Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Gianluca Gaidano | -- | 2326 | 2023-02-03 10:18:33 | | | |
| 2 | Rita Xu | Meta information modification | 2326 | 2023-02-03 10:44:05 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Mouhssine, S.; Gaidano, G. Richter Syndrome. Encyclopedia. Available online: https://encyclopedia.pub/entry/40805 (accessed on 08 May 2026).
Mouhssine S, Gaidano G. Richter Syndrome. Encyclopedia. Available at: https://encyclopedia.pub/entry/40805. Accessed May 08, 2026.
Mouhssine, Samir, Gianluca Gaidano. "Richter Syndrome" Encyclopedia, https://encyclopedia.pub/entry/40805 (accessed May 08, 2026).
Mouhssine, S., & Gaidano, G. (2023, February 03). Richter Syndrome. In Encyclopedia. https://encyclopedia.pub/entry/40805
Mouhssine, Samir and Gianluca Gaidano. "Richter Syndrome." Encyclopedia. Web. 03 February, 2023.
Copy Citation
Richter syndrome (RS) represents the occurrence of an aggressive lymphoma, most commonly diffuse large B-cell lymphoma (DLBCL), in patients with chronic lymphocytic leukemia (CLL).
Richter syndrome
chronic lymphocytic lymphoma
pathogenesis
genetic lesions
1. Definition of Richter Syndrome
Richter Syndrome (RS) was reported for the first time by Maurice N. Richter as “reticular cell sarcoma” in 1928 [1]. Currently, according to the World Health Organization (WHO) classification of Tumours of Haematopoietic and Lymphoid Tissues, RS is defined as the occurrence of an aggressive lymphoma in patients with a previous or concomitant diagnosis of chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) [2]. RS is currently divided into two recognized pathological variants: diffuse large B-cell lymphoma (DLBCL) variant, with confluent sheets of large neoplastic post-germinal center B lymphocytes, and Hodgkin lymphoma (HL) variant [2][3].
The neoplastic cells of DLBCL-type RS express CD20, and less commonly CD5 and CD23 [2]. PD-1 expression is documented in up to 80% of DLBCL-type RS, whereas in de novo DLBCL, this marker is poorly expressed. Another difference between DLBCL-type RS and de novo DLBCL is the low rate of BCL2 genetic lesions, compared with the prevalence of BCL2 translocations and somatic mutations that are commonly found in de novo DLBCL [4][5][6]. The analysis of immunoglobulin genes has shown that ~80% of the cases of DLBCL-type RS are clonally related to the CLL phase, thus documenting that this histologic shift is a true transformation event from the previous indolent phase. However, a minority (~20%) of DLBCL-type RS cases are characterized by a rearrangement of immunoglobulin genes that is distinct from that of the CLL phase, documenting a clonally unrelated origin of RS [3].
The HL variant is similar to its de novo counterpart, with the presence of Hodgkin and Reed–Sternberg cells in a typical background of reactive T cells, epithelioid histiocytes, eosinophils and plasma cells or, eventually, interspersed in a background of CLL cells [7][8][9]. Hodgkin and Reed–Sternberg cells are characterized by a CD30+/CD15+/CD20− immunophenotype and are often found to be EBV positive [3][9].
2. Epidemiology
CLL is the most frequent leukemia in adults, with an incidence of 4.7/100,000 per year in the US [10]. In the chemo-immunotherapy (CIT) era, data mainly based on retrospective studies showed an incidence of RS transformation ranging from 1 to 7% [11]. Consistently, the CALGB 9011 clinical trial demonstrated a RS transformation rate of ~7% in treatment-naïve CLL patients after at least 15 years after treatment with fludarabine or chlorambucil [12]. Remarkably, in the CLL8 trial evaluating the effect of fludarabine and cyclophosphamide with or without the anti-CD20 monoclonal antibody (mAb) rituximab, the use of rituximab proved to be a protective factor against RS transformation, leading to lower rates of progression to RS in patients receiving rituximab [13]. A retrospective study suggested a potential role played by prolymphocytes in RS development, underlining that deaths due to RS were significantly more common in CLL patients who had ≥10% circulating prolymphocytes [14].
More recently, a pooled analysis of the German CLL Study Group (GCLLSG) considering frontline treatment trials with both CIT and pathway inhibitors, including Bruton tyrosine kinase (BTK) and BCL2 inhibitors, has documented a 3% prevalence of RS transformation among 2975 CLL patients monitored after their enrolment in clinical trials, recruited from 1999 to 2016, with a median observation time of 53 months [15]. Data from the Surveillance, Epidemiology and End Results (SEER) database of CLL patients diagnosed between 2000 and 2016 have documented that the incidence of RS transformation was 0.7% [16].
The issue of RS epidemiology in the era of novel agents has been partially answered by results collected from the first clinical trials with pathway inhibitors: in first-line treatment, novel agents showed RS transformation rates comparable to those of the CIT era, suggesting that pathway inhibitors are neither harmful nor fully protective [17][18]. Among relapsed/refractory (R/R) CLL patients treated with novel agents, the RS incidence was higher than the overall incidence of RS in the CIT era, probably due to the biological behavior and genetic profile of R/R CLL [19][20][21][22]. Because pathway inhibitors have been approved for the treatment of CLL relatively recently (the first BTK inhibitor, ibrutinib, was approved in 2014), further investigation is needed to assess the RS transformation rate with these novel agents in a real-life setting. Currently available data on RS frequency in cases treated with pathway inhibitors are summarized in Table 1.
