005, 58, 495–505.
- Patten, D.A.; Germain, M.; Kelly, M.A.; Slack, R.S. Reactive oxygen species: Stuck in the middle of neurodegeneration. J. Alzheimers Dis. 2010, 20, 357–367.
- Lorenzetti, V.; Allen, N.B.; Fornito, A.; Yücel, M. Structural brain abnormalities in major depressive disorder: A selective review of recent MRI studies. J. Affect. Disord. 2009, 117, 1–17.
- Videbech, P.; Ravnkilde, B. Hippocampal volume and depression: A meta-analysis of MRI studies. Am. J. Psychiatry 2004, 161, 1957–1966.
- Kim, N.-R.; Kim, H.-Y.; Kim, M.-H.; Kim, H.-M.; Jeong, H.-J. Improvement of depressive behavior by Sweetme Sweet Pumpkin™ and its active compound, β- carotene. Life Sci. 2016, 147, 39–45.
- Bocchio-Chiavetto, L.; Bagnardi, V.; Zanardini, R.; Molteni, R.; Gabriela Nielsen, M.; Placentino, A.; Giovannini, C.; Rillosi, L.; Ventriglia, M.; Riva, M.A.; et al. Serum and plasma BDNF levels in major depression: A replication study and meta-analyses. World J. Biol. Psychiatry 2010, 11, 763–773.
- Molendijk, M.L.; Bus, B.A.; Spinhoven, P.; Penninx, B.W.; Kenis, G.; Prickaerts, J.; Oude Voshaar, R.C.; Elzinga, B.M. Serum levels of brain-derived neurotrophic factor in major depressive disorder: State-trait issues, clinical features and pharmacological treatment. Mol. Psychiatry 2011, 16, 1088–1095.
- Zhou, X.; Gan, T.; Fang, G.; Wang, S.; Mao, Y.; Ying, C. Zeaxanthin improved diabetes-induced anxiety and depression through inhibiting inflammation in hippocampus. Metab. Brain Dis. 2018, 33, 705–711.
- Park, S.J.; Jaiswal, V.; Lee, H.J. Dietary intake of flavonoids and carotenoids is associated with anti-depressive symptoms: Epidemiological study and in silico-mechanism analysis. Antioxidants 2021, 11, 53.
- Forlenza, M.J.; Miller, G.E. Increased serum levels of 8-hydroxy-2’-deoxyguanosine in clinical depression. Psychosom. Med. 2006, 68, 1–7.
More
Information
Subjects:
Biochemistry & Molecular Biology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.2K
Entry Collection:
Neurodegeneration
Revisions:
2 times
(View History)
Update Date:
17 Mar 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