Table 1. RS frequency in patients treated with therapeutic regimens based on pathway inhibitors.
| Number of CLL Patients | Study Population | Treatment | RS Prevalence (%) | Reference |
|---|---|---|---|---|
| 391 | Relapsed | Ibrutinib, ofatumumab | 1 | Byrd, 2014 [23] |
| 29 | Progressive untreated | Ibrutinib | 3 | O’Brien, 2014 [24] |
| 194 | R/R | Venetoclax-rituximab | 3 | Seymour, 2018 [19] |
| 127 | R/R | Ibrutinib | 5 | Jain, 2015 [25] |
| 84 | 17p deleted or ≥65 years | Ibrutinib | 6 | Ahn, 2017 [21] |
| 358 | Treatment-naïve | Acalabrutinib, Obinutuzumab | 2 | Sharman, 2020 [18] |
| 51 | 17p deleted | Ibrutinib | 6 | Farooqui, 2015 [26] |
| 178 | BCRi treated | Ibrutinib, idelalisib | 7 | Mato, 2016 [27] |
| 113 | Treatment-naïve | Ibrutinib-obinutuzumab | 0 | Moreno, 2019 [17] |
| 85 | R/R | Ibrutinib | 8 | Byrd, 2013 [28] |
| 116 | R/R | Venetoclax | 16 | Roberts, 2016 [29] |
| 67 | R/R, 17p deleted | Venetoclax | 25 | Anderson, 2017 [30] |
| 2975 | R/R | B, F, C, Clb, rituximab, obinutuzumab, ibrutinib, venetoclax | 3 | Al-Sawaf, 2021 [15] |
| 195 | R/R | Ibrutinib | 10 | Munir, 2019 [20] |
3. Molecular Pathways in RS
Several molecular alterations associated with DLBCL-type RS have been described, whereas the development of HL-type RS has been studied less extensively and is thought to be similar to that of de novo HL and possibly linked to EBV-mediated immunosuppression, thus favoring CLL progression to HL [7][31][32].
Genetic lesions of RS. The pathogenesis of DLBCL-type RS is linked to the dysregulation of intracellular pathways involved in DNA damage response, tumor suppression, apoptosis, modulation of the cell cycle and proliferation. The main genetic lesions associated with DLBCL-type RS are represented by somatic mutations or disruptions of the TP53, CDKN2A, NOTCH1 and c-MYC genes (Figure 1) [2][5][6][7][33][34].
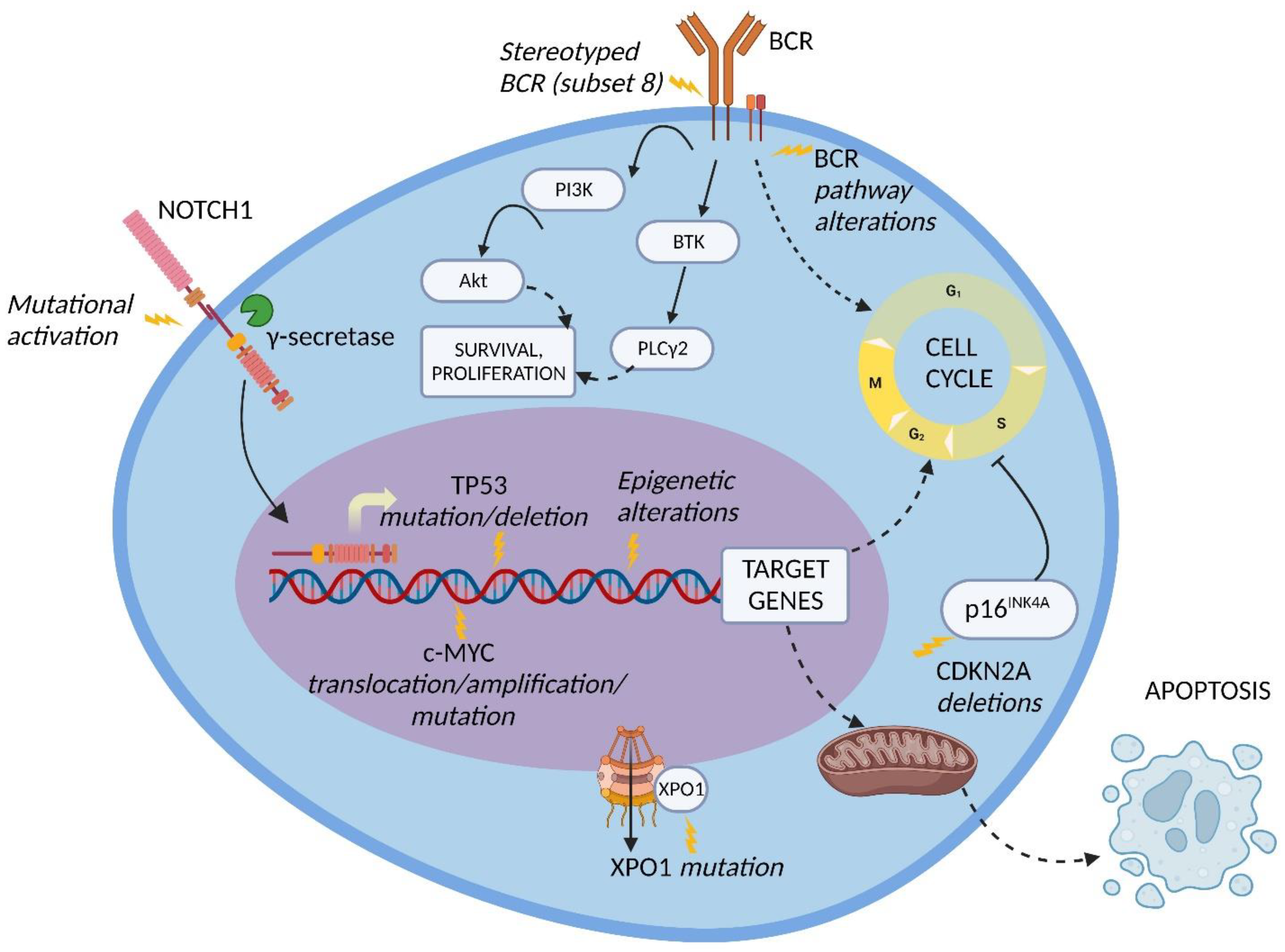
Figure 1. Molecular pathway alterations in DLBCL-type RS. The pathogenesis of DLBCL-type RS is due to the dysregulation of multiple molecular pathways due to genetic lesions of proto-oncogenes and tumor suppressor genes, stereotyped B-cell receptor (BCR) configuration, and BCR signaling alterations; these lead to the enhanced cell survival and proliferation typical of DLBCL-type RS cells. Inhibition of apoptosis may also be involved. BTK, Bruton tyrosine kinase; PI3K, phosphatidylinositol-3 kinase; PLCγ2, phospholipase C gamma 2; XPO1, exportin 1. Image created with Biorender.com (accessed on 4 August 2022).
TP53 encodes for one of the main regulators of the DNA-damage-response pathway, and its disruption, generally acquired at the time of transformation, leads to the chemorefractoriness characteristic of RS and, therefore, favors the positive selection and expansion of mutated tumor cells [34][35]. Disruption of TP53 by mutation and/or deletion has been documented in a large fraction of DLBCL-type RS, including 60 to 80% of clonally related RS cases, which represent the overwhelming majority of RS events, and in 20% of clonally unrelated RS cases [6]. The high recurrence of TP53 disruption explains, at least in part, the frequency of chemorefractoriness in this condition.
CDKN2A deletion occurs in ~30% of DLBCL-type RS and is commonly acquired at the time of transformation [5]. The CDKN2A gene is responsible for the negative regulation of the G1 to S transition of the cell cycle and for the activation of the p53 transcriptional program through its transcripts p16INK4A and p14ARF, respectively, leading to tumor suppression [5][34][36]. Importantly, both positive and negative cell cycle regulators are induced by B-cell receptor (BCR) signaling in murine models. On these grounds, the concomitant loss of the negative cell cycle regulators TP53 and CDKN2A/B shifts BCR-dependent signaling toward the promotion of positive cell cycle regulators, leading to an aggressive proliferation compatible with RS transformation [37].
NOTCH1 encodes for a surface receptor that, after being triggered by a ligand belonging to the SERRATE/JAGGED or DELTA families, is cleaved by γ-secretase, migrates into the nucleus and activates the transcription of several genes involved in cell proliferation and survival [38]. At the time of diagnosis, the frequency of NOTCH1 mutational activation in RS is significantly higher compared to the frequency in CLL (31% vs. 8.3%, respectively) [39]. These data, together with the noticeably higher reported risk of developing DLBCL-type RS in NOTCH1 mutated CLL (45% at 15 years) vs. NOTCH1 wild type CLL (4.6% at 15 years), indicate that mutations of NOTCH1 are a significant risk factor for developing RS transformation [40].
The c-MYC proto-oncogene and its transcriptional product take part in many crucial cellular pathways, and a dysregulation of this network results in altered cell survival, proliferation, metabolism, self-renewal, and genomic instability [41]. The main genetic lesions deregulating c-MYC expression in DLBCL-type RS are represented by chromosomal translocations between the c-MYC locus and the IGHV regulatory regions, and gene amplifications as well as gain-of-function mutations of the c-MYC promoter. An alternative mechanism of c-MYC deregulation is represented by loss-of-function mutations of the MGA gene, which encodes for a protein inhibiting c-MYC heterodimerization with its partner MAX. Overall, by combining genetic lesions of c-MYC and MGA, ~40% of DLBCL-type RS cases harbor c-MYC deregulation [42].
The NF-κB pathway is also involved in RS pathogenesis [43]. TRAF3, a gene implicated in the negative regulation of signaling through the NF-κB and mitogen-activated protein kinase (MAPK) pathways, is disrupted by heterozygous deletions and frameshift mutations in a fraction of RS cases [43]. Inactivation of TRAF3 leads to NF-κB activation, promoting B cell survival and, in particular, enhancing the expression of c-MYC and PIM-2 [44]. PIM-2 maintains high levels of NF-κB, which are required for its antiapoptotic function. The pathogenetic role of PIM-2 in B-cell neoplasia is documented by its overexpression, translocation, or amplification in a fraction of B-cell lymphomas [43][44][45][46].
Other molecular alterations detected in DLBCL-type RS include (i) overexpression and amplification of PTPN11, a positive regulator of the MAPK-RAS-ERK signaling pathway; (ii) deletion of the SETD2 histone methyltransferase, which plays a major role in chromatin epigenetic remodeling; and (iii) disrupting mutations of the tumor suppressor gene PTPRD, which encodes for a receptor-type protein, tyrosine phosphatase, regulating cell growth and found to be inactivated also in other types of B-cell neoplasia and solid cancers [43][47][48][49][50].
Modification of immune regulators in RS. Recent studies have underlined the importance of immune checkpoints and of the tumor microenvironment in lymphatic tissues of DLBCL-type RS [4][51]. The main immune checkpoints involved in RS are PD-1, LAG3 and TIGIT.
PD-1 is a T cell surface molecule which stimulates effector T-cell apoptosis and Treg survival through its binding with the PD-L1 ligand, which is expressed mainly on the surface of antigen-presenting cells (APC), such as macrophages, B cells and dendritic cells (DC) [52]. Augmented levels of PD-1 in RS cells and enhanced expression of PD-L1 in histiocytes and dendritic cells of the RS microenvironment have been reported [4][51]. Altered expression of the PD-1/PD-L1 axis leads to RS tumor-cell resistance to the cytotoxicity exerted by T cells [52].
LAG3 is a T cell surface protein with a structure similar to the T helper antigen CD4, and its main ligand is the major histocompatibility complex (MHC) class II, typically expressed by APC. Additional ligands have been identified, namely Galectin-3 (Gal-3), which is expressed by several cell types [53]. The interaction between LAG3 and its ligands promotes tumor escape from apoptosis through the recruitment of tumor-specific CD4+ T cells (through the interaction with MHC class II) and the inhibition of CD8+ T cells’ cytotoxic function by Gal-3 binding [53]. Higher LAG3 levels have been observed in RS neoplastic and tumor-infiltrating lymphocytes, suggesting its potential role in promoting tumor immune escape and neoplastic cell survival [54].
TIGIT, expressed on normal T and NK cells and overexpressed in RS, is capable of immune suppression as a consequence of its binding with the CD155 ligand, exposed on the cell membrane of various cell types, such as dendritic cells, T cells, B cells, and macrophages [54][55]. The mechanism of action of TIGIT is supposed to be linked to the transduction of immune-suppressive stimuli on T and NK cells and to the promotion of tolerogenic DC that downregulate T cell responses [55]. The finding that immune checkpoints are overexpressed in RS cells and tumor-infiltrating lymphocytes suggests the potential role of these molecules in the promotion of a permissive immune microenvironment, resulting in immune suppression and tumor escape.
The BCR pathway in RS. Several BCR pathway alterations related to RS transformation have been documented. The BCR is a transmembrane complex expressed in B cells, composed by a surface immunoglobulin linked to a signal transduction subunit and responsible for antigen recognition and B cell activation (Figure 1) [56].
The variable part of the BCR IGHV subunit is characterized by a molecular pattern typical of mature B cells, the VDJ rearrangement, which causes a considerable diversity across the BCR expressed by different B cell clones. Approximately thirty percent of CLL patients carry stereotyped BCR, which are characterized by almost identical VDJ rearrangement across patients and are groupable in well-codified subsets identified by progressive numbers [57][58].
CLL patients carrying BCR subset 8 (characterized by IGHV4-39/IGHD6-13/IGHJ5 rearrangement) display a significantly higher risk of developing DLBCL-type RS, especially in combination with NOTCH1 mutations. [59][60]. From a mechanistic perspective, CLL cells harboring BCR subset 8 tend to overreact to multiple autoantigens and immune stimuli derived from the microenvironment (Figure 1). The propensity of these cells to undergo RS transformation can be explained by this promiscuous antigen reactivity [61].
In the BCR signal transduction, a key role is played by Bruton tyrosine kinase (BTK) and phosphatidylinositol-3 kinase (PI3K) [62]. BTK is phosphorylated subsequently to BCR stimulation and leads to the activation of phospholipase C gamma 2 (PLCγ2), causing calcium mobilization and activation of cell survival, proliferation and differentiation pathways, including MAPK and NF-κB signaling (Figure 1) [63]. PI3K, which is responsible for the activation of the serine/threonine kinase Akt and for the delta isoform of protein kinase C (PKC), was observed to be constitutively active in CLL patients, resulting in an enhanced anti-apoptotic effect [64].
Akt signaling in RS. Akt takes part in cell-survival signaling through mTOR and is constitutively active in high-risk CLL (i.e., TP53 or NOTCH1 mutated CLL) and in >50% of cases of RS [64][65]. In an Eµ-TCL1 murine model of CLL with constitutively active Akt alleles in B cells, the excessive Akt activation led to an aggressive DLBCL-type lymphoma with histological and biological features coherent with human RS [65]. Additionally, this murine model enlightened the correlation between hyperactivation of Akt and NOTCH1 signaling, since mice with constitutively active Akt alleles presented an expansion of CD4+ T cells expressing the NOTCH1 ligand DLL1 in the tumor microenvironment, implying a higher engagement of NOTCH1 by its ligands in neoplastic cells [65].
References
- Richter, M.N. Generalized Reticular Cell Sarcoma of Lymph Nodes Associated with Lymphatic Leukemia. Am. J. Pathol. 1928, 4, 285–292.287.
- Swerdlow, S.H.; Campo, E.; Harris, N.L.; Jaffe, E.S.; Pileri, S.A.; Stein, H.; Thiele, J. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues, Revised, Revised 4th ed.; International Agency for Research on Cancer: Lyon, France, 2017; Volume 2.
- Mao, Z.; Quintanilla-Martinez, L.; Raffeld, M.; Richter, M.; Krugmann, J.; Burek, C.; Hartmann, E.; Rudiger, T.; Jaffe, E.S.; Müller-Hermelink, H.K.; et al. IgVH mutational status and clonality analysis of Richter’s transformation: Diffuse large B-cell lymphoma and Hodgkin lymphoma in association with B-cell chronic lymphocytic leukemia (B-CLL) represent 2 different pathways of disease evolution. Am. J. Surg. Pathol. 2007, 31, 1605–1614.
- He, R.; Ding, W.; Viswanatha, D.S.; Chen, D.; Shi, M.; Van Dyke, D.; Tian, S.; Dao, L.N.; Parikh, S.A.; Shanafelt, T.D.; et al. PD-1 Expression in Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma (CLL/SLL) and Large B-cell Richter Transformation (DLBCL-RT): A Characteristic Feature of DLBCL-RT and Potential Surrogate Marker for Clonal Relatedness. Am. J. Surg. Pathol. 2018, 42, 843–854.
- Fabbri, G.; Khiabanian, H.; Holmes, A.B.; Wang, J.; Messina, M.; Mullighan, C.G.; Pasqualucci, L.; Rabadan, R.; Dalla-Favera, R. Genetic lesions associated with chronic lymphocytic leukemia transformation to Richter syndrome. J. Exp. Med. 2013, 210, 2273–2288.
- Rossi, D.; Spina, V.; Deambrogi, C.; Rasi, S.; Laurenti, L.; Stamatopoulos, K.; Arcaini, L.; Lucioni, M.; Rocque, G.B.; Xu-Monette, Z.Y.; et al. The genetics of Richter syndrome reveals disease heterogeneity and predicts survival after transformation. Blood 2011, 117, 3391–3401.
- Condoluci, A.; Rossi, D. Biology and Treatment of Richter Transformation. Front. Oncol. 2022, 12, 829983.
- Gupta, N.; Mittal, A.; Duggal, R.; Dadu, T.; Agarwal, A.; Handoo, A. Hodgkin Variant of Richter’s Transformation in Chronic Lymphocytic Leukemia (CLL): An Illustrative Case Report and Literature Review. Int. J. Hematol. Oncol. Stem Cell Res. 2021, 15, 249–254.
- Xiao, W.; Chen, W.W.; Sorbara, L.; Davies-Hill, T.; Pittaluga, S.; Raffeld, M.; Jaffe, E.S. Hodgkin lymphoma variant of Richter transformation: Morphology, Epstein-Barr virus status, clonality, and survival analysis-with comparison to Hodgkin-like lesion. Hum. Pathol. 2016, 55, 108–116.
- Chronic Lymphocytic Leukemia-Cancer Stat Facts. Surveillance, Epidemiology, and End Results Program. 2022. Available online: https://seer.cancer.gov/statfacts/html/clyl.html (accessed on 7 September 2022).
- Rossi, D.; Gaidano, G. Richter syndrome. Adv. Exp. Med. Biol. 2013, 792, 173–191.
- Solh, M.; Rai, K.R.; Peterson, B.L.; Kolitz, J.E.; Appelbaum, F.R.; Tallman, M.S.; Belch, A.; Larson, R.A.; Morrison, V.A. The impact of initial fludarabine therapy on transformation to Richter syndrome or prolymphocytic leukemia in patients with chronic lymphocytic leukemia: Analysis of an intergroup trial (CALGB 9011). Leuk Lymphoma 2013, 54, 252–254.
- Fischer, K.; Bahlo, J.; Fink, A.M.; Goede, V.; Herling, C.D.; Cramer, P.; Langerbeins, P.; von Tresckow, J.; Engelke, A.; Maurer, C.; et al. Long-term remissions after FCR chemoimmunotherapy in previously untreated patients with CLL: Updated results of the CLL8 trial. Blood 2016, 127, 208–215.
- Oscier, D.; Else, M.; Matutes, E.; Morilla, R.; Strefford, J.C.; Catovsky, D. The morphology of CLL revisited: The clinical significance of prolymphocytes and correlations with prognostic/molecular markers in the LRF CLL4 trial. Br. J. Haematol. 2016, 174, 767–775.
- Al-Sawaf, O.; Robrecht, S.; Bahlo, J.; Fink, A.M.; Cramer, P.; Tresckow, V.T.; Lange, E.; Kiehl, M.; Dreyling, M.; Ritgen, M.; et al. Richter transformation in chronic lymphocytic leukemia (CLL)-a pooled analysis of German CLL Study Group (GCLLSG) front line treatment trials. Leukemia 2021, 35, 169–176.
- Elnair, R.; Ellithi, M.; Kallam, A.; Shostrom, V.; Bociek, R.G. Outcomes of Richter’s transformation of chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL): An analysis of the SEER database. Ann. Hematol. 2021, 100, 2513–2519.
- Moreno, C.; Greil, R.; Demirkan, F.; Tedeschi, A.; Anz, B.; Larratt, L.; Simkovic, M.; Samoilova, O.; Novak, J.; Ben-Yehuda, D.; et al. Ibrutinib plus obinutuzumab versus chlorambucil plus obinutuzumab in first-line treatment of chronic lymphocytic leukaemia (iLLUMINATE): A multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2019, 20, 43–56.
- Sharman, J.P.; Egyed, M.; Jurczak, W.; Skarbnik, A.; Pagel, J.M.; Flinn, I.W.; Kamdar, M.; Munir, T.; Walewska, R.; Corbett, G.; et al. Acalabrutinib with or without obinutuzumab versus chlorambucil and obinutuzmab for treatment-naive chronic lymphocytic leukaemia (ELEVATE TN): A randomised, controlled, phase 3 trial. Lancet 2020, 395, 1278–1291.
- Seymour, J.F.; Kipps, T.J.; Eichhorst, B.; Hillmen, P.; D’Rozario, J.; Assouline, S.; Owen, C.; Gerecitano, J.; Robak, T.; De la Serna, J.; et al. Venetoclax-Rituximab in Relapsed or Refractory Chronic Lymphocytic Leukemia. N. Engl. J. Med. 2018, 378, 1107–1120.
- Munir, T.; Brown, J.R.; O’Brien, S.; Barrientos, J.C.; Barr, P.M.; Reddy, N.M.; Coutre, S.; Tam, C.S.; Mulligan, S.P.; Jaeger, U.; et al. Final analysis from RESONATE: Up to six years of follow-up on ibrutinib in patients with previously treated chronic lymphocytic leukemia or small lymphocytic lymphoma. Am. J. Hematol. 2019, 94, 1353–1363.
- Ahn, I.E.; Underbayev, C.; Albitar, A.; Herman, S.E.; Tian, X.; Maric, I.; Arthur, D.C.; Wake, L.; Pittaluga, S.; Yuan, C.M.; et al. Clonal evolution leading to ibrutinib resistance in chronic lymphocytic leukemia. Blood 2017, 129, 1469–1479.
- Brown, J.R.; Hillmen, P.; O’Brien, S.; Barrientos, J.C.; Reddy, N.M.; Coutre, S.E.; Tam, C.S.; Mulligan, S.P.; Jaeger, U.; Barr, P.M.; et al. Extended follow-up and impact of high-risk prognostic factors from the phase 3 RESONATE study in patients with previously treated CLL/SLL. Leukemia 2018, 32, 83–91.
- Byrd, J.C.; Brown, J.R.; O’Brien, S.; Barrientos, J.C.; Kay, N.E.; Reddy, N.M.; Coutre, S.; Tam, C.S.; Mulligan, S.P.; Jaeger, U.; et al. Ibrutinib versus ofatumumab in previously treated chronic lymphoid leukemia. N. Engl. J. Med. 2014, 371, 213–223.
- O’Brien, S.; Furman, R.R.; Coutre, S.E.; Sharman, J.P.; Burger, J.A.; Blum, K.A.; Grant, B.; Richards, D.A.; Coleman, M.; Wierda, W.G.; et al. Ibrutinib as initial therapy for elderly patients with chronic lymphocytic leukaemia or small lymphocytic lymphoma: An open-label, multicentre, phase 1b/2 trial. Lancet Oncol. 2014, 15, 48–58.
- Jain, P.; Keating, M.; Wierda, W.; Estrov, Z.; Ferrajoli, A.; Jain, N.; George, B.; James, D.; Kantarjian, H.; Burger, J.; et al. Outcomes of patients with chronic lymphocytic leukemia after discontinuing ibrutinib. Blood 2015, 125, 2062–2067.
- Farooqui, M.Z.H.; Valdez, J.; Martyr, S.; Aue, G.; Saba, N.; Niemann, C.U.; Herman, S.E.M.; Tian, X.; Marti, G.; Soto, S.; et al. Ibrutinib for previously untreated and relapsed or refractory chronic lymphocytic leukaemia with TP53 aberrations: A phase 2, single-arm trial. Lancet Oncol. 2015, 16, 169–176.
- Mato, A.R.; Nabhan, C.; Barr, P.M.; Ujjani, C.S.; Hill, B.T.; Lamanna, N.; Skarbnik, A.P.; Howlett, C.; Pu, J.J.; Sehgal, A.R.; et al. Outcomes of CLL patients treated with sequential kinase inhibitor therapy: A real world experience. Blood 2016, 128, 2199–2205.
- Byrd, J.C.; Furman, R.R.; Coutre, S.E.; Flinn, I.W.; Burger, J.A.; Blum, K.A.; Grant, B.; Sharman, J.P.; Coleman, M.; Wierda, W.G.; et al. Targeting BTK with ibrutinib in relapsed chronic lymphocytic leukemia. N. Engl. J. Med. 2013, 369, 32–42.
- Roberts, A.W.; Davids, M.S.; Pagel, J.M.; Kahl, B.S.; Puvvada, S.D.; Gerecitano, J.F.; Kipps, T.J.; Anderson, M.A.; Brown, J.R.; Gressick, L.; et al. Targeting BCL2 with Venetoclax in Relapsed Chronic Lymphocytic Leukemia. N. Engl. J. Med. 2016, 374, 311–322.
- Anderson, M.A.; Tam, C.; Lew, T.E.; Juneja, S.; Juneja, M.; Westerman, D.; Wall, M.; Lade, S.; Gorelik, A.; Huang, D.C.S.; et al. Clinicopathological features and outcomes of progression of CLL on the BCL2 inhibitor venetoclax. Blood 2017, 129, 3362–3370.
- Kaźmierczak, M.; Kroll-Balcerzak, R.; Balcerzak, A.; Czechowska, E.; Gil, L.; Sawiński, K.; Szczepaniak, A.; Komarnicki, M. Hodgkin lymphoma transformation of chronic lymphocytic leukemia: Cases report and discussion. Med. Oncol. 2014, 31, 800.
- Tsimberidou, A.M.; Keating, M.J. Richter syndrome: Biology, incidence, and therapeutic strategies. Cancer 2005, 103, 216–228.
- Chigrinova, E.; Rinaldi, A.; Kwee, I.; Rossi, D.; Rancoita, P.M.; Strefford, J.C.; Oscier, D.; Stamatopoulos, K.; Papadaki, T.; Berger, F.; et al. Two main genetic pathways lead to the transformation of chronic lymphocytic leukemia to Richter syndrome. Blood 2013, 122, 2673–2682.
- Rossi, D.; Spina, V.; Gaidano, G. Biology and treatment of Richter syndrome. Blood 2018, 131, 2761–2772.
- Duffy, M.J.; Synnott, N.C.; Crown, J. Mutant p53 as a target for cancer treatment. Eur. J. Cancer 2017, 83, 258–265.
- Sherr, C.J. Ink4-Arf locus in cancer and aging. Wiley Interdiscip. Rev. Dev. Biol. 2012, 1, 731–741.
- Chakraborty, S.; Martines, C.; Porro, F.; Fortunati, I.; Bonato, A.; Dimishkovska, M.; Piazza, S.; Yadav, B.S.; Innocenti, I.; Fazio, R.; et al. B-cell receptor signaling and genetic lesions in TP53 and CDKN2A/CDKN2B cooperate in Richter transformation. Blood 2021, 138, 1053–1066.
- Rosati, E.; Baldoni, S.; De Falco, F.; Del Papa, B.; Dorillo, E.; Rompietti, C.; Albi, E.; Falzetti, F.; Di Ianni, M.; Sportoletti, P. NOTCH1 Aberrations in Chronic Lymphocytic Leukemia. Front. Oncol. 2018, 8, 229.
- Fabbri, G.; Rasi, S.; Rossi, D.; Trifonov, V.; Khiabanian, H.; Ma, J.; Grunn, A.; Fangazio, M.; Capello, D.; Monti, S.; et al. Analysis of the chronic lymphocytic leukemia coding genome: Role of NOTCH1 mutational activation. J. Exp. Med. 2011, 208, 1389–1401.
- Rossi, D.; Rasi, S.; Spina, V.; Fangazio, M.; Monti, S.; Greco, M.; Ciardullo, C.; Famà, R.; Cresta, S.; Bruscaggin, A.; et al. Different impact of NOTCH1 and SF3B1 mutations on the risk of chronic lymphocytic leukemia transformation to Richter syndrome. Br. J. Haematol. 2012, 158, 426–429.
- Dhanasekaran, R.; Deutzmann, A.; Mahauad-Fernandez, W.D.; Hansen, A.S.; Gouw, A.M.; Felsher, D.W. The MYC oncogene—The grand orchestrator of cancer growth and immune evasion. Nat. Rev. Clin. Oncol. 2022, 19, 23–36.
- De Paoli, L.; Cerri, M.; Monti, S.; Rasi, S.; Spina, V.; Bruscaggin, A.; Greco, M.; Ciardullo, C.; Famà, R.; Cresta, S.; et al. MGA, a suppressor of MYC, is recurrently inactivated in high risk chronic lymphocytic leukemia. Leuk Lymphoma 2013, 54, 1087–1090.
- Klintman, J.; Appleby, N.; Stamatopoulos, B.; Ridout, K.; Eyre, T.A.; Robbe, P.; Pascua, L.L.; Knight, S.J.L.; Dreau, H.; Cabes, M.; et al. Genomic and transcriptomic correlates of Richter transformation in chronic lymphocytic leukemia. Blood 2021, 137, 2800–2816.
- Whillock, A.L.; Mambetsariev, N.; Lin, W.W.; Stunz, L.L.; Bishop, G.A. TRAF3 regulates the oncogenic proteins Pim2 and c-Myc to restrain survival in normal and malignant B cells. Sci. Rep. 2019, 9, 12884.
- Gómez-Abad, C.; Pisonero, H.; Blanco-Aparicio, C.; Roncador, G.; González-Menchén, A.; Martinez-Climent, J.A.; Mata, E.; Rodríguez, M.E.; Muñoz-González, G.; Sánchez-Beato, M.; et al. PIM2 inhibition as a rational therapeutic approach in B-cell lymphoma. Blood 2011, 118, 5517–5527.
- Mondello, P.; Cuzzocrea, S.; Mian, M. Pim kinases in hematological malignancies: Where are we now and where are we going? J. Hematol. Oncol. 2014, 7, 95.
- Dance, M.; Montagner, A.; Salles, J.P.; Yart, A.; Raynal, P. The molecular functions of Shp2 in the Ras/Mitogen-activated protein kinase (ERK1/2) pathway. Cell Signal. 2008, 20, 453–459.
- Wagner, E.J.; Carpenter, P.B. Understanding the language of Lys36 methylation at histone H3. Nat. Rev. Mol. Cell Biol. 2012, 13, 115–126.
- Veeriah, S.; Brennan, C.; Meng, S.; Singh, B.; Fagin, J.A.; Solit, D.B.; Paty, P.B.; Rohle, D.; Vivanco, I.; Chmielecki, J.; et al. The tyrosine phosphatase PTPRD is a tumor suppressor that is frequently inactivated and mutated in glioblastoma and other human cancers. Proc. Natl. Acad. Sci. USA 2009, 106, 9435–9440.
- Spina, V.; Khiabanian, H.; Messina, M.; Monti, S.; Cascione, L.; Bruscaggin, A.; Spaccarotella, E.; Holmes, A.B.; Arcaini, L.; Lucioni, M.; et al. The genetics of nodal marginal zone lymphoma. Blood 2016, 128, 1362–1373.
- Wang, Y.; Sinha, S.; Wellik, L.E.; Secreto, C.R.; Rech, K.L.; Call, T.G.; Parikh, S.A.; Kenderian, S.S.; Muchtar, E.; Hayman, S.R.; et al. Distinct immune signatures in chronic lymphocytic leukemia and Richter syndrome. Blood Cancer J. 2021, 11, 86.
- Salmaninejad, A.; Valilou, S.F.; Shabgah, A.G.; Aslani, S.; Alimardani, M.; Pasdar, A.; Sahebkar, A. PD-1/PD-L1 pathway: Basic biology and role in cancer immunotherapy. J. Cell Physiol. 2019, 234, 16824–16837.
- Ruffo, E.; Wu, R.C.; Bruno, T.C.; Workman, C.J.; Vignali, D.A.A. Lymphocyte-activation gene 3 (LAG3): The next immune checkpoint receptor. Semin. Immunol. 2019, 42, 101305.
- Gould, C.; Lickiss, J.; Kankanige, Y.; Yerneni, S.; Lade, S.; Gandhi, M.K.; Chin, C.; Yannakou, C.K.; Villa, D.; Slack, G.W.; et al. Characterisation of immune checkpoints in Richter syndrome identifies LAG3 as a potential therapeutic target. Br. J. Haematol. 2021, 195, 113–118.
- Harjunpää, H.; Guillerey, C. TIGIT as an emerging immune checkpoint. Clin. Exp. Immunol. 2020, 200, 108–119.
- Treanor, B. B-cell receptor: From resting state to activate. Immunology 2012, 136, 21–27.
- Gerousi, M.; Laidou, S.; Gemenetzi, K.; Stamatopoulos, K.; Chatzidimitriou, A. Distinctive Signaling Profiles With Distinct Biological and Clinical Implications in Aggressive CLL Subsets With Stereotyped B-Cell Receptor Immunoglobulin. Front. Oncol. 2021, 11, 771454.
- Stamatopoulos, K.; Belessi, C.; Moreno, C.; Boudjograh, M.; Guida, G.; Smilevska, T.; Belhoul, L.; Stella, S.; Stavroyianni, N.; Crespo, M.; et al. Over 20% of patients with chronic lymphocytic leukemia carry stereotyped receptors: Pathogenetic implications and clinical correlations. Blood 2007, 109, 259–270.
- Rossi, D.; Spina, V.; Cerri, M.; Rasi, S.; Deambrogi, C.; De Paoli, L.; Laurenti, L.; Maffei, R.; Forconi, F.; Bertoni, F.; et al. Stereotyped B-cell receptor is an independent risk factor of chronic lymphocytic leukemia transformation to Richter syndrome. Clin. Cancer Res. 2009, 15, 4415–4422.
- Rossi, D.; Spina, V.; Bomben, R.; Rasi, S.; Dal-Bo, M.; Bruscaggin, A.; Rossi, F.M.; Monti, S.; Degan, M.; Ciardullo, C.; et al. Association between molecular lesions and specific B-cell receptor subsets in chronic lymphocytic leukemia. Blood 2013, 121, 4902–4905.
- Gounari, M.; Ntoufa, S.; Apollonio, B.; Papakonstantinou, N.; Ponzoni, M.; Chu, C.C.; Rossi, D.; Gaidano, G.; Chiorazzi, N.; Stamatopoulos, K.; et al. Excessive antigen reactivity may underlie the clinical aggressiveness of chronic lymphocytic leukemia stereotyped subset #8. Blood 2015, 125, 3580–3587.
- Burger, J.A.; Wiestner, A. Targeting B cell receptor signalling in cancer: Preclinical and clinical advances. Nat. Rev. Cancer 2018, 18, 148–167.
- Cheng, S.; Ma, J.; Guo, A.; Lu, P.; Leonard, J.P.; Coleman, M.; Liu, M.; Buggy, J.J.; Furman, R.R.; Wang, Y.L. BTK inhibition targets in vivo CLL proliferation through its effects on B-cell receptor signaling activity. Leukemia 2014, 28, 649–657.
- Ringshausen, I.; Schneller, F.; Bogner, C.; Hipp, S.; Duyster, J.; Peschel, C.; Decker, T. Constitutively activated phosphatidylinositol-3 kinase (PI-3K) is involved in the defect of apoptosis in B-CLL: Association with protein kinase Cdelta. Blood 2002, 100, 3741–3748.
- Kohlhaas, V.; Blakemore, S.J.; Al-Maarri, M.; Nickel, N.; Pal, M.; Roth, A.; Hövelmeyer, N.; Schäfer, S.C.; Knittel, G.; Lohneis, P.; et al. Active Akt signaling triggers CLL toward Richter transformation via overactivation of Notch1. Blood 2021, 137, 646–660.
More
Information
Subjects:
Hematology; Biochemistry & Molecular Biology; Immunology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.1K
Revisions:
2 times
(View History)
Update Date:
03 Feb 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





